Abstract
Protein arginine methyltransferase 1 (PRMT1), the predominant type I protein arginine methyltransferase, plays a crucial role in normal biological functions by catalyzing the methylation of arginine side chains, specifically monomethylarginine (MMA) and asymmetric dimethylarginine (ADMA), within proteins. Recent investigations have unveiled an association between dysregulated PRMT1 expression and the initiation and progression of tumors, significantly impacting patient prognosis, attributed to PRMT1’s involvement in regulating various facets of tumor cell biology, including DNA damage repair, transcriptional and translational regulation, as well as signal transduction. In this review, we present an overview of recent advancements in PRMT1 research across different tumor types, with a specific focus on its contributions to tumor cell proliferation, metastasis, invasion, and drug resistance. Additionally, we expound on the dynamic functions of PRMT1 during distinct stages of cancer progression, elucidating its unique regulatory mechanisms within the same signaling pathway and distinguishing between its promotive and inhibitory effects. Importantly, we sought to provide a comprehensive summary and analysis of recent research progress on PRMT1 in tumors, contributing to a deeper understanding of its role in tumorigenesis, development, and potential treatment strategies.
Similar content being viewed by others
Introduction
Cancer poses a significant threat to human health globally, given its high prevalence and complex nature. Despite substantial progress in understanding and treating tumors over the past few decades, eight key indicators of human tumors have been identified, encompassing sustained proliferation ability, evasion of growth inhibitory factors, resistance to death, replication immortality, angiogenesis induction, activation of invasion and metastasis, reprogramming of energy metabolism, and evasion of immune damage [1]. However, challenges persist in tumor treatment, including drug resistance, recurrence, and metastasis. Hence, the discovery of new therapeutic targets and strategies remains crucial to enhance the efficacy of tumor treatment.
As a pivotal post-translational modification enzyme, PRMT1 plays a crucial role in the occurrence and development of tumors by catalyzing protein arginine methylation modifications. Its impact spans various cellular processes, including gene expression regulation, cell cycle regulation, and DNA repair, significantly influencing tumor cell proliferation, invasion, metastasis, and antiapoptotic abilities [2]. The intricate involvement of PRMT1 in these processes underscores its significance in understanding the fundamental mechanisms of cancer onset and advancement, as well as its potential in predicting prognosis. Therefore, the study of PRMT1 in tumors holds both theoretical and practical importance. This review aims to comprehensively examine recent research progress on PRMT1 in tumors, summarizing its potential applications in tumor treatment. By exploring the function and regulatory mechanisms of PRMT1, we sought to deepen our understanding of the mechanisms underlying tumor occurrence, identify new therapeutic targets and strategies, and enhance the efficacy of tumor treatment. The review will be organized into three sections: first, an introduction to the structure and function of PRMT1; second, a review of the expression and function of PRMT1 in different types of tumors; and finally, a summary of the potential application value of PRMT1 as a therapeutic target, outlining future research directions.
Structure and function of PRMT1
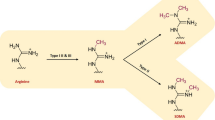
PRMTs play a pivotal role in transferring methyl groups from S-Adenosyl methionine (SAM) to the guanidine nitrogen of protein arginine, resulting in the production of methyl arginine and SAH (S-adenosylhomocysteine) [3]. There are nine PRMT members in total, categorized into three types. Type I PRMTs (PRMT-1, 2, 3, 4, 6, and 8) catalyze the formation of MMA and ADMA. Type II PRMTs (PRMT 5 and 9) generate MMA and SDMA. Lastly, Type III PRMTs (PRMT7) exclusively produce MMA [4]. Additionally, a Type IV PRMT produces d-NG-monomethylarginine in fungi [5].
The PRMT1 gene is situated on human chromosome 19, precisely at 19q13.32. Comprising 12 exons and 11 introns, this gene is present in all eukaryotes and displays a high degree of conservation [6]. The homology among mammals, zebrafish, and Xenopus is over 90%, while the homology between humans and Saccharomyces cerevisiae is approximately 50% [7]. The complex genomic structure at the 5′ terminus of the PRMT1 gene results in the expression of seven PRMT1 isoforms, denoted as PRMT1v1-v7 (Fig. 1A). These isoforms vary in different aspects, such as molecular weight, N-terminal structure, substrate specificity, tissue specificity, subcellular localization, and tissue expression. Biochemical characterization has revealed that all isoforms, except PRMT1v7, are active [8]. Notably, the PRMT1v2 isoform contains amino acids that regulate its cytoplasmic localization and significantly contribute to the survival and invasion of breast cancer cells [9].
The genomic and protein structure of PRMT1. A The genomic structure of the PRMT1 gene consists of 12 constitutive exons, with exon 1 further divided into 4 alternative exons. The PRMT1 gene generates PRMT1 isoforms (v1 to v7) through transcription and splicing, along with the exon composition of these isoforms. Intron boundaries sequences are represented by the black boxes. B The protein structure of PRMT1-v2, which is the signature motifs (I, Post-I (PI), II, III), double E motif, and THW loop (adapted from [6])
PRMT1, the earliest discovered protein arginine methyltransferase and the predominant Type I PRMT in mammalian cells, accounts for 85% of cellular PRMT activity [10]. Over the years, the structure of PRMT1 has undergone extensive study and characterization, consisting of two domains: the N-terminal AdoMet-binding domain and the C-terminal β-barrel domain. The N-terminal end of the AdoMet-binding domain contains a dynamic α-helix, which plays a critical role in AdoMet binding, and the active site pocket, where the methyltransfer reaction occurs, is located between these two domains [11]. Mutagenesis studies have underscored the essential role of two active site glutamates in the enzymatic activity of PRMT1. Furthermore, the α helical dimerization arm, which is responsible for PRMT1 dimerization, extends from the N-terminal of the β barrel domain and connects to the N-terminal of the AdoMet-binding domain [11]. This creates the core form of PRMT1, which is a doughnut-shaped homodimer arranged in a head-to-tail pattern that is imperative for its binding to the co-factor S-Adenosyl methionine [12]. The catalytic core of PRMT1, comprising approximately 310 highly conserved amino acids, is responsible for its methyltransferase activity. This catalytic core consists of six highly conserved peptide motifs: motif I (VLDVGSGTG), motif II (VDI), motif III (LAPDG), post-motif I (VIGIE), double E motif (SEWMGYCLFYESM), and THW loop (YTHWK), which are essential for methyltransferase activity of PRMT1 (Fig. 1B). The site that binds AdoMet is delimited by motif I, which is stabilized by motifs II and III. The glutamic acid residue of the post-motif I forms hydrogen bonds with AdoMet, which is crucial for AdoMet binding in this pocket. The substrate pocket for arginine residues is defined collectively by the double E motif, which is composed of two negatively charged glutamic acid residues (E144 and E153) that can neutralize the positively charged guanidium group of the target arginine, and the THW loop, which stabilizes the three dynamic α-helices (αX, αY, αZ) located at the N-terminus of the Rossmann fold and participates in the recognition of arginine residues [6].
PRMT1 is a pivotal enzyme involved in various cellular processes. Extensive research has revealed that PRMT1 primarily targets substrates containing conserved glycine- and arginine-rich (GAR) motifs, typically located at the C-terminus of proteins. However, Coactivator-associated arginine methyltransferase 1 (CARM1), a notable exception, predominantly methylates patterns rich in PGM motifs situated at the N-terminus or intermediate region of proteins [13]. The arginine methylation of the GAR motif by PRMT1 is particularly vital for regulating the DNA damage response of 53BP1 [14], evidenced by the resistance to γ-IR sensitivity observed in mice with lysine substituted for arginine in the MRE11 GAR motif [15]. Certain proteins that contain RGG motifs, such as FUS and nucleolin, can be specific targets for PRMT1 [16, 17]. Recent research has shown that PRMT1 mediates arginine methylation within the C-terminal RGG repeats of METTL14, thereby regulating m6A modification and promoting tumorigenesis [18]. Importantly, PRMT1 recognizes not only RGG and GAR sequences but also other amino acid sequences, suggesting that the characteristics of these substrate sequences may influence the substrate selectivity and functional regulation of PRMT1 [19].
It is now understood that in normal cells, PRMT1 plays a critical role in various physiological processes [20]. PRMT1 deletion in mice leads to embryonic lethality, highlighting its indispensable nature for survival until birth [21]. Furthermore, PRMT1-mediated histone H4R3me2a methylation is crucial for regulating cell proliferation in various organs and serves as a key regulator of normal vertebrate development and growth [22]. Studies have demonstrated PRMT1’s involvement in regulating the self-renewal of hematopoietic stem cells and normal hematopoiesis [23], as well as in the normal development of lymphocytes [24, 25], the differentiation and proliferation of intestinal cells [26], myelin regeneration [27], and spermatogenesis [28]. Notably, the absence of PRMT1 in mice results in a significant decrease in the proliferation of palate mesenchymal cells, ultimately preventing the palatal shelves from reaching the midline and resulting in a complete cleft palate phenotype [29]. Additionally, the PRMT1-P53 pathway plays a crucial role in the formation of epicardium-derived mesenchymal lineages during cardiac development, supporting ventricular morphogenesis and coronary artery formation [30]. In the context of vascular health, transcriptome data of abdominal aortic aneurysm patients and elderly aortas have shown decreased expression of PRMT1, revealing its role in the necessary epigenetic modification of chromatin to maintain vascular smooth muscle contraction [31]. Mice with PRMT1 knockout exhibit characteristics of heart failure, highlighting the importance of PRMT1 in regulating alternative splicing in vivo and maintaining cardiac homeostasis [32]. Moreover, PRMT1’s modulation of cardiac IKs activity may be a crucial target for preventing excessive prolongation of action potential duration and arrhythmias in heart failure patients [33]. PRMT1 is also essential for the maintenance of normal white adipose tissue function under diet-induced obesity conditions, preventing lipid accumulation in peripheral tissues and contributing to the regulation of metabolic homeostasis [34]. Moreover, PRMT1 reduces high-fat diet-induced hepatic steatosis by increasing PGC-1α expression through the recruitment of HNF-4α to the PGC-1α promoter, thereby promoting fatty acid oxidation [35]. However, the dysregulation of PRMT1 expression has been linked to numerous diseases, with its overexpression identified as a significant factor in the development of allergic rhinitis by increasing the levels of epithelial-derived cytokines [36]. Targeting PRMT1 has also shown promise in improving patient outcomes in neurodegenerative diseases and diabetic nephropathy [37, 38]. PRMT1 overexpression has been associated with tumor progression, making it a significant target for therapeutic intervention [6]. In conclusion, PRMT1 plays an essential role in maintaining normal physiological function and, due to its diverse roles in various pathological processes, has potential as an important therapeutic target in a wide range of diseases.
The roles of PRMT1 in cancers
The extensive involvement of PRMT1 in the development and progression of cancer has spurred extensive research to unravel its functions and regulatory mechanisms. The ultimate goal is to unveil groundbreaking opportunities and strategies for cancer treatment. In the subsequent section, we present a comprehensive review of research findings on PRMT1 in tumors, exploring its potential value in tumor therapy.
Breast cancer
Breast cancer incidence rates have consistently risen by approximately 0.5% per year, establishing it as the predominant cancer type among women [39]. Investigations conducted on mouse models have unveiled the significant role of PRMT1 overexpression in the mammary gland in tumorigenesis, fostering PI3K-AKT pathway activity [40]. Recent research has demonstrated heightened PRMT1 expression in breast cancer specimens compared to normal tissues, establishing a correlation between elevated PRMT1 levels and malignancy in breast cancer patients [41, 42]. Moreover, escalated PRMT1 levels have been linked to unfavorable clinical outcomes in these patients [43]. Within the breast cancer microenvironment, tumor-associated macrophages (TAMs) secrete the IL-6 cytokine, which triggers PRMT1 to mediate the formation of asymmetric dimethylation of EZH2 at arginine 342 [44]. This process reinforces EZH2 stability by hindering EZH2 phosphorylation through both CDK1-mediated and AMPK-mediated pathways, thus impeding EZH2 degradation via the TRAF6 ubiquitin-proteasome pathway [43]. Additionally, PRMT1-mediated EZH2 methylation (R342) associates with SUZ12, enhancing PRC2 assembly. This, in turn, results in the suppression of P16 and P21 transcription due to elevated EZH2 expression and increased H3K27me3 enrichment at their respective promoters. Consequently, elevated EZH2 levels promote metastasis of breast cancer cells by suppressing the expression of target genes such as E-cadherin, DAB2IP, and CSTA. The diminished levels of P16 and P21 in this context accelerate cell cycle progression and cellular proliferation [45].
In breast cancer cells, PRMT1 plays a crucial role in mediating the asymmetric dimethylation of histone H4 at arginine 3 within the ZEB1 promoter, facilitating the transcriptional expression of ZEB1, a key factor in the EMT process [46]. The maintenance of proliferative signaling and the disruption of negative feedback mechanisms are integral to tumorigenesis. For instance, in triple-negative breast cancer, PRMT1 regulates the EGFR and Wnt signaling pathways, thus sustaining proliferative signaling [42, 47]. Additionally, PRMT1 reverses the repression of cyclin D1 transcription mediated by C/EBPα through the inhibition of HDAC3 corepressor activity via the weakened interaction between HDAC3 and unmethylated C/EBPα, achieved through the methylation of three arginine residues (R35, R156, and R165) on C/EBPα [41].
Under hypoxic conditions, PRMT1 upregulates the expression of circTBC1D14 and methylates FUS, forming a complex with circTBC1D14. This complex helps maintain a balance between stress granule (SG) formation and autophagy, thus preserving cellular homeostasis [48]. PRMT1 also inhibits the transcriptional activity of P53 through multi-region methylation, preventing breast cancer cells from undergoing apoptosis or senescence [49]. Radiation-induced damage to breast cancer cells increases levels of S-Adenosyl methionine and enhances PRMT1 enzymatic activity, leading to the methylation of BRCA1. This methylated BRCA1, facilitated by BARD1, translocates to the nucleus and upregulates the antiapoptotic protein BCL2 [50]. Furthermore, PRMT1 is involved in DNA homologous recombination repair, a process regulated by the PRMT1/c-Myc network [51]. These mechanisms collectively contribute to the epigenetic defense of breast cancer cells against ionizing radiation, conferring resistance to olaparib, a PARP inhibitor. As a co-factor, PRMT1 forms a repressive multiprotein complex with HP1γ, HDAC1/2, and LSD1. This complex anchors the PR promoter, inducing local chromatin closure and maintaining the silent state of PR target genes in the absence of progesterone [52]. Upon exposure to progesterone, PRMT1 acts as a coactivator by methylating PR at arginine 637. This process promotes the expression of genes involved in cell proliferation and metastasis by dissociating the repressive multiprotein complex. However, the stability of R637me-PR diminishes, resulting in PR degradation, intensifying the PR cycle, and ultimately accelerating PR transcriptional activity [53]. Moreover, PRMT1 effectively methylates ERα when triggered by IGF-1, which enables ERα to bind with IGF-1R. Upon binding, IGF-1R phosphorylates ERα on residue Y219, strengthening their interaction. Notably, IGF-1R also phosphorylates tyrosine residues of IRS1 and Shc, creating docking platforms for PI3K and Grb2, which activate the Akt and ERK pathways, respectively [54]. In conclusion, recent insights into the multifaceted role of PRMT1 in breast cancer underscore the need for further investigation, positioning PRMT1 as a promising therapeutic target in breast cancer treatment. The complexity of its involvement in various pathways suggests its potential as a focal point for therapeutic intervention in breast cancer (Fig. 2).
Pancreatic cancer
By mediating ADMA of substrate proteins, the post-translational modification enzyme PRMT1 plays a significant role in pancreatic cancer (Fig. 3). Recent research conducted by Virginia et al. has illuminated the importance of PRMT1-generated ADMA in regulating RNA processing and controlling gene expression associated with DNA damage response (DDR). Such regulation is critical for maintaining genome stability and promoting tumor growth [55]. Previous studies have established that the overactivation of β-catenin/TCF signaling contributes to the growth, migration, and metastasis of pancreatic cancer cells. Notably, PRMT1 plays a pivotal role in this context by facilitating the expression of the β-catenin protein through its binding to the CTNNB1 promoter region [56]. Additionally, PRMT1 enhances the oncogenic function of Gli1, a transcription factor, by methylating it at the R597 site. This methylation event promotes the accumulation of Gli1 on its target gene promoter, enhancing its transcriptional activity and subsequently increasing the expression of target genes [57]. Another noteworthy discovery is the PRMT1-HSP70-BCL2 pathway, which has been associated with drug resistance in cancer cells, including resistance to the chemotherapy drug gemcitabine. The mechanism underlying this pathway involves PRMT1 methylation of HSP70 at the R416 and R447 sites. This methylation event enhances the binding and stabilization of HSP70 with BCL2 mRNA, leading to increased BCL2 protein expression. The accumulation of BCL2, a crucial antiapoptotic protein, can benefit pancreatic cancer cells as it protects them from apoptosis induced by cellular stresses and therapeutic interventions [58]. These findings offer valuable insights into the intricate mechanisms underlying pancreatic cancer development and underscore the potential role of PRMT1 as a therapeutic target in combating this formidable disease.
Lung cancer
In the field of tumor cell metabolism, it is well-established that both glycolysis, which provides rapid energy production, and aerobic glycolysis, which offers efficient energy production, play a pivotal role in tumor progression [59, 60]. The proper concentration of calcium (Ca2+) within the mitochondria is essential for facilitating aerobic oxidation in tumor cells [61]. To counter mitochondrial Ca2+ overload, PRMT1 methylates MICU1, reducing its activity. Moreover, PRMT1 overexpression, and UCP2 collectively normalize mitochondrial Ca2+ uptake, ensuring oxidative phosphorylation (OXPHOS), aligning with the observed elevated levels of mitochondrial respiration in lung carcinoma cells. By regulating mitochondrial Ca2+ uptake, PRMT1 plays a crucial role in sustaining the vitality and proliferation of tumor cells [62]. In addition to its involvement in mitochondrial regulation, PRMT1 exerts effects on other cellular processes relevant to cancer. PRMT1 methylates INCENP at arginine 887, a region binding to AURKB. This methylation is advantageous for the activation of AURKB, promoting normal cell division and growth in cancer cells [63]. Furthermore, PRMT1 has been found to methylate Twist1, a known E-cadherin repressor, at arginine 34. This methylation is crucial for actively repressing E-cadherin, unveiling a novel regulatory mechanism of EMT in non-small cell lung cancer (NSCLC) [64]. The upregulation of PRMT1 in lung cancer has also been associated with chemotherapeutic drug resistance. Specifically, PRMT1 regulates the expression of FEN1, a major component of the Base Excision Repair pathway critical for DNA repair ability and chemotherapeutic drug response [65, 66]. Additionally, PRMT1 methylation of SOX2 at arginine 43 has been linked to increased stemness and chemotherapy resistance in small cell lung cancer (SCLC) [67]. Collectively, these findings underscore the multifaceted role of PRMT1 in cancer development and progression, impacting metabolism, cell division, EMT, and chemotherapeutic response (Fig. 4). Targeting PRMT1 holds promise as a potential therapeutic strategy in lung cancer.
Liver cancer
PRMT1 has emerged as a significant player in liver cancer (Fig. 5), particularly hepatocellular carcinoma (HCC). Extensive research reveals significant overexpression of PRMT1 in HCC tissues compared to normal liver samples, as evidenced by the analysis of 50 normal liver samples and 371 HCC samples from The Cancer Genome Atlas (TCGA) database. Moreover, multiple patient cohorts have validated the prognostic value of PRMT1 in HCC, establishing it as a novel prognostic marker for assessing disease progression and patient outcomes [68]. This observation gains particular relevance given the underlying role of chronic inflammation in hepatocarcinogenesis, where hepatocytes, incited by chronic inflammation, transition from chronic hepatitis to cirrhosis, ultimately leading to liver cancer. Notably, myeloid-specific PRMT1 knockout mice exhibited reduced levels of IL-6 and suppressed STAT3 activity, protecting against alcohol-induced HCC development [69]. PRMT1 overexpression has been shown to activate the STAT3 signaling pathway by elevating STAT3 phosphorylation. Interestingly, inhibition of STAT3 phosphorylation using crypto tanshinone, a STAT3 inhibitor, reversed this process, highlighting PRMT1’s involvement in multiple regulatory mechanisms of the STAT3 pathway and suggesting its potential as a therapeutic target in HCC [70]. The deregulation of the mTORC1 pathway, linked to hepatocellular carcinoma cell proliferation, is influenced by GATOR2-mediated suppression of the GATOR1 protein. It has been reported that PRMT1 interacts with WDR24, a critical component of the GATOR2 complex, and enhances GATOR2’s inhibitory effect on GATOR1 by methylating it at arginine 329 [71]. Beyond these contributions, PRMT1 performs various cellular functions beyond its role as a transcriptional co-activator in epigenetic regulation, encompassing transcription, DNA repair, and signal transduction. The overexpression of PRMT1 in fresh HCC tissues has been associated with EMT, considered one of the hallmarks of cancer [72]. Mechanistically, activated Smad signaling due to PRMT1 activation fosters EMT through the TGF-β1/Smad pathway in hepatic carcinoma cells [73]. Notably, an inconsistency between HCC metabolomic and transcriptomic profiles has been observed, revealing that serine synthesis is activated despite downregulated mRNA and protein levels of PHGDH (phosphoglycerate dehydrogenase). Besides, PRMT1 plays a crucial role in augmenting PHGDH activity by methylating arginine 236, activating serine synthesis, enhancing the oxidative stress response, and promoting HCC growth [74]. Furthermore, a study by Ryu et al. unveiled the relationship between PRMT1 and CDKN1A (P21), whereby co-transfecting cells with siPRMT1 and siCDKN1A resulted in spheroid formation and restored cell growth, suggesting the involvement of CDKN1A downstream of PRMT1 in regulating tumor growth and formation in HCC [68]. In summary, elevated PRMT1 expression in HCC tissues, coupled with its prognostic value and involvement in various molecular pathways, underscores its potential as both a diagnostic marker and therapeutic target in liver cancer.
Leukemia
Optimal cell growth and differentiation heavily rely on the proper functioning of various epigenetic regulators, with PRMT1 being of significant importance. PRMT1 plays a crucial role in normal hematopoiesis, regulating myeloid differentiation and lymphocyte development [24, 75]. However, it has also been identified as a therapeutic target due to its involvement in hindering megakaryocyte differentiation in conditions like myelodysplastic syndrome (MDS) and megakaryocytic leukemia. PRMT1 hinders megakaryocyte terminal differentiation through the downregulation of RBM15 protein levels, affecting RNA splicing. RBM15, an RNA-binding protein, controls post-transcriptional processing [76, 77]. Additionally, PRMT1 is involved in the ubiquitination and degradation of DUSP4, a dual-specificity phosphatase in the P38 MAPK signaling pathway, leading to heightened P38 MAPK activity and the blockage of megakaryocyte differentiation [78]. Chromosomal translocations, such as MLL-GAS7 and MOZ-TIF2, produce abnormal oncogenic fusion proteins, highlighting PRMT1’s significance in leukemia pathogenesis. Studies indicate that MLL fusions and MOZ-TIF2-transformed cells recruit PRMT1 for mediating H4R3me2a. Besides, these cells also recruit KDM4C, a KDM, facilitating the removal of the H3K9me3 repressive mark. This synergistic effect enhances the transcription and transformation capability of these genes, contributing to acute myeloid leukemia (AML) development [79]. Moreover, methylation of PRMT1 at specific residues on FLT3-ITD protein amplifies its carcinogenic signal [80]. Inhibiting PRMT1 has promising potential to enhance the efficacy of FLT3-ITD+ AML kinase inhibitors, as well as in MLL-rearranged acute lymphoblastic leukemia [81]. PRMT1’s interaction with β-Catenin and HOXA9 in LSK-MLL-ENL cells plays a crucial role in mediating leukemia self-renewal, highlighting the significance of the HOXA9/PRMT1 molecular network beyond β-Catenin dependence [82]. Moreover, in the presence of GFI1, PRMT1 plays a pivotal role in the DNA damage repair pathway by methylating key molecules like MRE11 and 53BP1, contributing to efficient DNA damage repair and the development of lymphoid leukemia [83]. In summary, PRMT1 is a key epigenetic regulator in leukemia with diverse effects on cell growth and differentiation (Fig. 6). Understanding its mechanisms and interactions offers insights into potential therapeutic interventions for various hematological disorders.
Colorectal cancer
The epidermal growth factor receptor (EGFR) family, an extensively researched receptor tyrosine kinase (RTK) system, plays crucial roles in normal development. Abnormal expression of EGFR is tightly correlated with various pathological conditions, particularly cancer [84, 85]. Cetuximab, a targeted therapy specifically designed for EGFR, has shown remarkable efficacy in treating colon cancer patients, resulting in improved prognosis and prolonged survival [86]. However, drug resistance remains a significant challenge for many patients. Recent studies have provided insights into the involvement of PRMT1 in the mechanisms of drug resistance within the EGFR signaling pathway. In a groundbreaking discovery by Liao et al., it was found that PRMT1 could methylate specific arginine residues, R198 and R200, located in the extracellular domain of EGFR. This arginine methylation by PRMT1, occurring in the endoplasmic reticulum (ER)/Golgi, subsequently leads to the transportation of methylated EGFR to the cell membrane. This facilitates receptor dimerization and signal activation, ultimately promoting cancer cell growth and conferring resistance to cetuximab treatment [87]. Additionally, PRMT1 enhances the transcriptional activity of EGFR, further fostering colon cancer cell proliferation, clonogenicity, and migration. Mechanistically, PRMT1 initiates asymmetric dimethylation of histone H4 on arginine 3, attracting SMARCA4. This process results in the formation of a complex that collaboratively activates the transcription of TNS4 and EGFR [88]. Furthermore, the SMARCA4R1157W mutation increases SMARCA4 binding to PRMT1-mediated H4R3me2a, enhancing complex formation [89]. However, the presence of KRAS gene mutations presents another significant obstacle in treatment. In a related context, PRMT1-mediated methylation of NONO at position R251 has been shown to facilitate the growth and metastasis of colorectal cancer (CRC). Inhibiting PRMT1 demonstrated the potential to reduce NONO arginine methylation and suppress CRC progression, regardless of the mutation status of another crucial gene, KRAS [90]. These findings collectively suggest that inhibiting PRMT1 may hold significant therapeutic potential for CRC treatment, particularly in overcoming drug resistance mechanisms associated with EGFR-targeted therapies (Fig. 7).
Esophageal cancer
Recent studies have unveiled that PRMT1, a protein arginine methyltransferase, is highly expressed in esophageal squamous cell carcinoma (ESCC) and is associated with poor prognosis. The role of PRMT1 in ESCC is twofold: firstly, it facilitates histone H4 methylation, thereby preserving the stem cell characteristics of ESCC [91]; secondly, it mediates Gli1 methylation, leading to an enhancement in its transcriptional activity [92]. The overexpression of PRMT1 triggers the activation of target genes downstream of the Hedgehog signaling pathway. Mechanistically, PRMT1 methylates the DNA-binding domain (DBD) of Gli1, known as glioma-associated oncogene homolog 1, thereby potentiating its transcriptional activity, promoting the growth and migration of esophageal squamous cell carcinoma cells. However, further validation is warranted to confirm the methylation site of Gli1, specifically R618 [92, 93]. Silencing PRMT1 has been found to significantly inhibit ESCC progression through the transcriptional activation mediated by histone H4R3me2a. Additionally, silencing PRMT1 reduces the self-renewal, tumorigenicity, and chemoresistance of OV6+ cells. RNA sequencing (RNA-seq) transcriptome analysis has further revealed that the overexpression of PRMT1 in ESCC cell lines activates the Wnt/β-catenin and Notch signaling pathways [91]. In conclusion, PRMT1, as a novel effector, plays a crucial role in promoting the stem cell characteristics of esophageal cancer while also maintaining tumor cell proliferation and migration (Fig. 8).
Osteosarcoma
Osteosarcoma is a type of bone cancer primarily observed in children and adolescents. The understanding of PRMT1’s mechanism in osteosarcoma tumorigenesis and the identification of exploitable vulnerabilities for targeted therapy are of utmost importance in the quest for effective treatment strategies (Fig. 9). Studies utilizing a mouse model with P53/RB deletion have highlighted the pivotal role of PRMT1 in osteosarcoma tumorigenesis. Deletion of PRMT1 in this context led to a significant increase in the lifespan of mice, emphasizing its significance in disease progression. PRMT1 exerts regulatory influence on the translation initiation complex in P53/RB-null cells by methylating eIF4G1 at positions R689 and/or R698, suggesting PRMT1’s involvement in promoting tumor initiation and maintenance through modulation of translation-associated genes at the translation level [94]. Li et al.’s study provides additional evidence of PRMT1’s role in promoting osteosarcoma at the translational level. It was found that PRMT1-mediated H4R3me2a could enhance c-Myc expression, activating the mTOR signaling pathway and promoting osteosarcoma progression [95]. Methylation of STAT3 at arginine 688 by PRMT1 promoted its transcriptional activation, contributing to osteosarcoma malignancy. TIPE1, by targeting the catalytic domain of PRMT1, inhibited its methyltransferase activity and suppressed osteosarcoma progression [96]. Moreover, it has been noted that the methylation and activation of STAT3 could prevent the transcription of FAS, thus hindering the apoptotic pathway initiated by FAS and supporting OS’s progression. The degradation of PRMT1 by GGA through the HSP70-CHIP-mediated proteasomal pathway was found to induce cell apoptosis triggered by FAS, suggesting potential therapeutic approaches for osteosarcoma [97]. In conclusion, understanding PRMT1’s role in osteosarcoma tumorigenesis, its interactions with translation-associated genes, and its involvement in the apoptosis pathway provides insights for developing targeted therapies against osteosarcoma.
Prostate cancer
Recent studies have highlighted the involvement of epigenetic networks, particularly PRMT1, in the progression of prostate cancer (PCa) (Fig. 10). The correlation between PRMT1 and CARM1 expression in PCa has been demonstrated, with PRMT1 overexpression associated with the EMT process [98]. PRMT1, when bound to hsa_circ_0094606, promotes M2 macrophage polarization by methylating ILF3 at R609, leading to the upregulation of IL-8 and enhanced proliferation, migration, and EMT processes in PCa cells [99]. A genome-scale CRISPR screen identified PRMT1 as a key mediator in the androgen receptor (AR) signaling pathway. Interestingly, it was found that PRMT1 could regulate the activity of AR enhancers, controlling the transcription and expression of the AR gene [100]. Targeting PRMT1 reduces the H4R3me2a modification at the AR locus, inhibiting AR-FL and AR-V7 transcription [101]. These findings offer insights into the resistance mechanisms of AR and AR-V7-driven castration-resistant prostate cancer, providing potential avenues for treatment.
Ovarian cancer
PRMT1 has also been identified as an oncogene in ovarian cancer (Fig. 11). It is typically expressed in various ovarian cancer cells, such as ES2 and SKOV3, and has been found to methylate FAM98A, a novel substrate of PRMT1. This methylation process promotes colony formation, in vivo proliferation, migration, and invasion of cancer cells [102]. PRMT1 also acts as a protective factor against cisplatin-induced apoptosis in ovarian cancer cells. When exposed to cisplatin, PRMT1 interacts with DNA-PK, undergoes phosphorylation at position 291, enhances its affinity for substrates, increases enzymatic activity, and accumulates in chromatin. The increased accumulation establishes a foundation for subsequent processes, facilitating the transcription of SASP (senescence-associated secretory phenotype) genes through the mediation of increased H4R3me2a levels. This modification plays a crucial role in maintaining the activation of the NF-κB -SASP axis. By inducing H4R3me2a, PRMT1 facilitates the expression of genes associated with the senescence-related secretory phenotype, blocking the sensitivity of cancer cells to CDDP [103]. Importantly, PRMT1 expression could serve as a predictive marker for sensitivity to platinum-based chemotherapy in patients with ovarian serous carcinoma [104].
Gastric cancer
Bioinformatics analysis revealed significantly upregulated PRMT1 expression in gastric cancer tissue compared to normal tissue [105]. Wang et al. discovered the role of KTN1 in stabilizing PRMT1 protein expression by inhibiting K48-linked polyubiquitination through decreased TRIM48-PRMT1 interaction. Current evidence suggests that PRMT1 facilitates the recruitment of MLXIP to the promoter region of the β-catenin gene, triggering the transcription of the β-catenin signaling pathway. This activation promotes the proliferation, migration, and invasion of gastric cancer cells, leading to tumor growth and metastasis [106]. PRMT1 also plays a role in promoting the EMT process through the Hippo signaling pathway and enhances the migration and invasive ability of gastric cancer cells [107]. The direct interaction between c-Fos and PRMT1 synergistically enhances c-Fos-mediated AP-1 activity by methylating the R287 residue, protecting it from autophagy degradation. The increased stability of the c-Fos protein leads to increased AP-1 function, promoting gastric tumorigenesis [108]. These findings highlight PRMT1 as a potential therapeutic target in gastric cancer treatment (Fig. 12).
Glioma
Glioma is recognized as the most aggressive type of brain tumor, and its highly infiltrative nature presents a significant challenge for achieving maximal safe resection. Temozolomide has become a crucial postoperative treatment for glioblastoma. Recent studies have elucidated the importance of arginine methylation, a post-translational protein modification, in the pathogenesis and progression of GBM. Dong et al. established the selective role of PRMT2 in facilitating the invasive phenotype of GBM [109]. PRMT5 regulates the proliferation and self-renewal of GBM neurospheres and is essential for the growth of GBM differentiated cells in serum [110]. Furthermore, PRMT6 was found to enhance mitotic activity [111] and cell cycle progression [112], thereby promoting tumor formation and progression in GBM models.
PRMT1 has been found to be overexpressed in both human glioma tissue and cell lines compared to normal brain tissue (Fig. 13). Studies indicated that downregulating PRMT1 levels effectively arrested the cell cycle’s progression from the G1 to S phase in glioma cells exhibiting high PRMT1 levels, leading to the inhibition of proliferation and induction of apoptosis [113]. However, patients diagnosed with IDH1R132H typically exhibit a noticeable decrease in PRMT1 expression. Previous studies have established that gliomas carrying the IDH1 mutant (IDH1R132H) exhibited antiproliferative characteristics due to hypermethylation of DNA and chromatin. Lathoria et al. recently identified additional mechanisms affecting cell death pathways in IDH1 mutant gliomas. Downregulated PRMT1 expression could hinder PTX3 transcription, resulting in escalated amalgamation between autophagosomes and lysosomes, along with an upsurge in ferritinophagy within glioma cells [114]. Ultimately, this leads to apoptosis in mutant glioma cells, holding the potential to improve patient prognosis.
Others
In nasopharyngeal carcinoma, the expression of PRMT1 and RRM2 is significantly elevated in CNE-2 cells. PRMT1 was found to inhibit CNE-2 cell apoptosis by promoting RRM2 expression. The presence of PRMT1 in these cells could inhibit apoptosis by promoting the expression of RRM2 [115]. In renal cancer, inhibiting PRMT1 resulted in decreased deposition of H3R4me2a at the Lcn2 promoter, attenuating the transcription of Lcn2 and resulting in a decrease in its expression. Ultimately, the Akt-RB signaling pathway is mitigated. Inhibition of ccRCC cell proliferation is characterized by G1 phase cell cycle arrest, and it enhances the sensitivity of ccRCC cells to Sunitinib treatment [116].
Tumor suppressive roles of PRMT1 in cancers
Many studies have shown that PRMT1 also acts as a tumor suppressor to prevent the occurrence of cancer (Fig. 14). In alcohol-induced liver cancer, alcohol inhibits the ability of PRMT1 to methylate arginine residues on the HNF4A promoter. This reduces HNF4A expression and promotes alcohol-related pathogenesis and proliferation of liver cancer cells [117]. The asymmetric dimethylarginine produced by PRMT1 inhibits the inducible nitric oxide synthase, reducing the production of nitrogen oxides and sulfur oxides, thereby weakening the oxidative stress and inflammatory response of the liver under the influence of alcohol, which in turn reduces cell death, inflammation, and the Wnt/β-Catenin signaling pathway activation, as well as tumor growth [118]. In addition, it has been revealed that PRMT1 could assist in the reversal of RIP3 necrotic colon cancer immune evasion. Mechanistically, PRMT1 methyltransferase is reportedly accountable for the methylation of RIP3, a central factor that accelerates necroptosis. This process of methylation obstructs the initiation of necroptosis by hindering the phosphorylation of RIP3, ultimately preventing the progression of tumor immune escape and necrotic colon cancer due to the initiation of necroptosis [119]. It has been found that PRMT1 could alleviate anemia symptoms in MDS patients [120]. Wang et al. elucidated the role of PRMT1 in mediating methylation of ME2 at the R67 site, which inhibited the conversion of ME2 monomers into dimers, resulting in decreased DUT activity, which mitigated thymidine and mtDNA generation. It has also been observed that the interaction between the MRPL45 and ME2 monomers could weaken the assembly of mitochondrial ribosomes and impair the production of mitochondrial proteins [121]. As a result, the progression of leukemia is significantly slowed. PRMT1 binds to ATF4 in a BTG1-dependent manner, and PRMT1 methylates ATF4, promoting transcription of some target genes of ATF4, leading to increased apoptosis in cancer cells [122]. In contrast to the function of PRMT5, PRMT1 overexpression has been found to modulate the protein level of CFLARL, an antiapoptotic protein, through physical interactions. This interaction enhances the binding between CFLARL and the E3 ligase ITCH, resulting in changes in ubiquitination levels and ultimately leading to the degradation of CFLARL. This degradation process increases the apoptosis of NSCLC cells [123]. Furthermore, under stress conditions, such as DNA damage in pancreatic cancer cells, PRMT1 has been observed to bind and methylate with P14ARF. This methylation caused ADMA at specific sites of the NLS/NoLS of P14ARF, namely R87/88/96/99. As a result, P14ARF and NPM were separated in the nucleolus and redistributed to the nucleus and cytoplasm, which promoted non-dependent P53 apoptosis and improved the prognosis of patients [124]. It is worth noting that PRMT1 exhibits a dual role, as it not only promotes the EMT process but also inhibits the proliferation of gastric cancer cells [107]. Low expression of PRMT1 inhibits the accumulation of FOXO1 in the nucleus and reduces sensitivity to chemotherapy drugs, leading to relapse after adjuvant chemotherapy and a poor prognosis [125].
PRMT1 inhibitors
A growing body of evidence from recent literature highlights the significance of PRMT1 in cancer research. Numerous studies have revealed that PRMT1 exhibits oncogene-like properties, playing a crucial role in regulating tumor cell proliferation, metastasis, and drug resistance. Consequently, it has been observed that PRMT1 is frequently overexpressed in tumor tissues, and its overexpression is often associated with poor prognosis. Given these findings, PRMT1 has emerged as a promising new research target for tumor therapy. As a result, there has been a surge of interest in the development of PRMT1 inhibitors as researchers strive to explore the potential of these inhibitors as cancer therapeutics (Table 1). The study of PRMT1 inhibitors has thus become a prominent area of focus in the quest for effective treatments against cancer.
Selective PRMT1 inhibitors
AMI-1
AMI-1, also known as Disodium 7,7′-(carbonyldiimino) bis(4-hydroxynaphthalene-2-sulfonate), is the first discovered protein arginine methyltransferase inhibitor [126]. With a chemical formula of C21H14N2Na2O9S2, it also acts as an effective NADPH oxidase-derived superoxide scavenger [127]. AMI-1 exhibits some similarity to peptidyl arginine but functions as a SAM non-competitive inhibitor since AMI-1 may bind to the co-substrate binding site rather than interact with the substrate pocket [126, 128]. By blocking the entry of protein/peptide substrates, it selectively inhibits the interaction between the substrate (arginine residue) and the enzyme without competing with the AdoMet binding site [129]. AMI-1 has been widely studied and applied in various research experiments. Yin et al. demonstrated through Duolink PLA that NONO ADMA levels were significantly reduced in AMI-1-treated KM12 and HCT8 CRC cells compared to control cells. In xenograft models, mice treated with AMI-1 exhibited smaller tumors [90]. Following AMI-1 treatment, PRMT1-mediated proliferation of gastric cancer cells was inhibited, exhibiting a dose-dependent effect [106]. Due to the effective inhibition of arginine methyltransferase activity by AMI-1, further analogs of AMI-1 are currently being developed [130].
Tc-e 5003
TC-E 5003 is a selective PRMT1 inhibitor, also known as N, N′-(sulfonyl-4,1-phenylene) bis(2-chloroacetamide). In the study by Kim et al., TC-E-5003 demonstrated promising anti-inflammatory effects by modulating the AP-1 and NF-κB signaling pathways induced by LPS, suggesting its potential as an anti-inflammatory compound [131]. In vitro experiments have shown that TC-E-5003 exhibits significant anti-tumor activity against lung cancer and breast cancer. Furthermore, by utilizing the INEI drug delivery system to deliver TC-E-5003-loaded INEI (40% NBCA) in an animal model, the average growth inhibition rate of xenografted human lung cancer cells was 68.23%, surpassing the inhibition rate achieved by TC-E-5003 alone (31.76%). This study further confirms the potential of the inhibitor TC-E-5003 as an anti-tumor drug and highlights INEI as an effective technique for enhancing anti-tumor effects [132].
C-7280948
C-7280948 is a selective PRMT1 inhibitor that inhibits the activity of PRMT1 by binding to the substrate-binding pocket, with an IC50 value of 12.8 μM [133]. Studies have shown that treatment with C-7280948 can eliminate radiation resistance induced by PRMT1-mediated methylation of PKP2, making it a potential radiosensitizer in lung cancer [134]. Additionally, it can reduce the levels of ADMA in KM12 and HCT8 cells, thereby inhibiting the proliferation, migration, and invasion of colorectal cancer cells [90].
ZJG51 and ZJG58
ZJG51 and ZJG58, selective inhibitors of type I PRMT, were designed and synthesized using molecular modeling to discover a new sub-binding pocket and occupy it by introducing a third substituent on the thiazole moiety. Molecular dynamics simulations revealed that ZJG51 - Pose 1 (where the ethylenediamine moiety is bound to the sublayer arginine binding site) emerged as the only stable binding site in the ZJG51 complex with PRMT1. Its additional aromatic substituent maintains its biological activity as a PRMT1 inhibitor by occupying the newly discovered sub-binding site. In comparison to ZJG58, ZJG51 exhibited potent inhibitory activity against all four tested tumor cells, with a remarkable effect against HeLa cells (IC50 = 9.43 ± 0.10 μM). Moreover, ZJG51 demonstrated efficacy in inducing apoptosis and inhibiting the migration of HeLa cells. Its mechanism of action may involve the activation of Caspase 9 and inhibition of EMT, both of which hold significant clinical implications for future cervical cancer treatment [135].
Non-selective PRMT1 inhibitors
MS023
The rational design and synthesis of MS023 involve the incorporation of the ethylenediamine side chain from EPZ020411 and CMPD-1, with the replacement of the pyrazole ring in EPZ020411 by a 1,2,3-triazole or pyrrole ring. MS023 is a selective inhibitor of type I PRMTs that exerts dose-dependent inhibition of PRMT1-mediated H4R3 methylation activity in MCF7 cells and suppresses the overexpression of PRMT6 in HEK293 cells [136]. MS023 can reportedly antagonize the methylation-promoting effect of PRMT1-mediated METTL14 on cell proliferation [18]. The combination of MS023 with PARP inhibitors could potentially serve as a novel therapeutic approach for MTAP-negative NSCLC and certain cancer cells that are resistant to PARP inhibitors [137]. In animal models treated with MS023, AML mice exhibited effective therapeutic outcomes [80], while significant inhibition of MM mice tumor growth was observed [138]. These findings indicate that MS023 has clinical value and the potential to be a valuable compound in the field of clinical oncology therapy.
GSK3368715
GSK3368715 is a potent and reversible inhibitor of type I PRMTs that binds to the peptide site adjacent to the SAM pocket, making it a non-competitive peptide-mixed inhibitor of SAM [139]. As a monotherapy, GSK3368715 has shown increased sensitivity to ferroptosis in AML [140] and has the potential to enhance anti-tumor immune responses [141, 142]. Promising preclinical results and peripheral target engagement were observed at higher doses in a clinical study (TRIAL REGISTRATION NUMBER: NCT03666988). However, the occurrence of treatment-emergent adverse events (TEEs), variable target engagement at the tumor level, and limited clinical efficacy observed led to the premature termination of the trial [143].
II757
The pan-inhibitor of arginine methyltransferases, II757, was carefully designed and synthesized by incorporating m-bromophenyl onto a guanidine moiety linked to a thioadenosine scaffold, inspired by the known inhibitor AH237. Through kinetic studies, it has been demonstrated that II757 exhibits competitive binding to the SAM binding site of PRMT1, thereby functioning as a potent SAM-competitive inhibitor of PRMT1 with an IC50 value of 16.4 nM. Importantly, II757 displays selectivity towards PRMTs over a range of other methyltransferases, making it a valuable tool and precursor for further exploration of PRMTs [144].
Conclusions and perspectives
Methylation is a fundamental type of epigenetic regulation that involves the transfer of active methyl groups to target chemical substances under the catalysis of methyltransferases without altering the composition of the DNA sequence. Methylation can occur in DNA, RNA, and proteins, with arginine methylation being a frequently studied post-translational modification of proteins [145]. PRMT1 is the most prevalent arginine methyltransferase, and its expression is dysregulated in cancer. Numerous studies have demonstrated that PRMT1 is significantly expressed in different cancer types and is linked to tumor malignancy and prognosis. PRMT1 contributes to tumorigenesis by regulating transcription factors and signaling pathways, which also paves the way for the occurrence and development of tumors. Furthermore, the excessive expression of PRMT1 is linked to the conspicuous proliferation, invasion, and metastasis of tumorous cells, hence making it a crucial protein in cancer development.
A review of the literature reveals several key points about PRMT1. Firstly, PRMT1 exhibits different roles in different stages of tumor development, as demonstrated in alcohol-induced liver cancer. Under the stimulation of high-risk factors associated with alcohol consumption, PRMT1 functions normally to prevent tumor formation. However, varying degrees of alcohol intake can lead to different outcomes, such as decreased enzymatic activity of PRMT1. In alcohol-induced liver cancer cells, the normal biological function of PRMT1 may not be maintained, and instead, it may exhibit characteristics that promote cancer cell proliferation, invasion, and metastasis, thereby exerting a carcinogenic effect. The stimulatory and inhibitory effects of PRMT1 on growth may vary in different situations, while its ability to inhibit tumor growth depends on the stage of tumor development. Secondly, PRMT1 exhibits different mechanisms of action in regulating the same signaling pathway in different types of tumor cells. In the regulation of the EGFR signaling pathway, PRMT1 can act as a transcriptional regulator, promoting the expression of EGFR and enhancing signal transduction. It can also act as a methyltransferase, directly interacting with the EGFR molecule, leading to methylation of arginine residues in the extracellular domain of EGFR, thereby affecting the EGFR signaling pathway. Thirdly, the anticancer effect of PRMT1 is undeniable. Studies have found that compared to apoptosis, necroptosis is immunogenic, indicating that necroptosis in tumor cells can potentially be used as a therapeutic method by activating the immune system [146]. However, in necrotic colorectal cancer, a unique subtype of colon cancer, necroptosis can also provide a barrier for tumor cells, promoting tumor growth by creating an immune evasion microenvironment during the process of necroptosis. PRMT1 can reverse the immune evasion of tumor cells caused by necroptosis. Additionally, PRMT1 binds to ATF4 in a BTG1-dependent manner, methylating ATF4 and promoting the transcription of certain target genes of ATF4, leading to increased apoptosis of cancer cells. Under genotoxic stress conditions, PRMT1 promotes P53-independent cell apoptosis, improving the prognosis of pancreatic cancer patients, suggesting that PRMT1 also plays different mechanisms in inhibiting tumors.
Given the role of PRMT1 in tumors, it is imperative to further investigate the development and utilization of PRMT1 inhibitors. Table 1 presents the PRMT1 inhibitors that have been developed thus far, showcasing their ability to inhibit tumor cell proliferation and invasion, among other effects, underscoring their anti-tumor activity. Notably, GSK3368715 underwent a clinical trial that was prematurely terminated due to the occurrence of a TEE, variable engagement with the tumor target level, and limited clinical effectiveness. Additionally, GGA [97], spermine [101], and sodium propionate [147] have demonstrated inhibition of PRMT1. Therefore, it is crucial to explore more selective PRMT1-targeted inhibitors that have fewer side effects and improved efficacy. Furthermore, it is equally important to consider new therapeutic avenues of research, such as prime editing [148], proteolysis targeting chimera (PROTAC) [149], and CAR-T cell therapy [150].
It is well-established that PRMT1 plays a crucial role in regulating gene transcription, protein expression, and tumor cell differentiation, which contribute to the proliferative and metastatic behaviors exhibited by tumors. Additionally, PRMT1 is involved in promoting DNA damage repair, leading to the acquisition of antiapoptotic and drug-resistant characteristics in tumor cells. The role of PRMT1 in regulating the tumor microenvironment has also been gradually recognized, although its potential involvement in promoting tumor angiogenesis remains an area of unknown research. Furthermore, the exact mechanisms by which PRMT1 functions in various signaling pathways, such as the Hedgehog and Hippo signaling pathways, warrant further investigation.
Herein, we provide a systematic introduction to the role of PRMT1 in tumors (Fig. 15). The dysregulated expression of PRMT1 in tumors underscores its considerable impact. Additionally, emerging research indicates that PRMT1 is implicated in inflammatory diseases [151], cardiovascular diseases [152], neurological disorders [38], and immune disorders [153], emphasizing its pivotal role in the development of various diseases. As a highly valuable arginine methyltransferase, PRMT1 holds substantial research potential, providing crucial insights into the progression of diverse diseases. In summary, while PRMT1 has demonstrated a significant impact on tumor development, a comprehensive understanding of its role and potential therapeutic applications remains a work in progress.
Availability of data and materials
Not applicable.
Abbreviations
- PRMTs:
-
Protein arginine methyltransferases
- MMA:
-
Monomethylarginine
- ADMA:
-
Asymmetric dimethylarginine
- SDMA:
-
Symmetric dimethylarginine
- SAM:
-
S-Adenosyl methionine
- SAH:
-
S-adenosylhomocysteine
- GAR:
-
Glycine- and arginine-rich
- PGM:
-
Post-translational glycine methylation
- RGG:
-
Arg-Gly-Gly
- 53BP1:
-
P53-binding protein 1
- MRE11:
-
Meiotic recombination 11 homolog A
- γ-IR:
-
Gamma ionizing radiation
- METTL14:
-
Methyltransferase like 14
- PR:
-
Progesterone receptor
- EMT:
-
Epithelial-mesenchymal transition
- BCL2:
-
B-cell lymphoma 2
- PI3K:
-
Phosphatidylinositide 3-kinases
- AKT (PKB):
-
Protein kinase B
- TAMs:
-
Tumor-associated macrophages
- IL-6:
-
Interleukin-6
- EZH2:
-
Enhancer of zeste homolog 2
- PRC2:
-
Polycomb repressive complex 2
- C/EBPα:
-
CCAAT/enhancer binding proteins α
- SG:
-
Stress granule
- PARP:
-
Poly (ADP-ribose) polymerase
- HP1γ:
-
Heterochromatin protein 1γ
- HDAC1/2:
-
Histone deacetylases 1 and 2
- LSD1:
-
Lysine-specific demethylase 1
- CDK1:
-
Cyclin-dependent kinase 1
- AMPK:
-
Adenosine monophosphate-activated protein kinase
- TRAF6:
-
TNF receptor-associated factor 6
- SUZ12:
-
Suppressor of zeste 12 protein homolog
- DAB2IP:
-
Disabled homolog 2-interacting protein
- CSTA:
-
Cystatin A
- P16:
-
Cyclin-dependent kinase inhibitor 2A
- P21:
-
Cyclin-dependent kinase inhibitor 1A
- ZEB1:
-
Zinc finger E-Box binding homeobox 1
- EGFR:
-
Epidermal growth factor receptor
- HDAC3:
-
Histone deacetylase 3
- circTBC1D14:
-
Circular RNA TBC1D14
- P53:
-
Tumor protein p53
- BRCA1:
-
Breast cancer type 1 susceptibility protein
- BARD1:
-
BRCA1-associated RING domain protein 1
- c-Myc:
-
Cellular myelocytomatosis oncogene
- ERα:
-
Estrogen receptor alpha
- IGF-1:
-
Insulin-like growth factor 1
- IGF-1R:
-
Insulin-like growth factor 1 receptor
- IRS1:
-
Insulin receptor substrate 1
- Shc:
-
Src homology 2 domain-containing transforming protein
- Grb2:
-
Growth factor receptor-bound protein 2
- ERK:
-
Extracellular signal-regulated kinase
- MICU1:
-
Mitochondrial Ca2+ uptake 1
- UCP2:
-
Uncoupling protein 2
- OXPHOS:
-
Oxidative phosphorylation
- INCENP:
-
Inner centromere protein
- AURKB:
-
Aurora kinase B
- SCLC:
-
Small cell lung cancer
- FEN1:
-
Flap endonuclease 1
- NSCLC:
-
Non-small cell lung cancer
- HCC:
-
Hepatocellular carcinoma
- TCGA:
-
The Cancer Genome Atlas
- DDR:
-
DNA damage response
- TCF:
-
T-cell factor
- CTNNB1:
-
Catenin beta 1
- Gli1:
-
Glioma-associated oncogene 1
- HSP70:
-
Heat shock protein 70
- EphA2:
-
Erythropoietin-producing hepatocellular A2
- SOX2:
-
SRY-Box transcription factor 2
- STAT3:
-
Signal transducer and activator of transcription 3
- mTORC1:
-
Mammalian target of rapamycin complex 1
- GATOR1:
-
GTPase-activating protein (GAP) activity towards rags 1
- WDR24:
-
WD repeat-containing protein 24
- TGF-β1:
-
Transforming growth factor beta 1
- CDKN1A:
-
Cyclin-dependent kinase inhibitor 1A
- MDS:
-
Myelodysplastic syndrome
- RBM15:
-
RNA-binding motif protein 15
- DUSP4:
-
Dual-specificity phosphatase 4
- P38 MAPK:
-
p38 mitogen-activated protein kinase
- MLL-GAS7:
-
Mixed-lineage leukemia-growth arrest-specific 7
- MOZ-TIF2:
-
Monocytic leukemia zinc finger protein- transcriptional intermediary factor 2
- H4R3me2a:
-
Asymmetric dimethylation of histone H4 on arginine 3
- H3K9me3:
-
Trimethylation of histone H3 on lysine 9
- KDM4C:
-
Lysine-specific demethylase 4C
- AML:
-
Acute myeloid leukemia
- MLL:
-
Mixed-lineage leukemia
- FLT3:
-
FMS-like tyrosine kinase 3
- ITD:
-
Internal tandem duplications
- HOXA9:
-
Homeobox A9
- LSK:
-
Lin− Sca-1+ c-kit+
- ENL:
-
Eleven-nineteen-leukemia;
- GFI1:
-
Growth factor independence 1
- SMARCA4:
-
SWI/SNF related matrix-associated actin-dependent regulator of chromatin subfamily A member 4
- TNS4:
-
Tensin-4
- KRAS:
-
Kirsten rat sarcoma viral oncogene homolog
- NONO:
-
Non-POU domain-containing octamer-binding protein
- P53/RB:
-
Tumor protein 53/retinoblastoma protein
- eIF4G1:
-
Eukaryotic translation initiation factor 4 gamma 1
- TIPE1:
-
Tumor necrosis factor alpha-induced protein 8-like 1
- CHIP:
-
Carboxyl terminus of HSP70-interacting protein
- FAS:
-
Tumor necrosis factor receptor superfamily member 6
- hsa_circ_0094606:
-
Homo sapiens circular RNA 0094606
- CRISPR:
-
Clustered regularly interspaced short palindromic repeats
- ILF3:
-
Interleukin-enhanced binding factor 3
- FAM98A:
-
Family with sequence similarity 98 member A
- DNA-PK:
-
DNA-dependent protein kinase
- NF-κB:
-
Nuclear factor kappa-light-chain-enhancer of activated B cells
- CDDP:
-
Cisplatin
- KTN1:
-
Kinectin 1
- TRIM48:
-
Tripartite motif-containing 48
- MLXIP:
-
MLX-interacting protein
- AP-1:
-
Activator protein 1
- GBM:
-
Glioblastoma
- IDH1:
-
Isocitrate dehydrogenase (NADP+) 1
- PTX3:
-
Pentraxin 3
- RRM2:
-
Ribonucleotide reductase regulatory subunit M2
- Lcn2:
-
Lipocalin 2
- ccRCC:
-
Clear cell renal cell carcinoma
- HNF4A:
-
Hepatocyte nuclear factor 4 alpha
- RIP3:
-
Receptor interacting protein kinase 3
- ME2:
-
Malic enzyme 2
- DUT:
-
Deoxyuridine 5′-triphosphate nucleotidohydrolase
- mtDNA:
-
Mitochondrial DNA
- MRPL45:
-
Mitochondrial ribosomal protein L45
- BTG1:
-
B-cell translocation gene 1
- ATF4:
-
Activating transcription factor 4
- CFLARL :
-
CASP8 and FADD-like apoptosis regulator
- P14ARF :
-
p14 Alternate Reading Frame (ARF) protein
- NLS/NoLS:
-
Nuclear localization signal/nucleolar localization signal
- FOXO1:
-
Forkhead box O1
- NADPH:
-
Nicotinamide adenine dinucleotide phosphate
- CRC:
-
Colorectal cancer
- INEI:
-
Injectable NBCA ethyl oleate implant
- NBCA:
-
N-butyl-2-cyanoacrylate
- MTAP:
-
Methylthioadenosine phosphorylase
- MM:
-
Multiple myeloma
- CAR-T:
-
Chimeric antigen receptor T-cell therapy
- RTK:
-
Receptor tyrosine kinase
- ESCC:
-
Esophageal squamous cell carcinoma
- DBD:
-
DNA binding domain
- LPS:
-
Lipopolysaccharide
- PKP2:
-
Plakophilin 2
- GGA:
-
Geranylgeranylacetone
- TEEs:
-
Treatment-emergent adverse events
- NPM:
-
Nucleophosmin
- PROTAC:
-
Proteolysis targeting chimera
- 3-PG:
-
3-phosphoglycerate
- 3-PPyr:
-
3-phosphopyruvate
- HUWEI:
-
HECT, UBA and WWE domain containing E3 ubiquitin protein ligase 1
- ITCH:
-
Itchy E3 ubiquitin protein ligase
- iNOS:
-
Inducible nitric oxide synthase.
References
Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011;144:646–74.
Blanc RS, Richard S. Arginine methylation: the coming of age. Mol Cell. 2017;65:8–24.
Stopa N, Krebs JE, Shechter D. The PRMT5 arginine methyltransferase: many roles in development, cancer and beyond. Cell Mol Life Sci. 2015;72:2041–59.
Yang Y, Bedford MT. Protein arginine methyltransferases and cancer. Nat Rev Cancer. 2013;13:37–50.
Chen Z, Gan J, Wei Z, Zhang M, Du Y, Xu C, et al. The emerging role of PRMT6 in Cancer. Front Oncol. 2022;12:841381.
Thiebaut C, Eve L, Poulard C, Le Romancer M. Structure, activity, and function of PRMT1. Life (Basel). 2021;11:1147.
Zhang X, Cheng X. Structure of the predominant protein arginine methyltransferase PRMT1 and analysis of its binding to substrate peptides. Structure. 2003;11:509–20.
Goulet I, Gauvin G, Boisvenue S, Côté J. Alternative splicing yields protein arginine methyltransferase 1 isoforms with distinct activity, substrate specificity, and subcellular localization. J Biol Chem. 2007;282:33009–21.
Baldwin RM, Morettin A, Paris G, Goulet I, Côté J. Alternatively spliced protein arginine methyltransferase 1 isoform PRMT1v2 promotes the survival and invasiveness of breast cancer cells. Cell Cycle. 2012;11:4597–612.
Lin WJ, Gary JD, Yang MC, Clarke S, Herschman HR. The mammalian immediate-early TIS21 protein and the leukemia-associated BTG1 protein interact with a protein-arginine N-methyltransferase. J Biol Chem. 1996;271:15034–44.
Tewary SK, Zheng YG, Ho MC. Protein arginine methyltransferases: insights into the enzyme structure and mechanism at the atomic level. Cell Mol Life Sci. 2019;76:2917–32.
Tang J, Frankel A, Cook RJ, Kim S, Paik WK, Williams KR, et al. PRMT1 is the predominant type I protein arginine methyltransferase in mammalian cells. J Biol Chem. 2000;275:7723–30.
Bedford MT. Arginine methylation at a glance. J Cell Sci. 2007;120:4243–6.
Boisvert FM, Rhie A, Richard S, Doherty AJ. The GAR motif of 53BP1 is arginine methylated by PRMT1 and is necessary for 53BP1 DNA binding activity. Cell Cycle. 2005;4:1834–41.
Yu Z, Vogel G, Coulombe Y, Dubeau D, Spehalski E, Hébert J, et al. The MRE11 GAR motif regulates DNA double-strand break processing and ATR activation. Cell Res. 2012;22:305–20.
Wang H, Straubinger RM, Aletta JM, Cao J, Duan X, Yu H, et al. Accurate localization and relative quantification of arginine methylation using Nanoflow liquid chromatography coupled to Electron transfer dissociation and Orbitrap mass spectrometry. J Am Soc Mass Spectrom. 2009;20:507–19.
Yamaguchi A, Kitajo K. The effect of PRMT1-mediated arginine methylation on the subcellular localization, stress granules, and detergent-insoluble aggregates of FUS/TLS. PLoS One. 2012;7:e49267.
Wang J, Wang Z, Inuzuka H, Wei W, Liu J. PRMT1 methylates METTL14 to modulate its oncogenic function. Neoplasia. 2023;42:100912.
Wooderchak WL, Zang T, Zhou ZS, Acuña M, Tahara SM, Hevel JM. Substrate profiling of PRMT1 reveals amino acid sequences that extend beyond the “RGG” paradigm. Biochem. 2008;47:9456–66.
Sudhakar SRN, Khan SN, Clark A, Hendrickson-Rebizant T, Patel S, Lakowski TM, et al. Protein arginine methyltransferase 1, a major regulator of biological processes. Biochem Cell Biol. 2023;
Pawlak MR, Scherer CA, Chen J, Roshon MJ, Ruley HE. Arginine N-methyltransferase 1 is required for early postimplantation mouse development, but cells deficient in the enzyme are viable. Mol Cell Biol. 2000;20:4859–69.
Shibata Y, Okada M, Miller TC, Shi YB. Knocking out histone methyltransferase PRMT1 leads to stalled tadpole development and lethality in Xenopus tropicalis. Biochim Biophys Acta Gen Subj. 2020;1864:129482.
Zhu L, He X, Dong H, Sun J, Wang H, Zhu Y, et al. Protein arginine methyltransferase 1 is required for maintenance of normal adult hematopoiesis. Int J Biol Sci. 2019;15:2763–73.
Mizutani S, Yoshida T, Zhao X, Nimer SD, Taniwaki M, Okuda T. Loss of RUNX1/AML1 arginine-methylation impairs peripheral T cell homeostasis. Br J Haematol. 2015;170:859–73.
Infantino S, Light A, O’Donnell K, Bryant V, Avery DT, Elliott M, et al. Arginine methylation catalyzed by PRMT1 is required for B cell activation and differentiation. Nat Commun. 2017;8:891.
Xue L, Bao L, Roediger J, Su Y, Shi B, Shi YB. Protein arginine methyltransferase 1 regulates cell proliferation and differentiation in adult mouse adult intestine. Cell Biosci. 2021;11:113.
Lee J, Villarreal OD, Wang YC, Ragoussis J, Rivest S, Gosselin D, et al. PRMT1 is required for the generation of MHC-associated microglia and remyelination in the central nervous system. Life Sci All. 2022;5:e202201467.
Waseem S, Kumar S, Lee K, Yoon BH, Kim M, Kim H, et al. Protein arginine methyltransferase 1 is essential for the meiosis of male germ cells. Int J Mol Sci. 2021;22:7951.
Gou Y, Li J, Jackson-Weaver O, Wu J, Zhang T, Gupta R, et al. Protein arginine methyltransferase PRMT1 is essential for Palatogenesis. J Dent Res. 2018;97:1510–8.
Jackson-Weaver O, Ungvijanpunya N, Yuan Y, Qian J, Gou Y, Wu J, et al. PRMT1-p53 pathway controls Epicardial EMT and invasion. Cell Rep. 2020;31:107739.
Pyun JH, Ahn BY, Vuong TA, Kim SW, Jo Y, Jeon J, et al. Inducible Prmt1 ablation in adult vascular smooth muscle leads to contractile dysfunction and aortic dissection. Exp Mol Med. 2021;53:1569–79.
Murata K, Lu W, Hashimoto M, Ono N, Muratani M, Nishikata K, et al. PRMT1 deficiency in mouse juvenile heart induces dilated cardiomyopathy and reveals cryptic alternative splicing products. iScience. 2018;8:200–13.
An X, Lee J, Kim GH, Kim HJ, Pyo HJ, Kwon I, et al. Modulation of IKs channel-PIP2 interaction by PRMT1 plays a critical role in the control of cardiac repolarization. J Cell Physiol. 2022;237:3069–79.
Choi S, Choi D, Lee YK, Ahn SH, Seong JK, Chi SW, et al. Depletion of Prmt1 in adipocytes impairs glucose homeostasis in diet-induced obesity. Diabetes. 2021;70:1664–78.
Xu L, Huang Z, Lo TH, Lee JTH, Yang R, Yan X, et al. Hepatic PRMT1 ameliorates diet-induced hepatic steatosis via induction of PGC1α. Theranostics. 2022;12:2502–18.
Park JY, Choi JH, Lee SN, Cho HJ, Ahn JS, Kim YB, et al. Protein arginine methyltransferase 1 contributes to the development of allergic rhinitis by promoting the production of epithelial-derived cytokines. J Allergy Clin Immunol. 2021;147:1720–31.
Chen YY, Peng XF, Liu GY, Liu JS, Sun L, Liu H, et al. Protein arginine methyltranferase-1 induces ER stress and epithelial-mesenchymal transition in renal tubular epithelial cells and contributes to diabetic nephropathy. Biochim Biophys Acta Mol basis Dis. 2019;1865:2563–75.
Hashimoto M, Fukamizu A, Nakagawa T, Kizuka Y. Roles of protein arginine methyltransferase 1 (PRMT1) in brain development and disease. Biochim Biophys Acta Gen Subj. 2021;1865:129776.
Siegel RL, Miller KD, Fuchs HE, Jemal A. Cancer statistics, 2021. CA Cancer J Clin. 2021;71:7–33.
Bao J, Di Lorenzo A, Lin K, Lu Y, Zhong Y, Sebastian MM, et al. Mouse models of overexpression reveal distinct oncogenic roles for different type I protein arginine methyltransferases. Cancer Res. 2019;79:21–32.
Suresh S, Huard S, Brisson A, Némati F, Dakroub R, Poulard C, et al. PRMT1 regulates EGFR and Wnt signaling pathways and is a promising target for combinatorial treatment of breast Cancer. Cancers (Basel). 2022;14:306.
Liu LM, Sun WZ, Fan XZ, Xu YL, Cheng MB, Zhang Y. Methylation of C/EBPα by PRMT1 inhibits its tumor-suppressive function in breast Cancer. Cancer Res. 2019;79:2865–77.
Li Z, Wang D, Lu J, Huang B, Wang Y, Dong M, et al. Methylation of EZH2 by PRMT1 regulates its stability and promotes breast cancer metastasis. Cell Death Differ. 2020;27:3226–42.
Li Z, Wang D, Wang W, Chen X, Tang A, Hou P, et al. Macrophages-stimulated PRMT1-mediated EZH2 methylation promotes breast cancer metastasis. Biochem Biophys Res Commun. 2020;533:679–84.
Li Z, Wang D, Chen X, Wang W, Wang P, Hou P, et al. PRMT1-mediated EZH2 methylation promotes breast cancer cell proliferation and tumorigenesis. Cell Death Dis. 2021;12:1080.
Gao Y, Zhao Y, Zhang J, Lu Y, Liu X, Geng P, et al. The dual function of PRMT1 in modulating epithelial-mesenchymal transition and cellular senescence in breast cancer cells through regulation of ZEB1. Sci Rep. 2016;6:19874.
Nakai K, Xia W, Liao HW, Saito M, Hung MC, Yamaguchi H. The role of PRMT1 in EGFR methylation and signaling in MDA-MB-468 triple-negative breast cancer cells. Breast Cancer. 2018;25:74–80.
Liu Y, Liu Y, He Y, Zhang N, Zhang S, Li Y, et al. Hypoxia-induced FUS–circTBC1D14 stress granules promote autophagy in TNBC. Adv Sci. 2023;10:e2204988.
Liu LM, Tang Q, Hu X, Zhao JJ, Zhang Y, Ying GG, et al. Arginine methyltransferase PRMT1 regulates p53 activity in breast Cancer. Life (Basel). 2021;11:789.
Montenegro MF, González-Guerrero R, Sánchez-Del-Campo L, Piñero-Madrona A, Cabezas-Herrera J, Rodríguez-López JN. PRMT1-dependent methylation of BRCA1 contributes to the epigenetic defense of breast cancer cells against ionizing radiation. Sci Rep. 2020;10:13275.
Hsu WJ, Chen CH, Chang YC, Cheng CH, TsaI YH, Lin CW. PRMT1 confers resistance to Olaparib via modulating MYC signaling in triple-negative breast Cancer. J Pers Med. 2021;11:1009.
Malbeteau L, Jacquemetton J, Languilaire C, Corbo L, Le Romancer M, Poulard C. PRMT1, a key modulator of Unliganded progesterone receptor signaling in breast Cancer. Int J Mol Sci. 2022;23:9509.
Malbeteau L, Poulard C, Languilaire C, Mikaelian I, Flamant F, Le Romancer M, et al. PRMT1 Is Critical for the Transcriptional Activity and the Stability of the Progesterone Receptor. iSci. 2020;23:101236.
Choucair A, Pham TH, Omarjee S, Jacquemetton J, Kassem L, Trédan O, et al. The arginine methyltransferase PRMT1 regulates IGF-1 signaling in breast cancer. Oncogene. 2019;38:4015–27.
Giuliani V, Miller MA, Liu CY, Hartono SR, Class CA, Bristow CA, et al. PRMT1-dependent regulation of RNA metabolism and DNA damage response sustains pancreatic ductal adenocarcinoma. Nat Commun. 2021;12:4626.
Song C, Chen T, He L, Ma N, Li JA, Rong YF, et al. PRMT1 promotes pancreatic cancer growth and predicts poor prognosis. Cell Oncol (Dordr). 2020;43:51–62.
Wang Y, Hsu JM, Kang Y, Wei Y, Lee PC, Chang SJ, et al. Oncogenic functions of Gli1 in pancreatic adenocarcinoma are supported by its PRMT1-mediated methylation. Cancer Res. 2016;76:7049–58.
Wang L, Jia Z, Xie D, Zhao T, Tan Z, Zhang S, et al. Methylation of HSP70 orchestrates its binding to and stabilization of BCL2 mRNA and renders pancreatic Cancer cells resistant to therapeutics. Cancer Res. 2020;80:4500–13.
Hu Q, Qin Y, Ji S, Xu W, Liu W, Sun Q, et al. UHRF1 promotes aerobic glycolysis and proliferation via suppression of SIRT4 in pancreatic cancer. Cancer Lett. 2019;452:226–36.
Yang J, Ren B, Yang G, Wang H, Chen G, You L, et al. The enhancement of glycolysis regulates pancreatic cancer metastasis. Cell Mol Life Sci. 2020;77:305–21.
Vilas-Boas EA, Cabral-Costa JV, Ramos VM, Caldeira da Silva CC, Kowaltowski AJ. Goldilocks calcium concentrations and the regulation of oxidative phosphorylation: too much, too little, or just right. J Biol Chem. 2023;299:102904.
Madreiter-Sokolowski CT, Győrffy B, Klec C, Sokolowski AA, Rost R, Waldeck-Weiermair M, et al. UCP2 and PRMT1 are key prognostic markers for lung carcinoma patients. Oncotarget. 2017;8:80278–85.
Deng X, Von Keudell G, Suzuki T, Dohmae N, Nakakido M, Piao L, et al. PRMT1 promotes mitosis of cancer cells through arginine methylation of INCENP. Oncotarget. 2015;6:35173–82.
Avasarala S, Van Scoyk M, Karuppusamy Rathinam MK, Zerayesus S, Zhao X, Zhang W, et al. PRMT1 is a novel regulator of epithelial-mesenchymal-transition in non-small cell lung Cancer. J Biol Chem. 2015;290:13479–89.
He L, Hu Z, Sun Y, Zhang M, Zhu H, Jiang L, et al. PRMT1 is critical to FEN1 expression and drug resistance in lung cancer cells. DNA Repair (Amst). 2020;95:102953.
Guo Z, Zheng L, Xu H, Dai H, Zhou M, Pascua MR, et al. Methylation of FEN1 suppresses nearby phosphorylation and facilitates PCNA binding. Nat Chem Biol. 2010;6:766–73.
Liang S, Wang Q, Wen Y, Wang Y, Li M, Wang Q, et al. Ligand-independent EphA2 contributes to chemoresistance in small-cell lung cancer by enhancing PRMT1-mediated SOX2 methylation. Cancer Sci. 2023;114:921–36.
Ryu JW, Kim SK, Son MY, Jeon SJ, Oh JH, Lim JH, et al. Novel prognostic marker PRMT1 regulates cell growth via downregulation of CDKN1A in HCC. Oncotarget. 2017;8:115444–55.
Zhao J, O’Neil M, Vittal A, Weinman SA, Tikhanovich I. PRMT1-dependent macrophage IL-6 production is required for alcohol-induced HCC progression. Gene Expr. 2019;19:137–50.
Zhang XP, Jiang YB, Zhong CQ, Ma N, Zhang EB, Zhang F, et al. PRMT1 promoted HCC growth and metastasis in vitro and in vivo via activating the STAT3 Signalling pathway. Cell Physiol Biochem. 2018;47:1643–54.
Yin S, Liu L, Ball LE, Wang Y, Bedford MT, Duncan SA, et al. CDK5-PRMT1-WDR24 signaling cascade promotes mTORC1 signaling and tumor growth. Cell Rep. 2023;42:112316.
Gou Q, He S, Zhou Z. Protein arginine N-methyltransferase 1 promotes the proliferation and metastasis of hepatocellular carcinoma cells. Tumour Biol. 2017;39:1010428317691419.
Wei H, Liu Y, Min J, Zhang Y, Wang J, Zhou M, et al. Protein arginine methyltransferase 1 promotes epithelial-mesenchymal transition via TGF-β1/Smad pathway in hepatic carcinoma cells. Neoplasma. 2019;66:918–29.
Wang K, Luo L, Fu S, Wang M, Wang Z, Dong L, et al. PHGDH arginine methylation by PRMT1 promotes serine synthesis and represents a therapeutic vulnerability in hepatocellular carcinoma. Nat Commun. 2023;14:1011.
Zhao X, Jankovic V, Gural A, Huang G, Pardanani A, Menendez S, et al. Methylation of RUNX1 by PRMT1 abrogates SIN3A binding and potentiates its transcriptional activity. Genes Dev. 2008;22:640–53.
Zhang L, Tran NT, Su H, Wang R, Lu Y, Tang H, et al. Cross-talk between PRMT1-mediated methylation and ubiquitylation on RBM15 controls RNA splicing. Elife. 2015;4:e07938.
Jin S, Mi Y, Song J, Zhang P, Liu Y. PRMT1-RBM15 axis regulates megakaryocytic differentiation of human umbilical cord blood CD34+ cells. Exp Ther Med. 2018;15:2563–8.
Su H, Jiang M, Senevirathne C, Aluri S, Zhang T, Guo H, et al. Methylation of dual-specificity phosphatase 4 controls cell differentiation. Cell Rep. 2021;36:109421.
Cheung N, Fung TK, Zeisig BB, Holmes K, Rane JK, Mowen KA, et al. Targeting aberrant epigenetic networks mediated by PRMT1 and KDM4C in acute myeloid leukemia. Cancer Cell. 2016;29:32–48.
He X, Zhu Y, Lin YC, Li M, Du J, Dong H, et al. PRMT1-mediated FLT3 arginine methylation promotes maintenance of FLT3-ITD+ acute myeloid leukemia. Blood. 2019;134:548–60.
Zhu Y, He X, Lin YC, Dong H, Zhang L, Chen X, et al. Targeting PRMT1-mediated FLT3 methylation disrupts maintenance of MLL-rearranged acute lymphoblastic leukemia. Blood. 2019;134:1257–68.
Siriboonpiputtana T, Zeisig BB, Zarowiecki M, Fung TK, Mallardo M, Tsai CT, et al. Transcriptional memory of cells of origin overrides β-catenin requirement of MLL cancer stem cells. EMBO J. 2017;36:3139–55.
Vadnais C, Chen R, Fraszczak J, Yu Z, Boulais J, Pinder J, et al. GFI1 facilitates efficient DNA repair by regulating PRMT1 dependent methylation of MRE11 and 53BP1. Nat Commun. 2018;9:1418.
Sasaki T, Hiroki K, Yamashita Y. The role of epidermal growth factor receptor in cancer metastasis and microenvironment. Biomed Res Int. 2013;2013:546318.
Patel R, Leung HY. Targeting the EGFR-family for therapy: biological challenges and clinical perspective. Curr Pharm Des. 2012;18:2672–9.
Jonker DJ, O’Callaghan CJ, Karapetis CS, Zalcberg JR, Tu D, Au HJ, et al. Cetuximab for the treatment of colorectal cancer. N Engl J Med. 2007;357:2040–8.
Liao HW, Hsu JM, Xia W, Wang HL, Wang YN, Chang WC, et al. PRMT1-mediated methylation of the EGF receptor regulates signaling and cetuximab response. J Clin Invest. 2015;125:4529–43.
Yao B, Gui T, Zeng X, Deng Y, Wang Z, Wang Y, et al. PRMT1-mediated H4R3me2a recruits SMARCA4 to promote colorectal cancer progression by enhancing EGFR signaling. Genome Med. 2021;13:58.
Zeng X, Yao B, Liu J, Gong GW, Liu M, Li J, et al. The SMARCA4R1157W mutation facilitates chromatin remodeling and confers PRMT1/SMARCA4 inhibitors sensitivity in colorectal cancer. NPJ Precis Oncol. 2023;7:28.
Yin XK, Wang YL, Wang F, Feng WX, Bai SM, Zhao WW, et al. PRMT1 enhances oncogenic arginine methylation of NONO in colorectal cancer. Oncogene. 2021;40:1375–89.
Zhao Y, Lu Q, Li C, Wang X, Jiang L, Huang L, et al. PRMT1 regulates the tumour-initiating properties of esophageal squamous cell carcinoma through histone H4 arginine methylation coupled with transcriptional activation. Cell Death Dis. 2019;10:359.
Zhou W, Qin W, Qiao F, Xu Z, Yuan Y, Chen H. Protein arginine methyltransferase 1 interacts with Gli1 and regulates its transcriptional activity. Tumor Biol. 2016;37:9071–6.
Zhou W, Yue H, Li C, Chen H, Yuan Y. Protein arginine methyltransferase 1 promoted the growth and migration of cancer cells in esophageal squamous cell carcinoma. Tumour Biol. 2016;37:2613–9.
Hsu JH, Hubbell-Engler B, Adelmant G, Huang J, Joyce CE, Vazquez F, et al. PRMT1-mediated translation regulation is a crucial vulnerability of Cancer. Cancer Res. 2017;77:4613–25.
Li Z, Meng D, Liu Y, Bi F, Tian K, Xu J, et al. Knockdown of PRMT1 suppresses the malignant biological behavior of osteosarcoma cells and increases cisplatin sensitivity via c-Myc-mediated BCAT1 downregulation. J Biochem Mol Toxicol. 2024;38:e23537.
Yang M, Zhang Y, Liu G, Zhao Z, Li J, Yang L, et al. TIPE1 inhibits osteosarcoma tumorigenesis and progression by regulating PRMT1 mediated STAT3 arginine methylation. Cell Death Dis. 2022;13:815.
Jiang L, Liao J, Liu J, Wei Q, Wang Y. Geranylgeranylacetone promotes human osteosarcoma cell apoptosis by inducing the degradation of PRMT1 through the E3 ubiquitin ligase CHIP. J Cell Mol Med. 2021;25:7961–72.
Grypari IM, Logotheti S, Zolota V, Troncoso P, Efstathiou E, Bravou V, et al. The protein arginine methyltransferases (PRMTs) PRMT1 and CARM1 as candidate epigenetic drivers in prostate cancer progression. Medicine (Baltimore). 2021;100:e27094.
Zhang Y, Wang K, Yang D, Liu F, Xu X, Feng Y, et al. Hsa_circ_0094606 promotes malignant progression of prostate cancer by inducing M2 polarization of macrophages through PRMT1-mediated arginine methylation of ILF3. Carcinogenesis. 2023;44:15–28.
Tang S, Sethunath V, Metaferia NY, Nogueira MF, Gallant DS, Garner ER, et al. A genome-scale CRISPR screen reveals PRMT1 as a critical regulator of androgen receptor signaling in prostate cancer. Cell Rep. 2022;38:110417.
Li X, Li F, Ye F, Guo H, Chen W, Jin J, et al. Spermine is a natural suppressor of AR signaling in castration-resistant prostate cancer. Cell Rep. 2023;42:112798.
Akter KA, Mansour MA, Hyodo T, Ito S, Hamaguchi M, Senga T. FAM98A is a novel substrate of PRMT1 required for tumor cell migration, invasion, and colony formation. Tumour Biol. 2016;37:4531–9.
Musiani D, Giambruno R, Massignani E, Ippolito MR, Maniaci M, Jammula S, et al. PRMT1 is recruited via DNA-PK to chromatin where it sustains the senescence-associated secretory phenotype in response to cisplatin. Cell Rep. 2020;30:1208–1222.e9.
Matsubara H, Fukuda T, Awazu Y, Nanno S, Shimomura M, Inoue Y, et al. PRMT1 expression predicts sensitivity to platinum-based chemotherapy in patients with ovarian serous carcinoma. Oncol Lett. 2021;21:162.
Meng X, Zhao Y, Liu J, Wang L, Dong Z, Zhang T, et al. Comprehensive analysis of histone modification-associated genes on differential gene expression and prognosis in gastric cancer. Exp Ther Med. 2019;18:2219–30.
Wang F, Chen S, Peng S, Zhou X, Tang H, Liang H, et al. PRMT1 promotes the proliferation and metastasis of gastric cancer cells by recruiting MLXIP for the transcriptional activation of the β-catenin pathway. Genes Diseases. 2023;10:2622–38.
Zhang Y, Wang D, Zhang M, Wei H, Lu Y, Sun Y, et al. Protein arginine methyltransferase 1 coordinates the epithelial-mesenchymal transition/proliferation dichotomy in gastric cancer cells. Exp Cell Res. 2018;362:43–50.
Kim E, Rahmawati L, Aziz N, Kim HG, Kim JH, Kim KH, et al. Protection of c-Fos from autophagic degradation by PRMT1-mediated methylation fosters gastric tumorigenesis. Int J Biol Sci. 2023;19:3640–60.
Dong F, Li Q, Yang C, Huo D, Wang X, Ai C, et al. PRMT2 links histone H3R8 asymmetric dimethylation to oncogenic activation and tumorigenesis of glioblastoma. Nat Commun. 2018;9:4552.
Banasavadi-Siddegowda YK, Russell L, Frair E, Karkhanis VA, Relation T, Yoo JY, et al. PRMT5-PTEN molecular pathway regulates senescence and self-renewal of primary glioblastoma neurosphere cells. Oncogene. 2017;36:263–74.
Huang T, Yang Y, Song X, Wan X, Wu B, Sastry N, et al. PRMT6 methylation of RCC1 regulates mitosis, tumorigenicity, and radiation response of glioblastoma stem cells. Mol Cell. 2021;81:1276–1291.e9.
Wang J, Xiao Z, Li P, Wu C, Li Y, Wang Q, et al. PRMT6-CDC20 facilitates glioblastoma progression via the degradation of CDKN1B. Oncogene. 2023;42:1088–100.
Wang S, Tan X, Yang B, Yin B, Yuan J, Qiang B, et al. The role of protein arginine-methyltransferase 1 in gliomagenesis. BMB Rep. 2012;45:470–5.
Lathoria K, Gowda P, Umdor SB, Patrick S, Suri V, Sen E. PRMT1 driven PTX3 regulates ferritinophagy in glioma. Autophagy. 2023;19:1997–2014.
Zhou L, Wu J, Zhang M, Zhao B, Ma S. PRMT1 inhibits apoptosis of nasopharyngeal carcinoma cells by promoting RRM2 expression. Nan Fang Yi Ke Da Xue Xue Bao. 2022;42:1783–90.
Wang J, Wang C, Xu P, Li X, Lu Y, Jin D, et al. PRMT1 is a novel molecular therapeutic target for clear cell renal cell carcinoma. Theranostics. 2021;11:5387–403.
Zhao J, Adams A, Roberts B, O’Neil M, Vittal A, Schmitt T, et al. Protein arginine methyl transferase 1- and Jumonji C domain-containing protein 6-dependent arginine methylation regulate hepatocyte nuclear factor 4 alpha expression and hepatocyte proliferation in mice. Hepatol. 2018;67:1109–26.
Zhao J, O’Neil M, Schonfeld M, Komatz A, Weinman SA, Tikhanovich I. Hepatocellular protein arginine methyltransferase 1 suppresses alcohol-induced hepatocellular carcinoma formation by inhibition of inducible nitric oxide synthase. Hepatol Commun. 2020;4:790–808.
Zhang L, He Y, Jiang Y, Wu Q, Liu Y, Xie Q, et al. PRMT1 reverts the immune escape of necroptotic colon cancer through RIP3 methylation. Cell Death Dis. 2023;14:233.
Jin S, Su H, Tran NT, Song J, Lu SS, Li Y, et al. Splicing factor SF3B1K700E mutant dysregulates erythroid differentiation via aberrant alternative splicing of transcription factor TAL1. PLoS One. 2017;12:e0175523.
Wang YP, Sharda A, Xu SN, van Gastel N, Man CH, Choi U, et al. Malic enzyme 2 connects the Krebs cycle intermediate fumarate to mitochondrial biogenesis. Cell Metab. 2021;33:1027–1041.e8.
Yuniati L, van der Meer LT, Tijchon E, van Ingen SD, van Emst L, Levers M, et al. Tumor suppressor BTG1 promotes PRMT1-mediated ATF4 function in response to cellular stress. Oncotarget. 2016;7:3128–43.
Li M, An W, Xu L, Lin Y, Su L, Liu X. The arginine methyltransferase PRMT5 and PRMT1 distinctly regulate the degradation of anti-apoptotic protein CFLARL in human lung cancer cells. J Exp Clin Cancer Res. 2019;38:64.
Repenning A, Happel D, Bouchard C, Meixner M, Verel-Yilmaz Y, Raifer H, et al. PRMT1 promotes the tumor suppressor function of p14ARF and is indicative for pancreatic cancer prognosis. EMBO J. 2021;40:e106777.
Altan B, Yokobori T, Ide M, Mochiki E, Toyomasu Y, Kogure N, et al. Nuclear PRMT1 expression is associated with poor prognosis and chemosensitivity in gastric cancer patients. Gastric Cancer. 2016;19:789–97.
Cheng D, Yadav N, King RW, Swanson MS, Weinstein EJ, Bedford MT. Small molecule regulators of protein arginine methyltransferases *. J Biol Chem. 2004;279:23892–9.
Chen F, Fulton DJR. An inhibitor of protein arginine methyltransferases, 7,7’-carbonylbis (azanediyl)bis(4-hydroxynaphthalene-2-sulfonic acid (AMI-1), is a potent scavenger of NADPH-oxidase-derived superoxide. Mol Pharmacol. 2010;77:280–7.
Spannhoff A, Heinke R, Bauer I, Trojer P, Metzger E, Gust R, et al. Target-based approach to inhibitors of histone arginine methyltransferases. J Med Chem. 2007;50:2319–25.
Feng Y, Xie N, Wu J, Yang C, Zheng YG. Inhibitory study of protein arginine methyltransferase 1 using a fluorescent approach. Biochem Biophys Res Commun. 2009;379:567–72.
Castellano S, Milite C, Ragno R, Simeoni S, Mai A, Limongelli V, et al. Design, synthesis and biological evaluation of Carboxy analogues of arginine methyltransferase inhibitor 1 (AMI-1). Chem Med Chem. 2010;5:398–414.
Kim E, Jang J, Park JG, Kim KH, Yoon K, Yoo BC, et al. Protein arginine methyltransferase 1 (PRMT1) selective inhibitor, TC-E 5003, has anti-inflammatory properties in TLR4 signaling. Int J Mol Sci. 2020;21:3058.
Zhang P, Tao H, Yu L, Zhou L, Zhu C. Developing protein arginine methyltransferase 1 (PRMT1) inhibitor TC-E-5003 as an antitumor drug using INEI drug delivery systems. Drug Deliv. 2020;27:491–501.
Heinke R, Spannhoff A, Meier R, Trojer P, Bauer I, Jung M, et al. Virtual screening and biological characterization of novel histone arginine methyltransferase PRMT1 inhibitors. Chem Med Chem. 2009;4:69–77.
Cheng C, Pei X, Li SW, Yang J, Li C, Tang J, et al. CRISPR/Cas9 library screening uncovered methylated PKP2 as a critical driver of lung cancer radioresistance by stabilizing β-catenin. Oncogene. 2021;40:2842–57.
Zhao Z, Zhang J, Ren Y, Dong L, Wu H, Hong W, et al. Discovery of 2,4-diphenyl-substituted thiazole derivatives as PRMT1 inhibitors and investigation of their anti-cervical cancer effects. Bioorg Med Chem. 2023;92:117436.
Eram MS, Shen Y, Szewczyk M, Wu H, Senisterra G, Li F, et al. A potent, selective, and cell-active inhibitor of human type I protein arginine methyltransferases. ACS Chem Biol. 2016;11:772–81.
Dominici C, Sgarioto N, Yu Z, Sesma-Sanz L, Masson JY, Richard S, et al. Synergistic effects of type I PRMT and PARP inhibitors against non-small cell lung cancer cells. Clin Epigenetics. 2021;13:54.
Nguyen HP, Le AQ, Liu E, Cesarano A, DiMeo F, Perna F, et al. Protein arginine methyltransferase 1 is a therapeutic vulnerability in multiple myeloma. Front Immunol. 2023;14:1239614.
Fedoriw A, Rajapurkar SR, O’Brien S, Gerhart SV, Mitchell LH, Adams ND, et al. Anti-tumor activity of the type I PRMT inhibitor, GSK3368715, synergizes with PRMT5 inhibition through MTAP loss. Cancer Cell. 2019;36:100–114.e25.
Zhou L, Jia X, Shang Y, Sun Y, Liu Z, Liu J, et al. PRMT1 inhibition promotes ferroptosis sensitivity via ACSL1 upregulation in acute myeloid leukemia. Mol Carcinog. 2023;62:1119–35.
Fedoriw A, Shi L, O’Brien S, Smitheman KN, Wang Y, Hou J, et al. Inhibiting type I arginine methyltransferase activity promotes T cell-mediated antitumor immune responses. Cancer Immunol Res. 2022;10:420–36.
Liu J, Bu X, Chu C, Dai X, Asara JM, Sicinski P, et al. PRMT1 mediated methylation of cGAS suppresses anti-tumor immunity. Nat Commun. 2023;14:2806.
El-Khoueiry AB, Clarke J, Neff T, Crossman T, Ratia N, Rathi C, et al. Phase 1 study of GSK3368715, a type I PRMT inhibitor, in patients with advanced solid tumors. Br J Cancer. 2023;129:309–17.
Iyamu ID, Al-Hamashi AA, Huang R. A Pan-inhibitor for protein arginine methyltransferase family enzymes. Biomolecules. 2021;11:854.
Dai X, Ren T, Zhang Y, Nan N. Methylation multiplicity and its clinical values in cancer. Expert Rev Mol Med. 2021;23:e2.
Krysko O, Aaes TL, Kagan VE, D'Herde K, Bachert C, Leybaert L, et al. Necroptotic cell death in anti-cancer therapy. Immunol Rev. 2017;280:207–19.
Ryu TY, Kim K, Son MY, Min JK, Kim J, Han TS, et al. Downregulation of PRMT1, a histone arginine methyltransferase, by sodium propionate induces cell apoptosis in colon cancer. Oncol Rep. 2019;41:1691–9.
Fu Y, He X, Gao XD, Li F, Ge S, Yang Z, et al. Prime editing: current advances and therapeutic opportunities in human diseases. Sci Bull (Beijing). 2023;68:3278–91.
Chen S, Cui J, Chen H, Yu B, Long S. Recent progress in degradation of membrane proteins by PROTACs and alternative targeted protein degradation techniques. Eur J Med Chem. 2023;262:115911.
Uscanga-Palomeque AC, Chávez-Escamilla AK, Alvizo-Báez CA, Saavedra-Alonso S, Terrazas-Armendáriz LD, Tamez-Guerra RS, et al. CAR-T cell therapy: from the shop to Cancer therapy. Int J Mol Sci. 2023;24:15688.
Kim JH, Yoo BC, Yang WS, Kim E, Hong S, Cho JY. The Role of Protein Arginine Methyltransferases in Inflammatory Responses. Mediators Inflamm. 2016;2016:4028353.
Zheng S, Zeng C, Huang A, Huang F, Meng A, Wu Z, et al. Relationship between protein arginine methyltransferase and cardiovascular disease (Review). Biomed Rep. 2022;17:90.
Zhao X, Chong Z, Chen Y, Zheng XL, Wang QF, Li Y. Protein arginine methyltransferase 1 in the generation of immune megakaryocytes: A perspective review. J Biol Chem. 2022;298:102517.
Acknowledgements
The illustrations in this study were created with BioRender.com (https://biorender.com), for which we would like to express our gratitude.
Funding
This work was supported by the National Natural Science Foundation of China (82303851), Natural Science Foundation of Jiangxi Province (20202BABL216025), Guangdong Basic and Applied Basic Research Foundation (2022A1515111065), and the China Postdoctoral Science Foundation (2023M740840).
Author information
Authors and Affiliations
Contributions
S.Q.S, H.L.Z, Z.Y.X, and S.F.Z collected relevant literature. Y.H.T, D.M.C, and X.P prepared figures and tables. S.Q.S wrote the first draft of this manuscript. Y.Z.W and J.W conceptualized the project and revised the article. All authors examined and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Shen, S., Zhou, H., Xiao, Z. et al. PRMT1 in human neoplasm: cancer biology and potential therapeutic target. Cell Commun Signal 22, 102 (2024). https://doi.org/10.1186/s12964-024-01506-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12964-024-01506-z