Abstract
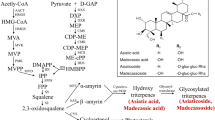
Valerenic acid (VA) is a pharmacologically-active sesquiterpene found in valerian (Valeriana officinalis L., Valerianaceae) roots and rhizomes. The plant produces only small amounts of this metabolite naturally. So, induction of hairy roots as well as elicitation can be useful to increase its commercial production. In this study, Wild-type strain ‘A13’ of Agrobacterium rhizogenes was used to induce hairy roots in valerian. The influence of three different elicitors including Fusarium graminearum extract (FE), methyl jasmonate (MJ) and salicylic acid (SA) on VA production in the selected hairy root line ‘LeVa-C4’ was also investigated. The 23-day-old cultures were treated with different concentrations of the elicitors at exposure time of 3 and 7 days. FE (1%) and MJ (100 µM L−1) highly promoted VA production at 7 days after elicitation, to a level of 12.31- and 6-fold higher than that of non-elicited controls, respectively, and FE did not exert any negative effects on biomass yield of hairy root. SA did not significantly increase the production of VA. This is the first time study to assess the elicitation of hairy root cultures to promote VA biosynthesis in valerian and the resulting experiments demonstrated that F. graminearum extract and MJ were indeed a potent inducer of VA biosynthesis.
Similar content being viewed by others
References
Hazelhoff B., Malingre T.M., Meijer D.K.F., Antispasmodic effects of valeriana compounds: an in vivo and in vitro study on the guinea-pig ileum, Arch. Int. Pharmacodyn., 1982, 257, 274–287
Rucker G., Tautges J., Sieck A., Wenzl H., Graf E., Isolation and pharmacodynamic activity of the sesquiterpene valeranone from Nardostachys jatamansi DC [in German], Arzneimittelforschung, 1987, 28, 7–13
Oliveria D.M., Barreto G., De Andrade G.V.D., Saraceno E., Bertolino L.A., Capani F., Dos Santos El Bacha R., Giraldez L.D., Cytoprotective Effect of Valeriana officinalis Extract on an in vitro Experimental Model of Parkinson Disease, Neurochem. Res., 2009, 34, 215–220
Bos R., Woerdenbag H.J., De Smet P.A.G.M., Scheffer J.J.C., Valeriana Species, In: De Smet P.A.G.M., Keller K., Chandler R.F. (Eds.), Adverse effects of herbal drugs, Springer-Verlag, 1997
Khom S., Baburin I., Timin N., Hohaus A., Trauner G., Kopp B., Hering S., Valerenic acid potentiates and inhibits GABAA receptors: molecular mechanism and subunit specificity, Neuropharmacol., 2007, 53, 178–187
Trauner G., Khom S., Baburin I., Benedek B., Hering S., Kopp B., Modulation of GABAA receptors by valerian extracts is related to the content of valerenic acid. Planta Med., 2008, 74, 19–24
Hansel R., Schulz J., Valerens auren und Valerenal als Leitstoffe des offizinellen Baldrians. Bestimmung mittels HPLC-Technik, Z. Phytother, 1982, 3, 333–340
Verpoorte R., Contin A., Memelink J., Biotechnology for the production of plant secondary metabolites, Phytochem. Rev., 2002 1, 13–25
Gerth A., Schmidt D., Wilken D., The production of plant secondary metabolites using bioreactors. The 27th International Horticultural Congress, Symposium on Plant Biotechnology: From Bench to Commercialization (13–19 August 2006, Seoul, Korea), Seoul, 2006
Srivastava S., Srivastava A.K., Hairy root culture for mass-production of high-value secondary metabolites, Crit. Rev. Biotech., 2007, 27, 29–43
Kim C.H., Park J.H., Chung I.S., Kim S.R., Lee S.W., Enhanced anthocyanin production in hairy root culture of daucus carota by fungal elicitor, Hort. Sci. J., 1992, 27, 694
Guillon S., Tremouillaux-Guiller J., Pati P.K., Rideau M., Gantet P., Harnessing the potential of hairy roots: dawn of a new era, Trends Biotechnol, 2006, 24, 403–409
Rhee H.S., Cho H.Y., Son S.Y., Yoon S.Y.H., Park J.M., Enhanced accumulation of decursin and decursinol angelate in root cultures and intact roots of Angelica gigas Nakai following elicitation, Plant Cell Tiss. Org. Cult., 2010, 1, 295–302
Wielanek M., Urbanek H., Glucotropaeolin and myrosinase production in hairy root cultures of Tropaeolum majus, Plant Cell Tiss. Org. Cult., 1999, 57, 39–45
Bauer N., Kiseljak D., Jelaska S., The effect of yeast extract and methyl jasmonate on rosmarinic acid accumulation in Coleus blumei hairy roots, Biol. Plantarum, 2009, 53, 650–656
Sun J., Xiao J., Wang X., Yuan X., Zhao B., Improved cardenolide production in Calotropis gigantea hairy roots using mechanical wounding and elicitation, Biotechnol. Lett., 2012, 34, 563–569
Kim O.T., Yoo N.H., Kim G.S., kim Y.C., Bang K.H., Hyun D.Y., Kim S.H., Kim M.Y., Stimulation of Rg3 ginsenoside biosynthesis in ginseng hairy roots elicited by methyl jasmonate, Plant Cell Tiss. Org. Cult., 2013, 112, 87–93
Chotikadachanarong K., Dheeranupattana S., Jatisatienr C., Wangkarn S., Mungkornasawakul P., Pyne S.G., Ung A.T., Sastraruji T., Influence of salicylic acid on alkaloid production by root cultures of Stemona curtisii Hook, Cur. Res. J. Bio. Sci., 2011, 3, 322–325
Esmaeilzadeh S., Sharifi M., Safaie N., Murata J., Yamagaki T., Satake H., Increased lignin biosynthesis in the suspension cultures of Linum album by fungal extracts, Plant Biotechnol. Rep., 2011, 5, 367–373
Harsh P.B., Dattatreya B.S., Ravishankar G.A., Production of volatile compounds by hairy root cultures of Cichorium intybus L under the influence of fungal elicitors and their analysis using solidphase micro extraction gas chromatography-mass spectrometry, J. Sci. Food Agri., 2003, 83, 769–774
Karwasara V.S., Tomar P., Dixit V.K., Influence of fungal elicitation on glycyrrhizin production in transformed cell cultures of Abrus precatorius Linn., Phcog. Mag., 2011, 7, 307–313
Kim Y.J., Weathers P.J., Wyslouzil B.E., The growth of Artemisia annua hairy roots in liquid and gas phase reactors, Biotechnol. Bioeng., 2002, 80, 454–464
Banerjee S., Rahman L., Uniyal G.C., Ahuja P.S., Enhanced production of valepotriates by Agrobacterium rhizogenes induced hairy root cultures of Valeriana wallichii DC., Plant Sci., 1998, 131, 203–208
Caetano L.C., Charlwood B.V., Gahan P.B., The localization and accumulation of valepotriates in hairy roots of Valerianella discoidea (L.) Loisel, Phytochem. Anal., 1999, 10, 181–186
Gränicher F., Christen P., Kapetanidis I., High-yield production of valepotriates by hairy root cultures of Valeriana officnalis L. var. sambucifolia Mikan. Plant Cell. Rep., 1992, 11, 339–342
Kittipongpatana N., Davis D.L., Porter J.R., Methyl jasmonate increases the production of valepotriates by transformed root cultures of Valerianella locusta, Plant Cell Tiss. Org. Cult., 2002, 71, 65–75
Maurmann N., Carvalho C.M.B., Silva L.A., Fett-Neto A.G., von Poser G.L., Rech S.B., Valepotriates accumulation in callus, suspended cells and untransformed root cultures of Valeriana glechomifolia, In Vitro Cell Dev. Biol. Plant, 2006, 42, 50–53
Tousi S.E., Radjabianb T., Ebrahimzadeha H., Niknama V., Enhanced production of valerenic acids and valepotriates by in vitro cultures of Valeriana officinalis L., Int. J. Plant Prod., 2010, 4, 209–222
Murashige T., Skoog F., A revised medium for rapid growth and bioassays with tobacco tissue cultures, Physiol. Plant, 1962, 15, 473–476
Doyle J.J., Doyle J.L., A rapid isolation procedure for small quantities of fresh leaf tissue, Phytochem. Bull., 1987, 19, 11–15
Nilsson O., Olsson O., The role of the Agrobacterium rhizogenes rol genes in the formation of hairy roots, Physiol. Plantarum, 1997, 100, 463–473
Lee S.Y., Kim S.G., Song W.S., Kim Y.K., Park N.I., Park S.U., Influence of different strains of Agrobacterium rhizogenes on hairy root induction and production of alizarin and purpurin in Rubia akane Nakai, Romanian Biotechnol. Lett., 2010, 15, 5405–5409
Afsaneh S., Carapetian J., Heidari R., Jafari M., Hassanzadeh Gorttapeh A., Hairy Root Induction in Linum mucronatum ssp. mucronatum, an Anti-Tumor Lignans Producing Plant, Not. Bot. Horti. Agrobo., 2012, 40, 125–131
Ooi C.T., Syahida A., Stanslas J., Maziah M., Efficiency of different Agrobacterium rhizogenes strains on hairy roots induction in Solanum mammosum, World J. Microb. Biotechnol., 2013, 29, 421–430
Hu Y.H., Yu Y.T., Piao C.H., Liu J.M., Yu H.S., Methyl jasmonate- and salicylic acid-induced D-chiro-inositol production in suspension cultures of buckwheat (Fagopyrum esculentum), Plant Cell Tiss. Org. Cult., 2011, 106, 419–424
Lee K.T., Hirano H., Yamakawa T., Kodama T., Igarashi Y., Shimomura K., Responses of transformed root culture of Atropa belladonna to salicylic acid stress, J. Biosci. Bioeng., 2001, 91, 586–589
Ali M.B., Hahn E.J., Paek K.Y., Methyl jasmonate and salicylic acid induced oxidative stress and accumulation of phenolics in panax ginseng bioreactor root suspension cultures. Molecules, 2007, 12, 607–621
Lukasz K., Elzbieta B., Halina W., Methyl jasmonate effect diterpenoid accumulation in Salvia sclarea hairy root culture in shake flasks and sprinkle bioreactor, Enzyme Microb. Tech., 2009, 44, 406–410
Xu M.J., Dong J.F., Zhu M.Y., Nitric oxide mediates the fungal elicitor-induced hypericin production of hypericum perforatum cell suspension cultures through a jasmonic-acid-dependent signal pathway, Plant Physiol., 2005, 139, 991–998
Chen X.M., Guo S.X., Effects of four species of endophytic fungi on the growth and polysaccharide and alkaloid contents of Dendrobium nobile, China J. Chin. Mat. Med., 2005, 30, 253–257
Van der Fits L., Memelink J., ORCA3 a jasmonate-responsive transcriptional regulator of plant primary and secondary metabolism, Science, 2000, 289, 295–297
Cui L., Wang Z.Y., Zhou X.H., Optimization of elicitors and precursors to enhance valtrate production in adventitious roots of Valeriana amurensis Smir ex Kom, Plant Cell Tiss. Org. Cult., 2012, 108, 411–420
Yousefzadi M., Sharifi M., Behmanesh M., Ghasempour A., Moyano E., Palazon J., Salicylic acid improves podophyllotoxin production in cell cultures of Linum album by increasing the expression of genes related with its biosynthesis, Biotechnol. Lett., 2010, 32, 1739–1743
Pieterse C.M.J., Ton J., Van Loon L.C., Cross-talk between plant defense signaling pathways: boost or burden? Ag. Biotech. Net., 2001, 3, 1–8
Ding C.K., Wang C.Y., Gross K.C., Smith D.L., Jasmonate and salicylate induce the expression of pathogenesis-related-proteins genes and increase resistance to chilling injury in tomato fruit, Planta, 2002, 214, 895–901
Xu M., Dong J., Wang H., Huang L., Complementary action of jasmonic acid on salicylic acid in mediating fungal elicitor-induced flavonol glycoside accumulation of Ginkgo biloba cells, Plant Cell Environ., 2009, 32, 960–967
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Torkamani, M.R.D., Jafari, M., Abbaspour, N. et al. Enhanced production of valerenic acid in hairy root culture of Valeriana officinalis by elicitation. cent.eur.j.biol. 9, 853–863 (2014). https://doi.org/10.2478/s11535-014-0320-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.2478/s11535-014-0320-3