Abstract
Drought is one of the major threats to food security. Among several mechanisms involved in plant stress tolerance, one protein family—the plant metallothioneins (MTs)—shows great promise for enhancing drought resistance. Plant metallothioneins in oat (Avena sativa L.) have not yet been deeply analysed, and the literature lacks a comprehensive study of the whole family of plant MTs in response to drought. In this study, we showed that the number and nature of cis-elements linked with stress response in promoters of AsMTs1–3 differed depending on the MT type. Drought stress in oat plants caused an increase in the expression of AsMT2 and AsMT3 and a decrease in the expression of AsMT1 compared to well-watered plants. Moreover, the low values of relative water content, water use efficiency, net photosynthesis (PN), transpiration (E), stomatal conductance (gs), chlorophyll a, and carotenoid were accompanied by high levels of electrolyte leakage, internal CO2 concentration (Ci) and abscisic acid content, and high activity of antioxidants enzymes in plants under drought stress. The present study puts forward the idea that AsMTs are crucial for oat response to drought stress not only by regulating antioxidant activity but also by changing the plant water regime and photosynthesis. Our results support the hypothesis that structural differences among types of plant MTs reflect their diversified physiological roles.
Similar content being viewed by others
Introduction
Anthropogenic activities have raised the level of CO2 and other greenhouse gasses in the atmosphere by 50% since the eighteenth century. As a result, Earth’s temperature is rising and rainfall patterns are changing1. Studies show that the Earth’s surface temperature is expected to exceed the limit of 2 °C above pre-industrial levels (1850–1900) by the end of the twenty-first century2. As Earth warms, incidents of drought will be longer and more severe3.
Water deficit is one of the crucial factors limiting plant productivity and thus threatening food security. Agricultural or ecological drought occurs when water demand exceeds supply1. When subjected to drought stress, plants adopt one or more of the four main survival strategies, i.e. (1) drought avoidance, (2) drought escape, (3) drought resistance, and (4) drought recovery. To avoid drought, plants reduce water loss by partial stomatal closure, increased leaf wax accumulation, and leaf rolling4. Moreover, a well-developed root system enhances water uptake ability. Drought escape is the natural adjustment of the growth period and life cycle of a plant or artificial changes in planting time by farmers in order to decrease the possible harmful effects of drought. Drought resistance and recovery are the ability of plants to sustain a certain level of physiological activities under drought-stress conditions and then resume growth in non-drought conditions4.
In response to drought, several mechanisms are activated in plant cells. To maintain cell turgor pressure, plants produce osmolytes such as proline, soluble sugars, spermine, and betaine5,6,7,8. In drought conditions, the production of abscisic acid (ABA) is induced. ABA is a stress-response phytohormone and functions as a crucial signal molecule in plant response to drought9. ABA triggers a range of physiological processes including induction of stomatal closure, modulation of root development, and inhibition of plant growth10,11. Several lines of evidence have shown that drought-responsive genes can be classified into two groups: ABA-dependent and ABA-independent12,13. Uno et al.12 showed that many ABA-dependent drought-related genes possess an ABA-responsive cis-element (ABRE) in the promoter region. On the other hand, genes induced by drought that are not induced by ABA possess other cis-elements including drought-responsive elements (DRE) and C-repeat (CTR)14.
Drought stress leads to increased production of reactive oxygen species (ROS). This causes oxidative damage to lipids, proteins, and DNA, which can lead to cell death15. Plants possess enzymatic and non-enzymatic antioxidant systems. The enzymatic antioxidant system consists of superoxide dismutase (SOD), which catalyses the dismutation of superoxide anion radical (O2·−) to hydrogen peroxide (H2O2) and oxygen (O2). Hydrogen peroxide is converted to water and oxygen by ascorbate peroxidase (PX) in the presence of a reducing agent such as ascorbic acid or by catalase (CAT)16. The non-enzymatic antioxidants include various reducing compounds, such as tocopherols, glutathione, flavonoids, carotenoids, and ascorbic acid. Moreover, under drought, plants accumulate phenolic compounds, which can function as sources of electrons and protons for reactive oxygen species17,18,19.
Metallothioneins (MTs) are a family of small cysteine-rich proteins present in eukaryotes20 and some prokaryotes21. In plants, MTs (pMTs) are divided into four types depending on the amino-acid sequence, i.e. pMTs belonging to type 1 (MT1) have 12 cysteine residues, type-2 MTs (MT2) contain 14 cysteines, type-3 MTs (MT3) have ten cysteines, and type-4 pMTs (MT4) contain 17 cysteines22,23. MTs have been shown to bind a variety of heavy metal ions (in particular Cu+, Zn2+, and Cd2+) via thiol groups of cysteine residues24,25,26,27,28,29,30. Some plant MTs have one or more histidine residues, which can also play a role in binding metal ions31,32. The expression of pMT genes is spatiotemporal and induced by various stimuli, including drought, which suggests that pMTs have a role that goes beyond the maintenance of micronutrient homeostasis and toxic metal detoxication33,34. Several lines of evidence have shown that thiol groups of pMTs are powerful antioxidants and can protect plants from oxidative stress33. Moreover, MTs can, by binding Cu+ ions, stop the Fenton reaction35,36,37,38.
One of the most cultivated cereals worldwide is oat (Avena sativa L.)39. This plant is mostly used as livestock feed, but every year it is increasing in popularity as human food. Oat has many nutritional benefits due to its high levels of calcium, soluble fibre, oil, and protein40,41,42. Oat is also a popular and proven-to-work ingredient in various skincare cosmetics. There are some clear indicators that pMTs play a role in drought tolerance, i.e. the increased expression of pMTs in water-limiting conditions has been observed for various plant species43,44,45,46, and the expression of several pMTs is regulated by ABA47,48,49. We propound the hypothesis that certain pMTs might play essential roles in plant drought resistance. The literature information concerning pMTs and drought is rather scarce and usually limited to only one type of pMT. Therefore, we aimed to analyse and compare the possible roles that MTs of types 1–3 play in response to drought in single plant species. We chose economically important oat since the growth and yield of this crop plant are significantly limited by drought stress15. Moreover, we examined the physiological and biochemical parameters reflecting the water regime, photosynthesis efficiency, and antioxidant activity of oat plants subjected to drought stress. The knowledge generated in this study that allows us to gain deeper insight into the mechanisms of oat response to drought stress may enable us to obtain oat varieties more tolerant to drought stress and to reduce yield losses.
Results and discussion
A. sativa is a crop of increasing interest as it is well-adapted to a wide range of soil types. It can perform better than other small-grain cereals on marginal soils. However, oat is sensitive to hot, dry weather, and hence, in several regions of the world, drought is the main factor limiting the yield of oat39. To succeed in breeding programmes, the selection of plants with complex traits such as drought resistance should be based upon a comprehensive understanding of innate tolerance mechanisms50. In the face of global warming and a growing world population, an understanding of the cellular mechanisms underlying drought tolerance seems to be crucial for food security.
Since their discovery in wheat germs in 198751, plant metallothioneins have been linked with various physiological roles including micronutrient homeostasis52, toxic metal detoxication53, reactive oxygen species scavenging54, senescence55, and stress response56. Plant MTs have been investigated in various plants such as Arabidopsis thaliana (L.) Heynh.57,58,59,60, Nicotiana tabacum L.61, Ipomoea nil (L.) Roth22, Brassica napus L.24,38,62, Cucumis sativus L.63, Oryza sativa L.64, and Zea mays L.65. However, only one report on A. sativa metallothioneins has been published so far66. This may be because oat is an allohexaploid species (2n = 6× = 42, AACCDD) with a large genome (12.5 Gb), which makes it difficult to work with on the gene level67. Beginning in 2016, attempts were made to sequence the oat genome68, and in March 2022 the oat genome was published69. This will significantly accelerate the research on oat.
In silico analyses
Plant metallothioneins have been divided into four types based on the number and arrangement of cysteine residues. In all those angiosperm genomes analysed to date, all four types of pMTs are present. There is no clear picture of the possible physiological roles of each type of pMT. It is possible that there is no single unifying role of plant metallothioneins and that one type fulfils different functions depending on the stage of plant development, plant organ, and environmental conditions70. Type 4 pMTs are the best-known type of pMTs. This type of pMTs was excluded from this study because pMT4s are seed-specific proteins and in most analysed up-to-date species the expression of this type of pMTs is restricted to developing and mature seeds.
The putative amino acid sequences of oat metallothioneins analysed in this study have a high cysteine content. The number and arrangement of cysteines allow oat MTs to be classified into three types—AsMT1, AsMT2, and AsMT3 (Table 1). The predicted proteins were 64 (AsMT3) to 79 (AsMT2) amino acids long, and the molecular masses ranged from 6814.56 to 7594.64 kDa. The isoelectric point (pI) of AsMT1–3 was similar for all three proteins, and it ranged from 4.85 to 5.10 pI (Table 1). AsMT1 shares the highest homology with MT1 from Festuca rubra (87.93%, O24528.1) and MT1 from Hordeum vulgare (78.67%, CAD54078.1), AsMT2 is most similar to MT2a from Lolium rigidum (84.51%, XP_047054761.1) and MT2 from Poa secunda (84.72%, AAK38824.1), and AsMT3 is most similar to MT3 from Oryza coarctata (73.44%, AAF68995.1) and MT3 from Carica papaya (70.77%, XP_021894753.1). Comparison of the oat MT sequences cloned by our group (Bingo cultivar) with the sequences deposited in the PanOat database revealed 100% identity for AsMT1, AsMT2 and AsMT3 (AVESA.00001b.r3.7Cg0001922, AVESA.00001b.r3.1Cg0000164 and AVESA.00001b.r1.3Ag0000786, respectively). Similarly to MTs from other plant species, AsMT1–3 had two Cys-rich domains separated by one Cys-free stretch. Two His residues were present in AsMT3 and one in AsMT1 (Fig. 1). His residues are involved in the binding of metal ions by MTs, which has been confirmed for bacterial metallothioneins71 and type 4 plant MTs32. As in AsMT3, it is common for type-3 pMTs to have one or more His residues: one His residue located at the C-terminus of the protein, and a second His residue located in the spacer region of MT. In AsMT1, His residue is located in the middle of the Cys-free region, which is rather uncommon for this type of pMT70. The potential involvement of histidines in metal binding has been suggested also for type 3 pMTs31.
Promoter analysis is a powerful tool that can provide an insight into the regulatory mechanisms of genes of interest. Moreover, studies on cis-regulatory elements (CREs) provide a foundation for future experiments54,72,73. Regulatory elements play a crucial role in plant responses to various stresses, including drought stress74. There are not many studies comparing promoters of different types of MTs in one plant species64,65,72. Our analyses of oat metallothionein promoters revealed the presence of several cis-acting elements involved in response to light, phytohormones, biotic and abiotic stress, and plant development (Fig. 2, Supplementary Table S1). CREs are not distributed equally among promoters of oat MTs. A similar observation was made for MT promoters of B. napus, N. tabacum, and Z. mays61,65,72. The highest number of CREs was found in AsMT2 (104) and the lowest in AsMT3 (92), whereas AsMT1 promoter contains 97 CREs (Supplementary Table S2). Elements involved in abscisic acid (ABA), jasmonic acid (MeJA), gibberellin, and auxin response were found, of which the first two were the most numerous. ABA-responsive cis-elements, also called ABRE, were present in all oat metallothionein promoters: the promoter of AsMT2 had seven ABRE elements, AsMT1 had four and AsMT3 had only one (Supplementary Table S1). In N. tabacum, ABRE elements were the most abundant regulatory motifs and were present in all 12 NtMT promoters61. The promoter of AsMT2 had the highest number of elements involved in light response (20), while AsMT1 and AsMT3 had six and four light-responsive elements, respectively (Supplementary Table S2). Cis-regulatory sequences related to the response to light have been identified in MT promoters in other plants, e.g. in MT2 promoter of L. esculentum75. AtMT1B and AtMT1C in A. thaliana, OsMT1F, OsMT2A, and OsMT2B in O. sativa72, EgMT3A and EgMT3B in Elaeis guineensis76, CgMT1 in Casuarina glauca77, and BrMT1 and BrMT2 in B. rapa78. The analysed promoters also contained several development-related elements, i.e. seven elements in AsMT2 and ten elements in AsMT3. Interestingly, AsMT1 had only two development-related CREs (Supplementary Table S1).
Stress responsive elements were numerous in promoters of oat MTs, i.e. AsMT1 (11.3%), AsMT2 (8.6%), and AsMT3 (6.5%) (Fig. 2). Crucially, drought-related CREs were the most common stress-responsive elements among promoters of AsMT1–3. AsMT1 had nine drought-responsive elements, AsMT2 had six and AsMT3 had two (Supplementary Table S1). A regulatory element associated with drought response has been identified in the promoter of the rice OsMT2b gene79. Interestingly, elements involved in response to other stresses, such as fungal elicitor response, wounding response, and anaerobic conditions response were not distributed evenly among oat metallothioneins. In the promoter of AsMT3, there were no elements associated with wounding response, however; only in the promoter of this gene CREs involved in the response to fungal elicitors and anaerobic conditions were present (Supplementary Table S1).
Experimental research confirming the functionality of in silico identified CREs in plant MT promoters is scarce. The function of MT promoters has usually been studied using transgenic A. thaliana plants, where the promoter was fused with β-glucuronidase (GUS). For example, in the study by Ren and Zhao79, a rice type 2 MT promoter was especially induced by wounding, ABA, gibberellin, cytokinin, PEG, cold, hot, NaCl and Zn treatment, and, in that promoter, respective CREs were found. In another, similar study, the promoter of another rice type 2 MT had ABA and metal-responsive CREs, and the application of ABA, Zn, and Cu caused an increase in GUS levels80. The promoter of rice type 1 metallothionein has been shown to be responsive to ABA, drought, dark, Zn, Cu, Pb, and Al, and respective CRE motifs have been found in the promoter sequence81. In an analysis of type 1 MT promoter from C. glauca, CREs involved in the response to metals and wounding were found. The researchers found that the promoter was indeed responsive to wounding, but did not find the responsiveness to metals that the promoter analysis suggested. In transgenic A. thaliana, levels of GUS did not increase significantly after Cu, Zn, and Cd treatment, whereas wounding and H2O2 treatments led to an increase in levels of the reporter gene activity82.
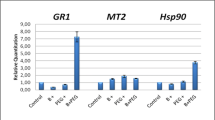
AsMT1–3 expression in response to drought stress
In a limited number of studies, the upregulation of pMTs in response to drought has been shown, e.g. during drought stress, a higher expression of type 2 MT in watermelon83 and a three-fold increase in the expression of MT3 in leaves of buckwheat (Fagopyrum esculentum Moench) were observed84. Here, the exposure of oat seedlings to drought stress caused significant changes in AsMT1–3 expression in oat shoots and roots. As mentioned before, in promoters of AsMTs, we found elements involved in ABA and drought response, but each AsMT promoter had a different number of those elements. In drought-stressed plants, the expression of AsMT1 in the shoots did not change, and the AsMT1 expression level in the roots was half that of control plants (Fig. 3A,B). The highest upregulation by drought was observed for AsMT2, i.e. it was 12-fold higher in the shoots and 27-fold higher in the roots in comparison to control plants (Fig. 3C,D). The expression of AsMT3 in the shoots of drought-stressed plants was 2.6 times lower (Fig. 3E), but in the roots of drought-stressed plants a 2.6-fold increase was detected (Fig. 3F). Interestingly, the total number of ABA-responsive and drought-responsive CREs in the promoter regions of AsMT1 and AsMT2 is the same (13), but AsMT2 had more ABA-responsive elements than drought-responsive elements, whereas the opposite is observed for AsMT1 (Supplementary Table S1). AsMT3 has the fewest drought-related and ABA-responsive cis-elements. These results indicate different roles of oat MT1–3 in drought response and suggest that the expression of oat MTs in drought-stressed plants is regulated via the ABA-dependent pathway. Previous studies have revealed that the expression levels of some MT genes, such as OsMT1a44, OsMT2b85, GhMT3a43, and BrMT178, are increased by ABA treatment, while the transcription levels of BrMT2 and BrMT3 were downregulated78.
Relative gene expression of AsMT1 (A, B), AsMT2 (C, D) and AsMT3 (E, F) in shoots and roots of oat seedlings in control and drought conditions. AsMT1-3 genes were quantified with RT-qPCR and normalised using housekeeping gene EIF4A. Bars represent mean values ± SE of three replicates. Student’s t-Test, p ≤ 0.05.
Contrary to our results, Jaiswal et al.86 showed that, under drought stress, the expression of genes of MT2 and MT3 from guar (Cyamopsis tetragonoloba L.) was unchanged in roots or shoots, and the MT1 gene was upregulated in both organs of the plant. Exposing Citrullus lanatus (Thunb.) Mansf. to drought stress resulted in increased expression of 32 genes, one of which was homologous to Lycopersicon esculentum L. MT283. Overexpression of metallothioneins confers drought stress tolerance in plants. For example, drought-stressed A. thaliana L. plants overexpressing type 1 metallothionein from chickpea had longer roots, higher biomass, and higher levels of enzymatic and non-enzymatic antioxidants in comparison to WT87. Similar results were obtained for A. thaliana L. plants overexpressing the MT2A gene from date palm88 and OsMT3-a from rice89. The regulation of MT genes expression in response to stress is multi-dimensional. Our results confirm the hypothesis that different types of MTs act differently and have different functions in plant cells. MTs exhibit a strong antioxidant property against oxidative damage via the neutralization of O2·− and enhanced H2O2 scavenging ability83,90,91. According to a study done by Li et al.90, overexpression of MT genes can significantly improve drought tolerance and is accompanied by elevated antioxidant enzyme activities, supporting the view that the MTs are involved in the ROS scavenging pathway.
Water status and photosynthetic efficiency of oat seedlings under soil drought
Water deficiency is an important factor affecting the growth and yield of plants subjected to drought. During the drought period, disturbances of many metabolic processes such as photosynthesis are observed92. The ability to retain stability of cell membrane under drought stress is one of the key physiological indices widely used to evaluate the drought tolerance of plants93. Measurements of relative water content (RWC), water loss (WL), electrolyte leakage (EL), and photosynthetic water use efficiency (WUE) are parameters frequently used as a selection test for the assessment of plant cultivar tolerance to various stresses94,95,96,97.
According to Hsiao96, the level of RWC drop corresponds to the severity of the water stress. In our study, drought caused a significant decrease in leaf RWC (Fig. 4A), i.e. from 85.3% in well-watered plants to 56.1% in drought-treated plants. In response to drought, a significant decrease in WL (Fig. 4B) and a significant increase in EL (Fig. 4C) were observed. Both parameters show the loss of cell membrane permeability and are changed under many stresses95. The photosynthetic water use efficiency (WUE), defined as the ratio of carbon assimilation to transpiration, was considerably lower in drought-stressed plants (1.4 µmol mmol−1) in comparison to well-watered plants (4.6 µmol mmol−1) (Fig. 4D). WUE is controlled by synchronising the relation between carbon assimilation and water intake, which is a significant strategy used by plants to survive drought. In the study by Liang et al.98, WUE and each of the gas exchange parameters of tomato leaves decreased in response to low levels of soil moisture. It is a common phenomenon that drought limits plant growth by reducing the photosynthetic rate. The key reasons for decreased photosynthesis are stomatal closure caused by decreased CO2 levels and reduced photosynthetic activity in the mesophyll98. At the beginning of drought stress, the stomata close first to reduce water transpiration, and as a result, the level of CO2 in the leaves decreases. When the decrease in net photosynthesis (PN) as a result of drought is accompanied by increased (or unchanged) internal CO2 concentration (Ci), non-stomatal factors are the main cause of reduced photosynthetic rate; meanwhile, when decreased PN is accompanied by decreased Ci, stomatal factors are the main cause.
Usually, under moderate and severe drought stress, the Ci gradually increases as the PN and stomatal conductance (gs) decrease. This indicates that non-stomatal restriction is the main factor of the decrease in the photosynthetic rate as the drought stress extends, which could lead to damage to the chloroplast structure99. In our study, drought significantly reduced the content of chlorophyll a and carotenoids in oat leaves (Fig. 5). Moreover, the net photosynthesis (PN), transpiration (E), and stomatal conductance (gs) drastically decrease in drought-treated plants (Fig. 6A–C). These changes accompany a substantial increase in internal CO2 concentration (Ci) (D) in oat leaves under drought (Fig. 6D). These observations suggest that nonstomatal restriction was accountable for reduced photosynthesis in oat leaves. As described by Zhao et al.100 and Zhang et al.99, PN, E, gs decreased significantly and were strictly associated with the degree and duration of drought stress in Avena nuda L. and A. sativa. A drought-prompted decrease in the photosynthetic activity of wheat leaves was also reported by Todorova et al.101. A large decrease in PN and gs has been observed in drought-stressed oat plants in comparison to control plants, while a lower decrease has been observed for E and WUE. Also, PN rate has been closely related to chlorophyll loss102 and all photosynthetic pigments, as well as the disruption or loss of thylakoid membranes103.
The activity of the antioxidant system of oat seedlings
Biochemical responses of crops associated with tolerance to drought are linked to changes in metabolic pathways, leading to the production of, e.g., sugars and phenolic compounds104. These metabolites mainly act as osmolytes, which reduce cellular dehydration and participate in the stabilization of enzymes and cellular membranes105. In our study, soil drought markedly increased the content of soluble sugars in roots and shoots of oat seedlings (Fig. 7A), whereas the amount of phenolic compounds was unaffected (Fig. 7B). As reported by Arabzadeh106, the accumulation of sugars by plants enhances water-holding capacity in cells and can thus reduce drought stress via regulation of the plant's osmotic potential.
An oxidative burst commonly occurs in response to various stress conditions107. The question of whether plant MTs are general stress proteins because of their potential to scavenge radicals108 or whether they are involved in response to some limited stress conditions is still open. Figure 8 shows the activities of antioxidant enzymes: catalase (CAT), superoxide dismutase (SOD), and peroxidase (PX) in shoots and roots of oat plants in response to drought. Compared to well-watered plants, CAT activity increases significantly after drought treatment in oat roots and shoots (Fig. 8A), while SOD activity decreases (Fig. 8B). PX activity was significantly higher in shoots and lower in roots of drought-treated plants in comparison to control plants (Fig. 8C). In our previous study66, in oat seedlings subjected to osmotic stress, CAT and PX had the highest activity in the treated plants and SOD had the lowest. A similar observation was reported by Chakraborty and Pradhan109 in Triticum aestivum L., where SOD activity showed a general decline in activity and the activity of PX increased greatly during water deficit. This is in line with Gratão et al.110, who hypothesised that SOD acts as the first line of defence against. H2O2 produced by SOD is then metabolized by the next enzyme, CAT. In our study we also observed a slight, but insignificant, increase in phenolic compounds in both roots and shoots of drought-treated plants (Fig. 7B). Soluble sugars and phenolics eliminate H2O2, and thus reduce the harmful effects of oxidative stress111.
Many studies emphasize the well-established role of ABA in physiological processes and acclimation to abiotic stresses, thereby assigning it the role of a positive regulator of plant drought resistance112,113. Here, high levels of ABA were observed in shoots (142.8 ng/g FW) and roots (121.1 ng/g FW) of drought-treated plants comparing to control (5.0 ng/g FW and 30.2 ng/g FW, respectively) (Fig. 7C). In research by Peltonen-Sainio and Mäkelä114 on 19 oat cultivars, it was determined that drought stress significantly increased the accumulation of ABA, whereas Ci and RWC decreased due to water deficit. It is known that ABA is a key regulator of abiotic stress resistance in plants. It mediates many stress-responsive genes, including genes regulating the efficiency of photosynthesis. ABA-induced stress tolerance is partly associated with the action of antioxidant systems, which protects plant cells from oxidative damage14.
Correlations between gene expressions, water status and stress responses
In our study, the expressions of AsMT2 and AsMT3 were significantly negatively correlated to each other in shoots but positively correlated in roots. The expression of AsMT1 was independent in shoots but negatively correlated with the expression of AsMT2 and AsMT3 in roots (Fig. 9). AsMT2 expression was positively correlated with EL, Ci, sugars, and ABA in shoots, conversely to AsMT3, which was negatively correlated with these parameters. A negative correlation between the expression of AsMT2 and RWC, WL, WUE, Car, PN, E, and gs was observed in shoots. Inverse correlations were observed for AsMT3 expression and the mentioned parameters. In both shoots and roots, the expression of AsMT2 was positively correlated with ABA. This might confirm that AsMT2 is involved in oat response to drought and is regulated via an ABA-dependent pathway. The expression of AsMT3 negatively correlated with ABA in shoots, but in roots the correlation was positive. Interestingly, AsMT3 had significantly fewer ABA-responsive CREs than AsMT1 and AsMT2. The expression level of AsMT1 was positively correlated with chlorophyll a content in shoots and negatively correlated with ABA in roots (Fig. 9). Interestingly, no correlation between AsMTs expression and antioxidant enzymes was observed. A negative correlation was observed between PX activity and levels of chlorophyll in oat shoots. In oat roots, a negative correlation was observed between SOD activity and levels of sugars.
Pearson correlations between AsMT gene expressions and measured plant traits in shoots (A) and in roots (B). Only significant relations are demonstrated (p < 0.05). Abbreviations: AsMT1—oat metallothionein type 1, AsMT2—oat metallothionein type 2, AsMT3—oat metallothionein type 3, RWC—relative water content, WL—water loss, EL—electrolyte leakage, WUE—photosynthetic water use efficiency, Chl a—chlorophyll a, Chl b—chlorophyll b, Car—carotenoids, Pn—net photosynthesis, E—transpiration, gs—stomatal conductance, Ci—internal CO2 concentration, ABA—abscisic acid, CAT—catalase activity, SOD—superoxide dismutase activity, PX—peroxidase activity.
Drought stress is extensively investigated in plants of industrial importance, including oat. However, still little is known about the molecular mechanisms underlying oat’s tolerance of or susceptibility to drought50. Thus, it is necessary to conduct further physiological and molecular research concerning the responses of oat to drought stresses. Plant MTs seem to participate in plant drought tolerance, but the exact pathways are still unclear, and more in-depth research is needed. Our results showed that oat metallothioneins type 1–3 have different roles in plant cells in response to drought stress. During drought stress in oat plants, the efficiency of photosynthesis decreased and the content of ABA significantly increased. We hypothesised that the expression of AsMT2 was induced via ABA in drought-stressed plants. Metallothioneins together with sugars and antioxidant enzymes (CAT and PX) protect cells from a high level of ROS. We propound the hypothesis that a higher amount of MTs is necessary to provide elevated levels of zinc in cells. Zinc is a crucial cofactor of several enzymes and structural element of countless transcription factors. Moreover, prolonged stress leads to the activation of apoptosis, which is also regulated by Zn ions. Moreover, MTs are crucial for the translocation of zinc and possibly other metal ions to different parts of plants115.
In conclusion, the conducted research provides important new information on the response of plants to stress mediated by metallothioneins. This knowledge about the role of AsMTs in drought stress response will enable the creation of plants via conventional or transgenic breeding that will be resistant to stresses, including drought. This will allow for greater yield from crops even in adverse environmental conditions.
Materials and methods
In silico analyses of promoters of A. sativa L. MT genes
We have previously cloned three oat metallothionein partial cDNA sequences66. Since then, the genome assembly of A. sativa has been published in the PanOat database (https://wheat.pw.usda.gov/GG3/PanOat)116. For each AsMT gene, a 1500-bp-long fragment of genomic DNA upstream of the start codon was retrieved from the PanOat database116. The promoters were analysed using the PlantCARE database117. Molecular masses and pI of putative oat MT proteins were calculated using the Compute pI/Mw tool (ExPaSy)118.
Plant material
Grains of oat cv. Bingo, purchased from Plant Breeding Strzelce Ltd., PBAI Group, Strzelce, Łódź Voivodeship, Poland, were sown individually to 3-dm3 pots filled with a mixture of soil and sand (3/1 v/v). Plants were grown at 25 °C under a 16-h photoperiod and 800 μmol (hν) m2 s−1 PAR. Drought stress was induced by the cessation of watering the soil when the plants reached the four-leaf stage. The degree of soil moisture was determined by the gravimetric method and set at 70% field water capacity (FWC) for control conditions and 20% FWC for drought conditions. After 14 days, leaves and roots of control and drought-treated plants were collected (Fig. 10). The authors confirm that all methods used were performed in accordance with the relevant guidelines and legislation.
Isolation of nucleic acid and analysis of AsMT1–3 expression in response to drought stress
The oat plants were washed several times in nuclease-free water. Shoots and roots were ground separately in liquid nitrogen. Total RNA was isolated using RNeasy Plant Mini Kit (QIAGEN, Germany) according to the manufacturer’s protocol. The quality and quantity of the extracted RNA were checked by agarose gel electrophoresis and by spectrophotometric measurement using a NanoDrop Lite Spectrophotometer (Thermo Fisher Scientific, USA). To remove any DNA contamination from RNA samples, 1.5 μg of total RNA was treated with 1 U of DNase I (Thermo Fisher Scientific, US) and incubated at 37 °C for 30 min. The reaction was stopped by the addition of 1 μL 50 mM EDTA and incubation at 65 °C for 10 min. Reverse transcription reaction was performed using a RevertAid Reverse Transcriptase (Thermo Fisher Scientific, US) according to the manufacturer’s protocol using 250 ng oligo (dT)20 primer and 200 ng random hexamers.
RT-qPCR was performed in a total volume of 10 μL using a Maxima SYBR Green/ROX qPCR Master Mix (Thermo Fisher Scientific, US)66. The reaction mixture contained 4 μL of 10 × diluted cDNA and 0.3 μM gene-specific primers (Table 2). Three replicates were performed for each reaction. The qPCR reaction was conducted in a LightCycler 480 Instrument (Roche, Germany). The thermal cycling conditions were as follows: 95 °C for 10 min for initial denaturation, 40 cycles of 95 °C for 15 s, 60 °C for 30 s, and 72 °C for 30 s66,119. Differences in the target gene expression were evaluated by a relative quantification method normalising the data to the reference genes for eukaryotic initiation factor 4A-3 (EIF4A) and heterogeneous nuclear ribonucleoprotein 27C (HNR)120. The fold-change in gene expression was calculated using LightCycler 480 Software (ver. 1.5.1.62).
Biochemical analyses
Relative water content (RWC)
RWC was determined in leaves according to Ober et al.121. Samples were collected from the second fully developed leaf. RWC was calculated according to the equation: RWC (%) = (Wf − Wd)/(Wt − Wd) × 100, where Wf, Wd and Wt represent fresh weight, dry weight, and turgid weight, respectively. The experiment was repeated three times with five plants.
Water loss (WL) test
WL in leaves was determined using Clarke and McCaig’s122 method. Plants were grown in a greenhouse under well-watered conditions at 21 °C until the fourth leaf had fully emerged. This leaf was cut and placed in a growth chamber at 20 °C, 50% relative humidity, and continuous light of 250 µmol m−2 s−1. The mass of leaf was recorded after cutting (0 h), 6 h later, and after drying at 70 °C for 48 h. WL was calculated as water loss per unit of initial water content according to the equation: WL (%) = (FW0 − FW6)/(FW0 − DW), where FW0 and FW6 are fresh weights after cutting and 6 h later, respectively, and DW is the dry weight after drying at 70 °C. The experiment was repeated three times with five plants.
Electrolyte leakage (EL)
Three leaf discs (1 cm in diameter) were placed into a plastic tube containing 10 mL of redistilled water. They were shaken (100 rpm) at room temperature and the initial electrolyte leakage (EL0) was measured with a conductivity meter (CI 317, Elmetron, Poland) after 24 h. The tubes with leaves were stored at − 70 °C overnight, shaken after thawing, and then their conductivity, and total content of ions (EL1) were measured. The permeability of cell membranes was represented as a percentage of total electrolyte leakage according to the equation: EL = EL0 × 100/EL1. The experiment was repeated three times with five plants.
Leaf gas-exchange parameters
The rate of gas exchange was measured in the fully developed second leaf using a portable CIRAS-2 photosynthesis system (PP System, Hitchin, UK). The rate of net photosynthesis (PN) and transpiration (E), stomatal conductance (gs), and internal CO2 concentration (Ci) were measured between 9:00 and 11:00 a.m. The photosynthetic water use efficiency (WUE) was also expressed as PN/E. The experiment was repeated three times with five plants. The measurements included three replicates per plant.
Photosynthetic pigments content
The 100 mg of leaves was homogenized in 1 mL of 80% ethanol and then centrifuged at 2800 rpm for 10 min. The absorbance of the samples was measured at λ = 470 nm, λ = 648 nm, and λ = 664 nm on a micro-plate reader (Synergy 2, Bio-Tek, Winooski, VT, USA). Concentrations of photosynthetic pigments (chlorophylls a, b and carotenoids) were determined using a Lichtenthaler and the Wellburn method123. The experiment was repeated three times with five plants.
Soluble sugar content
The 100 mg of leaves was homogenized in 1 mL of 80% ethanol, then centrifuged at 2800 rpm for 10 min. The amounts of total soluble sugars were estimated by the phenol–sulphuric acid method124. Briefly, the supernatant was mixed with 5% phenol and 96% sulphuric acid. The absorbance of the samples was measured spectrophotometrically at λ = 490 nm on a micro-plate reader (Synergy 2, Bio-Tek, Winooski, VT, USA). The amount of soluble sugars was expressed as milligrams of glucose per 100 g of fresh mass (FW) of plant tissue. The experiment was repeated three times with five plants.
Total phenolics content
The 100 mg of leaves was homogenized in 1 mL of 80% ethanol, then centrifuged at 2800 rpm for 10 min. To estimate the phenolics content, the supernatant was mixed with 20% Na2CO3 and Folin–Ciocalteu reagent125. The absorbance of samples was measured spectrophotometrically at λ = 760 nm on a micro-plate reader (Synergy 2, Bio-Tek, Winooski, VT, USA). The total phenolic content was calculated as milligrams of chlorogenic acid per gram of FW of plant tissue. The experiment was repeated three times with five plants.
Abscisic acid (ABA) content
The leaves were frozen in liquid nitrogen, lyophilised and homogenised. Then, 50 mg of plant material was extracted with a 1-mL mixture of methanol/water/formic acid (15/4/1; v/v/v) according to Dobrev and Kaminek126. An internal isotopic standard of ABA was added to each sample. The extract was then centrifuged, the supernatant was collected, and the extraction procedure was repeated. The combined supernatant was dried and reconstituted in 1 mL of 1 M formic acid. This extract was fractionated with SPE columns Oasis MCX 1 cc/30 mg (Waters, Milford, MA, USA). The acidic fraction was eluted from the SPE column with 1 mL methanol, evaporated to dryness, and reconstituted in 50 µL methanol. Samples prepared in this manner were analysed on a Supelco Ascentis RP-Amide HPLC column (Saint Louis, MO, USA) (7.5 cm, 4.6 mm, 2.7 µm). Mobile phases were 0.1% formic acid solution in water (solvent A) and acetonitrile/methanol (1/1) mixture. Gradient elution was applied under the flow rate of 0.5 mL/min. The HPLC apparatus was an Agilent Technologies 1260 equipped with an Agilent Technologies 6410 Triple Quad LC/ MS with ESI (Electrospray Interface, Agilent Technologies, Santa Clara, CA, USA). The two most abundant secondary ions were monitored: abscisic acid (ABA)—m/z 265.2 primary, m/z 229.1, 247.1 secondary; D-ABA (deuterium labelled ABA used as internal standard)—m/z 271.2 primary, m/z 167.1 secondary. Ten-point calibration curves were prepared for the analysed compounds. The experiment was repeated three times with five plants.
Antioxidant enzymes activities
The leaves were homogenised with 0.05 M phosphate buffer (pH 7.0) containing 0.1 mM EDTA at 4 °C and centrifuged at 2800 rpm for 10 min. Superoxide dismutase (SOD) activity was assayed according to McCord and Fridovich127. The reaction mixture consisted of 0.05 M phosphate buffer, 0.013 mM cytochrome c, 0.1 mM xanthine, 0.024 U per ml xanthine oxidase, and supernatant. Absorbance was measured at λ = 550 nm.
Catalase (CAT) activity was determined according to Aebi128. The reaction mixture contained 0.05 M phosphate buffer, 0.1 mM H2O2, and supernatant. The rate of H2O2 decomposition was measured at λ = 240 nm.
The activity of peroxidase (PX) was measured by the method of Lűck129. The amount of oxidation product of 1% p-phenylenediamine in the presence of 0.03 M H2O2 was measured at λ = 485 nm.
The reaction kinetics of all enzymes were measured spectrophotometrically using a micro-plate reader (Synergy 2, Bio-Tek, Winooski, VT, USA). Enzyme activities were calculated per milligram of protein measured by Bradford method with bovine serum albumin as a protein standard130. The experiment was repeated three times with five plants for each enzyme.
Statistical analysis
The results are expressed as mean values, and error bars represent standard error (SE). Before statistical assessment, data normality was tested by the Shapiro–Wilk test. The majority of parameters were normally distributed, and statistical analysis of the experimental data was done with the analysis of variance (ANOVA). Student’s t-test (p value ≤ 0.05) was applied to determine differences between expression levels of control and drought-stressed plants. To demonstrate relations between measured traits, Pearson correlations were calculated131. The programs Past 4.0132, STATISTICA 13.0 (Stat-soft, Inc., USA), and RStudio133 were applied for calculations.
Data availability
The datasets generated during the current study are available from the corresponding author on reasonable request.
References
Swann, A. L. S. Plants and drought in a changing climate. Curr. Clim. Change Rep. 4, 192–201 (2018).
Mukherjee, S., Mishra, A. & Trenberth, K. E. Climate change and drought: a perspective on drought indices. Curr. Clim. Change Rep. 4, 145–163 (2018).
Dai, A., Zhao, T. & Chen, J. Climate change and drought: a precipitation and evaporation perspective. Curr. Clim. Change Rep. 4, 301–312 (2018).
Fang, Y. & Xiong, L. General mechanisms of drought response and their application in drought resistance improvement in plants. Cell. Mol. Life Sci. 72, 673–689 (2015).
Hanson, A. D. et al. Osmoprotective compounds in the Plumbaginaceae: A natural experiment in metabolic engineering of stress tolerance. Proc. Natl. Acad. Sci. U. S. A. 91, 306–310 (1994).
Mohanty, A. K., Misra, M. & Drzal, L. T. Sustainable bio-composites from renewable resources: Opportunities and challenges in the green materials world. J. Polym. Environ. 10, 19–26 (2002).
Gong, D. S. et al. Early activation of plasma membrane H+-ATPase and its relation to drought adaptation in two contrasting oat (Avena sativa L.) genotypes. Environ. Exp. Bot. 69, 1–8 (2010).
Ashraf, M. & Foolad, M. R. Roles of glycine betaine and proline in improving plant abiotic stress resistance. Environ. Exp. Bot. 59, 206–216 (2007).
Shinozaki, K. & Yamaguchi-Shinozaki, K. Gene networks involved in drought stress response and tolerance. J. Exp. Bot. 58, 221–227 (2007).
Bhargava, S. & Sawant, K. Drought stress adaptation: metabolic adjustment and regulation of gene expression. Plant Breed. 132, 21–32 (2013).
Mahmood, T. et al. Insights into drought stress signaling in plants and the molecular genetic basis of cotton drought tolerance. Cells 9, 105 (2019).
Uno, Y. et al. Arabidopsis basic leucine zipper transcription factors involved in an abscisic acid-dependent signal transduction pathway under drought and high-salinity conditions. Proc. Natl. Acad. Sci. U. S. A. 97, 11632–11637 (2000).
Yamaguchi-Shinozaki, K. & Shinozaki, K. Organization of cis-acting regulatory elements in osmotic- and cold-stress-responsive promoters. Trends Plant Sci. 10, 88–94 (2005).
Liu, S., Lv, Z., Liu, Y., Li, L. & Zhang, L. Network analysis of ABA-dependent and ABA-independent drought responsive genes in Arabidopsis thaliana. Genet. Mol. Biol. 41, 624–637 (2018).
Islam, M. R. et al. Effects of water-saving superabsorbent polymer on antioxidant enzyme activities and lipid peroxidation in oat (Avena sativa L.) under drought stress. J. Sci. Food Agric. 91, 680–686 (2011).
Dąbrowska, G., Kata, A., Goc, A., Szechyńska-Hebda, M. & Skrzypek, E. Characteristics of the plant ascorbate peroxidase family. Acta Biol. Crac. Ser. Bot. 49, 7–17 (2007).
Hura, T., Hura, K. & Grzesiak, S. Contents of total phenolics and ferulic acid, and pal activity during water potential changes in leaves of maize single-cross hybrids of different drought tolerance. J. Agron. Crop Sci. 194, 104–112 (2008).
Latif, F. et al. Effects of salicylic acid on growth and accumulation of phenolics in Zea mays L. under drought stress. Acta Agric. Scand. Soil Plant Sci. 66, 325–332 (2016).
Skrzypek, E., Szechyńska-Hebda, M., Dąbrowska, G. B. & Goc, A. The role of osmotic stress during in vitro regeneration of Triticum aestivum L. and Vicia faba ssp. minor. Zesz. Prob. Post. Nauk Rol. 524, 221–230 (2008).
Blindauer, C. A. & Leszczyszyn, O. I. Metallothioneins: Unparalleled diversity in structures and functions for metal ion homeostasis and more. Nat. Prod. Rep. 27, 720–741 (2010).
Blindauer, C. A. Bacterial metallothioneins: Past, present, and questions for the future. J. Biol. Inorg. Chem. 16, 1011–1024 (2011).
Mierek-Adamska, A., Znajewska, Z., Goc, A. & Dąbrowska, G. B. Molecular cloning and characterization of Ipomoea nil metallothioneins. Turk. J. Bot. 42, 247–256 (2018).
Koszucka, A. M. & Dąbrowska, G. Plant metallothioneins. Adv. Cell Biol. 33, 285–302 (2006).
Mierek-Adamska, A., Dąbrowska, G. B. & Blindauer, C. A. The type 4 metallothionein from Brassica napus seeds folds in a metal-dependent fashion and favours zinc over other metals. Metallomics 10, 1430–1443 (2018).
Zhang, H., Xu, W., Dai, W., He, Z. & Ma, M. Functional characterization of cadmium-responsive garlic gene AsMT2b: A new member of metallothionein family. Chin. Sci. Bull. 51, 409–416 (2006).
Jin, S. et al. Functional characterization of a type 2 metallothionein gene, SsMT2, from alkaline-tolerant Suaeda salsa. Sci. Rep. 7, 1–11 (2017).
Bellion, M. et al. Metal induction of a Paxillus involutus metallothionein and its heterologous expression in Hebeloma cylindrosporum. New Phytol. 174, 151–158 (2007).
Evans, K. M. et al. Expression of the pea metallothionein-like gene PsMTA in Escherichia coli and Arabidopsis thaliana and analysis of trace metal ion accumulation: Implications for PsMTA function. Plant Mol. Biol. 20, 1019–1028 (1992).
Cobbett, C. S. Phytochelatins and their roles in heavy metal detoxification. Plant Physiol. 123, 825–832 (2000).
Freisinger, E. Structural features specific to plant metallothioneins. J. Biol. Inorg. Chem. 16, 1035–1045 (2011).
Tomas, M. et al. His-containing plant metallothioneins: Comparative study of divalent metal-ion binding by plant MT3 and MT4 isoforms. J. Biol. Inorg. Chem. 19, 1149–1164 (2014).
Leszczyszyn, O. I., Schmid, R. & Blindauer, C. A. Toward a property/function relationship for metallothioneins: Histidine coordination and unusual cluster composition in a zinc-metallothionein from plants. Proteins 68, 922–935 (2007).
Mierek-Adamska, A. et al. Potential involvement of rapeseed (Brassica napus L.) metallothioneins in the hydrogen peroxide-induced regulation of seed vigour. J. Agron Crop Sci. 205, 598–607 (2019).
Sekhar, S. et al. Comparative transcriptome profiling of low light tolerant and sensitive rice varieties induced by low light stress at active tillering stage. Sci. Rep. 9, 1–14 (2019).
Mir, G. et al. A plant type 2 metallothionein (MT) from cork tissue responds to oxidative stress. J. Exp. Bot. 55, 2483–2493 (2004).
Hrynkiewicz, K., Dąbrowska, G., Baum, C., Niedojadlo, K. & Leinweber, P. Interactive and single effects of ectomycorrhiza formation and Bacillus cereus on metallothionein MT1 expression and phytoextraction of Cd and Zn by willows. Water Air Soil Pollut. 223, 957–968 (2012).
Lee, J., Shim, D., Song, W. Y., Hwang, I. & Lee, Y. Arabidopsis metallothioneins 2a and 3 enhance resistance to cadmium when expressed in Vicia faba guard cells. Plant Mol. Biol. 54, 805–815 (2004).
Dąbrowska, G., Mierek-Adamska, A. & Goc, A. Characterisation of Brassica napus L. metallothionein genes (BnMTs) expression in organs and during seed germination. Aust. J. Crop Sci. 7, 1324–1332 (2013).
Stevens, E. J., Armstrong, K. W., Bezar, H. J., Griffin, W. B. & Hampton, J. G. Fodder Oats: A World Overview (Food and Agriculture Organization of The United Nations, 2004).
Andon, M. B. & Anderson, J. W. The oatmeal-cholesterol connection: 10 years later. Am. J. Lifestyle Med. 2, 51–57 (2008).
Jenkins, A. L., Jenkins, D. J. A., Zdravkovic, U., Würsch, P. & Vuksan, V. Depression of the glycemic index by high levels of β-glucan fiber in two functional foods tested in type 2 diabetes. Eur. J. Clin. Nutr. 56, 622–628 (2002).
Peterson, D. M. Oat antioxidants. J. Cereal Sci. 33, 115–129 (2001).
Xue, T. et al. Cotton metallothionein GhMT3a, a reactive oxygen species scavenger, increased tolerance against abiotic stress in transgenic tobacco and yeast. J. Exp. Bot. 60, 339–349 (2009).
Yang, Z., Wu, Y., Li, Y., Ling, H. Q. & Chu, C. OsMT1a, a type 1 metallothionein, plays the pivotal role in zinc homeostasis and drought tolerance in rice. Plant Mol. Biol. 70, 219–229 (2009).
Seki, M. et al. Monitoring the expression profiles of 7000 Arabidopsis genes under drought, cold and high-salinity stresses using a full-length cDNA microarray. Plant J. 31, 279–292 (2002).
Kumar, S. et al. Metallothionein (MT1): A molecular stress marker in chickpea enhances drought and heavy metal stress adaptive efficacy in transgenic plants. Environ. Exp. Bot. 199, 104871 (2022).
Ren, Y. et al. Type 4 metallothionein genes are involved in regulating Zn ion accumulation in late embryo and in controlling early seedling growth in Arabidopsis. Plant Cell Environ. 35, 770–789 (2012).
Cheng, M. et al. Genome-wide identification and analysis of the metallothionein genes in Oryza genus. Int J. Mol Sci. 22, 9651 (2021).
Feng, M. et al. ScMT10, a metallothionein-like gene from sugarcane, enhances freezing tolerance in Nicotiana tabacum transgenic plants. Environ. Exp. Bot. 194, 104750 (2022).
Sánchez-Martín, J. et al. A metabolomic study in oats (Avena sativa) highlights a drought tolerance mechanism based upon salicylate signalling pathways and the modulation of carbon, antioxidant and photo-oxidative metabolism. Plant Cell Environ. 38, 1434–1452 (2015).
Lane, B., Kajioka, R. & Kennedy, T. The wheat-germ Ec protein is a zinc-containing metallothionein. Biochem. Cell Biol. 65, 1001–1005 (1987).
Cobbett, C. & Goldsbrough, P. Phytochelatins and metallothioneins: roles in heavy metal detoxification and homeostasis. Annu. Rev. Plant Biol. 53, 159–182 (2002).
Saeed-Ur-Rahman, K. M., Hui, N., Kayani, S. I. & Tang, K. Diversity and versatile functions of metallothioneins produced by plants: A review. Pedosphere 30, 577–588 (2020).
Hassinen, V. H., Tervahauta, A. I., Schat, H. & Kärenlampi, S. O. Plant metallothioneins - metal chelators with ROS scavenging activity?. Plant Biol. 13, 225–232 (2011).
Buchanan-Wollaston, V. Isolation of cDNA clones for genes that are expressed during leaf senescence in Brassica napus (identification of a gene encoding a senescence-specific metallothionein-like protein). Plant Physiol. 105, 839–846 (1994).
Kumar, S. et al. Plant metallothioneins as regulators of environmental stress responses. Int. J. Plant Environ. 7, 27–38 (2021).
Guo, W.-J., Bundithya, W. & Goldsbrough, P. B. Characterization of the Arabidopsis metallothionein gene family: Tissue-specific expression and induction during senescence and in response to copper. New Phytol. 159, 369–381 (2003).
Imam, H. T. & Blindauer, C. A. Differential reactivity of closely related zinc(II)-binding metallothioneins from the plant Arabidopsis thaliana. J. Biol. Inorg. Chem. 23, 137–154 (2017).
Murphy, A., Zhou, J., Goldsbrough, P. B. & Taiz, L. Purification and immunological identification of metallothioneins 1 and 2 from Arabidopsis thaliana. Plant Physiol. 113, 1293–1301 (1997).
Zhu, W. et al. Arabidopsis thaliana metallothionein, AtMT2a, Mediates ROS balance during oxidative stress. J. Plant Biol. 52, 585–592 (2009).
Yu, Q. et al. Genome-wide identification and expression analysis of heavy metal stress–responsive metallothionein family genes in Nicotiana tabacum. Plant Mol. Biol. Rep. 39, 443–454 (2020).
Pan, Y. et al. Genome-wide characterization and analysis of metallothionein family genes that function in metal stress tolerance in Brassica napus L.. Int J. Mol. Sci. 19, 1–18 (2018).
Zhou, Y., Liu, J., Liu, S., Jiang, L. & Hu, L. Identification of the metallothionein gene family from cucumber and functional characterization of CsMT4 in Escherichia coli under salinity and osmotic stress. Biotech 9, 1–11 (2019).
Zhou, G., Xu, Y., Li, J., Yang, L. & Liu, J. Y. Molecular analyses of the metallothionein gene family in rice (Oryza sativa L.). J. Biochem. Mol. Biol. 39, 595–606 (2006).
Gao, C. et al. Genome-wide analysis of metallothionein gene family in maize to reveal its role in development and stress resistance to heavy metal. Biol. Res. 55, 1–13 (2022).
Konieczna, W., Mierek-Adamska, A., Warchoł, M., Skrzypek, E. & Dąbrowska, G. The involvement of metallothioneins and stress markers in response to osmotic stress in Avena sativa L. J. Agron. Crop Sci. (In press)
Yan, H. et al. Genome size variation in the genus Avena. Genome 59, 209–220 (2016).
Chaffin, A. S. et al. A consensus map in cultivated hexaploid oat reveals conserved grass synteny with substantial subgenome rearrangement. Plant Genome 9, 1–21 (2016).
Kamal, N. et al. The mosaic oat genome gives insights into a uniquely healthy cereal crop. Nature 606, 113–119 (2022).
Leszczyszyn, O. I., Imam, H. T. & Blindauer, C. A. Diversity and distribution of plant metallothioneins: A review of structure, properties and functions. Metallomics 5, 1146–1169 (2013).
Blindauer, C. A. et al. Multiple bacteria encode metallothioneins and SmtA-like zinc fingers. Mol. Microbiol. 45, 1421–1432 (2002).
Dąbrowska, G., Mierek-Adamska, A. & Goc, A. Plant metallothioneins: Putative functions identified by promoter analysis in silico. Acta Biol. Crac. Ser. Bot. 54, 109–120 (2012).
Dąbrowska, G. B., Turkan, S., Tylman-Mojżeszek, W. & Mierek-Adamska, A. In silico study of the rsh (Rela/spot homologs) gene family and expression analysis in response to PGPR bacteria and salinity in Brassica napus. Int J. Mol. Sci. 22, 10666 (2021).
Javadi, S. M., Shobbar, Z. S., Ebrahimi, A. & Shahbazi, M. New insights on key genes involved in drought stress response of barley: Gene networks reconstruction, hub, and promoter analysis. J. Genet. Eng. Biotechnol. 19, 1–12 (2021).
Whitelaw, C. A., le Huquet, J. A., Thurman, D. A. & Tomsett, A. B. The isolation and characterisation of type II metallothionein-like genes from tomato (Lycopersicon esculentum L.). Plant Mol. Biol. 33, 503–511 (1997).
Omidvar, V., Abdullah, S. N. A., Izadfard, A., Ho, C. L. & Mahmood, M. The oil palm metallothionein promoter contains a novel AGTTAGG motif conferring its fruit-specific expression and is inducible by abiotic factors. Planta 232, 925–936 (2010).
Laplaze, L. et al. Symbiotic and non-symbiotic expression of cgMT1, a metallothionein-like gene from the actinorhizal tree Casuarina glauca. Plant Mol. Biol. 49, 81–92 (2002).
Ahn, Y. O. et al. Three Brassica rapa metallothionein genes are differentially regulated under various stress conditions. Mol. Biol. Rep. 39, 2059–2067 (2012).
Ren, Y. & Zhao, J. Functional analysis of the rice metallothionein gene OsMT2b promoter in transgenic Arabidopsis plants and rice germinated embryos. Plant Sci. 176, 528–538 (2009).
Lü, S. et al. The GUS reporter-aided analysis of the promoter activities of a rice metallothionein gene reveals different regulatory regions responsible for tissue-specific and inducible expression in transgenic Arabidopsis. Transgenic Res. 16, 177–191 (2007).
Dong, C.-J.J., Wang, Y., Yu, S.-S.S. & Liu, J.-Y.Y. Characterization of a novel rice metallothionein gene promoter: Its tissue specificity and heavy metal responsiveness. J. Integr. Plant Biol. 52, 914–924 (2010).
Obertello, M. et al. Functional analysis of the metallothionein gene cgmt1 isolated from the actinorhizal tree Casuarina glauca. Mol. Plant-Microbe Interact. 20, 1231–1240 (2007).
Akashi, K., Nishimura, N., Ishida, Y. & Yokota, A. Potent hydroxyl radical-scavenging activity of drought-induced type-2 metallothionein in wild watermelon. Biochem. Biophys. Res. Commun. 323, 72–78 (2004).
Samardžić, J. T. et al. Tissue expression analysis of FeMT3, a drought and oxidative stress related metallothionein gene from buckwheat (Fagopyrum esculentum). J. Plant Physiol. 167, 1407–1411 (2010).
Yuan, J., Chen, D., Ren, Y., Zhang, X. & Zhao, J. Characteristic and expression analysis of a metallothionein gene, OsMT2b, down-regulated by cytokinin suggests functions in root development and seed embryo germination of rice. Plant Physiol. 146, 1637–1650 (2008).
Jaiswal, P. S., Mittal, N. & Randhawa, G. S. Cyamopsis tetragonoloba type 1 metallothionein (CtMT1) gene is upregulated under drought stress and its protein product has an additional C-X-C motif and unique metal binding pattern. Int. J. Biol. Macromol. 119, 1324–1334 (2018).
Dubey, A. K. et al. Over-expression of CarMT gene modulates the physiological performance and antioxidant defense system to provide tolerance against drought stress in Arabidopsis thaliana L. Ecotoxicol. Environ. Saf. 171, 54–65 (2019).
Patankar, H. V. et al. Overexpression of a metallothionein 2a gene from date palm confers abiotic stress tolerance to yeast and Arabidopsis thaliana. Int. J. Mol. Sci. 20, 2871 (2019).
Mekawy, A. M. M., Assaha, D. V. M. & Ueda, A. Constitutive overexpression of rice metallothionein-like gene OsMT-3a enhances growth and tolerance of Arabidopsis plants to a combination of various abiotic stresses. J. Plant Res. 133, 429–440 (2020).
Li, Z. et al. The alterations of endogenous polyamines and phytohormones induced by exogenous application of spermidine regulate antioxidant metabolism, metallothionein and relevant genes conferring drought tolerance in white clover. Environ. Exp. Bot. 124, 22–38 (2016).
Ruttkay-Nedecky, B. et al. The role of metallothionein in oxidative stress. Int. J. Mol. Sci. 14, 6044–6066 (2013).
Wang, Z. et al. Effects of drought stress on photosynthesis and photosynthetic electron transport chain in young apple tree leaves. Biol. Open 7, bio035279 (2018).
Farooq, M., Wahid, A., Kobayashi, N., Fujita, D. & Basra, S. M. A. Plant drought stress: Effects, mechanisms and management. Agron. Sustain. Dev. 29, 185–212 (2009).
Grzesiak, M. T. et al. Impact of soil compaction stress combined with drought or waterlogging on physiological and biochemical markers in two maize hybrids. Acta Physiol. Plant 38, 1–15 (2016).
Bajji, M., Kinet, J. M. & Lutts, S. The use of the electrolyte leakage method for assessing cell membrane stability as a water stress tolerance test in durum wheat. Plant Growth Reg. 36, 61–70 (2002).
Hsiao, T. C. Plant responses to water stress. Annu. Rev. Plant Physiol. 24, 519–570 (1973).
Ostrowska, A. & Hura, T. Physiological comparison of wheat and maize seedlings responses to water stresses. Sustainability 14, 7932 (2022).
Liang, G., Liu, J., Zhang, J. & Guo, J. Effects of drought stress on photosynthetic and physiological parameters of tomato. J. Am. Soc. Hort. Sci. 145, 12–17 (2020).
Zhang, X. et al. Effects of drought stress during critical periods on the photosynthetic characteristics and production performance of naked oat (Avena nuda L.). Sci. Rep. 12, 1–11 (2022).
Zhao, B., Ma, B. L., Hu, Y. & Liu, J. Source–sink adjustment: a mechanistic understanding of the timing and severity of drought stress on photosynthesis and grain yields of two contrasting oat (Avena sativa L.) genotypes. J. Plant Growth Regul. 40, 263–276 (2021).
Todorova, D., Aleksandrov, V., Anev, S. & Sergiev, I. Photosynthesis alterations in wheat plants induced by herbicide, soil drought or flooding. Agronomy 12, 390 (2022).
Marcińska, I. et al. Application of photochemical parameters and several indices based on phenotypical traits to assess intraspecific variation of oat (Avena sativa L.) tolerance to drought. Acta Physiol. Plant 39, 1–13 (2017).
Esteban, R. et al. Internal and external factors affecting photosynthetic pigment composition in plants: A meta-analytical approach. New Phytol. 206, 268–280 (2015).
Seki, M., Umezawa, T., Urano, K. & Shinozaki, K. Regulatory metabolic networks in drought stress responses. Curr. Opin. Plant Biol. 10, 296–302 (2007).
Guy, C., Kaplan, F., Kopka, J., Selbig, J. & Hincha, D. K. Metabolomics of temperature stress. Physiol. Plant 132, 220–235 (2008).
Arabzadeh, N. The effect of drought stress on soluble carbohydrates (sugars) in two species of Haloxylon persicum and Haloxylon aphyllum. Asian J. Plant Sci. 11, 44–51 (2012).
Bhattacharjee, S. Reactive oxygen species and oxidative burst: Roles in stress, senescence and signal transducation in plants. Curr. Sci. 89 (2005).
Si, M. & Lang, J. The roles of metallothioneins in carcinogenesis. J. Hematol. Oncol. 11, 1–20 (2018).
Chakraborty, U. & Pradhan, B. Oxidative stress in five wheat varieties (Triticum aestivum L.) exposed to water stress and study of their antioxidant enzyme defense system, water stress responsive metabolites and H2O2 accumulation. Braz. J. Plant Physiol. 24, 117–130 (2012).
Gratão, P. L., Polle, A., Lea, P. J. & Azevedo, R. A. Making the life of heavy metal-stressed plants a little easier. Funct. Plant Biol. 32, 481–494 (2005).
Bolouri-Moghaddam, M. R., le Roy, K., Xiang, L., Rolland, F. & van den Ende, W. Sugar signalling and antioxidant network connections in plant cells. FEBS J. 277, 2022–2037 (2010).
Gietler, M., Fidler, J., Labudda, M. & Nykiel, M. Abscisic acid—Enemy or savior in the response of cereals to abiotic and biotic stresses?. Int. J. Mol. Sci. 21, 4607 (2020).
Bharath, P., Gahir, S. & Raghavendra, A. S. Abscisic acid-induced stomatal closure: An important component of plant defense against abiotic and biotic stress. Front. Plant Sci. 12, 324 (2021).
Peltonen-Sainio, P. & Mäkelä, P. Comparison of physiological methods to assess drought tolerance in oats. Acta Agric. Scand. B Soil Plant Sci. 45, 32–38 (1995).
Formigari, A., Irato, P. & Santon, A. Zinc, antioxidant systems and metallothionein in metal mediated-apoptosis: Biochemical and cytochemical aspects. Comp. Biochem. Physiol. 146, 443–459 (2007).
PepsiCo. Avena sativa - OT3098 v2, PepsiCo, https://wheat.pw.usda.gov/jb?data=/ggds/oat-ot3098v2-pepsico. (2021).
Lescot, M. et al. PlantCARE, a database of plant cis-acting regulatory elements and a portal to tools for in silico analysis of promoter sequences. Nucl. Acids Res. 30, 325–327 (2002).
Gasteiger, E. et al. ExPASy: The proteomics server for in-depth protein knowledge and analysis. Nucl. Acids Res. 31, 3784–3788 (2003).
Cárdenas-Pérez, S. et al. Maternal salinity influences anatomical parameters, pectin content, biochemical and genetic modifications of two Salicornia europaea populations under salt stress. Sci. Rep 12, 1–16 (2022).
Yang, Z., Wang, K., Aziz, U., Zhao, C. & Zhang, M. Evaluation of duplicated reference genes for quantitative real-time PCR analysis in genome unknown hexaploid oat (Avena sativa L.). Plant Meth. 16, 1–14 (2020).
Ober, E. S. et al. Evaluation of physiological traits as indirect selection criteria for drought tolerance in sugar beet. Field Crops Res. 91, 231–249 (2005).
Clarke, J. M. & McCaig, T. N. Excised-leaf water retention capability as an indicator of drought resistance of Triticum genotypes. Can. J. Plant Sci. 62, 571–578 (2011).
Lichtenthaler, H. K. Chlorophylls and carotenoids: Pigments of photosynthetic biomembranes. Methods Enzymol. 148, 350–382 (1987).
Dubois, M., Gilles, K. A., Hamilton, J. K., Rebers, P. A. & Smith, F. Colorimetric method for determination of sugars and related substances. Anal. Chem. 28, 350–356 (1956).
Singleton, V. L. & Rossi, J. A. Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am. J. Enol. Vitic. 16, 144–158 (1965).
Dobrev, P. & Kamínek, M. Fast and efficient separation of cytokinins from auxin and abscisic acid and their purification using mixed-mode solid-phase extraction. J. Chromatogr. 950, 21–29 (2002).
McCord, J. & Fiodovich, I. Superoxide dismutase an enzimic function for erytrocuperein (hemocuperein). J. Biol. Chem. 244, 6049–6055 (1969).
Aebi, H. Catalase in vitro. Methods Enzymol. 105, 121–125 (1984).
Lűck, H. Methoden der enzymatischen Analyse. Verlag Chemie (GmbH Weinheim, 1962).
Bradford, M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 72, 248–254 (1976).
Zar, J. H. Biostatistical Analysis (Prentice-Hall/Pearson, 2010).
Hammer, D. A. T., Ryan, P. D., Hammer, Ø. & Harper, D. A. T. Past: Paleontological Statistics Software Package for Education and Data Analysis. Palaeon. Electron. 4, (2001).
RStudio Team. RStudio: integrated development for R. RStudio. http://www.rstudio.com/ (2020).
Acknowledgements
The authors acknowledge Dr. M. Grzesiak from the Franciszek Górski Institute of Plant Physiology, Polish Academy of Sciences for measurements of leaf gas-exchange parameters. The research was financed from the subsidy of the Ministry of Science and Higher Education of the Republic of Poland awarded to the Nicolaus Copernicus University in Toruń and the Franciszek Górski Institute of Plant Physiology, Polish Academy of Sciences in Kraków.
Author information
Authors and Affiliations
Contributions
G.B.D. and W.K. conceived and designed the experiment; G.B.D., W.K., A.M.-A., M.W., E.S., carried out the experiments; G.B.D., W.K., A.M.-A., M.W., E.S., P.W., A.P performed the analyses; G.B.D., W.K., A.M.-A., M.W., E.S., P.W., A.P analysed the data; W.K. writing—original draft preparation; G.B.D., W.K., A.M.-A., M.W., E.S., A.P. writing—revision; G.B.D., W.K., funding acquisition; G.B.D. Supervision. All authors read the manuscript and accepted it. All the authors read and approved the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Konieczna, W., Warchoł, M., Mierek-Adamska, A. et al. Changes in physio-biochemical parameters and expression of metallothioneins in Avena sativa L. in response to drought. Sci Rep 13, 2486 (2023). https://doi.org/10.1038/s41598-023-29394-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-29394-2
- Springer Nature Limited