Abstract
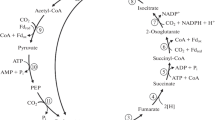
The strict anaerobe Desulfuromonas acetoxidans can oxidize acetate to CO2 with elemental sulfur as electron acceptor. 14C-labelling experiments and enzyme studies are described revealing that acetate oxidation proceeds via the citric acid cycle with the synthesis of oxaloacetate from acetate and 2 CO2 via pyruvate as anaplerotic reaction. An oxidation of acetate via one carbon unit intermediates as proposed for anaerobic bacteria fermenting acetate to 2 CO2 and 4 H2 was excluded.
Similar content being viewed by others
References
Aleem MIH (1977) Coupling of energy with electron transfer reactions in chemolithotrophic bacteria. In: Haddock BA, Hamilton WA (eds) Microbial energetics (27th Symposium of the Society for General Microbiology, London 1977). Cambridge University Press, Cambridge London New York Melbourne, pp 351–381
Bache R, Kroneck PMH, Merkle H, Beinert H (1983) A survey of EPR-detectable components in sulfur-reducing bacteria. Biochim Biophys Acta 722:417–426
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Brandis-Heep A, Gebhardt NA, Thauer RK, Widdel F, Pfennig N (1983) Anaerobic acetate oxidation to CO2 by Desulfobacter postgatei. 1. Demonstration of all enzymes required for the operation of the cytric acid cycle. Arch Microbiol 136:222–229
Buchanan BB (1972) Ferredoxin-linked carboxylation reactions. In: Boyer PD (ed) The enzymes, vol VI. Academic Press, New York London, pp 193–216
Diekert G, Fuchs G, Thauer RK (1984) Properties and function of carbon monoxide dehydrogenase from anaerobic bacteria. In: Pole RK (ed) Microbial gas metabolism. Academic Press, New York (in press)
Fuchs G, Stupperich E, Eden G (1980) Autotrophic CO2 fixation in Chlorobium limicola. Evidence for the operation, of a reductive tricarboxylic acid cycle in growing cells. Arch Microbiol 128:64–71
Fuchs G, Stupperich E, Thauer RK (1978) Acetate assimilation and the synthesis of alanine, aspartate and glutamate in Methanobacterium thermoautotrophicum. Arch Microbiol 117:61–66
Gebhardt NA, Linder D, Thauer RK (1983) Anaerobic acetate oxidation to CO2 by Desulfobacter postgatei 2. Evidence from 14C-labelling studies for the operation of the citric acid cycle. Arch Microbiol 136:230–233
Gottschalk G (1968) The stereospecificity of the citrate synthase in sulfate-reducing and photosynthetic bacteria. Eur J Biochem 5:346–351
Gottschalk G, Barker HA (1966) Synthesis of glutamate and citrate by Clostridium kluyveri. A new type of citrate synthase. Biochemistry 5:1125–1133
Gottschalk G, Barker HA (1967) Presence and stereospecificity of citrate synthase in anaerobic bacteria. Biochemistry 6:1027–1034
Gottschalk G, Dittbrenner S (1970) Properties of (R)-citrate synthase from Clostridium acidi-urici. Hoppe-Seyler's Z Physiol Chem 351:1183–1190
Hederstedt L, Rutberg L (1981) Succinate dehydrogenase—a comparative review. Microbiol Rev 45:542–555
Holländer R, Wolf G, Mannheim W (1977) Lipoquinone of some bacteria and mycoplasms, with consideration on their functional significance. Antonie van Leeuwenhoek 43:177–185
Kaulfers PM (1976) Untersuchungen über Enzyme des Intermediärstoffwechsels von Desulfuromonas acetoxidans Stamm 11070. Diploma Thesis, Univ Göttingen
Kerscher L, Oesterhelt D (1981) The catalytic mechanism of 2-oxoacid: ferredoxin oxidoreductases from Halobacterium halobium. One-electron transfer at two distinct steps of the catalytic cycle. Eur J Biochem 116:595–600
Kerscher L, Oesterhelt D (1982) Pyruvate: ferredoxin oxidoreductase—new findings on an ancient enzyme. TIBS 7:371–374
Klingenberg M, Schollmeyer P (1960) Zur Reversibilität der oxydativen Phosphorylierung. Adenosintriphosphat-abhängige Atmungskontrolle und Reduktion von Diphosphopyridin-nucleotid in Mitochondrien. Biochem Z 333:335–350
Knaff DB (1978) Reducing potentials and the pathway of NAD+ reduction. In: Clayton RK, Sistrom WR (eds) The photosynthetic bacteria. Plenum Press, New York London, pp 629–640
Kornberg HL (1966) Anaplerotic sequences and their role in metabolism. In: Campbell PN, Greville GD (eds) Assays in biochemistry, vol. 2. Academic Press, New York London, pp 1–31
Krebs HA, Johnson WA (1937) The role of citric acid in intermediate metabolism in animal tissues. Enzymologia 4:148–156
Kröger A (1978) Determination of contents and redox states of ubiquinone and menaquionone. In: Colowick SP, Kaplan NO (eds) Methods in enzymology, vol. LIII. Academic Press, New York San Francisco London, pp 579–591
Ljungdahl LG, Wood HG (1982) Acetate biosynthesis. In: Dolphin D (ed) B12: Biochemistry and medicine. John Wiley& Sons, New York Chichester Brisbane Toronto Singapore, pp 166–202
Lowenstein JM (1967) The tricarboxylic acid cycle. In: Greenberg DM (ed) Metabolic pathways, vol I. Academic Press, New York London, pp 146–270
Ormerod JG, Gest H (1960) Symposium on metabolism of inorganic compounds. IV. Hydrogen photosynthesis and alternative metabolic pathways in photosynthetic bacteria. Bacteriol Rev 24:51–66
Payne WJ (1973) Reduction of nitrogenous oxides by microorganisms. Bacteriol Rev 37:409–452
Pfennig N (1967) Photosynthetic bacteria. In: Clifton CE, Raffel, S, Starr MP (eds) Ann Rev Microbiol, vol. 21. Annual Reviews Inc. Palo Alto, California, pp 285–324
Pfennig N, Biebl H (1976) Desulfuromonas acetoxidans gen. nov. and sp. nov., a new anaerobic, sulfur-reducing, acetate-oxidizing bacterium. Arch Microbiol 110:2–12
Pfennig N, Widdel F (1981) Ecology and physiology of some anaerobic bacteria from the microbial sulfur cycle. In: Bothe H, Trebst A (eds) Biology of inorganic nitrogen and sulfur. Springer, Berlin Heidelberg New York, pp 169–177
Probst I, Bruschi M, Pfennig N, Le Gall J (1977) Cytochrome c-551.5 (c7) from Desulfuromonas acetoxidans. Biochim Biophys Acta 460:58–64
Probst I, Moura JJG, Moura I, Bruschi M, Le Gall J (1978) Isolation and characterization of a rubredoxin and an (8Fe-8S) ferredoxin from Desulfuromonas acetoxidans. Biochim Biophys Acta 502: 38–44
Simon H, Floss HG (1967) Bestimmung der Isotopenverteilung in markierten Verbindungen. Springer, Berlin Heidelberg New York, pp 53–64
Spangler WJ, Gilmour CM (1966) Biochemistry of nitrate respiration in Pseudomonas stutzeri. I. Aerobic and nitrate respiration routes of carbohydrate catabolism. J Bacteriol 91:245–250
Thauer RK, Jungermann K, Decker K (1977) Energy conservation in chemotrophic anaerobic bacteria. Bacteriol Rev 41:100–180
Thauer RK, Morris JG (1984) Metabolism of chemotrophic anaerobes: Old views and new aspects. In: Kelly DP, Carr NG (eds) The microbe 1984, part II (Symposium at the 100th meeting of the Society for General Microbiology, Warwick, April 1984). Cambridge University Press, Cambridge London New York Melbourne, pp 123–168
Weitzman PDJ (1981) Unity and diversity in some bacterial citric acid-cycle enzymes. In: Rose AH, Morris JG (eds) Advances in microbial physiology, vol 22. Academic Press, London New York Toronto Sydney San, Francisco, pp 185–244
Widdel F, Pfennig N (1981) Studies on dissimilatory sulfate-reducing bacteria that decompose fatty acids. 1. Isolation of new sulfate-reducing bacteria enriched with acetate from saline environments. Description of Desulfobacter postgatei gen. nov., sp. nov. Arch Microbiol 129:395–400
Wood HG, Drake HL, Hu SI (1982) Studies with Clostridium thermoaceticum and the resolution of the pathway used by acetogenic bacteria that grow on carbon monoxide or carbon dioxide and hydrogen. In: Snell EE (ed) Amino acids, fermentations, and nucleic acids: a symposium. Ann Rev Monograph, Ann Rev Inc, Palo Alto, California, pp 29–56
Zinder, SH, Koch M (1984) Non-acetoclastic methanogenesis from acetate: acetate oxidation by a thermophilic synthrophic coculture. Arch Microbiol 138:263–272
Author information
Authors and Affiliations
Additional information
Dedicated to Professor Dr. Gerhart Drews on the occasion of his 60th birthday
Rights and permissions
About this article
Cite this article
Gebhardt, N.A., Thauer, R.K., Linder, D. et al. Mechanism of acetate oxidation to CO2 with elemental sulfur in Desulfuromonas acetoxidans . Arch. Microbiol. 141, 392–398 (1985). https://doi.org/10.1007/BF00428855
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00428855