Abstract
The in-situ management of rice straw incorporation has become an essential practice in the rice–wheat cropping system with the growing adoption of super-seeder among farmers. While this equipment facilitates straw incorporation, nutrient immobilization from organic residues during decomposition poses a challenge to production. This study investigates the effect of bio-decomposer, nitrogen (N) level, and N scheduling on on various parameters in super-seeder sown wheat over two consecutive years (2020–21 and 2021–22). The experiment was laid in split-split-plot design without (B0) and with (B1) bio-decomposer, two nitrogen levels 150 (N1) and 180 (N2) kg ha−1 and three N splits (%) viz., 50: 25:25 (S1), 40: 30: 30 (S2), and 30:35:35 (S3) and replicated thrice. Results indicate significantly higher N concentration and uptake at pre- and post-anthesis with N2 and S1 compared to N1, S2, and S3. Post-anthesis N accumulation (post-NA) and remobilization (NR) were notably higher with N2. Dry matter accumulation (DMA) at anthesis and harvest significantly increased with N2 and S1 compared to N1 and S3, leading to significantly improved grain yield by 8.3% and 10.9% (two years mean). Soil urease activity (UA) significantly increased with bio-decomposer, N2, and S1 compared to B0, N1, and S3 at 30 and 60 DAS. Moreover, bio-decomposer enhanced soil microbial biomass carbon (SMBC). These findings underscore the importance of higher N levels with a greater basal N proportion in mitigating N stress and promoting sustainable wheat productivity. It also highlight the significance of effectively utilizing and conserving rice residue in the rice–wheat system.
Article Highlights
-
Evaluation of nitrogen immobilization status by assessing its absorption and concentration in plants.
-
Application of higher nitrogen level coupled with higher proportion of basal nitrogen overcome early nitrogen stress in wheat crop under rice straw incorporation.
-
Impact of bio-decomposer on soil urease enzymes and soil microbial biomass carbon.
Similar content being viewed by others
1 Introduction
Wheat (Triticum aestivum L.) is one of the most extensively grown cereal crops worldwide, and serves as a staple food for about 205 million of world population. Globally, wheat occupies around 217 million hectares of land with the annual production of 713 million tonnes [1], and provides half of the calories in the region of North Africa, West and Central Asia. The rice–wheat cropping system (RWCS) of Indo-Gangetic plain falling in South Asia covers 12.3 million ha in India [2]. This system is of immense importance for ensuring food and nutritional securities in India and contributes about 75% to the national food chain [3]. However, the conventional RWCS has been deemed unsustainable under the ever-changing climatic conditions, due to receding groundwater table [4], increasing labour, capital, and energy requirements [5]. In addition, conventional transplanted rice has negative impact on the succeeding wheat, resulting in soil structural degradation [6], and delayed seeding of the wheat crop [7]. Hence, the conventional RWCS needs to re-orient to a more sustainable management to safeguard the food-security of millions of south Asian families.
The highly mechanized harvesting and threshing of rice using combine harvesters is a common practice in North-West (NW) India, wherein huge residues are left behind in the forms of standing stubbles and loose residue in the field. Timely management of this residue in the short span of 10–20 days for timely planting of wheat crop is a difficult task. Therefore, the farmers commonly opt for burning of rice residue in the combine-harvested fields due to lack of access to user-friendly, cost- and time-effective management practices. It was estimated that in NW states of India, about 23 MT of rice residues were burnt annually [8]. Extensive residue burning results in production of copious amount of harmful gases; an hectare (10,000 m2) of paddy field produces around 6.2 tonnes of stubble, whereby on burning, releases 18.5 kg of particulate matter, 370.7 kg of carbon monoxide, 9,019.5 kg of carbon dioxide, 1230.6 kg of ash and 12.4 kg of sulphur dioxide [9], and the black carbon emitted during residue burning warms the lower atmosphere and is the second most important contributor to global warming after CO2 [10]. Burning of rice residue degrades the soil health due to loss of soil organic matter and plant nutrients. About 90% of nitrogen (N) and sulphur (S), and 15–20% of phosphorus (P) and potassium (K) contained in rice residue are lost during burning [2]. Therefore, the need for providing a cost-effective and farmer friendly option for management of rice residue is a major challenge for the sustainability of intensive RWCS in developing countries.
The on-farm management of rice straw viz., surface retention (mulching), incorporation (in-situ) and composting (ex-situ) and the recent practice i.e., use of bio-decomposer, are the promising strategy to address the issue of burning as well as maintaining the soil health for long-term sustainability of RWCS. Composting of rice residue management is not adopted on large scale by the farmers as it is energy and cost intensive coupled with time limitation. Over the years, different in-situ RS management technologies have been notified and adopted under the RWCS, such as zero-tillage, happy seeder, and super seeder. The recent development of machinery like super-seeder (rota-till-drill) has greatly simplified the incorporation of rice residues into the soil by crushing and evenly spreading the straw over the field leading to clean cultivation [11].
In addition, incorporation of residues increases hydraulic conductivity, cation exchange capacity, and reduces bulk density of soil by modifying soil structure and aggregate stability, surface crust formation, water evaporation from the topsoil layer [12]. Besides, residues incorporation can prevent nutrients leaching and increase crop yield and reduce significantly soil greenhouse gases [13]. Therefore, the use of super seeder could be a more sustainable alternative to conventional RWCS. Furthermore, the soil biological properties viz., microbial biomass, urease activity, dehydrogenase and alkaline phosphatase can be enhanced through incorporation of residues [14].
Rice residues are a potential source of organic carbon and plant nutrients to enhance the soil organic matter dynamics, nutrient cycling, and soil physical environment. Rice straw contains around 0.7% N, 0.23% P and 1.75% K and it is also an important source of micronutrients (Zinc) and rich in Silicon [15]. Soil enzymes are a major index for soil microbial activity and soil organic carbon status [16], particularly α- and β-glucosidase and urease [17]. Moreover, it has been reported that urease activity is very crucial for soil N metabolism and it can be positively enhanced by the application of organic manure and chemical N fertilizers [18, 19]. Application of inoculum consortium under rich incorporated organic residues could provide substrate availability, moisture, and temperature for better microbial growth [20]. The use of bio-decomposer i.e., inoculum consortium applied for crop residue decomposition, is an interesting way to enhance soil microbial population.
The development of crop yield can be determined through the accumulation and partitioning of dry matter [21]. Further, it has been reported that, the accumulation, distribution, and remobilization of dry matter in plant organs differs with different management practices such as fertilization [22]. In wheat, it has been reported that during the grain filling period, the dry matter for grain is mainly supported by the vegetative organs, and strongly influences the grain yield formation [23]. Therefore, the vegetative organs (non-grain components) growth post flowering in wheat is very crucial to carry out the photosynthetic activity and continue to support the translocation of soluble carbohydrate [24]. In addition, the supply of macronutrients particularly nitrogen (N) greatly influenced the production and distribution of dry matter [25]. The variation in N uptake is primarily associated with the dry matter [26] apart from the soil N status and other management practices.
Considering the great significance of in-situ management of available nutrient resources, the present study was performed under field conditions during 2020–21 and 2021–22 to (i) assess the effect of bio-decomposer, nitrogen level, and nitrogen split on productivity of wheat crop under rice straw incorporation, (ii) find out the N immobilization status through its uptake and content in plants coupled with soil urease activity, and iii) assess dry matter and nitrogen accumulation, remobilization, and contribution to grain yield and N.
2 Materials and methods
2.1 Experimental site description and climatic conditions
A fixed-plot field experiment was carried out during the winter (rabi) season for two years (2020–21 and 2021–22) at Norman E. Borlaug Crop Research Centre of G.B. Pant University of Agriculture and Technology, Pantnagar, Uttarakhand, India (29° N; 79°3′ E; 244 m. a.s.l.). The region falls under sub-humid, sub-tropical climatic zone, on the foot-hills of Himalayas (Tarai region). The region experiences warm and humid summer (April-June), with cold winter (November-February). Generally, south-west monsoon commences in the second fortnight of June and continues sparsely till September. The highest weekly mean rainfall are 18.6 mm and 54.7 mm during January in both the years. The daily average air temperature during the experimental period (December–April) varies from 1.0 to 9.0 °C (min.) and 30–43 °C (max.), respectively and the crop received an average of 6.2 sunshine hour (Supplementary Table 1). The soil of the experimental site was sandy clay loam with 49.4, 27.3, and 23.3% sand, silt, and clay, respectively, typic Ustochrept classification, soil pH of 7.2 [27], EC of 0.31 dSm−1 [28], soil organic carbon − 0.80% [29], available nitrogen 0.11 g kg−1 [30], available phosphorous 0.01 g kg−1 [31], and available potassium 0.08 g kg−1 [27].
2.2 Cultural operation, design, and crop management
The preceding Kharif rice was harvested by combine harvester, and rice straw equivalent to 7 t ha−1 was retained and spread uniformly in all the plots. The experiment was laid in split-split-plot design with main-plot: without (B0) and with (B1) application of bio-decomposer; nitrogen levels 150 (N1) and 180 (N2) kg ha−1 in sub-plot; and three N splits (%) in sub-sub-plot viz., 50: 25:25 (S1), 40: 30: 30 (S2), and 30:35:35 (S3) and replicated thrice with gross plot size 8.8 m × 5.0 m = 44.0 m2. The wheat variety in the present study (PBW 373) was a late sown variety developed by Punjab Agricultural University, India. The details of the cropping history and treatment are given in Tables 1 and 2, respectively. Prior to sowing (4–5 days) of wheat crop, the microbial formulation (pusa bio-decomposer) (25 L ha−1) was prepared and then sprayed uniformly over the retained residue. The microbial formulation used in the present study was developed by the ICAR-Indian Agricultural Research Institute, New Delhi, India containing beneficial microorganisms which enhance the enzymatic activities in soil and straw for fast decomposition of biomass [32]. The crop was sown 20 days after the harvest of the preceding rice using super-seeder (rota-till-drill), wherein the residue is incorporated to the depth of about 0.15 m while the seed was drill to a depth of 0.05–0.07 m in a single operation. The crop was sown on 24th November (2020–21), and 26th November (2021–22) at a row-spacing of 0.20 m and seed rate of 110 kg ha−1. Pre-sowing irrigation was not applied since good amount of residual moisture was present. The fertilizer, nitrogen (N), phosphorus (P), and potassium (K) were applied in the form of NPK mixture (12:32:16% of N, P and K) and urea (46% N). Two levels of nitrogen as 150 kg ha−1 (recommended) and 180 kg ha−1 were taken to test the optimization N rates under residues incorporation. Full dose of P and K were applied as basal, while N was applied in three different splits ratios as described in Table 2. Irrigation was applied based on the critical crop growth stages and taking rainfall into consideration. A total of four and three irrigations were given in the first (2020–21) and second year (2021–22) respectively (Supplementary Table 2). Both years, a post-emergence (35 DAS) herbicide clodinafop-propargyl 15% + metsulfuron methyl 1% (broad spectrum) at 400 g a.i. ha−1 was applied. In the second year (2021–22), propiconazole 25 EC @ 500 mL ha−1 was sprayed during the active tillering stage to control yellow rust (Puccinia triticina f.sp. tritici). The other diseases and pest were controlled following the standard management practices.
2.3 Soil sampling
A composite soil sample was collected randomly from a depth of 0.00–0.15 m, both initially before wheat sowing and at wheat harvest, using a tube auger, with 5 replicates in all plots. The sample was shade dried, processed, passed through 80 mesh sieves, and analyzed for determination of various soil chemical properties viz., pH, EC, OC, available N, P and K. Further, to assess soil urease activity, fresh soil samples were collected from a depth of 0.00–0.15 m with 5 replications at 30 and 60 DAS in both years. Similarly, for soil microbial biomass carbon (SMBC) analysis, fresh soil samples from the same depth with 5 replications were collected at crop maturity during the second season.
2.4 Nitrogen concentration and uptake in plants
The plant samples for N concentration and its uptake were collected from an area of 1 m2 at 30, 60 DAS, anthesis, and harvest. The samples were shade-dried for 48 h, then oven-dried at 65 °C to a constant weight and the oven-dried biomass was recorded. The dried samples were grinded through Wiley-mill having 40-mesh sieve, and appropriate amount (0.5 g) of the ground sample with catalyst mixture was digested on hot plate in processing lab. Subsequently, the amount of NH4-N liberated during distillation with an alkali (NaoH) was absorbed by boric acid containing mixed indicator and titrated against sulphuric acid. This method, outlined by [27] was used to determine the total N concentration. The total shoot N uptake was worked out by multiplying the N concentration with the shoot biomass for all the stages of the crop. The nitrogen accumulation, remobilization, remobilization efficiency, and contribution to grain nitrogen was calculated as given by [33, 34]:
-
1)
Post-anthesis nitrogen accumulation (Post-NA, g m−2) = Total above-ground N uptake at maturity- Total N uptake at anthesis
-
2)
Nitrogen remobilization (NR, g m−2) = Total N uptake at anthesis – Vegetative N uptake at maturity
-
3)
Nitrogen remobilization efficiency (NRE, %) = (NR/ Total N uptake at anthesis) × 100
-
4)
NR contribution to grain N (%) = (NR / grain N) × 100
2.5 Dry matter accumulation
The plant samples were collected at 30 DAS, 60 DAS, anthesis, and harvest in both years. In each plot, the plants were cut at the base from an area of 1 m2 in the mid-rows. The samples were placed in a perforated paper bag, air-dried for 48 h, and then oven-dried at 65 °C till constant dry weight. The dried biomass values were used for determining dry matter accumulation, remobilization, remobilization efficiency, and contribution of remobilization to grain yield as described by [35, 36].
-
1)
Post-anthesis dry matter accumulation (Post-DMA, g m−2) = Total above-ground dry matter at maturity–dry matter at anthesis
-
2)
Dry matter remobilization (DMR, g m−2) = Dry matter at anthesis–vegetative dry matter at maturity
-
3)
Dry matter remobilization efficiency (DMRE, %) = (DMR/Dry matter at anthesis) × 100
-
4)
DMR contribution to grain yield (%) = (DMR / grain yield) × 100
2.6 Yields and yield components
The wheat was harvested during the second fortnight of April in both years. The crop was harvested manually from the net plot (5 m × 4 m, 20 m2) leaving the border rows from both the sides. The harvested samples were sun-dried, weighed for biological yield (total above-ground biomass), threshed using pullman thresher, and the grain yield was recorded for each plot. The straw yield for each plot was calculated by subtracting the grain yield from the biological yield. The yield attributes, such as productive tillers (tillers bearing productive spike), spike weight (average of 10 spikes), grains spike−1 (10 spikes average), and 1000-GW (grain weight) were recorded by harvesting the crop from 1 m2 area at maturity.
2.7 Urease activity
The activity of urease was determined following the method of [37] at 30 and 60 DAS. In this method, one gram of fresh soil was combined with 1 ml of toluene and then supplemented with 10 ml of pH 7 buffer solution and 5 ml of 10% urea solution. After shaking and incubating for 3 h at 37 °C, 0.5 ml of the filtrate was taken and mixed with 5 ml of distilled water. This mixture was then treated with 2 ml of phenolate solution and 1.5 ml of sodium hypochlorite solution containing 5% active chlorine. The volume was adjusted to 25 ml with distilled water, and the optical density was measured at 630 nm using a spectrophotometer. The amount of NH4+-N released was calculated by reference- calibrated curve and was expressed as NH4+-N µg per gram dry soil per three hours.
2.8 Soil microbial biomass carbon
Soil microbial biomass carbon in soil samples was estimated following chloroform fumigation extraction procedure as described by [38]. Biomass carbon in soil was calculated from the relationship, Biomass carbon in soil (Bc) = Fc/Kc, Where, Fc = Difference between extractable carbon from fumigated soil and non-fumigated soil. Kc = Efficiency factor, which is 0.45 [39] and express as µg g−1 soil.
2.9 Statistical analysis
The data was analyzed by using analysis of variance technique [40], with SAS 9.4 for testing the significance of difference between any two means wherever F-test was significant. Tukey’s honestly significant difference test (Tukey’s HSD) at 0.05% probability was performed to compare the mean effects.
3 Results
3.1 Weather conditions
The highest mean precipitation of 98.9 mm was received in January 2022 followed by 48.2 mm in February 2022 whereas, the other months of the crop season remained dry. The total amount of rainfall received in 2021 was 25.7 mm, while in 2022 it was 154.7. During the study period, about 90–95% of total rainfall was received between January and February. The average minimum and maximum relative humidity (RH) ranged between 48–89% and 51–89%, respectively in 2020–21 and 2021–22. (Supplementary Table 1).
3.2 Nitrogen concentration and uptake
The shoot nitrogen concentration at 30 DAS remains similar between B0 and B1, while with the N-level, N2 had significantly higher shoot N concentration compared to N1 by 16.5% and 14.5% in 2020–21 and 2021–22, respectively (Table 3). Among the N-splits, maximum shoot N concentration was observed with S1, wherein it was similar to S2 in both years but significantly higher than S3 to the tune of 7% and 6%. Similarly, the shoot N concentration at 60 DAS did not observe significant difference between B0 and B1 in both years, but with N-levels, N2 recorded significantly higher shoot N concentration of 12.9% (2020–21) and 13.4% (2021–22), respectively (Table 4). Further, among the N-splits, S1 had the highest shoot N concentration wherein, it was similar S2 in but statistically more than S3 in both years. The increment of S1 over S3 in shoot N concentration was to the tune of 5% and 5.8% in 2020–21 and 2021–22. The total shoot N concentration at flowering remains similar between B0 and B1 in both years, but with N-level, N2 resulted in significantly higher shoot N concentration than N1 by 12.9% and 15.8%, respectively in 2020–21 and 2021–22. Further, among the N-splits, maximum shoot N concentration was observed with S1 in both years, wherein it was significantly higher than S3 to the tune of 7.7% (2020–21) and 8.0% (2021–22) but at par with S2. Similarly, at harvest total N concentration in straw with B0 and B1 were at par, but with N-level, N2 had significantly higher content of 18.3% and 21.2% over N1 in 2020–21 and 2021–22. While, among the N-splits S1 resulted in highest N concentration which was similar to N2 but significantly more than S3 to the tune of 19.4% (2020–21) and 21.2% (2021–22). Furthermore, the N concentration in grain did not have significant impact with or without bio-decomposer application in both years. Nevertheless, with N-level, N2 had significantly higher N concentration in grain over N1 by 13.9% and 15.5% in 2020–21 and 2021–22. While, among the N-splits, maximum grain N concentration was observed with S1 in both years, wherein it was significantly more than S2 and S3 by 6.9% and 13.4% in 2020–21, while in 2021–22 it was significantly higher by 5.7% and 8.6%, respectively (Table 4).
The total shoot N uptake in 2020–21 at 30 and 60 DAS under B1 was significantly higher than B0 by 10.3% and 8.9%, while with N-level N2 resulted in statistically higher N-uptake to the tune of 24.2% and 17.7% at 30 and 60 DAS. Among the N-splits, N-uptake at 30 DAS with S1 was significantly higher by 10–15% over S2 and S3, while at 60 DAS S1 was significantly higher by 12.7% over S3 but similar to S2 (Fig. 1a). Further, in 2021–22 at 30 DAS B0 and B1 were found similar in N-uptake, while at 60 DAS B1 was statistically higher by 8.9% over B0. While, between N-level, N2 had significantly higher N uptake both at 30 and 60 DAS to the tune of 21.3% and 18.5%, respectively. Further, among the N-splits, highest N uptake was recorded with S1 both at 30 and 60 DAS which was significantly higher by 9–17% and 5–13% compared to S2 and S3 (Fig. 1b). The total N uptake in shoot at flowering with B1 was significantly higher than B0 by 7.3% and 5.5%, in 2020–21 and 2021–22. With, N-level, N2 resulted in significantly higher N uptake to the tune of 18.4% (2020–21) and 20.7% (2021–22), while among the N-splits maximum N uptake was recorded with S1 in both years, wherein it was significantly more than S3 by 12.8% in 2020–21, and 8% and 13.1% than S2 and S3 in 2021–22 (Fig. 2). Further, the N uptake in straw at harvest remains similar between B0 and B1 in 2020–21, while in 2021–22 significantly higher N uptake was observed with B1 (6.5%) over B0. While, with N-level, N2 resulted in significantly higher N uptake of 25.3% (2020–21) and 21.6% (2021–22) over N1. Among the N-splits, S1 observed maximum N uptake in both years which was significantly higher than S2 and S3 by 13.6% and 22.6% in 2020–21, and 17.5% and 25.9% in 2021–22 (Fig. 3). In addition, the N uptake in grain remains similar between B0 and B1 in 2020–21, but B1 had significantly higher uptake of 9% in 2021–22. While, the N uptake with N2 had significantly 20.2% and 20.3% more compared to N1 in 2020–21 and 2021–22, respectively. The highest N uptake in grain was recorded with S1 among the N-splits, wherein it was significantly more than S2 and S3 to the tune of 11–19% in 2020–21 and 10–13% in 2021–22 (Fig. 3).
Wheat N uptake (g m−2) of shoot determined at 30 and 60 days after sowing (DAS) in (a) 2020–21 and (b) 2021–22 season, as effected by the bio-decomposer application (B0- without application; B1- with bio-decomposer application), the N rate (N1- nitrogen at 150 kg ha−1; N2- nitrogen at 180 kg ha−1) and the N splitting (S1- 50:25:25; S2- 40:30:30; S3-30:35:35) (N applied at sowing, crown root initiation, and booting). Similar letters above the bar are not significantly different at p ≤ 0.05 as analysed by Tukey’s honest significant difference (Tukey’s HSD) test
Wheat N uptake (g m−2) of shoot determined at flowering in 2020–21 and 2021–22 seasons, as effected by the bio-decomposer application (B0- without application; B1- with bio-decomposer application), the N rate (N1- nitrogen at 150 kg ha−1; N2- nitrogen at 180 kg ha−1) and the N splitting (S1- 50:25:25; S2- 40:30:30; S3-30:35:35) (N applied at sowing, crown root initiation, and booting). Similar letters above the bar are not significantly different at p ≤ 0.05 as analysed by Tukey’s honest significant difference (Tukey’s HSD) test
Wheat N uptake (g m−2) at harvest both in straw and grain in 2020–21 and 2021–22 seasons, as effected by the bio-decomposer application (B0- without application; B1- with bio-decomposer application), the N rate (N1- nitrogen at 150 kg ha−1; N2- nitrogen at 180 kg ha−1) and the N splitting (S1- 50:25:25; S2- 40:30:30; S3-30:35:35) (N applied at sowing, crown root initiation, and booting). Similar letters above the bar are not significantly different at p ≤ 0.05 as analysed by Tukey’s honest significant difference (Tukey’s HSD) test
3.3 Post-anthesis N accumulation, remobilization, and contribution to grain N
The post-anthesis N accumulation (post-NA) with B0 and B1 were at par in both years. While, the Post-NA with N2 had significantly 4.5 g m−2 and 3.5 g m−2 more compared to N1 in 2020–21 and 2021–22 (Table 5). Among the N-splits, S1 had highest post-NA which was significantly higher than S2 and S3 by 17–27% in 2020–21, and S3 by 20.5% in 2021–22. The N remobilization under B1 was significantly higher by 8.1% in 2020–21 but remains similar in 2021–22. Further, between the N-level, N2 was significantly higher than N1 by 13.6% and 20.2% in 2020–21 and 2021–22, respectively. While, N remobilization remains at par among the N-splits in both years. The N remobilization efficiency (NRE) in both years were at par between B0 and B1, while with N1 the NRE was enhanced by 5.3% over N2 in 2020–21 but were found similar in 2021–22. Further, among the N-splits, there observed no significant difference in 2020–21, while in 2021–22 significantly higher NRE of 10.7% was recorded with S3 compared to S1 but remains at par with S2. The contribution of N remobilization to grain N among the treatments did not observe significant difference in both years.
3.4 Dry matter accumulation
The data pertaining to dry matter accumulation of wheat at 30 and 60 DAS is given in Supplementary Table 3, while for anthesis and harvest under different treatments have been furnished in Table 6. The application of bio-decomposer did not affect the dry matter accumulation at 30 and 60 DAS in both years. While, between the nitrogen levels, N2 produced significantly higher dry matter of 7.9–9.7% (30 DAS) and 5.8–5.9% (60 DAS), respectively over N1. Among the N-splits, S1 resulted in statistically higher dry matter accumulation compared to S3 but at par with S2 in both the stages of the crop and years. The increment of S1 over S3 was to the tune of 8.5–12.2% (30 DAS) and 7.8–7.9% (60 DAS). Further, the dry matter accumulation at flowering did not differ with and without bio-decomposer application, but with the N-level, N2 had significantly higher dry matter of 5.8% and 5.5% in 2020–21 and 2021–22, respectively over the N1. Similarly, among the N-splits, S1 produced maximum dry matter in both years which was at par with S2 but significantly more than S3 by 5.2% (2020–21) and 7.1(2021–22). Similarly, at harvest the total dry matter remains similar to each other between B0 and B1 in both years, but with N-level N2 had significantly higher dry matter accumulation of 5.2% and 5.6% in 2020–21 and 2021–22. Among the N-splits, maximum dry matter accumulation was recorded with S1 in both years, wherein it was similar to S2 but significantly higher than S3 by 5.8% and 5.9% in 2020–21 and 2021–22, respectively (Table 6).
3.5 Post-anthesis dry matter accumulation, remobilization, and contribution to grain yield
Bio-decomposer did not influence the post-anthesis dry matter accumulation (Post-anthesis-DMA) in both years (Table 6). However, between the N-level, N2 had significantly 11.3% more post-DMA over N1 in 2020–21, though it remains similar in 2021–22. Further, among the N-splits, maximum post-DMA was recorded with S1 in both years, wherein it was significantly higher by 12.1% and 7.4% compared to N3 in 2020–21 and 2021–22, but at par with S2. The dry matter remobilization (DMR) in both years remains similar with or without bio-decomposer application, while with the N-level, N2 had significantly higher DMR to the tune of 21.4% and 26.5% compared to N1 during 2020–21 and 2021–22. Further, among the N-splits, a significantly higher DMR was observed with S1, wherein it was at par with S2 but statistically (p < 0.05) more than S3 by 33.2% (2020–21) and 32.6% (2021–22), respectively. The dry matter remobilization efficiency (DMRE) with B1 was significantly more than B0 in 2020–21, but remain similar in 2021–22. While, the DMRE with N2 was significantly higher compared to N1 in both years, and among the N-splits, the highest DMRE was recorded with S1 which was at par with S2 but significantly more than S3 in both years. Further, the contribution of dry matter remobilization to grain yield remains similar for all the treatments irrespective of the years.
3.6 Yield components
The number of effective tillers with and without bio-decomposer application were at par in both years, while between the N-levels, N2 produced significantly higher effective tillers of 5.7% in both years compared to N1 (Table 7). Further, among the N-splits, the effective tillers with S1 was maximum in both years, wherein it was at par with S2 but significantly more than S3 by 7.3% in 2020–21, while in 2021–22 S1 resulted in significantly higher effective tillers over S2 and S3 to the tune of 2.5–7.3%. The spike length remains similar between the treatments during both years of study. Further, the number of grains spike−1 did not differ with the bio-decomposer in both years. Nevertheless, the grains spike−1 with N2 produced 10.8% (2020–21) and 11.2% (2021–22) more compared to N1. The maximum grain spike−1 was recorded with S1 among the N-splits, wherein it remains similar to S2 in both years but significantly higher than S3 to the tune of 16.5% and 14.4% in 2020–21 and 2021–22, respectively. The grain weight spike−1 was not influenced by bio-decomposer in both years, but between the N-levels, N2 produced significantly heavier grain weight over N1 by 12.7% and 12.4% in 2020–21 and 2021–22. While, among the N-splits, S1 resulted in heaviest grain weight spike−1 in both years, wherein it was statistically at par with S2 but significantly more than S3 by 17.6% (2020–21) and 15.4% (2021–22). The 1000-grain weight in both years remains similar between the treatments.
3.7 Grain and straw yields
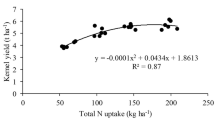
Bio-decomposer did not have significant impact on grain yield of wheat in both years, but with N-levels, N2 resulted in significant yield improvement of 8% and 8.5% respectively in 2020–21 and 2021–22 compared to N1 (Table 8). Further, among the N-splits, S1 produced maximum grain yield which was similar to S2, but significantly higher than S3 to the tune of 10.1% (2020–21) and 11.8% (2021–22). Similarly, straw yield in both years remains at par with and without bio-decomposer, while with N-levels, N2 had significantly higher straw yield of 7.4% and 7.8% over N1 in 2020–21 and 2021–22. Among the N-splits, the highest straw yield was recorded with S1 in both years, wherein it was statistically higher than S3 by 6.9% and 9.9% in 2020–21 and 2021–22, though at par with S2. In addition, the biological yield did not observe significant impact with the bio-decomposer though there is 4.4% and 4.7% higher with B1 over B0 in 2020–21 and 2021–22. The N-levels had significant impact on biological yield with N2 resulted in significantly higher yield of 7.7% (2020–21) and 7.6% (2021–22) compared to N1. Furthermore, the N-splits with S1 produced maximum biological yield in both years, which was at par to S2 but significantly higher than S3 by 8.2% and 10.6% in 2020–21 and 2021–22, respectively.
3.8 Urease activity and soil microbial biomass carbon
The urease activity (UA) in the upper soil layer (0.00–0.15 m) at 30 and 60 DAS with B1 was significantly higher by 6.8% and 6.2% over B0 in 2020–21 (Fig. 4a). While, between the N-level, N2 had significantly higher UA of 10.8% and 9.4% compared to N1, while among the N-splits, S1 resulted in maximum UA wherein it was at par with S2 but significantly more than S3 to the tune of 8.2% and 12.3% at 30 and 60 DAS. Similarly, in 2021–22 (Fig. 4b), the soil UA in the soil depth of 0.00–0.15 m with B1 was statistically higher by 7.8% and 9.1% at 30 and 60 DAS. Further, with N2 the UA was increased by 9.5% and 8.8% over N1, respectively at 30 and 60 DAS. Among the N-splits, S1 produced the highest UA which was significantly more than S3 by 13.8% and 12.8%, but found similar to S2 at 30 and 60 DAS. Furthermore, the SMBC at harvest in 2021–22 (Fig. 5) from 0.00–0.15 m soil depth with B1 was significantly higher than B0 by 8.9%. Indeed, different N-level and N-splits did not have significant impact on soil-MBC.
Soil urease activity determined at 30 and 60 DAS in (a) 2020–21 and (b)2021–22 seasons, as effected by the bio-decomposer application (B0- without application; B1- with bio-decomposer application), the N rate (N1- nitrogen at 150 kg ha−1; N2- nitrogen at 180 kg ha−1) and the N splitting (S1- 50:25:25; S2- 40:30:30; S3-30:35:35) (N applied at sowing, crown root initiation, and booting). Similar letters above the bar are not significantly different at p ≤ 0.05 as analysed by Tukey’s honest significant difference (Tukey’s HSD) test
# SMBC- Soil microbial biomass carbon determined after wheat harvest in 2021–22 season, as effected by the bio-decomposer application (B0- without application; B1- with bio-decomposer application), the N rate (N1- nitrogen at 150 kg ha−1; N2- nitrogen at 180 kg ha−1) and the N splitting (S1- 50:25:25; S2- 40:30:30; S3-30:35:35) (N applied at sowing, crown root initiation, and booting). Similar letters above the bar are not significantly different at p ≤ 0.05 as analysed by Tukey’s honest significant difference (Tukey’s HSD) test
4 Discussion
4.1 Nitrogen concentration and uptake and post-anthesis accumulation
The incorporation of rice straw results in soil nitrates immobilization due to high C:N ratio, thereby reducing the plants N uptake leading to N deficiency [41, 42]. Hence, to accelerate the decomposition process, the application of bio-decomposer (beneficial microbial formulation) coupled with a timely supply of optimum N fertilizer could create a congenial environment for better crop growth and development. The lower N concentration and uptake with N1(150 kg ha−1) as compared to N2 (180 kg ha−1) could be due to low N availability in rice crop residues, resulting in temporary immobilization of inorganic N [43, 44], wherein the soil decomposers have to obtain the inorganic N from the N- fertilizer applied for building their cellular components [44, 45]. Meanwhile, with a higher rate of N-application (N2), the excess nitrogen could meet the N requirement of the microbes for the breakdown of the organic matter resulting in higher available N to crop particularly in the early stages. Further, higher proportion of N as basal might have subdued the temporary N lock-up in the soil [46] thereby enhancing the availability of N to the crop and improving its concentration and uptake.
Further, previous studies reported that the total N concentration was primarily associated with the variation in dry matter [26, 47]. The N concentration in grain at maturity has been propounded to be greatly influenced by the remobilization of N accumulated before anthesis [48, 49], while its contribution from post-anthesis uptake depends on the soil N status, genotype, and water availability during the grain filling period [50,51,52].
4.2 Dry matter accumulation and post-anthesis dry matter accumulation, remobilization, and contribution to grain yield
The dry matter and nutrient accumulation and remobilization for wheat grown with an increased N rate and higher basal N were comparatively higher than the treatments with recommended dose of nitrogen and lower nitrogen at basal which could lead to a larger accumulation of assimilates available for remobilization with higher assimilate demand during the grain filling. In addition, dry matter partitioning and remobilization have been noted to be affected by factors such as climatic conditions, varieties, planting patterns, and management practices [53]. The higher dry matter remobilization (DMR) could be due to an increase in mineralization of organic residue which increases the nutrient concentrations in the soil and attributes to better early crop establishment and improved root growth and volume thereby enhancing the plant uptake resulting in increasing cellular activities ultimately lead to a higher synthesis of photosynthates remobilization. The total dry matter remobilized under the higher nitrogen dose (N2) was enhanced by 21.4% and 26.5% in both years (Table 6). In wheat, current photosynthesis, and the remobilization of reserves from the stem and other plant parts provide the carbon required during the grain filling phase [54].
The dry matter remobilization efficiency (DMRE) in the present study varied with different treatments. This could be the result of decreased in dry matter accumulation and the photosynthetic rate at anthesis due to stress at the initial stage of the crop without decomposer and lesser dose at basal application, yet improved the remobilization of photosynthates to the spikes. Similar findings under the water stress conditions were reported by [24, 55]. Further, it has been reported in wheat that the contribution from remobilization of dry matter to grain yield varies from 7 to 57% [56], which was in line with our study, wherein it ranged between 15–29% among the treatments (Table 6). Nevertheless, the contribution of dry matter remobilization to grain yield among the treatments was found to be at par with each other. Nitrogen stress, particularly due to the temporary lock-up of applied N during the initial stage of the crop has been found to decrease the remobilization and contribution to grain yield [57, 58]. Meanwhile, because of the stress during post-anthesis, the accumulation and assimilation might have been reduced, as it is demonstrated in the current study, where post-anthesis dry matter accumulation with the treatments B0, N1, S3 decreased by 15 to 29% (Table 6). Hence, more the dry matter and N accumulated before anthesis, higher the rates of dry matter translocation to grain and the risk of net N losses at maturity [59].
4.3 Yield components and yields
Application of bio-decomposer caused grain yield benefits of 4.6% over without bio-decomposer application, which could be attributed to hastening the composting process of RS through microbial bioaugmentation [60, 61] thereby augmenting the nutrients available to the crop and eventually resulting in an improved yield of the crop. In addition, the combined use of RS along with inorganic fertilizers particularly N has been reported to enhance the crop yield under the RWCS [62]. Previous studies revealed that the application of higher N at 180 kg ha−1 over 60 kg ha−1 when the RS is incorporated had a lesser reduction in wheat yield [63]. The present study further confirmed that higher N needs to be applied when residue is incorporated to achieve greater yield, as N2 had 9% (two years mean) higher grain yield compared to N1. The higher rate of N application when residue is incorporated is to overcome the temporary immobilization of applied fertilizer and makes it more available to the crop in early growth stages [64]. Subsequently, the higher grain yield under N2 and S1 could be attributed to higher nitrogen mineralization and overcoming the stress (N immobilization) at the initial stage of the crop thereby increasing the dry matter accumulation (Supplementary Table 3), and yield components viz., number of grains/spike and grain weight/spike (Table 7). Similar findings were reported by [65, 66]. Furthermore, N dose and N split influenced the number of grains/spike significantly during both years. The reason could be better mineralization of inorganic residues in the initial growing period resulted in supplying of available nutrients to the crop and nutrient uptake which might help in the efficient partitioning and allocation of photosynthates from source to sink [67]. Moreover, the increase in grain weight/spike was observed with the higher dose of nitrogen [68]. The lower grain yield in the second year (Table 7) as compared to the first year could be due to heavy rain (Supplementary Table 1) during the anthesis of the crop which led to chlorosis, and reduced photosynthetic ability of the crop. Besides, due to water logging, leaching and denitrification of N caused chlorosis and thereby reduced photosynthetic capacity and N availability to the crop [69]. The higher straw yield with higher N dose (N2) and 50% N (S1) applied as basal may be ascribed to enhance the growth of the crop, especially the tillers number, which ultimately culminates in straw yield. Increasing the recommended N dose by 30 kg ha−1 and increasing the proportion of total N as basal i.e., 50%, helped the crop to off-set the N stress experienced during the initial phase in residue loaded plots [67, 70, 71]. The higher straw yield with N2 and S1 is clearly outlined in this study (Table 8).
4.4 Urease activity and soil microbial biomass
Urease activity in soil was influenced by the application of bio-decomposer, nitrogen doses, and N-splits. Soil urease activity has been reported to increase with conservation tillage and residue incorporation [72]. The activity of the urease enzyme is directly proportional to the dose of soil applied urea [7, 73], and therefore, with the higher nitrogen dose more is the activity of the urease enzyme. It is a general observation that the activity of urease increased up to 60–80 DAS and decreases thereafter [74]. The probable reason might be due to N-splits of nitrogen which had been applied up to 70–80 DAS (i.e., booting stage in this study), and in response to this the activity of urea hydrolyzing microbes increased, thereby increasing the urease enzyme as well [75].
The functions of an agro-soil ecosystem are driven by interactions between soil organic matter and microbial communities, and the concept of soil microbial biomass can help us comprehend these interactions [76]. Soil microbial biomasses assist in regulating soil fertility through the process of immobilization and mineralization for nutrients like carbon and nitrogen [77] and are identified as sensitive indicators of crop management practices [78, 79]. Crop residue acts as a readily available food source and provides a wide range of nutrients and congenial environment to microbes which resulted in higher microbial biomasses [80]. Thus, resulting in higher SMBC (8.9%) in the soil with the application of bio-decomposer compared to without bio-decomposer application. Our result is concurrent with findings reported by [7]. Considering this, bio-decomposer treatment becomes essential for the long-term viability of the soil and ultimately the intensive RWCS, in addition to increasing crop yield.
5 Conclusion
With the innovative invention of technology such as super-seeder, the illicit and treacherous practice of residue burning could significantly reduce by adopting the alternative eco-friendly and sustainable management of crop residues without delaying the seeding of the next crop particularly in RWCS. Our study is unique in optimizing both rate of fertilizer N and its scheduling to overcome the limitation of immobilization that results to N stress during initial stage of the crop for higher and sustainable productivity. The results of the field experiments conducted in wheat sown by super-seeder revealed that in combine harvested rice field with residue retention, wheat should be supplied with 180 kg N ha−1 and N-splitting as 50%:25%:25% (Basal: CRI: Booting) for improving the dry matter accumulation, yield attributes, grain and straw yields and higher N-mineralization in soil during initial stage of the crop. Besides, use of bio-decomposer could be taken as a new path for efficient use of nitrogen, as it helped in improving soil urease activity and SMBC and early release of nitrogen thereby, improved the grain yield. Further investigations for long-term studies in nutrients content of soil and plants are necessary.
Data availability
The original contributions presented in the study are included in the article/supplementary material. Further inquiries can be directed to the corresponding author.
References
USDA (United States Department of Agriculture). 2019. http://www.fasusdagov.
Brar AS, Sharma P, Kahlon CS, Walia US. Influence of rice residue management techniques and weed control treatments on soil available plant nutrients in rice-wheat cropping system. Amer J Plant Sci. 2019. https://doi.org/10.4236/ajps.2019.101006.
Benbi DK, Senapati N. Soil aggregation and carbon and nitrogen stabilization in relation to residue and manure application in rice–wheat systems in northwest India. Nutr Cycl Agroecosyst. 2010. https://doi.org/10.1007/s10705-009-9331-2.
Mahajan G, Singh K, Gill MS. Scope for enhancing and sustaining rice productivity in Punjab (food bowl of India). Afr J Agric Res. 2012;7(42):5611–20.
Bhushan L, Ladha JK, Gupta RK, Singh S, Tirol-Padre A, Saharawat YS, Gathala M, Pathak H. Saving of water and labor in a rice–wheat system with no-tillage and direct seeding technologies. Agron J. 2007. https://doi.org/10.2134/agronj2006.0227.
Tripathi RP, Sharma P, Singh S. Tilth index: an approach to optimize tillage in rice–wheat system. Soil Tillage Res. 2005. https://doi.org/10.1016/j.still.2004.03.004.
Jat HS, Choudhary M, Datta A, Yadav AK, Meena MD, Devi R, Gathala MK, Jat ML, McDonald A, Sharma PC. Temporal changes in soil microbial properties and nutrient dynamics under climate smart agriculture practices. Soil Tillage Res. 2020. https://doi.org/10.1016/j.still.2020.104595.
Satyendra T, Singh RN, Shaishav S. Emissions from crop/biomass residue burning risk to atmospheric quality. Int Res J Earth Sci. 2013;1(1):1–5.
Mooventhan P, Singh SR, Venkatesan P, Dixit AN, Sharma KC, Sivalingam PN, Kumar AG, Singh U, Kaushal P. Happy seeder-a promising technology in conservation agriculture. Harit Dhara. 2018;1(1):27–9.
Singh P, Gautam BS, Yadav RK. Performance evaluation of happy seeder for sowing wheat crop in combined harvested. Int J Agric Eng. 2017. https://doi.org/10.15740/HAS/IJAE/10.2/643-646.
Kaur M, Malik DP, Malhi GS, Sardana V, Bolan NS, Lal R, Siddique KH. Rice residue management in the indo-Gangetic plains for climate and food security. Rev Agron Sustain Dev. 2022. https://doi.org/10.1007/s13593-022-00817-0.
Bechini L, Costamagna C, Zavattaro L, Grignani C, Bijttebier J, Ruysschaert G. Barriers and drivers towards the incorporation of crop residue in the soil. Analysis of Italian farmers’ opinion with the theory of planned behaviour. Italian J Agron. 2015. https://doi.org/10.4081/ija.2015.663.
Memon MS, Guo J, Tagar AA, Perveen N, Ji C, Memon SA, Memon N. The effects of tillage and straw incorporation on soil organic carbon status, rice crop productivity, and sustainability in the rice-wheat cropping system of eastern China. Sustain. 2018. https://doi.org/10.3390/su10040961.
Jat HS, Datta A, Sharma PC, Kumar V, Yadav AK, Choudhary M, Choudhary V, Gathala MK, Sharma DK, Jat ML, Yaduvanshi NP. Assessing soil properties and nutrient availability under conservation agriculture practices in a reclaimed sodic soil in cereal-based systems of North-West India. Archives Agron Soil Sci. 2018. https://doi.org/10.1080/03650340.2017.1359415.
Goswami SB, Mondal R, Mandi SK. Crop residue management options in rice–rice system: a review. Archives Agron Soil Sci. 2020. https://doi.org/10.1080/03650340.2019.1661994.
Singh G, Kumar D, Marwaha TS, Singh AK. Influence of tillage, water regimes and integrated nitrogen management practices on soil quality indices in rice (Oryza sativa L.) in the Indo-Gangetic plains. Arch Agron Soil Sci. 2009. https://doi.org/10.1080/03650340802553591.
Bandick AK, Dick RP. Field management effects on soil enzyme activities. Soil Bio Biochem. 1999. https://doi.org/10.1016/S0038-0717(99)00051-6.
Mikanova O, Javurek M, Simon T, Friedlova M, Vach M. The effect of tillage systems on some microbial characteristics. Soil Tillage Res. 2009. https://doi.org/10.1016/j.still.2009.05.010.
Gupta RK, Sidhu HS. Nitrogen and residue management effects on agronomic productivity and nitrogen use efficiency in rice–wheat system in Indian Punjab. Nutrient Cycling Agroecosyst. 2009. https://doi.org/10.1007/s10705-008-9233-8.
Ghimire B, Ghimire R, VanLeeuwen D, Mesbah A. Cover crop residue amount and quality effects on soil organic carbon mineralization. Sustain. 2017. https://doi.org/10.3390/su9122316.
Zheng YF, Xu JX, Wu RJ. Effects of different shading condition on dry matter accumulation and distribution of winter wheat. Chin J Agrometeorol. 2013;34(1):64–73.
Yan S, Wu Y, Fan J, Zhang F, Qiang S, Zheng J, Xiang Y, Guo J, Zou H. Effects of water and fertilizer management on grain filling characteristics, grain weight and productivity of drip-fertigated winter wheat. Agric Water Managt. 2019. https://doi.org/10.1016/j.agwat.2018.12.019.
Huang X, Wang C, Hou J, Du C, Liu S, Kang J, Lu H, Xie Y, Guo T, Ma D. Coordination of carbon and nitrogen accumulation and translocation of winter wheat plant to improve grain yield and processing quality. Sci Rep. 2020. https://doi.org/10.1038/s41598-020-67343-5.
Zhang H, Han K, Gu S, Wang D. Effects of supplemental irrigation on the accumulation, distribution and transportation of 13C-photosynthate, yield and water use efficiency of winter wheat. Agric Water Managt. 2019. https://doi.org/10.1016/j.agwat.2018.12.028.
Yan S, Wu Y, Fan J, Zhang F, Zheng J, Qiang S, Guo J, Xiang Y, Zou H, Wu L. Dynamic change and accumulation of grain macronutrient (N, P and K) concentrations in winter wheat under different drip fertigation regimes. Field Crops Res. 2020. https://doi.org/10.1016/j.fcr.2020.107767.
Masoni A, Ercoli L, Mariotti M, Arduini I. Post-anthesis accumulation and remobilization of dry matter, nitrogen and phosphorus in durum wheat as affected by soil type. Eur J Agron. 2007. https://doi.org/10.1016/j.eja.2006.09.006.
Jackson ML. Soil chemical analysis, Pentice hall of India Pvt. Ltd., New Delhi, India. 1973; 498:151–4.
Bower CA, Wilcox LV. Soluble salts. In: Black CA, editor. Methods of soil analysis, agronomy Madison. Wisconsin: Amer Society of Agron; 1965. p. 933–51.
Black CA. Methods of soil analysis: part 2 chemical and microbiological properties agronomy monograph. Soil Sci Society Amer. 1965;92:1367–78.
Subbaiah BV. A rapid procedure for estimation of available nitrogen in soil. Curr Sci. 1956;25:259–60.
Olsen SR. Estimation of available phosphorus in soils by extraction with sodium bicarbonate. Washington, DC: US Department of Agriculture; 1954.
Manu SM, Singh YV, Shivay YS, Shukla L, Sharma VK, Saha ND, Shekhawat K, Bandopadhyay KK, Gouda HS. Nitrogen budgeting under the influence of in situ rice residue management options in rice (Oryza sativa)–wheat (Triticum aestivum) cropping system. Indian J Agric Sci. 2023. https://doi.org/10.56093/ijas.v93i2.129506.
Zhang Y, Xue Q, Li J, Huang J, Yao D, Wang Z. Dry matter and nitrogen accumulation and remobilization in wheat as affected by genotype and irrigation. J Plant Nutr. 2017. https://doi.org/10.1080/01904167.2016.1264593.
Zhiipao RR, Pooniya V, Kumar D, Biswakarma N, Shivay YS, Dass A, Kumar Bainsla N, Lakhena KK, Pandey RK, Burman A, Bhatia A. Above and below-ground growth, accumulated dry matter and nitrogen remobilization of wheat (Triticum aestivum) genotypes grown in PVC tubes under well-and deficit-watered conditions. Front Plant Sci. 2023. https://doi.org/10.3389/fpls.2023.1087343.
Ntanos DA, Koutroubas SD. Dry matter and N accumulation and translocation for India and Japonica rice under Mediterranean conditions. Field Crops Res. 2002. https://doi.org/10.1016/S0378-4290(01)00203-9.
Zhiipao RR, Pooniya V, Biswakarma N, Kumar D, Shivay YS, Dass A, Mukri G, Lakhena KK, Pandey RK, Bhatia A, Govindasamy P. Timely sown maize hybrids improve the post-anthesis dry matter accumulation, nutrient acquisition and crop productivity. Sci Rep. 2023. https://doi.org/10.1038/s41598-023-28224-9.
McGarity JW, Myers MG. A survey of urease activity in soils of northern New South Wales. Plant Soil. 1967;1:217–38.
Brookes PC, Kragt JF, Powlson DS, Jenkinson DS. Chloroform fumigation and the release of soil nitrogen: the effects of fumigation time and temperature. Soil Bio Biochem. 1985. https://doi.org/10.1016/0038-0717(85)90143-9.
Vance ED, Brookes PC, Jenkinson DS. An extraction method for measuring soil microbial biomass C. Soil bio Biochem. 1987. https://doi.org/10.1016/0038-0717(87)90052-6.
Gomez KA, Gomez AA. Statistical procedures for agricultural research. Hoboken: John wiley & sons; 1984.
Thuy NH, Shan YuHua SY, Bijay-Singh BS, Kai Rong Wang KR, Zu Cong Cai ZC, Yadvinder-Singh YS, Buresh RJ. Nitrogen supply in rice-based cropping systems as affected by crop residue management. Soil Sci Soc Am J. 2008;72:514–23.
Sharma S, Singh P, Choudhary OP. Nitrogen and rice straw incorporation impact nitrogen use efficiency, soil nitrogen pools and enzyme activity in rice-wheat system in north-western India. Field Crops Res. 2021. https://doi.org/10.1016/j.fcr.2021.108131.
Beri V, Sidhu BS, Bahl GS, Bhat AK. Nitrogen and phosphorus transformations as affected by crop residue management practices and their influence on crop yield. Soil Use Manag. 1995. https://doi.org/10.1111/j.1475-2743.1995.tb00496.x.
Chen B, Liu E, Tian Q, Yan C, Zhang Y. Soil nitrogen dynamics and crop residues. A Review Agron Sustain Dev. 2014. https://doi.org/10.1007/s13593-014-0207-8.
Robertson GP, Groffman PM. Nitrogen transformations. Soil Microbio Eco Biochem. 2007;10:341–64.
Yadvinder-Singh B-S, Timsina J. Crop residue management for nutrient cycling and improving soil productivity in rice-based cropping systems in the tropics. Adv Agron. 2005;85:269–407.
Bänziger M, Feil B, Stamp P. Competition between nitrogen accumulation and grain growth for carbohydrates during grain filling of wheat. Crop sci. 1994. https://doi.org/10.2135/cropsci1994.0011183X003400020025x.
Cox MC, Qualset CO, Rains DW. Genetic variation for nitrogen assimilation and translocation in wheat. II. Nitrogen assimilation in relation to grain yield and protein 1. Crop Sci. 1985. https://doi.org/10.2135/cropsci1985.0011183X002500030003x.
Pilbeam DJ. The utilization of nitrogen by plants: a whole plant perspective. Annual plant reviews, nitrogen metabolism in plants in the post-genomic era. New York: Wiley; 2011. https://doi.org/10.1002/9781444328608.
Mi G, Tang L, Zhang F, Zhang J. Is nitrogen uptake after anthesis in wheat regulated by sink size? Field Crops Res. 2000. https://doi.org/10.1016/S0378-4290(00)00119-2.
Martre P, Jamieson PD, Semenov MA, Zyskowski RF, Porter JR, Triboi E. Modelling protein content and composition in relation to crop nitrogen dynamics for wheat. Eur J Agron. 2006. https://doi.org/10.1016/j.eja.2006.04.007.
Manschadi AM, Soltani A. Variation in traits contributing to improved use of nitrogen in wheat: implications for genotype by environment interaction. Field Crops Res. 2021. https://doi.org/10.1016/j.fcr.2021.108211.
Weichert H, Högy P, Mora-Ramirez I, Fuchs J, Eggert K, Koehler P, Weschke W, Fangmeier A, Weber H. Grain yield and quality responses of wheat expressing a barley sucrose transporter to combined climate change factors. J Exp Bot. 2017. https://doi.org/10.1093/jxb/erx366.
Pheloung PC, Siddique KH. Contribution of stem dry matter to grain yield in wheat cultivars. Funct Plant Bio. 1991. https://doi.org/10.1071/PP9910053.
Yan S, Wu Y, Fan J, Zhang F, Guo J, Zheng J, Wu L. Optimization of drip irrigation and fertilization regimes to enhance winter wheat grain yield by improving post-anthesis dry matter accumulation and translocation in northwest China. Agric Water Managt. 2022. https://doi.org/10.1016/j.agwat.2022.107782.
Ehdaie B, Waines JG. Sowing date and nitrogen rate effects on dry matter and nitrogen partitioning in bread and durum wheat. Field Crops Res. 2001. https://doi.org/10.1016/S0378-4290(01)00181-2.
Xue Q, Zhu Z, Musick JT, Stewart BA, Dusek DA. Physiological mechanisms contributing to the increased water-use efficiency in winter wheat under deficit irrigation. J plant physio. 2006. https://doi.org/10.1016/j.jplph.2005.04.026.
Zhang YP, Zhang YH, Xue QW, Wang ZM. Remobilization of water soluble carbohydrates in non-leaf organs and contribution to grain yield in winter wheat under reduced irrigation. Int J Plant Prod. 2013;7(1):97–116.
Papakosta DK, Gagianas AA. Nitrogen and dry matter accumulation, remobilization, and losses for Mediterranean wheat during grain filling. Agron J. 1991. https://doi.org/10.2134/agronj1991.00021962008300050018x.
Sharma A, Sharma R, Arora A, Shah R, Singh A, Pranaw K, Nain L. Insights into rapid composting of paddy straw augmented with efficient microorganism consortium. Int J Recycling Org Waste Agric. 2014. https://doi.org/10.1007/s40093-014-0054-2.
Fitriatin BN, Simarmata T. Straw composting with biological agent inoculation and application biofertilizer to increase rice production. Int J Sustain Agric Res. 2016;3(3):49–53.
Mahapatra BS, Sharma GL, Singh N. Integrated management of straw and urea nitrogen in lowland rice under a rice-wheat rotation. J Agric Sci. 1991. https://doi.org/10.1017/S0021859600077601.
Singh Y, Singh B. Efficient management of primary nutrients in the rice-wheat system. J Crop Prod. 2001. https://doi.org/10.1300/J144v04n01_02.
Mandal KG, Misra AK, Hati KM, Bandyopadhyay KK, Ghosh PK, Mohanty M. Rice residue-management options and effects on soil properties and crop productivity. J Food Agric Environ. 2004;1(2):224–31.
Dordas C. Nutrient management perspectives in conservation agriculture. Conserv Agric. 2015. https://doi.org/10.1007/978-3-319-11620-4_4.
Singh M, Sidhu HS, Humphreys E, Thind HS, Jat ML, Blackwell J, Singh V. Nitrogen management for zero till wheat with surface retention of rice residues in north-west India. Field Crops Res. 2015. https://doi.org/10.1016/j.fcr.2015.03.025.
Li C, Xie R, Huang G, Wu C, Li S, Tang Y. Effects of nitrogen management on rice growth and grain yield under conservation tillage in rice-wheat rotation system. Pl Nutri Fertil Sci. 2010;16(3):528–35.
Kumar A, Jat ML, Kumar A, Tomar J, Kumar S, Kushwaha SR. Rice residue management in wheat under different tillage practices and nitrogen doses. Annals Agric Res. 2016;37(1):49–55.
Jiang D, Fan X, Dai T, Cao W. Nitrogen fertiliser rate and post-anthesis waterlogging effects on carbohydrate and nitrogen dynamics in wheat. Plant Soil. 2008. https://doi.org/10.1007/s11104-008-9556-x.
Dotaniya ML. Impact of crop residue management practices on yield and nutrient uptake in rice-wheat system. Curr Adv Agricultural Sci (Int J). 2013;5(2):269–71.
Ali I, Nabi G, Gill SM, Mahmood-ul-Hassan M, Hussain I. Crop residue management in rice-wheat system of Pakistan and its impact on yield and nutrient uptake. Int J Biosci. 2019. https://doi.org/10.12692/ijb/14.3.221-236.
Qin S, Hu C, Wang Y, Li X, He X. Tillage effects on intracellular and extracellular soil urease activities determined by an improved chloroform fumigation method. Soil Sci. 2010. https://doi.org/10.1097/SS.0b013e3181fa2810.
Rai TN, Yadav J. Influence of inorganic and organic nutrient sources on soil enzyme activities. J Indian Society Soil Sci. 2011;59(1):54–9.
Juan LI, Zhao BQ, Li XY, Jiang RB, Bing SH. Effects of long-term combined application of organic and mineral fertilizers on microbial biomass, soil enzyme activities and soil fertility. Agric sci China. 2008. https://doi.org/10.1016/S1671-2927(08)60074-7.
Jingjing S, Mijia Z, Xiaoqia Y, Chi Z, Jun Y. Microbial, urease activities and organic matter responses to nitrogen rate in cultivated soil. The Open Biotech J. 2015. https://doi.org/10.2174/1874070701509010014.
Stockdale EA, Murphy DV. Managing soil microbial biomass for sustainable agro-ecosystems. In: Tate KR, editor. Microbial biomass: a paradigm shift in terrestrial biogeochemistry. London: World Scientific; 2017.
O’donnell AG, Seasman M, Macrae A, Waite I, Davies JT. Plants and fertilisers as drivers of change in microbial community structure and function in soils. Plant and Soil. 2001. https://doi.org/10.1023/A:1010394221729.
Benintende SM, Benintende MC, Sterren MA, De Battista JJ. Soil microbiological indicators of soil quality in four rice rotations systems. Ecol Ind. 2008. https://doi.org/10.1016/j.ecolind.2007.12.004.
Gosai K, Arunachalam A, Dutta BK. Tillage effects on soil microbial biomass in a rainfed agricultural system of northeast India. Soil Tillage Res. 2010. https://doi.org/10.1016/j.still.2010.04.006.
Masto RE, Chhonkar PK, Singh D, Patra AK. Soil quality response to long-term nutrient and crop management on a semi-arid Inceptisol. Agric Ecosyst Environ. 2007. https://doi.org/10.1016/j.agee.2006.05.008.
Acknowledgements
We acknowledge the G.B. Pant University of Agriculture and Technology for providing experimental field and laboratory facilities during the experimentation.
Funding
The authors have not disclosed any funding.
Author information
Authors and Affiliations
Contributions
L Peace Raising, Virendra Pratap Singh, and Subhash Chandra led the research work, planned, supervised, and conducted field experiments along with the collection and analysis of soil and plant samples. L Peace Raising and R Rustum Zhiipao wrote the initial manuscript, L Peace Raising and Subhash Chandra performed statistical analysis, Hriipulou Duo, Kadapa Sreenivas Reddy, Lham Dorjee, and Karthik Rayapati read and edited the manuscript, and also prepared figures and tables. Anil Shukla, V K Singh, and Navneet Pareek reviewed and edited the final manuscript. All the authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors have no competing interests to declare that are relevant to the content of this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Longshang, P.R., Singh, V.P., Chandra, S. et al. Optimization of nitrogen management reduce nitrogen stress and enhance productivity of super-seeder sown wheat under rice residue incorporation. Discov Appl Sci 6, 211 (2024). https://doi.org/10.1007/s42452-024-05823-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s42452-024-05823-3