Abstract
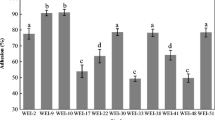
The gut is a source of lactic acid bacteria with remarkable functional and technologies properties as well as a potential source of probiotics. In the present study, 13 strains of Lactobacillus were isolated from poultry intestine and identified according to their 16S rDNA sequences, as well as the evaluation of their probiotic potential. The probiotic properties were tested in aspects of antibiotic susceptibility, antimicrobial activity, exopolysaccharide production, lysozyme tolerance, gut condition tolerance (low pH, bile salt tolerance) and adhesion to human colorectal adenocarcinoma cell line (Caco-2). Most isolates were resistance to streptomycin (10 μg/mL), gentamicin (10 μg/mL), kanamycin (30 μg/mL), penicillin (10 μg/mL) and chloramphenicol (30 μg/mL). Isolates shows strong abilities to adhere to Caco-2 cell in the range of (76 to 85%). Isolates SHA101 to SHA113 showed high survival rate under gastrointestinal tract condition (>80%), indicating their potential in application of probiotics. The results of these tests indicate that the lactic acid bacteria isolated from poultry intestine have potential use as probiotic in various products.
Similar content being viewed by others
References
Ahmed, F.E., Genetically modified probiotics in foods, Trends Biotechnol., 2003, vol. 21, no. 11, pp. 491–497. doi 10.1016/j.tibtech.2003.09.006
Bian, L., Molan, A.-L., Maddox, I., and Shu, Q., Antimicrobial activity of Lactobacillus reuteri DPC16 supernatants against selected food borne pathogens, World J. Microbiol. Biotechnol., 2010, vol. 27, no. 4, pp. 991–998. doi 10.1007/s11274-010-0543-z
Blajman, J., Gaziano, C., Zbrun, M.V., Soto, L., Astesana, D., Berisvil, A., Scharpen, A.R., Signorini, M., and Frizzo, L., In vitro and in vivo screening of native lactic acid bacteria toward their selection as a probiotic in broiler chickens, Res. Vet. Sci., 2015, vol. 101, pp. 50–56. doi 10.1016/j.rvsc.2015.05.017
Caggia, C., De Angelis, M., Pitino, I., Pino, A., and Randazzo, C., Probiotic features of Lactobacillus strains isolated from Ragusano and Pecorino Siciliano cheeses, Food Microbiol., 2015, vol. 50, pp. 109–117.
Chang, L., Zhang, Z.-Y., Ke, D., Jian-Ping, Y., and Xiao-Kui, G., Antibiotic resistance of probiotic strains of lactic acid bacteria isolated from marketed foods and drugs, Biomed. Environ. Sci., 2009, vol. 22, no. 5, pp. 401–412.
CLSI, Performance Standards for Antimicrobial Susceptibility Testing: Twenty-Forth Informational Supplement 34: M100-S124, 2014.
Collado, M.C., Gueimonde, M., Sanz, Y., and Salminen, S., Adhesion of selected Bifidobacterium strains to human intestinal mucus and the role of adhesion in enteropathogen exclusion, J. Food Prot., 2005, vol. 68, no. 12, pp. 2672–2678.
Collado, M.C., Jalonen, L., Meriluoto, J., and Salminen, S., Protection mechanism of probiotic combination against human pathogens: in vitro adhesion to human intestinal mucus, Asia Pacific J. Clin. Nutr., 2006, vol. 15, no. 4, pp. 570–575.
De Palencia, P.F., López, P., Corbí, A.L., Peláez, C., and Requena, T., Probiotic strains: survival under simulated gastrointestinal conditions, in vitro adhesion to Caco-2 cells and effect on cytokine secretion, Eur. Food Res. Technol., 2008, vol. 227, no. 5, pp. 1475–1484.
Degeest, B., Janssens, B., and De Vuyst, L., Exopolysaccharide (EPS) biosynthesis by Lactobacillus sakei 0–1: production kinetics, enzyme activities and EPS yields, J. Appl. Microbiol., 2001, vol. 91, pp. 470–477.
Dias, R., Vilas-Boas, E., Campos, F.M., Hogg, T., and Couto, J.A., Activity of lysozyme on Lactobacillus hilgardii strains isolated from Port wine, Food Microbiol., 2015, vol. 49, pp. 6–11.
Domingos-Lopes, M.F., Stanton, C., Ross, P.R., Dapkevicius, M.L., and Silva, C.C., Genetic diversity, safety and technological characterization of lactic acid bacteria isolated from artisanal Pico cheese, Food Microbiol., 2017, vol. 63, pp. 178–190. doi 10.1016/j.fm.2016.11.014
Drago, L., Rodighiero, V., Mattina, R., Toscano, M., and De Vecchi, E., In vitro selection of antibiotic resistance in the probiotic strain Lactobacillus rhamnosus GG ATCC 53103, J. Chemother., 2013, vol. 4, pp. 211–215.
FAO/WHO, Evaluation of health and nutritional properties of probiotics in food including powder milk with live lactic acid bacteria. Expert consultation report. Food and Agriculture Organization of the United Nations and World Health Organization, Córdoba, Argentina (1–4 October 2001).
Fontana, C., Cocconcelli, P.S., Vignolo, G., and Saavedra, L., Occurrence of antilisterial structural bacteriocins genes in meat borne lactic acid bacteria, Food Control, 2015, vol. 47, pp. 53–59.
Garcia-Hernandez, Y., Perez-Sanchez, T., Boucourt, R., Balcazar, J.L., Nicoli, J.R., Moreira-Silva, J., Rodriguez, Z., Fuertes, H., Nunez, O., Albelo, N., and Halaihel, N. Isolation, characterization and evaluation of probiotic lactic acid bacteria for potential use in animal production, Res. Vet. Sci., 2016, vol. 108, pp. 125–132. doi 10.1016/j.rvsc.2016.08.009
Handa, S. and Sharma, N., In vitro probiotic potential of Lactobacillus delbreuckii subsp. bulgaricus F18 isolated from homemade butter, Microbiology (Moscow), 2017, vol. 86, no. 1, pp. 143–149. doi 10.1134/s0026261716060114
Kaewiad, K., Kaewnopparat, S., and Kaewnopparat, N., In vitro comparison of probiotic properties of Lactobacillus fermentum SK54 isolated from new born baby with Lactobacillus rhamnosus GG ATCC 53103, Adv. Mat. Res., 2015, vol. 1060, pp. 215–218.
Kandola, S., Kumar, R., Mishra, A.K., and Singh, A., Investigation of acid tolerance attribute of various Lactobacillus casei group strains, Ind. J. Animal Res., 2016, vol. 50, no. 2, pp. 190–193.
Lavilla-Lerma, L., Pérez-Pulido, R., Martínez-Bueno, M., Maqueda, M., and Valdivia, E., Characterization of functional, safety, and gut survival related characteristics of Lactobacillus strains isolated from farmhouse goat’s milk cheeses, Int. J. Food Microbiol., 2013, vol. 163, no. 2, pp. 136–145.
Lee, Y.-K., Puong, K.-Y., Ouwehand, A.C., and Salminen, S., Displacement of bacterial pathogens from mucus and Caco-2 cell surface by lactobacilli, J. Med. Microbiol., 2003, vol. 52, no. 10, pp. 925–930.
Liu, C.F., Tseng, K.C., Chiang, S.S., Lee, B.H., Hsu, W.H., and Pan, T.M., Immunomodulatory and antioxidant potential of Lactobacillus exopolysaccharides, J. Sci. Food Agricul., 2011, vol. 91, no. 12, pp. 2284–2291.
Mandal, H., Jariwala, R., and Bagchi, T., Isolation and characterization of lactobacilli from human faeces and indigenous fermented foods for their potential application as probiotics, Canad. J. Microbiol., 2015, vol. 62, no. 4, pp. 349–359.
Manini, F., Casiraghi, M., Poutanen, K., Brasca, M., Erba, D., and Plumed-Ferrer, C., Characterization of lactic acid bacteria isolated from wheat bran sourdough, LWTFood Sci. Technol., 2016, vol. 66, pp. 275–283.
Minervini, F., De Angelis, M., Surico, R., Di Cagno, R., Gänzle, M., and Gobbetti, M., Highly efficient synthesis of exopolysaccharides by Lactobacillus curvatus DPPMA10 during growth in hydrolyzed wheat flour agar, Int. J. Food Microbiol., 2010, vol. 141, no. 1, pp. 130–135.
Oh, Y.J., and Jung, D.S., Evaluation of probiotic properties of Lactobacillus and Pediococcus strains isolated from Omegisool, a traditionally fermented millet alcoholic beverage in Korea, LWT-Food Sci. Technol., 2015, vol. 63, no. 1, pp. 437–444.
Panel, E.F., Guidance on the assessment of bacterial susceptibility to antimicrobials of human and veterinary importance, EFSA J., 2012, vol. 10, no. 6, p. 2740.
Pingitore, E.V., Pessione, A., Fontana, C., Mazzoli, R., and Pessione, E., Comparative proteomic analyses for elucidating metabolic changes during EPS production under different fermentation temperatures by Lactobacillus plantarum Q823, Int. J. Food Microbiol., 2016, vol. 238, pp. 96–102.
Rendueles, O., Kaplan, J.B., and Ghigo, J.M., Antibiofilm polysaccharides, Environ. Microbiol., 2013, vol. 15, no. 2, pp. 334–346.
Riaz Rajoka, M.S., Shi, J., Zhu, J., Shao, D., Huang, Q., Yang, H., and Jin, M., Capacity of lactic acid bacteria in immunity enhancement and cancer prevention, Appl. Microbiol. Biotechnol., 2017, vol. 101, no. 1, pp. 35–45. doi 10.1007/s00253-016-8005-7
Russo, P., López, P., Capozzi, V., De Palencia, P.F., Dueñas, M.T., Spano, G., and Fiocco, D., Beta-glucans improve growth, viability and colonization of probiotic microorganisms, Int. J. Mol. Sci., 2012, vol. 13, no. 5, pp. 6026–6039.
Saitou, N. and Nei, M., The neighbor-joining method: a new method for reconstructing phylogenetic trees, Mol. Biol. Evol., 1987, vol. 4, no. 4, pp. 406–425.
Salminen, S., von Wright, A., Morelli, L., Marteau, P., Brassart, D., de Vos, W.M., Fondén, R., Saxelin, M., Collins, K., and Mogensen, G., Demonstration of safety of probiotics—a review, Int. J. Food Microbiol., 1998, vol. 44, no. 1, pp. 93–106.
Schiffrin, E., Rochat, F., Link-Amster, H., Aeschlimann, J., and Donnet-Hughes, A., Immunomodulation of human blood cells following the ingestion of lactic acid bacteria, J. Dairy Sci., 1995, vol. 78, no. 3, pp. 491–497.
Shahid Riaz, M., Shaheen, T., Batool, N., Saleem, S., and Hayat, F., Lactic acid bacteria as probiotic candidate and their application, J. Biol. Today’s World, 2015, vol. 4, no. 12, pp. 209–216.
Solieri, L., Bianchi, A., Mottolese, G., Lemmetti, F., and Giudici, P., Tailoring the probiotic potential of non-starter Lactobacillus strains from ripened Parmigiano Reggiano cheese by in vitro screening and principal component analysis, Food Microbiol., 2014, vol. 38, pp. 240–249.
Tamura, K., Nei, M., and Kumar, S., Prospects for inferring very large phylogenies by using the neighbor-joining method, Proc. Natl. Acad. Sci. U. S. A., 2004, vol. 101, no. 30, pp. 11030–11035.
Tavakoli, M., Hamidi-Esfahani, Z., Hejazi, M.A., Azizi, M.H., and Abbasi, S., Characterization of probiotic abilities of lactobacilli isolated from Iranian koozeh traditional cheese, Polish J. Food Nutr. Sci., 2017, vol. 67, no. 1. doi 10.1515/pjfns-2016-0003
Tejero-Sariñena, S., Barlow, J., Costabile, A., Gibson, G.R., and Rowland, I., In vitro evaluation of the antimicrobial activity of a range of probiotics against pathogens: evidence for the effects of organic acids, Anaerobe, 2012, vol. 18, no. 5, pp. 530–538.
Todorov, S.D., Leblanc, J.G., and Franco, B.D., Evaluation of the probiotic potential and effect of encapsulation on survival for Lactobacillus plantarum ST16Pa isolated from papaya, World J. Microbiol. Biotechnol., 2012, vol. 28, no. 3, pp. 973–984. doi 10.1007/s11274-011-0895-z
Turchi, B., Mancini, S., Fratini, F., Pedonese, F., Nuvoloni, R., Bertelloni, F., Ebani, V.V., and Cerri, D., Preliminary evaluation of probiotic potential of Lactobacillus plantarum strains isolated from Italian food products, World J. Microbiol. Biotechnol., 2013, vol. 29, no. 10, pp. 1913–1922.
Verdenelli, M.C., Ghelfi, F., Silvi, S., Orpianesi, C., Cecchini, C., and Cresci, A., Probiotic properties of Lactobacillus rhamnosus and Lactobacillus paracasei isolated from human faeces, Eur. J. Nutr., 2009, vol. 48, no. 6, pp. 355–363.
Vesterlund, S., Paltta, J., Karp, M., and Ouwehand, A.C., Measurement of bacterial adhesion—in vitro evaluation of different methods, J. Microbiol. Methods, 2005, vol. 60, no. 2, pp. 225–233.
Yu, Z., Zhang, X., Li, S., Li, C., Li, D., and Yang, Z., Evaluation of probiotic properties of Lactobacillus plantarum strains isolated from Chinese sauerkraut, World J. Microbiol. Biotechnol., 2013, vol. 29, no. 3, pp. 489–498. doi 10.1007/s11274-012-1202-3
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is published in the original.
Rights and permissions
About this article
Cite this article
Rajoka, M.S.R., Hayat, H.F., Sarwar, S. et al. Isolation and evaluation of probiotic potential of lactic acid bacteria isolated from poultry intestine. Microbiology 87, 116–126 (2018). https://doi.org/10.1134/S0026261718010150
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0026261718010150