Abstract
The main objective of a plant breeding program is to deliver superior germplasm for farmers in a defined set of environments, or a target population of environments (TPE). Historically, CIMMYT has characterized the environments in which the developed germplasm will be grown. The main factors that determine when and where a wheat variety can be grown are flowering time, water availability and the incidence of pests and diseases. A TPE consists of many (population) environments and future years or seasons, that share common variation in the farmers’ fields, it can also be seen as a variable group of future production environments. TPEs can be characterized by climatic, soil and hydrological features, as well as socioeconomic aspects. Whereas the selection environments (SE) are the environments where the breeder does the selection of the lines. The SE are identified for predicting the performance in the TPE, but the SE may not belong to the TPE. The utilization of advanced statistical methods allows the identification of GEI to obtain higher precision when estimating the genetic effects. Multi-environmental testing (MET) is a fundamental strategy for CIMMYT to develop stable high grain yielding germplasm in countries with developing economies. An adequate MET strategy allows the evaluation of germplasm in stress hotspots and the identification of representative and correlated sites; thus, breeders can make better and targeted decisions in terms of crossing, selection and logistic operations.
You have full access to this open access chapter, Download chapter PDF
Similar content being viewed by others
Keywords
- Mega-environments (ME)
- Target population of environments (TPEs)
- Selection environments (SEs)
- Genotype-by-environment interaction (GEI)
1 Learning Objectives
-
Identify the factors that drive wheat adaptation for the classification of target environments.
-
Identify the statistical methods that can be used for defining TPEs.
-
Identify the importance of a multi-environmental testing strategy for wheat breeding with a global scope.
2 Introduction: Wheat Mega-environments in History and the Context of Global Wheat Breeding
The success of a breeding program and, particularly, of a program with international dimensions such as CIMMYT’s wheat breeding program, depends heavily on the characterization of the environments where the germplasm will be grown.
Historically, since its earliest efforts to breed wheat in the 1940s, CIMMYT has characterized the environments in which the developed germplasm will be grown. At that time, this characterization was restricted to the geographical areas in Mexico. However, as soon as CIMMYT’s mandate became global in the 1970s, 15 agroecological zones were defined, for instance, the region that encompasses India, Pakistan, Bangladesh and Nepal (South Asia), or the Nile Valley zone (Egypt and Sudan).
Those agroecological zones were redefined in the late 1980s. As it became evident that the sole geographical description was inadequate, given the diversification of the production systems, the need for high-yielding and stable germplasm and the simple fact that for specific conditions, certain traits were needed, particularly those related to stress tolerance and quality requirements. Hence, this redefinition led to the concept of Mega-Environment (ME), described by Rajaram et al. [1] as a “broad” but not necessarily “contiguous area” with similar biotic and abiotic constraints, cropping systems, consumer preferences and production volume. By 1992, twelve ME had been conceived, six for each spring and winter growth habit. Here we present only those corresponding to spring wheat (Table 3.1).
After CIMMYT’s target environment classification, and thanks to the historical data collected by the International Wheat Improvement Network (IWIN), this was followed by several reports on germplasm adaptation and performance in the context of ME (see Chap. 7). A study published by DeLacy et al. [4] demonstrated that the major discrimination factors for these ME were latitude and the presence/absence of stresses, plus the agreement between the ME-based locations and the location groups obtained from pattern analysis. Another study reported by Hodson and White [3] defined ME classification with the aid of GIS tools, and hence, a more quantitative and specific classification was proposed. In such a study, it was demonstrated that long term environmental variables, mainly temperature and precipitation in the coolest and wettest quarter of the year, were effective to separate environments based on abiotic stresses and growth habits, i.e., spring vs. winter growth type.
Additional studies derived from the historical data provided by the IWIN have shown how CIMMYT wheat germplasm performs and adapts throughout the locations within each ME [5,6,7]. These studies also demonstrated that CIMMYT’s main yield testing site located in northwest Mexico correlates positively with the locations that belong to each ME for spring wheat.
3 Major Factors That Broadly Impact the Definition of Target Environments
3.1 Flowering Time: Photoperiod and Vernalization
Flowering time is a fundamental adaptive trait, as it determines where and when a variety can be grown, and, in general, largely determines the reproductive success of a plant. Flowering must occur during an optimal environmental period that permits the full development of the reproductive organs. This period should also be long enough to allow optimal grain filling.
One factor that highly determines flowering time in wheat is photoperiod. In wheat there is a series of dominant genes (Ppd) located on chromosomes 2A, 2B and 2D that induce an insensitive reaction to photoperiod [8,9,10]. Photoperiod insensitivity means that plants reach flowering even under short days, provided that any vernalization requirements have been met. One characteristic of the wheat cultivars derived from the Green Revolution is their insensitivity to photoperiod, which along with their short stature and disease resistance, significantly contributed to their adaptation to a broad range of environments.
Various studies have been conducted to determine the advantages of photoperiod-sensitive (PS) and photoperiod-insensitive germplasm (PI). For high latitude locations, evidence indicates that PS germplasm may have an advantage over PI germplasm [11, 12]. High GEI in Northern Europe [11], North America [13], and other high latitude locations in Asia [14], indicates that regional adaptation plays a major role in breeding spring wheat.
The geographical division suggested by Worland et al. [11] in Europe where PI and PS spring wheat germplasm displays better adaptation is 45–46° N. For practical purposes, wheat grown north of Paris is frequently PS, while south of that latitude the germplasm that better adapts is PI due to the summer conditions in Southern Europe [11].
Another factor that largely determines flowering time is vernalization. In this context, vernalization is the exposure to cold temperatures after germination to acquire or accelerate the ability to flower [15]. In northern latitudes where winters are cold, vernalization sensitivity is required, as it delays floral initiation which consequently protects ear development when low temperatures can severely damage it [16], hence conferring adaptability to northern latitudes.
The distinction between spring and winter growth habits is determined by a series of genes that can express both sensitivity and insensitivity to vernalization. Of these series of genes—Vrn1, Vrn2 and Vrn3—Vrn1 and Vrn3 on the homoeologous groups 5 and 7, respectively, are dominant for spring growth habit, while Vrn2 on chromosome 5A is dominant for winter growth habit [17]. In winter wheat sown and germinated in the autumn, Vrn2 suppresses Vrn3, which in turn impedes the expression of Vrn1; then, as winter approaches, lower temperatures downregulate Vrn2, facilitating the upregulation of Vrn3, which in turn promotes Vrn1 transcription for the induction of flowering [18].
In geographical regions where wheat is grown during the winter and harvested late in the spring, the presence of Vrn1 dominant genes confers adaptability to those lower latitude regions. The Vrn-A1 and Vrn-D1 genes of the Vrn1 series are the most common, although all three (Vrn-A1, Vrn-B1 and Vrn-D1) are present in CIMMYT’s germplasm either alone or in combination [19].
The two previously mentioned factors—photoperiod and vernalization—alone, broadly determine the target breeding environments (high and low latitude regions) and, consequently, the type of germplasm that is required for each environment/s, since they guideline the planting and harvesting times.
The paradigm until the early years (1940s) of wheat breeding in Mexico dictated that breeding must be conducted in the environment where the future varieties will be cultivated [20]. However, given the need to accelerate the development of high-yielding and stem rust resistant germplasm, two generations per year started to be grown—using shuttle breeding—with the sole objective of speeding up the breeding process (Chap. 30 describes new technologies to speed up breeding). This paradigm shift took place years before any deep knowledge on the photoperiod in/sensitivity in wheat was available [21]. As germplasm exchange happened through the assembling of the first international yield trials during 1960s, the daylength effect on the materials became evident, since those shuttled-bred wheats developed in Mexico would adapt in most places in latitudes lower than 45° N [22].
3.2 Water Availability and Temperature
Water availability for the wheat crop is paramount to determine key traits in breeding. Water availability can favor optimal growing conditions, in the absence of high temperatures. However, drought stress is a constraint for wheat production in locations where water access is limited, either because of the lack of irrigation equipment or because the climate is dry (low rainfall).
Drought is one of the most severe factors that reduce wheat productivity (see Chap. 23 for details). In meteorological terms, it is defined as the absence of rain for a certain period, during which plants suffer from the lack of water in the soil. Yield losses of 20% can occur if plants are grown with 40% less water than required to avoid the stress [23]. This loss varies depending on the phenological stage at which the stress occurs, for instance, it can be larger if water is limited at the reproductive stage than if it occurs only at the vegetative stage [23].
Plants are drought stressed when water for the roots is limited and when the transpiration rate becomes higher. Drought can affect germination and plant establishment, growth, biomass accumulation, leaf senescence and, consequently, grain yield, but at the cellular level, it affects membrane integrity, pigment content, osmotic adjustment, photosynthetic activity, gas exchange and cell elongation [24].
Regions that are typically considered prone to drought stress are North Africa, some regions in West and Central Asia, and some locations in South America. Regions that are considered optimal in terms of water availability are the Nile Valley in Africa, the Northwestern Gangetic Plains and Northwestern Mexico.
Temperature is considered a stress factor that drastically influences wheat productivity once vernalization requirements—if any—have been met (see Chap. 22). Temperatures above optimum thresholds take high relevance, particularly in the context of climate change, since it determines the traits that the plants must carry to cope with the stress, such as earliness to avoid terminal heat stress [25]. It is estimated that for every °C increase above the a base temperature (13 °C) grain yield decreases by 6% [26]. Higher temperatures modify wheat phenology by reducing the number of days to reach flowering and maturity, consequently reducing the number of days in which plants can intercept light for photosynthesis, which leads to a reduction in biomass and grain yield. Larger yield reductions are expected in tropical and subtropical regions where wheat is grown, such as regions in India, which is a major wheat producer in the world [27].
3.3 Diseases
Following the fundamental paradigm in plant pathology (disease triangle), a disease outbreak occurs if there is (1) an adequate (susceptible) host, (2) a virulent pathogen, and if (3) favorable environmental conditions are present (see Chap. 19 for details). Hence, diseases tend to follow specific distribution patterns depending on the whether their environmental requirements are met.
While rusts, as a group of diseases, are found in all wheat growing areas, other leaf diseases occur in certain environments and crop management conditions (see Chaps. 8, 9 and 19 for details). Disease distribution and occurrence are dependent on both temporal and spatial variation, and these factors determine the resistance traits that cultivars must carry for certain environments. For instance, tan spot (caused by Pyrenophora tritici-repentis) incidence is linked to an expansion of zero-tillage practices (in Brazil, Argentina, Paraguay) or in places where climate does not allow fast stubble decomposition (Central Asia) and monocropping is common [28]. Septoria tritici blotch (caused by Mycosphaerella graminicola) is most common in temperate (15–20 °C) and humid wheat growing regions. Powdery mildew (caused by Blumeria graminis f. sp. tritici) is common in highly productive areas with maritime or semi-continental climate, particularly in China and South America [28].
4 Target Population of Environments
The main objective of a plant breeding program is to deliver superior germplasm for farmers in a defined set of environments, or a target population of environments (TPE). A TPE consists of many (population) environments and future years or seasons. A TPE is also a variable group of future production environments. Climatic (seasonal) variation in the same farmer’s field might change drastically year after year causing the exacerbation of GEI. GEI can have two components: (1) static and predictable (repeated) variability due to the location (site) where the trial has been established, and (2) dynamic and unpredictable variability due to the year effect.
Target environments should be characterized by climatic, soil and hydrological characteristics as well as by socioeconomic characteristics. There are different ways to group trials and environments into TPE. One is to group together sites where line means are highly correlated. A standard methodology is to use stratified hierarchical cluster analyses of the sites based on climatic variables and production traits [29].
The selection environments (SE) are the environments where the breeder does the selection of the lines. The SE are identified for predicting the performance in the TPE, but the SE may not belong to the TPE. If the lines in the SE predict those in the TPE, then (1) it is important to compute the genetic correlations between the lines in the SE versus the same (or related lines) in the TPE and show some relatively high correlations between lines in SE and in TPE; (2) for screening lines in the SE, the repeatability (broad-sense heritability) in the SE should be high; (3) SE should allow a large number of lines to be screened at a low cost, so SE should allow high selection intensity (i).
5 Multi-environmental Testing and Genotype-by-Environment Interactions
As CIMMYT’s mandate became international, the observations made between 1944 and the 1960s established the bases for the definition of target environments on a global scale. Along with this, the implementation of a breeding strategy based on ME targeted breeding, a diverse gene pool for crossing, shuttle breeding, selection under optimal conditions and multilocation testing have led to the enhancement of the adaptability and stability that characterize CIMMYT spring wheat germplasm to date.
Multi-environmental testing (MET) is a paramount strategy for CIMMYT to develop stable high grain yielding germplasm in countries with developing economies (see Chap. 7). An adequate MET strategy allows the evaluation of germplasm in stress hotspots and the identification of representative and correlated sites; thus, breeders can make better and targeted decisions in terms of crossing, selection and logistic operations. Another highly important aspect for CIMMYT’s MET strategy is that collaborators can directly evaluate CIMMYT’s elite germplasm, and hence they can make line selections for further evaluation and variety release, as well as utilize the germplasm as parental lines in their breeding programs to improve local adaptation.
Every year CIMMYT undertakes significant efforts to distribute international nurseries that comply with global and local seed health regulations, to collaborators within the IWIN, with the only request of returning the data to CIMMYT, for breeders to analyze them in a global context and support breeding decisions. The nurseries are of three different types: yield trials, observation nurseries (prior to yield trials), and trait specific nurseries (Table 3.2), Chap. 7 describes in detail international yield trials for bread wheat. Between 2013 and 2017, CIMMYT’s wheat germplasm was distributed to 350 collaborators in 80 countries per year.
Despite the large variability between MEs, it is possible to simulate the most significant ones at CIMMYT’s main testing site in northwest Mexico, Ciudad Obregon, a semi-arid location with suitable infrastructure for irrigation and available machinery for establishing the planting systems common around the globe. At this location in the Yaqui Valley, it is possible to mimic optimal, drought and heat stressed environments by applying the water management system corresponding to each ME, in combination with different sowing dates. This MET at one single location that is highly correlated with representative international locations [6, 7, 30] allows the breeders to select the elite germplasm that will most likely have an outstanding performance in international yield trials, and will consequently provide National Agricultural Research Centers a selection of CIMMYT’s best materials every year.
Analysis of these international trials requires the utilization of advanced statistical methods that are able to parsimoniously model the GEI, obtain higher precision when estimating the genetic effects, and allow the identification of GEI patterns, for instance, the Factor Analytic (FA) and Sites Regression (SREG) models [31,32,33]. The FA model utilizes the leading principal components of the GEI covariance matrix in a mixed model framework, and accounts for the maximum amount of variation with a reduced number of parameters [32]. In the SREG model, the genotype and the GEI are estimated together, which is useful for evaluating METs, as its first and second principal components account for the non-crossover and the crossover interaction, respectively [34]. This property allows a visual examination to discriminate genotypes and sites with and without crossover interactions [34]. The FA and SREG models have been used to identify the trend of genetic gains and site correlations in CIMMYT’s international nurseries [30, 35].
6 Example of TPE Definition
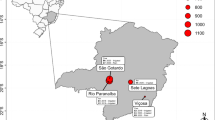
We applied the mentioned methodology to a set of locations in India with data from the Elite Spring Wheat Yield Trials that are distributed internationally by CIMMYT, upon request. Daily meteorological data for these locations in India were obtained from the NASA Langley Research Center POWER Project funded through the NASA Earth Science/Applied Science Program. Then we implemented a principal component analysis to infer the number of groups (TPEs) that would explain most of the variation and then perform hierarchical clustering with the Euclidean distance matrix of data. From our analysis we obtained three main TPEs for India, in agreement with the wheat producing zones determined by the Indian government (Table 3.3 and Fig. 3.1), and that together account for more than 97% of India’s wheat producing area. Finally, we obtained the correlated response to selection, between each TPE and SE in Mexico, by first obtaining the genetic correlations with Eq. 3.1.
where rA is the genetic correlation, p1, 2 represents the phenotypic correlation between site i and j, and \( {h}_i^2 \)and \( {h}_j^2 \) are the heritability of sites i and j, respectively.
Then, assuming that the same selection intensity applied in the SE is applied in the TPE (Eq. 3.2):
where CR is the correlated response to selection, \( \overline{r_A} \) is the genetic correlation averaged over years of testing, \( {h}_{SE}^2 \) is the heritability of the SE and \( {h}_{TPE}^2 \) is the heritability of the TPE.
Our results indicate that the centralized breeding efforts in combination with the MET can give a selection efficiency (CR) as high as in the TPE, assuming the same selection intensity (Table 3.4). However, several factors are in place to obtain this result: CIMMYT’s main yield testing site allows the simulation of various environments, the high heritability usually observed in the testing phase, a relatively stable (semi-arid) climate with favorable temperatures, water availability, irrigation infrastructure and mechanized operations. Furthermore, this result does not consider the fact that the selection intensity can be several times higher in the SE than in the TPE, given that several thousands of lines (~9000) are tested annually under optimal conditions (Stage 1 testing), from which ~1000 are selected to be tested in the SE (Stage 2), and ~300 are evaluated in the SE in the Stage 3 of testing, to finally distribute 46–48 new elite lines in each international yield trial nursery (Table 3.2).
7 Key Concepts and Conclusions
Characterizing TPEs is critical for any plant breeding endeavor to succeed. Determining the main factors that may limit wheat productivity in a determined set environments (TPEs) is fundamental to incorporate key traits in breeding. Such limitations include, but are not limited to: water availability, temperature and incidence of pests and diseases. Additionally, for a breeding program to succeed, it is important that the SE display relatively high correlations with the TPEs, allow a higher selection intensity and accuracy of selection (higher repeatability). At CIMMYT’s main testing location in northwest Mexico, it is possible to mimic optimal, drought and heat stressed environments to artificially create SE that, at one single location, are highly correlated with representative international locations to allow breeders the selection of elite germplasm with potential outstanding performance in international yield trials, and in so doing, to provide National Agricultural Research Centers a selection of CIMMYT’s best materials every year.
For a refined definition of TPEs, statistical methods such as the FA model and SREG coupled with the climatic, soil, hydrological and socioeconomic characteristics of the environments can be applied to allow the identification of GEI patterns. These models (FA and SREG) have the advantage of being parsimonious and allow to measure the extend of non-crossover and crossover GEI.
Multi-environmental testing is paramount to identify high yielding and climate reliance germplasm, as well as to determine the GEI patterns that conform potential TPEs. The CIMMYT MET strategy has the benefit of evaluating the germplasm in stress hotspots, identification of representative and correlated sites, rapid response to new constraints (see Chap. 9 for examples) and direct access to germplasm for CIMMYT collaborators, so the materials can be used as parents or directly released as varieties.
References
Rajaram S, Van Ginkel M, Fischer RA (1994) CIMMYT’s wheat breeding mega-environments (ME). In: Li ZS, Xin ZY (eds) Proceedings of the 8th International wheat genetic symposium. Beijing, pp 1101–1106
Lantican MA, Braun H-J, Payne TS, Singh RP, Sonder K, Michael B, van Ginkel M, Erenstein O (2016) Impacts of international wheat improvement research, pp 1994–2014
Hodson DP, White JW (2007) Use of spatial analyses for global characterization of wheat-based production systems. J Agric Sci 145:115
DeLacy IH, Fox PN, Corbett JD, Crossa J, Rajaram S, Fischer RA, van Ginkel M (1993) Long-term association of locations for testing spring bread wheat. Euphytica 72:95–106. https://doi.org/10.1007/BF00023777
Braun H-J, Pfeiffer WH, Pollmer WG (1992) Environments for selecting widely adapted spring wheat. Crop Sci 32:1420–1427. https://doi.org/10.2135/cropsci1992.0011183X003200060022x
Lillemo M, Van Ginkel M, Trethowan RM, Hernandez E, Rajaram S (2004) Associations among international CIMMYT bread wheat yield testing locations in high rainfall areas and their implications for wheat breeding. Crop Sci 44:1163–1169
Lillemo M, Van Ginkel M, Trethowan RM, Hernandez E, Crossa J (2005) Differential adaptation of CIMMYT bread wheat to global high temperature environments. Crop Sci 45:2443–2453
Mohler V, Lukman R, Ortiz-Islas S, William M, Worland AJ, Van Beem J, Wenzel G (2004) Genetic and physical mapping of photoperiod insensitive gene Ppd-B1 in common wheat. Euphytica 138:33–40. https://doi.org/10.1023/B:EUPH.0000047056.58938.76
Beales J, Turner A, Griffiths S, Snape JW, Laurie DA (2007) A pseudo-response regulator is misexpressed in the photoperiod insensitive Ppd-D1a mutant of wheat (Triticum aestivum L.). Theor Appl Genet 115:721–733. https://doi.org/10.1007/s00122-007-0603-4
Bentley AR, Turner AS, Gosman N, Leigh FJ, Maccaferri M, Dreisigacker S, Greenland A, Laurie DA (2011) Frequency of photoperiod-insensitive Ppd-A1a alleles in tetraploid, hexaploid and synthetic hexaploid wheat germplasm. Plant Breed 130:10–15. https://doi.org/10.1111/j.1439-0523.2010.01802.x
Worland AJ, Borner A, Korzun V, Li WM, Petrovic S, Sayers EJ (1998) The influence of photoperiod genes on the adaptability of European winter wheats. Euphytica 100:385–394. https://doi.org/10.1023/a:1018327700985
Dyck JA, Matus-Cádiz MA, Hucl P, Talbert L, Hunt T, Dubuc JP, Nass H, Clayton G, Dobb J, Quick J (2004) Agronomic performance of hard red spring wheat isolines sensitive and insensitive to photoperiod. Crop Sci 44:1976–1981. https://doi.org/10.2135/cropsci2004.1976
Lanning SP, Hucl P, Pumphrey M, Carter AH, Lamb PF, Carlson GR, Wichman DM, Kephart KD, Spaner D, Martin JM, Talbert LE (2012) Agronomic performance of spring wheat as related to planting date and photoperiod response. Crop Sci 52:1633–1639. https://doi.org/10.2135/cropsci2012.01.0052
Trethowan RM, Morgunov A, He Z, De Pauw R, Crossa J, Warburton M, Baytasov A, Zhang C, Mergoum M, Alvarado G (2006) The global adaptation of bread wheat at high latitudes. Euphytica 152:303–316. https://doi.org/10.1007/s10681-006-9217-1
Chouard P (1960) Vernalization and its relations to dormancy. Annu Rev Plant Physiol. https://doi.org/10.1146/annurev.pp.11.060160.001203
Law CN, Worland AJ (1997) Genetic analysis of some flowering time and adaptive traits in wheat. New Phytol 137:19–28. https://doi.org/10.1046/j.1469-8137.1997.00814.x
Kamran A, Iqbal M, Spaner D (2014) Flowering time in wheat (Triticum aestivum L.): a key factor for global adaptability. Euphytica 197:1–26. https://doi.org/10.1007/s10681-014-1075-7
Distelfeld A, Li C, Dubcovsky J (2009) Regulation of flowering in temperate cereals. Curr Opin Plant Biol 12:178–184
Beem J, Mohler V, Lukman R, Ginkel M, William M, Crossa J, Worland AJ (2005) Analysis of genetic factors influencing the developmental rate of globally important CIMMYT wheat cultivars. Crop Sci 45:2113–2119. https://doi.org/10.2135/cropsci2004.0665
Borlaug NE (1983) Contributions of conventional plant breeding to food production. Science 219(80):689–693
Mckinney HH, Sando WJ (1935) Earliness of sexual reproduction in wheat as influenced by temperature and light in relation to growth phases. J Agric Res 5:621–541
Rajaram S (1994) Wheat germplasm improvement: historical perspectives, philosophy, objectives, and missions_. In: Rajaram S, Hettel GP (eds) Wheat breeding at CIMMYT: commemorating 50years of research in mexico for global wheat improvement. CIMMYT, Texcoco, pp 1–10
Daryanto S, Wang L, Jacinthe P-A (2016) Global synthesis of drought effects on maize and wheat production. PLoS One 11:e0156362. https://doi.org/10.1371/journal.pone.0156362
Anjum SA, Xie X, Wang L, Saleem MF, Man C, Lei W (2011) Morphological, physiological and biochemical responses of plants to drought stress. Afr J Agric Res 6:2026–2032. https://doi.org/10.5897/ajar10.027
Mondal S, Singh RP, Crossa J, Huerta-Espino J, Sharma I, Chatrath R, Singh GP, Sohu VS, Mavi GS, Sukuru VSP, Kalappanavar IK, Mishra VK, Hussain M, Gautam NR, Uddin J, Barma NCD, Hakim A, Joshi AK (2013) Earliness in wheat: a key to adaptation under terminal and continual high temperature stress in South Asia. Field Crop Res 151:19–26. https://doi.org/10.1016/j.fcr.2013.06.015
Asseng S, Ewert F, Martre P, Rotter RP, Lobell DB, Cammarano D, Kimball BA, Ottman MJ, Wall GW, White JW, Reynolds MP, Alderman PD, Prasad PVV, Aggarwal PK, Anothai J, Basso B, Biernath C, Challinor AJ, De Sanctis G, Doltra J, Fereres E, Garcia-Vila M, Gayler S, Hoogenboom G, Hunt LA, Izaurralde RC, Jabloun M, Jones CD, Kersebaum KC, Koehler A-K, Muller C, Naresh Kumar S, Nendel C, O’Leary G, Olesen JE, Palosuo T, Priesack E, Eyshi Rezaei E, Ruane AC, Semenov MA, Shcherbak I, Stockle C, Stratonovitch P, Streck T, Supit I, Tao F, Thorburn PJ, Waha K, Wang E, Wallach D, Wolf J, Zhao Z, Zhu Y (2015) Rising temperatures reduce global wheat production. Nat Clim Chang 5:143–147
Asseng S, Cammarano D, Basso B, Chung U, Alderman PD, Sonder K, Reynolds M, Lobell DB (2017) Hot spots of wheat yield decline with rising temperatures. Glob Chang Biol 23:2464–2472. https://doi.org/10.1111/gcb.13530
Duveiller E, Singh RP, Nicol JM (2007) The challenges of maintaining wheat productivity: pests, diseases, and potential epidemics. Euphytica 157:417–430. https://doi.org/10.1007/s10681-007-9380-z
Cooper M, Hammer GL (1996) Plant adaptation and crop improvement. CAB International, Wallingford
Crespo-Herrera LA, Crossa J, Huerta-Espino J, Autrique E, Mondal S, Velu G, Vargas M, Braun HJHJ, Singh RPRP (2017) Genetic yield gains in CIMMYT’s international elite spring wheat yield trials by modeling the genotype × environment interaction. Crop Sci 57:789–801. https://doi.org/10.2135/cropsci2016.06.0553
Burgueño J, Crossa J, Cornelius PL, Yang R-C (2008) Using factor analytic models for joining environments and genotypes without crossover genotype × environment interaction. Crop Sci 48:1291. https://doi.org/10.2135/cropsci2007.11.0632
Meyer K (2009) Factor-analytic models for genotype × environment type problems and structured covariance matrices. Genet Sel Evol 41:21. https://doi.org/10.1186/1297-9686-41-21
Crossa J, Vargas M, Joshi AK (2010) Linear, bilinear, and linear-bilinear fixed and mixed models for analyzing genotype × environment interaction in plant breeding and agronomy. Can J Plant Sci 90:561–574. https://doi.org/10.4141/CJPS10003
Crossa J, Vargas M, Cossani CM, Alvarado G, Burgueño J, Mathews KL, Reynolds MP (2015) Evaluation and interpretation of interactions. Agron J 107:736. https://doi.org/10.2134/agronj/2012.0491
Crespo-Herrera L, Crossa J, Huerta-Espino J, Vargas M, Mondal S, Velu G, Payne TSS, Braun H, Singh RPP (2018) Genetic gains for grain yield in CIMMYT’s semi-arid wheat yield trials grown in suboptimal environments. Crop Sci 58:1890–1898. https://doi.org/10.2135/cropsci2018.01.0017
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Open Access This chapter is licensed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license and indicate if changes were made.
The images or other third party material in this chapter are included in the chapter's Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the chapter's Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder.
Copyright information
© 2022 The Author(s)
About this chapter
Cite this chapter
Crespo-Herrera, L.A., Crossa, J., Vargas, M., Braun, HJ. (2022). Defining Target Wheat Breeding Environments. In: Reynolds, M.P., Braun, HJ. (eds) Wheat Improvement. Springer, Cham. https://doi.org/10.1007/978-3-030-90673-3_3
Download citation
DOI: https://doi.org/10.1007/978-3-030-90673-3_3
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-90672-6
Online ISBN: 978-3-030-90673-3
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)