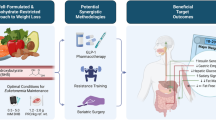
Abstract
Purpose
The purpose of this review is to examine the current evidence on the potential role of Mediterranean diet (MD) in the prevention and management of endocrine disorders and to highlight the importance of interdisciplinary collaboration between endocrinologists and nutritionists.
Methods
A literature search was conducted using PubMed and Google Scholar databases to identify relevant studies published in English. Studies were selected based on their relevance to the role of MD in the prevention and management of endocrine disorders. The search terms included "Mediterranean diet," "endocrine disorders," "thyroid disorders," "gonadal disorders," and "neuroendocrine tumors".
Results
The studies reviewed suggest that MD may have a beneficial effect in the prevention and management of various endocrine disorders, including thyroid disorders, gonadal disorders, and neuroendocrine tumors. MD has been associated with decreased risk of nodular thyroid disease and thyroid cancer, improved male and female reproductive health, and a potential role in the management of neuroendocrine tumors. MD's anti-inflammatory and antioxidant properties, as well as its high levels of phytochemicals, may play a role in its beneficial effects.
Conclusion
Interdisciplinary collaboration between endocrinologists and nutritionists is essential for the optimal management of endocrine disorders, including the potential role of MD in their prevention and management. While further research is needed, the current evidence suggests that MD may have a protective effect against endocrine disorders, and its incorporation into dietary recommendations may be beneficial.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Mediterranean diet (MD) is a dietary pattern characterized by high consumption of fruits, vegetables, legumes, whole grains, fish, and olive oil, and low consumption of red meat, processed foods, and simple sugar [1]. Actually, MD is recognized by United Nations Educational, Scientific and Cultural Organization (UNESCO) as an intangible cultural heritage of humanity, and it was defined as a social traditional practice ranging from the landscape to the cuisine of seven Mediterranean countries [2]. This dietary pattern has been associated with numerous health benefits, including a reduced risk of chronic diseases, such as type 2 diabetes mellitus (T2DM), cardiovascular disease, and certain types of cancer [3, 4].
Endocrine diseases are a group of disorders that affect the endocrine system, responsible for hormone production and regulation [5]. These disorders can have a significant impact on overall health and quality of life. Thyroid, gonads, and neuroendocrine systems are all important components of the endocrine system that play a crucial role in maintaining overall health [5]. Thyroid disorders such as thyroid nodules can affect metabolism, energy levels, and mood [6]. Disorders of the male gonadal system, such as hypogonadism, can lead to decreased libido, muscle mass, and bone density [7]. Similarly, disorders of the female gonadal system, such as polycystic ovary syndrome (PCOS) and menopause, can lead to hormonal imbalances, hirsutism and fertility issues [8]. Finally, neuroendocrine tumors (NETs) are a rare type of cancer that can affect various parts of the organism, including the pancreas, lungs, and gastrointestinal tract [9].
Of note, inflammation and oxidative stress play an important role in the pathogenesis and/or progression of various endocrine disorders, including thyroid disorders, gonadal disorders, and neuroendocrine tumors [10]. Chronic inflammation and high levels of reactive oxygen species (ROS) can lead to tissue damage and altered function, contributing to the development and progression of these disorders [10].
Research has shown that MD may have a beneficial effect for various endocrine disorders [9, 11,12,13,14]. For example, studies have suggested that higher adherence to MD could help to lower the risk of thyroid disorders, including autoimmune thyroiditis [15, 16]. Additionally, MD has been associated with improved male and female reproductive health [13, 14], including increased sperm quality [13] and improvement of the clinical picture of PCOS [11]. Studies have also suggested that MD may have a beneficial role in NETs [9].
Overall, MD appears to have a protective effect against endocrine disorders through a variety of mechanisms, including its anti-inflammatory and antioxidant properties [17, 18]. In fact, MD provides high levels of phytochemicals, including dietary polyphenols and flavonoids, which have been reported to exert beneficial biological effects, including antioxidant, anti-inflammatory, immunomodulatory, antitumoral, antidiabetic and anti-obesity activities [17]. In this regard, data from the INTERCATH study, an observational study of 1121 patients undergoing coronary angiography found that high sensitive C-reactive protein (hs-CRP), a marker of inflammation that has been linked to an increased risk of cardiovascular disease and other chronic conditions, correlated significantly with adherence to MD [19].
In this context, is clear that interdisciplinary collaboration between endocrinologists and nutritionists is essential for optimizing patient care and improving outcomes in the management of endocrine disorders.
The aim of this review was to provide an overview of the current evidence on the role of MD in various endocrine systems and highlights the importance of interdisciplinary collaboration in managing these conditions.
Endocrine axes and the Mediterranean diet
Thyroid axis and Mediterranean diet
Point of view of the endocrinologist
The thyroid gland is a crucial endocrine organ that regulates metabolic functions, male and female fertility, growth, and development [20, 21]. Thyroid diseases, including cancers, autoimmune diseases and thyroid dysfunctions, are becoming a serious social problem with rapidly increasing prevalence [22]. The most significant nutritional risk factor for developing thyroid nodular diseases is iodine deficiency [23, 24]. Additionally, iodine intake is associated with thyroid cancer risk [25], and there is a U-shaped relationship between iodine consumption and thyroid diseases, meaning that both low and high intake can lead to thyroid diseases [26]. Various factors, both modifying and non-modifying, could contribute to the increased incidence of benign/malign thyroid nodular diseases [27]. Unhealthy dietary patterns may play a role in the development of thyroid nodular disease, although it is unclear whether this is due to the direct effect of unhealthy nutrition or mediated by factors such as obesity, insulin resistance, and inflammation that often arise from unhealthy dietary habits [23]. Indeed, the thyroid gland is sensitive to inflammation and oxidative stress, which can impair its function and contribute to benign/malignant thyroid nodular diseases [28].
Both inflammation and oxidative stress can contribute to the formation of thyroid nodules [29]. Nodules are abnormal growths in the thyroid gland that can be benign or malignant. Chronic inflammation can lead to the formation of nodules by promoting the proliferation of thyroid cells and disrupting their normal function [30]. A clinical study involving 2722 subjects showed that individuals with high inflammation (assessed through the levels of white blood cell, neutrophil, lymphocyte and monocyte in subjects without obvious infection) had a higher prevalence of thyroid nodules and thyroid-stimulating hormone than those with low inflammation, even after adjusting for metabolic parameters and other confounders [31]. In particular, inflammation can inhibit the synthesis of thyroid hormone by affecting the conversion of thyroxine (T4) to triiodothyronine (T3) in peripheral tissues [32]. Cytokines, such as interleukin (IL)-1, IL-6, and tumor necrosis factor-alpha (TNF-α), can decrease the activity of 5′-deiodinase, which is responsible for the conversion. This results in a decrease in T3 levels and an increase in thyroid-stimulating hormone (TSH) levels as a compensatory mechanism. Cytokines can also decrease the expression of thyroglobulin, which further contributes to the decrease in thyroid hormone levels [32]. It is known that chronic inflammation may be also the result of a persistent immune response. For instance, Hashimoto's thyroiditis, an autoimmune disorder that causes chronic inflammation of the thyroid gland, is a common cause of nodules in the thyroid [33]. Oxidative stress can also contribute to the formation of nodules by promoting genetic mutations and DNA damage that lead to abnormal cell growth [30].
Thyroid cancer is a complex disease that can arise from various genetic and environmental factors [22]. Inflammation and oxidative stress are thought to play a role in its development [30]. Chronic inflammation can promote the growth and survival of cancer cells by providing them with a favorable microenvironment. For example, immune cells and cytokines can promote angiogenesis, the formation of new blood vessels, which is essential for the growth and spread of cancer cells. Inflammatory cytokines can also activate signaling pathways that promote cell proliferation and survival. Oxidative stress can contribute to the development of thyroid cancer by promoting genetic mutations and DNA damage that lead to the transformation of normal thyroid cells into cancer cells [30].
Several studies have investigated the relationship between inflammation, oxidative stress, and thyroid cancer [34,35,36]. In particular, chronic inflammation is linked to oxidative stress, which can cause DNA damage and thus contribute to the accumulation of cancer-initiating genetic alterations in cells [35]. For instance, a recent study found out that the serum levels of malondialdehyde, a well-established marker for screening and monitoring the oxidation stress level, were significantly higher in 55 patients with differentiated thyroid cancer compared to the healthy subjects [34]. Another study investigated the levels of ROS in 50 malignant and benign thyroid lesions and 41 normal tissues and correlated them with the thyroid differentiation score and the clinic-pathologic features [36]. In both malignant and benign thyroid tissue, the production of ROS and expression of the NADPH oxidase 4 (NOX4), a significant source of ROS, playing an important role in tumor cell proliferation and apoptosis, were found to be higher than in healthy tissues [36]. Follicular thyroid cancers (FTCs) and anaplastic/poorly differentiated cancers exhibited greater oxidative stress compared to papillary thyroid tumors (PTCs) [36]. Furthermore, oxidative stress was higher in FTCs than in follicular adenomas and mutated PTCs showed higher levels of oxidative stress than non-mutated PTCs. In malignant tumors, oxidative stress was negatively correlated with thyroid differentiation score and positively correlated with tumor stage and American Thyroid Association risk [36]. Overall, these findings suggested that thyroid tumors were subjected to increased oxidative stress compared to healthy tissues and that oxidative stress is associated with tumor aggressiveness and mutations in the MEK-ERK pathway in PTC [36].
In more detail, inflammatory mechanisms can contribute to thyroid alterations through the action of immune cells and cytokines [37]. Chronic inflammation can lead to the infiltration of immune cells, such as lymphocytes and macrophages, into the thyroid gland. These cells produce cytokines that can damage thyroid cells and alter their function. For example, pro-inflammatory cytokines like IL-1 and TNF-α can inhibit thyroid hormone synthesis and secretion, leading to hypothyroidism. Conversely, anti-inflammatory cytokines like IL-10 and transforming growth factor (TGF)-β can have a protective effect on thyroid cells and promote their survival [37]. Oxidative stress is another mechanism that can lead to thyroid alterations [32]. It occurs when there is an imbalance between the production of ROS and the body's ability to neutralize them with antioxidants. ROS can damage thyroid cells and contribute to the formation of nodules and thyroid cancer [32]. For example, hydrogen peroxide (H2O2) can inhibit thyroid hormone synthesis and secretion by damaging thyroid peroxidase, a key enzyme involved in the synthesis of thyroid hormones [38]. ROS can also cause DNA damage and promote genetic mutations that lead to the formation of nodules and thyroid cancer [32].
In conclusion, inflammation and oxidative stress are important mechanisms that can lead to thyroid alterations, the formation of nodules, and thyroid cancer. Chronic inflammation can promote thyroid cell damage, proliferation, and abnormal function, while oxidative stress can contribute to genetic mutations and DNA damage that lead to abnormal cell growth. Better understanding of the mechanisms underlying thyroid alterations and cancer can lead to the development of new diagnostic and therapeutic strategies to improve patient outcomes.
Point of view of the nutritionist
There is some evidence to suggest that the anti-inflammatory and antioxidant mechanisms of MD may be helpful in promoting thyroid health and reducing the risk of thyroid nodules and cancer [15, 16]. The formation of thyroid nodules is influenced by various factors, including genetics, iodine intake, and inflammation. The latter, together with oxidative stress, is a key driver of thyroid diseases.
First, since iodine intake, both high and low, is the main nutritional risk factor for nodular thyroid disease [39], MD provides a moderate amount of iodine, which can help promote optimal thyroid function without contributing to nodule formation. It is found in MD through the balanced consumption of whole grains, seafood, lean beef, poultry, milk and dairy products. In this regard, a cross-sectional observational study in 17.197 participants, aimed to analyze the nutrient intake adequacy of MD and Western diet patterns, found that a higher quintile of adherence to MD was associated with a lower prevalence of inadequacy for the intake of several vitamins and minerals, including iodine [40]. The quintiles of adherence to Western dietary pattern showed the opposite tendency. The highest quintile was associated with the highest percentage of individuals with noncompliance of recommendations for the iodine intake [40].
Second, MD is particularly rich in anti-inflammatory (such as polyphenols) and antioxidants compounds (such vitamin C and E) which are found in its variety of vegetable products like fruits, vegetables, legumes, and nuts. For fruits, three case–control studies exhibited a negative correlation between fruits intake and thyroid cancer risk and the authors of the various studies agreed that the cause of this association was due to their high levels of antioxidants, polyphenols, and fiber [41,42,43]. For vegetables, a metanalysis [44] of 19 case–control studies and a pooled analysis [45] of 11 case–control studies discovered a link between low vegetable consumption and the risk of thyroid cancer, showing a weak negative connection with the intake of all vegetables except cruciferous vegetables. Cruciferous vegetables that include broccoli, cauliflower, kale, cabbage, brussels sprouts and others, contain goitrogens that can block iodine uptake by the thyroid and have been shown to promote thyroid carcinogenesis in rats [46]; however, results from epidemiologic studies on the relationship between cruciferous vegetables and thyroid cancer risk have been inconsistent [47]. Of note, flavonoids are plants pigments with interesting pharmacological properties, such as antioxidant and anti-inflammatory [48]. They are widely present in the plant kingdom and include anthocyanins, flavones, and other pigments [48]. Recent studies conducted in vitro have shown that some flavonoids can be beneficial for thyroid cancer by reducing cell proliferation and increasing cell death [49, 50]. Flavonoids have been found to have effects on anti-proliferation and cell re-differentiation [51]. Specifically, apigenin and luteolin are the most powerful inhibitors of human thyroid carcinoma (papillary, follicular, and anaplastic carcinoma) cell lines in vitro, and also induce the re-expression of sodium iodide symporter (NIS) mRNA in anaplastic thyroid carcinoma cell lines [51]. Additionally, the regular administration of flavonoids for 5 days seems to increase not only the thyroid radioiodine uptake but also the NIS protein and mRNA levels in animal thyroids [52]. Despite the attention paid to the relationship between diet and thyroid cancer risk, it can be speculated that the beneficial anti-inflammatory and antioxidant effects of MD can be also extended to the complex pathogenesis of thyroid nodular disease.
However, it is well established that using a single food as the basis for study does not take into account the synergistic and/or antagonistic interactions that exist between nutrients and foods. Consequently, this approach may not have optimal statistical power to assess associations with thyroid diseases. In this regard, a recent study was conducted to examine the link between adherence to MD as a complete nutritional model and the incidence of thyroid nodular disease in 794 adults (aged 18–65 years; BMI 19.4–55.3 kg/m2) [15]. This study had also a second objective of assessing adherence to MD based on the cytological classification of thyroid nodules in the subgroup of study participants undergoing fine-needle aspiration. Authors found that the lowest adherence to MD was significantly associated with the presence of thyroid nodular disease, and in particular, with nodules at high-risk of malignancy. The study also found that patients with TIR5 (the highest risk category) had the lowest adherence to MD compared to other categories. In conclusion, adherence to MD was found to be associated with the presence of thyroid nodular disease and in particular with those at high-risk of malignancy [15].
Currently, the availability of randomized controlled trials (RCTs) specifically examining the effect of MD on thyroid disease is limited. Most research focuses on the general role of adherence to MD in influencing thyroid health but does not specifically examine a MD intervention in comparison with other nutritional approaches. However, observational studies suggest that MD may have benefits for thyroid health due to its nutrient and antioxidant content.
However, more research is needed to fully understand the relationship between diet and thyroid health.
Male gonadal axis and Mediterranean diet
Point of view of the endocrinologist
Infertility has emerged as a significant public health problem, now affecting around 15% of all couples trying to conceive [53]. Male factors attribute to this number with around 30%, while in 20% of couples there is a combination of male and female factor [54]. It is often mentioned in the media that the average male fertility has dropped significantly in the last century. Indeed, a meta-analysis performed in more than 40.000 men has clearly shown a deterioration in semen quality and almost 60% reduction in sperm count in the last 40 years in developed countries [55]. Furthermore, the span of relevant factors influencing male fertility is widening in recent years and now includes a multitude of genetic and lifestyle aspects. The influence of dietary habits becomes important from the very early stages of life, however with a less clear association between pubertal timing and obesity. Some studies show that peri-pubertal boys with overweight and obesity have a lower testicular volume than their age matched controls, which was predictive for a worse sperm production [56], while others have different results, with testicular enlargement occurring significantly earlier in boys with obesity in comparison to a normal-weight based reference cohort [57]. Whichever it may be, maintaining a healthy diet without significant metabolic alterations in childhood and adolescence is very significant to preserve male fertility and escape from the vicious cycle of obesity and male hypogonadism [58]. Apart from age, endocrine disorders and genetic conditions, environmental factors and personal behaviors could be seen as relevant contributors to these results [59,60,61,62,63]. Obesity, sedentary lifestyle, smoking, recreational drugs and alcohol intake, psychological stress, tight clothes, and xenobiotics (pesticides, toxins, pollutants, etc.) all create new challenges in male infertility treatment [59,60,61,62,63]. Novel analysis such as sperm DNA fragmentation, still not routinely assessed in fertility workup, improve fertility and in-vitro-fertilization (IVF) outcomes, and shed light on relevant mechanisms [64]. In males with no predisposing genetic factors, the aforementioned lifestyle factors are significantly associated with the generation of oxidative stress, regarded widely as one of the most significant contributors to deterioration of fertility [65]. A diet rich in simple carbohydrates, animal proteins, saturated and trans fats and poor in fiber, along with obesity is frequently observed in sub fertile and infertile males [66, 67]. High intake of red and processed meat, sugary drinks and alcohol can also lead to increased oxidative stress, which is reported in 30–80% of male infertility cases [68]. As ROS overcome the antioxidant barrier of the spermatozoa, they tend to deteriorate sperm morphology parameters [69], damage the cell membrane lipids, proteins and cell DNA and reduce motility, live sperm count, sperm concertation and oocyte connecting ability [70]. Spermatozoa are also exposed to oxidative stress during their passing in the seminal fluid and epididymis, and the degree of fragmentation is somewhat repairable in the oocyte but a pronounced DNA damage may not be, thus significantly influencing pregnancy outcomes and the fertilization process itself [71]. This in turn can lead to a vicious cycle, since continuous inadequate food choices keep increasing the oxidative stress while hyperinsulinemia and hyperglycemia also influence the hypothalamus-pituitary–gonadal (HPG) axis in separate mechanisms [62]. Excess weight can lead to hypogonadism, reduced testosterone levels and a reduced spermatozoa count [72]. The increased aromatase expression can convert testosterone to estrogen in turn decreasing follicle-stimulating hormone (FSH) and luteinizing hormone (LH) and disrupting the HPG axis further while low testosterone levels add to oxidative stress and mitochondrial disfunction in the Leydig cells [73]. Insulin resistance and impaired glucose tolerance also act in a multitude of ways, trough reducing sperm metabolism and glucose uptake and impairing their function and capacity [74]. It is known that patients with T2DM have an increased sperm DNA fragmentation and lower progressive sperm motility than non-diabetic patients along with a higher partner miscarriage rate [74]. Leptin is another relevant factor in this metabolic cascade, since, produced in excess by the adipose tissue in subjects with overweight/obesity, it affects FSH and LH pituitary release and pulsatility while also maintaining low grade inflammation, increasing ROS and decreasing sperm quality [75]. White adipose tissue is a source of production of ROS and low-grade inflammation mediated by ghrelin, IL-6 and TNF-α contributing to HPG axis disturbances [76]. A multitude of lipid profile alterations in dyslipidemia also contribute to infertility through several aforementioned mechanisms [77]. DNA fragmentation has been shown to be associated with obesity and is a good indicator of a poor spermatogenesis quality [78]. All this data shows us that diet has a major role in male infertility treatment, and a good dietary balance with plenty of foods containing antioxidants and antioxidant supplementation together with weight loss is always recommended [79].
MD embodies certain healthy food choices and food preparation techniques that significantly improve the aforementioned negative consequences of an inadequate diet, obesity and metabolic disbalances. The data on positive effects of MD on male reproductive health are not in abundance, but the existing research is mostly in line with other health benefits, since significant improvements in sperm quality, count and improved conception rates were shown [80]. A balanced diet rich in vegetables, fruit and whole grain food increases sperm count and motility [81, 82] and their bioactive compounds act via sperm mitochondria reducing oxidative stress and preserving optimal mitochondrial function [83, 84]. Resveratrol, for example, found in grapes, was shown to have an antioxidant and an anti-inflammatory effect [85]. Polyunsaturated fatty acids (PUFA) n-3 aid in metabolic function of the sperm cells while the addition of olive oil can have a positive effect in reducing the damage from oxidative stress and add to the sperm membrane lipid layer, thus improving the sperm quality [13]. Vitamins and other compounds found in these foods further reduce oxidative stress. The effect of MD on assisted reproductive technologies (ART) outcomes is also a point of discussion, however, there is a lot of variables and contributing factors influencing these results and further studies are necessary in this field. Even though the existing evidence do not support the theory that nutritional choices and food are a relevant factor for testicular cancer [86], several studies have shown certain associations between these types of tumors and dietary fat, cheese and even fiber intake, however, with very conflicting results [87].
All in all, due to the growing amount of research done in this field with various dietary choices and lifestyle alterations in relation to metabolic health, hormonal axis and male and female infertility, certain new compounds and novel dietary choices can emerge as relevant both in a positive and negative context. However, it is always necessary that this data is put in a broader perspective, since a well-balanced diet, with proper food choices, regular meals and adequate physical exercise remains a staple of metabolic, cardiovascular, reproductive, and overall health.
Point of view of the nutritionist
Diet exerts a relevant impact on the gonadal axis, in general, and, more specifically, on male fertility [88]. Such impact can be positive or negative, depending on dietary habits [13]. Besides consuming (or avoiding) specific foods or supplements, research focused on the effects of the global diet pattern, identifying MD as the best one. In this sense, MD is recognized for its health-promoting potential [89], as well as for its beneficial impact on male fertility [13]. This has been reported in various cross-sectional studies demonstrating the positive association between adherence to MD and semen quality in terms of sperm count, motility and concentration [80, 90,91,92]. Similarly, evidence from RCTs conducted on healthy young men demonstrated a positive effect on the semen quality of MD in comparison to a low-fat diet [93] or habitual diet [94]. Such beneficial effects are mainly due to both anti-inflammatory and antioxidant potential exerted by bioactive compounds contained in foods characterizing the MD [13, 95, 96]. Among these, dietary lipids play a prominent role.
In general, MD stands out for the low intake of saturated fatty acids (SFAs) and trans fatty acids, whose negative effects on sperm quality have been reported in animal-based studies [97,98,99]. On the contrary, MD ensures adequate intakes of both mono and polyunsaturated fatty acid (MUFA) and PUFA sources [100], able to modify the lipid composition of spermatozoa membranes, improving their energy metabolism [98]. More specifically, MUFA (mainly contained in extra virgin olive oil) exerts a marked antioxidant effect reducing the levels of ROS and restoring mitochondrial function [98]. Reduction of ROS levels is a key target since these oxidants can damage sperm membranes and alter DNA [101], causing epigenetic modifications responsible for the development, among the others, of cancer and infertility [102]. Interestingly, a higher MUFA/SFA ratio is positively associated with sperm concentration and total count, as reported in a cross-sectional analysis on men of sub-fertile couples [103].
The antioxidant activity is also exerted by PUFA n-3, whose action in increasing the aconitase/fumarate activity ratio has been reported [98]. These are two Krebs cycle key enzymes, whose activity ratio is recognized as an indicator of mitochondrial ROS production [104]. The antioxidant potential of PUFA (mainly contained in fish and nuts) is accompanied by their anti-inflammatory activity, as precursors of eicosanoids. Among PUFA, however, n-3 are precursors of anti-inflammatory eicosanoids (such as prostaglandins, thromboxanes, leukotrienes, and resolvins) [105], suggesting the importance to increase the intake of n-3 or increase the n-3/n-6 ratio, as ensured by following MD. In this sense, higher serum n-6/n-3 ratio have been found in infertile males than in fertile ones [106]. In addition, clinical evidence reported that the beneficial effect on male fertility of n-3 sources is weaker than the negative effect of higher consumption of n-6 sources [103]. This can be explained by the fact that testis cells are more able to convert C18 and C20 PUFA n-3 to C22 PUFA n-3 than to convert C18 and C20 PUFA n-3 to C22 PUFA n-3 [107].
Equally remarkable, is the ability of PUFA, in particular n-3, to modulate the activity of enzymes involved in spermatozoa energy pathways, including lactate dehydrogenase-C4 (LDH-C4) [98]. LDH-C4, catalyzing the pyruvate to lactate conversion (with consequent oxidization of NADH to NAD+), plays a pivotal role in sperm energy metabolism, allowing the progression of both glycolysis and oxidative phosphorylation [108, 109].
Data from an RCT on healthy men following a Western diet demonstrated that the inclusion of 60 g of a mixture of nuts (walnuts, almonds and hazelnuts) daily for 14 weeks resulted in significant improvements in sperm vitality, total motility, progressive motility, total count, and morphology. Such results were accompanied by a significant reduction of sperm DNA fragmentation, which can explain the observed improvements in sperm quality. Since the diet followed by the intervention group remained the same, except for the addition of nuts, it can be speculated that the beneficial effects observed might be attributed to the increased intake of PUFA, in particular, increased intake of n-3 and n-3/n-6 ratio [110].
The quality of lipids consumed following a Mediterranean-style dietary pattern is ensured by the choice of foods characterizing MD. For example, MD suggests the consumption of low-fat dairy products. Certainly, this suggestion aims to control the intake of cholesterol and SFA, but also it allows the consumption of foods that exert, among others, beneficial effects on male fertility, in terms of sperm vitality and concentration [111]. The consumption of low-fat milk, indeed, is associated with increased levels of insulin-like growth factor-1 (IGF-1) and insulin [112], both required by spermatogenesis to bind and activate the Leyding cell insulin receptors [113]. The consumption of low-fat dairy product typical of MD, thus, on one hand, contribute to reducing the intake of SFA and cholesterol, but on the other provides food alternatives with beneficial effects on fertility. Noteworthy, a not recent case–control study reported a significant association between high intake of SFA and cholesterol and increased risk of testicular cancer development [114].
The consumption of dairy products gained the attention of scientific opinion also for its potential effect in increasing the risk of testicular cancer development. This concern arose from the fact that such products may contain pesticides and veterinary drugs [115,116,117] acting as endocrine disruptors and female sex hormones [118], which may increase the risk of this cancer development. However, it is not clear whether the absorbed amount of these substances may be such that they effectively increase the risk. Also, there is no strong evidence of the association between high dairy product consumption and testicular cancer risk, and results are conflicting [119] However, MD suggests moderate consumption of such products [100] thus, in absence of evidence disproving it, this suggestion cannot be considered detrimental.
As well recognized, MD is also characterized by a high intake of fruit, vegetable, legumes and whole cereals [100], with positive effects on the control of inflammatory and oxidative status [95], mainly attributable to vitamins and antioxidants contained. Among the first ones, vitamins C and E emerge for their antioxidant activity [13], resulting in improving the overall sperm quality [120,121,122]. In particular, vitamin E (mainly contained in nuts, oils and seeds) protects spermatozoa membranes against ROS-caused lipid peroxidation, reducing the risk of structural and functional damage [123].
In addition to vitamins, several foods typically included in the Mediterranean dietary pattern are rich sources of bioactive compounds with antioxidant effects, mainly polyphenols. This, in addition to other features, allows attributing a nutraceutical potential to the MD [124].
In general, polyphenols are able to contrast inflammation and oxidative stress via the down-regulation of specific signaling pathways, including NF-κB and MAPK pathways [95]. Besides this principal activity (that per se elucidates a beneficial effect on male fertility of this class of bioactive compounds and by extension of MD), also relevant is their ability to target mitochondria, modulating their metabolism, as well as ROS homeostasis [13]. Although this can be interpreted as general activity, it is strictly linked to male fertility, since various mitochondrial parameters (i.e. respiratory activity, structure integrity, production of ROS and membrane potential) are related to sperm quality [109, 125, 126]. In this sense, the most important aspect refers to a peculiar biphasic activity of polyphenols, also defined as hormetic effect [13], where such molecules may target sperm mitochondria exerting positive or negative effects on their function, depending on their concentrations [127]. Specifically, ex-vivo experiments confirmed that polyphenols at low concentrations (0.1 nM) are able to stimulate the mitochondrial respiration active state, thus increasing the efficiency of this process. On the contrary, at higher concentrations (starting from 10 nM) this effect is lost, and the efficiency of mitochondrial respiration decreases [127]. This biphasic effect is due to the peculiar chemistry of polyphenols, i.e., quercetin. On one hand, they are able to interact directly with mitochondrial membranes [128] and complexes of the electron transport chain [129]. On the other hand, at high concentrations, they interfere with both lipid bilayer and membrane protein altering membrane electric properties [130]. These data are of relevant importance, highlighting the line between diet and supplementation. Digestion processes, indeed, can affect both the chemistry and bioactivity of polyphenols, varying their bioaccessibility and bioavailability [131]. In this context, as reported, the plasma concentration of polyphenols in the European population is around 10 nM [132]. Considering the hormetic effects of polyphenols, thus, the low absorption rate of polyphenols from foods (and not from supplements) may be considered a strength, at least referring to their effect on sperm mitochondria.
Another relevant aspect related to some polyphenols refers to their potential estrogen-like activity, due to their chemical structures, resulting in the interference with the activity of enzymes involved in the biosynthesis and degradation of steroids, acting as multi-functional endocrine disruptors [133]. Reducing the intake of sources of such polyphenols (also known as phytoestrogens) would be recommended in order to avoid potential negative effects on normal sex hormone balances.
In this sense, another advantage of MD is related to the regular consumption of fiber, able to reduce the circulating levels of estrogens or their metabolites [134], whose negative impact on normal semen production is clear [135]. Specifically, fiber may affect the enterohepatic reabsorption of estrogens excreted in bile directly or indirectly by reducing the activity of microbial β-glucuronidase [134].
Overall, this evidence demonstrates the relevant potential of MD in improving sperm quality, thus exerting a beneficial effect on male fertility. Although in vitro and animal-based studies elucidated the effects of Mediterranean food-derived bioactive compounds and their mechanisms, it should be noted that the majority of the available evidence on human are from observational studies. This is almost understandable since interventions with unhealthy dietary patterns are not ethical. However, only an association can be observed with these studies, but eventual cause-effect relationships cannot be proven [62]. Further prospective studies and RCT are needed to confirm the effect of MD or Mediterranean food consumption on male fertility and testis cancer.
Female gonadal axis and Mediterranean diet
Point of view of the endocrinologist
Infertility is defined as the failure to establish a clinical pregnancy after 12 months of regular, unprotected sexual intercourse. According to Borght and Wyns, infertility affects between 8 and 12% of couples of reproductive ages worldwide [136].
Female infertility contributes to 35% of overall infertility cases [137]. Several factors influence the possibility of spontaneous conception including the age of the female partner and infertility related to endocrine dysfunction [136]. In fact, regarding age, the possibility of achieving a spontaneous pregnancy decreases with age before conception: it is established that the decline in female fertility begins as early as 25–30 years of age, and the median age of last delivery is 40–41 years in most populations experiencing natural fertility [136].
Healthy nutrition and lifestyle play a major role on reproductive function in both women and men, representing a pivotal factor for all physiological processes related to fertility, such as ovulation, fertilization, implantation, placental growth, and nutrients supply to the fetus [138].
Importantly, alterations of female gonadal axis are often related to altered eating habits [139], since energy balance and reproduction are strictly regulated in hypothalamic neurons of the arcuate nucleus [140]. Different nutritional regimens have been associated with better fertility outcomes, even if the mechanistic links between diet and fertility is still a matter of debate [95]. Inflammation represents a key detrimental mechanism disrupting female fertility and represents a hallmark of the most common endocrine disorders of female gonadal axis, such as PCOS and endometriosis. The main anti-reproductive effects of inflammation can alter menstrual cyclicity, implantation and fetus growth [141].
ROS play a major role in the development of oocytes, corpus luteum function, embryo and its local environment. Therefore, oxidative stress is able to severely interfere with both natural and assisted fertility, and antioxidant therapies represent a hallmark in the counseling of the infertile couple. The most common endocrine alteration of female gonadal axis disruption is represented by PCOS, which is known to affect between 10 and 15% of the population [142]. The etiology of this syndrome is still controversial and debated: PCOS is the expression of a complex functional alteration of the reproductive system and not the consequence of a specific local ovarian or central defect. Insulin resistance represents a metabolic hallmark of PCOS [143, 144]. Hence, it can be concluded that PCOS is the resultant of different pathogenetic mechanisms. Treatment of PCOS should aim to normalize menstrual cycles, restore ovulation and thus fertility, reduce the clinical signs of hyperandrogenism and halt the tendency for progressive aggravation of the disease [145].
When the goal of the treatment is the seek for pregnancy, therapy should aim to correct anovulation, which is responsible for infertility. Weight reduction should be the first recommendation in patients with PCOS and with obesity: obesity, low-grade chronic inflammatory status, and insulin-resistance often coexist in PCOS [15]. In fact, weight loss of at least 5% from baseline weight can improve or even normalize ovarian function [146]. Weight loss reduces insulin, sex hormone-binding globulin (SHBG), and estrogen levels; moreover, excess weight before conception is a major risk factor for fertility outcomes and it is well established that weight loss improves fertility in women with overweight and obesity [137, 147].
Point of view of the nutritionist
The role that nutritional components and dietary habits play in modulating the risk of gynecologic diseases, such as PCOS, endometriosis, several gynecologic malignancies and infertility has long been debated [148]. According to Gaskins and Chavarro, identifying modifiable lifestyle factors, such as diet, that influence human fertility is of major clinical and public health significance [149]. Diet exerts an important effect on female fertility [150]. Unquestionably, among all the dietary styles that have been investigated, MD has a beneficial impact on female fertility [151]. Moreover, according to a review by Korre and colleagues, in which data from observational, longitudinal, and RCTs were examined, it has been demonstrated that Mediterranean-type diets can improve BMI and body weight, reduce the incidence of T2DM and metabolic syndrome risk factors, decrease cardiovascular morbidity and coronary heart disease mortality, and decrease all-cause mortality [152].
Additionally, as mentioned before, it is important to emphasize how crucial it is to maintain body weight and fat mass percentage in a normal range to improve fertility outcomes. In fact, it is well documented that obesity decreases natural fertility among men and women as well as pregnancy chances after conventional infertility and ART based treatments. Moreover, pregnancy complications are increased in women with overweight, obesity and PCOS [153]. PCOS is a very common endocrine disorder in women of reproductive age [143]. Although there is no cure for PCOS, first-line management includes conservative treatment through lifestyle interventions that emphasize weight loss and dietary modifications, with the goal of improving insulin sensitivity [148, 154].
Therefore, it is still debated how MD can have a positive impact on female fertility. A cohort study conducted by Chavarro and colleagues on 17.544 women who were planning a pregnancy or became pregnant during the study, an association was found between adherence to the pro-fertility diet (similar to MD) and a lower risk of infertility caused by ovulation disorders [155]. The pro-fertility diet was characterized by lower consumption of trans fatty acids and higher consumption of MUFAs and plant-based protein, as well as decreased consumption of animal protein, low glycemic index foods, and high-fiber foods. Women who followed the pro-fertility diet consumed more nonheme iron and more frequently, that is, at least 3 times a week, took multivitamins, particularly B vitamins (e.g., folic acid) [155].
In a review article by Gaskins and Chavarro, two studies were reported that revealed that adhering to MD resulted in a higher probability of engaging in both physiologic and post-IVF pregnancy [149]. The first case–control study on 485 women of the Seguimiento Universidad de Navarra project, found that women with the highest adherence to a Mediterranean-style diet, characterized by a higher intake of vegetables, fruits, fish, poultry, low-fat dairy products, and olive oil, were 0.56 (95% CI 0.35–0.95) more likely to seek medical attention for pregnancy difficulties [156]. In the second study, involving one 161 couples undergoing IVF/intracytoplasmic sperm injection (ICSI) treatment, it was observed that a higher adherence to MD before treatment was associated with a higher probability of pregnancy after IVF [157].
Lastly, in order to give a specific focus on the effects of MD and PCOS, in a study by Barrea and colleagues, adherence to MD and association with clinical severity of PCOS was assessed in a cohort of 112 treatment-naïve women with PCOS compared with a control group of 112 healthy women matched for age and BMI [15]. It was observed that women with PCOS consumed less extra virgin olive oil, legumes, fish/fish-based foods and nuts than the control group; it was also observed that women with PCOS consumed lower amounts of complex carbohydrates, fiber, MUFA and PUFA n-3 and higher amounts of simple carbohydrates, total fat, SFA and PUFA n-6 than the control group. Therefore, the authors conclude that there is an association between adherence to MD and clinical disease severity in women with PCOS. This association could support a therapeutic role of foods and nutrients from the Mediterranean dietary pattern in the pathogenesis of PCOS, likely involving inflammatory status, insulin resistance and hyperandrogenism [15].
Neuroendocrine tumors and Mediterranean diet
Point of view of the endocrinologist
NETs are tumors, which originate from neuroendocrine system. They generally occur in the digestive and respiratory systems [158]. A strong link between inflammation and neuroendocrine carcinogenesis has been widely reported [158, 159]. Chronic inflammation and pro-inflammatory cytokines contribute to the development and progression of NETs [160, 161]. The host immune system plays an interesting dual function in carcinogenesis, acting as pro- or anti-tumorigenic, depending by the balance of pro- and anti-inflammatory molecules and by the type of tumor-infiltrating immune cells [160, 161]. In the carcinogenesis process the host-mediated anti-tumor activity is inhibited, while the pro-inflammatory mediators are activated and contribute to shape the tumor microenvironment (TME) favouring tumor growth and progression [160].
TME is a compartment that changes during cancer process; it is composed by neoplastic cells, vascular components including blood and lymphatic vessels and proangiogenic factors, extracellular matrix with cancer-associated fibroblasts, infiltrating immune cells and pro-inflammatory and oncogenic molecules [162].
Chronic inflammation favours the activation of macrophages, dendritic cells and neutrophils with release of reactive oxygen species and pro-inflammatory cytokines, including TNF-α, IL-6, IL-8, IL-10, IL-17 and IL-1β and proangiogenic factors [163,164,165]. Notably, TNF-α, IL-6 and IL-17 are involved in tumor growth and promotion [166]. Pro-inflammatory cytokines activate NF-κB and signal transducers and activator of transcription (STAT3) signaling pathways [150] that are strongly involved in the development and maintenance of TME [167].
Chronic inflammation can favour the hyperplasia and dysplasia of neuroendocrine cells, leading to a neoplastic transformation. With a special regard to GEP-NET, chronic inflammation, but also the alteration of gut microbiota can contribute to carcinogenesis [168, 169]. Chronic atrophic gastritis induces hypergastrinemia that stimulating the proliferation of entero-chromaffin like cells favours gastric NET. In addition, hypochlorhydria induces a change in the gastric microbiota favouring colonization and increasing the risk of gastric malignancy [170]. Chronic atrophic gastritis affects the citric acid cycle, which is involved in gastric carcinogenesis [170].
The relationship between chronic pancreatitis and pancreatic NET has not been clearly established [171]. Capurso et al. reported T2DM and chronic pancreatitis as independent risk factors for pancreatic cancer [172]. However, this finding was not confirmed in other recent studies, that, by contrast, supported T2DM as a risk factor for pancreatic cancer [173]. Inflammatory bowel disease, including ulcerative colitis and Chron’s disease, was associated to an increased risk of small bowel NET [174]. Interestingly, large-scale studies reported that bowel NETs arose far from the area involved in the inflammatory disease, suggesting that they were the result of the effect of proinflammatory cytokines more than local inflammatory disease [175]. An interesting relationship between inflammation and appendiceal NETs has been reported in patients with Chron’s disease [176]. In addition, inflammatory bowel disease was also associated to increased risk of colonic and rectal NETs, causing the destruction of the intestinal epithelial barrier and therefore the release of several proinflammatory molecules. Similarly, to bowel NETs, colonic and rectal NETs developed in colon areas free from inflammatory bowel disease, as a likely result of a prolonged state of inflammation [177].
Finally, we must mention that obesity plays a significant role in NET carcinogenesis [178]. Indeed, obesity, that is associated with hyperplasia of white adipose tissue, in addition to favouring a chronic inflammation, also causes hyperinsulinism, predisposes to T2DM, increases IGF-1 secretion, induces oxidative stress, and impairs leptin and adiponectin secretion [179].
Point of view of the nutritionist
There is a correlation between low adherence to MD and cancer, and adherence to MD could influence the aggressiveness of different tumor types (i.e., prostate, breast cancer, and melanoma) [180].
MD may have a positive impact on cancer management by various mechanisms. It inhibits tumor cell growth by affecting hormones and intracellular pathways, and it has anti-inflammatory, anti-oxidative, and anti-aggregating effects. Anti-tumoral effects of MD can be attributed to various food components, such as fish, fruits, vegetables, cereals, olive oil, and legumes. These elements are rich in antioxidants, including vitamins A, C, and E, carotenoids, lycopene, flavonoids, PUFAs, and fibers, which are involved in cellular signaling pathways [180].
In particular, antioxidants scavenge ROS and reduce their production, which are involved in the stimulation of cancer development [181]. Flavonoids inhibit the activation of certain pathways, increase the expression of enzymes involved in the catabolism of carcinogens, and stimulate the AMP-activated protein kinase [182]. Fibers act on the gut microbiota, releasing short-chain fatty acids, which can reduce the expression of cancer-promoting factors [183]. Of note, higher dietary fibers intake was associated with a significant improvement in progression-free survival in 128 patients undergoing immune checkpoint blockade, with the most pronounced benefit observed in patients with sufficient dietary fibers intake and no probiotic use [183]. In this regard, MD is a nutritional pattern naturally characterized by a high fibers intake, provided by whole grains, vegetables, fruit and legumes. PUFAs help reduce chronic low-grade inflammation and the expression of inflammatory cytokines, including IL-6, IL-1β, and TNF-α [184]. Red wine is a source of resveratrol and quercetin, which can modulate the cell cycle, induce apoptosis in cancer cells, and act as an anti-inflammatory [185, 186]. However, the beneficial effects of red wine are controversial. In fact, it should be considered that other distinguishing features of the MD are the richness of virgin olive oil, fruit, cereals and vegetables, which are also good sources of polyphenols and other phytochemicals, and do not present the risks of wine [186].
For all these reasons, MD could be considered a useful model to adopt in patients with NET. However, there is still little scientific evidence on the role of MD in NET. Only one case–control, cross-sectional study, aimed to investigate the nutritional status and adherence to MD in patients with GEP-NETs [187]. A validated 14-item questionnaire called PREvención con DIeta MEDiterránea (PREDIMED) was used to assess adherence to MD. Authors found that patients with more aggressive disease had lower adherence to MD compared to those with G1, localized, and free/stable disease status. Additionally, patients with poor adherence to MD had a higher incidence of metastases and progressive disease. The study suggests that MD may be an important tool for preventing cancer aggressiveness in patients with neuroendocrine tumors, although further studies are necessary to confirm this finding [187].
In conclusion, MD has been found to have a positive impact on cancer management through various mechanisms, including inhibition of tumor cell growth, anti-inflammatory, anti-oxidative, and anti-aggregating effects. The food components of MD, such as antioxidants, flavonoids, fibers, and PUFAs, are involved in cellular signaling pathways that may help reduce cancer-promoting factors. While MD could be considered a useful model in patients with neuroendocrine tumors, there is still little scientific evidence on its role in this specific type of cancer. Therefore, future well-designed studies are needed to better explore the promising role of MD in neuroendocrine tumors.
Conclusion
Research has suggested that MD may have the potential to prevent and manage endocrine disorders, such as thyroid disorders, gonadal disorders, and neuroendocrine tumors. Studies have indicated that MD could potentially reduce the risk of nodular thyroid disease and thyroid cancer, enhance reproductive health in both men and women, and have a beneficial effect in the management of neuroendocrine tumors. The beneficial effects of MD could be attributed to its anti-inflammatory and antioxidant properties and its high levels of phytochemicals. It is important for endocrinologists and nutritionists to collaborate to optimize patient care and improve the management of endocrine disorders. While further research is necessary, current evidence supports the potential protective effects of MD against endocrine disorders and incorporating it into dietary recommendations could be advantageous.
References
Muscogiuri G, Verde L, Sulu C, Katsiki N, Hassapidou M, Frias-Toral E et al (2022) Mediterranean diet and obesity-related disorders: what is the evidence? Curr Obes Rep 11(4):287–304. https://doi.org/10.1007/s13679-022-00481-1. (Epub 2022/10/01)
United Nations Educational SaCOU. Representative list of the intangible cultural heritage of humanity. Available from: https://ich.unesco.org/en/RL/mediterranean-diet-00884
Dominguez LJ, Di Bella G, Veronese N, Barbagallo M (2021) Impact of Mediterranean diet on chronic non-communicable diseases and longevity. Nutrients. https://doi.org/10.3390/nu13062028. (Epub 2021/07/03)
Asoudeh F, Fallah M, Djafarian K, Shirzad N, Clark CCT, Esmaillzadeh A (2023) The effect of Mediterranean diet on inflammatory biomarkers and components of metabolic syndrome in adolescent girls. J Endocrinol Invest. https://doi.org/10.1007/s40618-023-02027-1. (Epub 2023/02/17)
Hiller-Sturmhofel S, Bartke A (1998) The endocrine system: an overview. Alcohol Health Res World 22(3):153–164 (Epub 2005/02/15)
Ioachimescu AG (2022) Thyroid disorders: an update. Endocrinol Metab Clin North Am 51(2):xiii–xiv. https://doi.org/10.1016/j.ecl.2022.02.007. (Epub 2022/06/07)
Ross A, Bhasin S (2016) Hypogonadism: its prevalence and diagnosis. Urol Clin North Am 43(2):163–176. https://doi.org/10.1016/j.ucl.2016.01.002. (Epub 2016/05/03)
Netzer I, Mustafa S, Lowenstein L (2010) Female sexual function and gynecological disorders. Minerva Ginecol 62(5):467–482 (Epub 2010/10/13)
Muscogiuri G, Barrea L, Cantone MC, Guarnotta V, Mazzilli R, Verde L et al (2022) Neuroendocrine tumors: a comprehensive review on nutritional approaches. Cancers (Basel). https://doi.org/10.3390/cancers14184402. (Epub 2022/09/24)
Rehman A, Pacher P, Hasko G (2021) Role of macrophages in the endocrine system. Trends Endocrinol Metab 32(4):238–256. https://doi.org/10.1016/j.tem.2020.12.001. (Epub 2021/01/19)
Barrea L, Arnone A, Annunziata G, Muscogiuri G, Laudisio D, Salzano C et al (2019) Adherence to the Mediterranean diet, dietary patterns and body composition in women with polycystic ovary syndrome (PCOS). Nutrients. https://doi.org/10.3390/nu11102278. (Epub 2019/09/25)
Bellastella G, Scappaticcio L, Caiazzo F, Tomasuolo M, Carotenuto R, Caputo M et al (2022) Mediterranean diet and thyroid: an interesting alliance. Nutrients. https://doi.org/10.3390/nu14194130. (Epub 2022/10/15)
Ferramosca A, Zara V (2022) Diet and male fertility: the impact of nutrients and antioxidants on sperm energetic metabolism. Int J Mol Sci. https://doi.org/10.3390/ijms23052542. (Epub 2022/03/11)
Garruti G, Depalo R, De Angelis M (2019) Weighing the impact of diet and lifestyle on female reproductive function. Curr Med Chem 26(19):3584–3592. https://doi.org/10.2174/0929867324666170518101008. (Epub 2017/05/20)
Barrea L, Muscogiuri G, de Alteriis G, Porcelli T, Vetrani C, Verde L et al (2022) Adherence to the Mediterranean diet as a modifiable risk factor for thyroid nodular disease and thyroid cancer: results from a pilot study. Front Nutr 9:944200. https://doi.org/10.3389/fnut.2022.944200. (Epub 2022/07/06)
Ruggeri RM, Giovinazzo S, Barbalace MC, Cristani M, Alibrandi A, Vicchio TM et al (2021) Influence of dietary habits on oxidative stress markers in hashimoto’s thyroiditis. Thyroid 31(1):96–105. https://doi.org/10.1089/thy.2020.0299. (Epub 2020/07/31)
Gantenbein KV, Kanaka-Gantenbein C (2021) Mediterranean diet as an antioxidant: the impact on metabolic health and overall wellbeing. Nutrients. https://doi.org/10.3390/nu13061951. (Epub 2021/07/03)
Barrea L, Tarantino G, Somma CD, Muscogiuri G, Macchia PE, Falco A et al (2017) Adherence to the Mediterranean diet and circulating levels of Sirtuin 4 in obese patients: a novel association. Oxid Med Cell Longev 2017:6101254. https://doi.org/10.1155/2017/6101254. (Epub 2017/07/15)
Waldeyer C, Brunner FJ, Braetz J, Ruebsamen N, Zyriax BC, Blaum C et al (2018) Adherence to Mediterranean diet, high-sensitive C-reactive protein, and severity of coronary artery disease: contemporary data from the INTERCATH cohort. Atherosclerosis 275:256–261. https://doi.org/10.1016/j.atherosclerosis.2018.06.877. (Epub 2018/07/07)
Jugan ML, Levi Y, Blondeau JP (2010) Endocrine disruptors and thyroid hormone physiology. Biochem Pharmacol 79(7):939–947. https://doi.org/10.1016/j.bcp.2009.11.006. (Epub 2009/11/17)
Mazzilli R, Medenica S, Di Tommaso AM, Fabozzi G, Zamponi V, Cimadomo D et al (2023) The role of thyroid function in female and male infertility: a narrative review. J Endocrinol Invest 46(1):15–26. https://doi.org/10.1007/s40618-022-01883-7. (Epub 2022/08/10)
Vanderpump MP (2011) The epidemiology of thyroid disease. Br Med Bull 99:39–51. https://doi.org/10.1093/bmb/ldr030. (Epub 2011/09/07)
Barrea L, Gallo M, Ruggeri RM, Giacinto PD, Sesti F, Prinzi N et al (2021) Nutritional status and follicular-derived thyroid cancer: an update. Crit Rev Food Sci Nutr 61(1):25–59. https://doi.org/10.1080/10408398.2020.1714542. (Epub 2020/01/31)
Ruggeri RM, Trimarchi F (2021) Iodine nutrition optimization: are there risks for thyroid autoimmunity? J Endocrinol Invest 44(9):1827–1835. https://doi.org/10.1007/s40618-021-01548-x. (Epub 2021/03/09)
Zimmermann MB, Galetti V (2015) Iodine intake as a risk factor for thyroid cancer: a comprehensive review of animal and human studies. Thyroid Res 8:8. https://doi.org/10.1186/s13044-015-0020-8. (Epub 2015/07/07)
Laurberg P, Cerqueira C, Ovesen L, Rasmussen LB, Perrild H, Andersen S et al (2010) Iodine intake as a determinant of thyroid disorders in populations. Best Pract Res Clin Endocrinol Metab 24(1):13–27. https://doi.org/10.1016/j.beem.2009.08.013. (Epub 2010/02/23)
Yildirim Simsir I, Cetinkalp S, Kabalak T (2020) Review of factors contributing to nodular goiter and thyroid carcinoma. Med Princ Pract 29(1):1–5. https://doi.org/10.1159/000503575. (Epub 2019/09/23)
Liu C, Wang LQ, Zhang M, Deng YL, Luo Q, Liu EN et al (2023) Oxidative stress mediates the associations between phthalate exposures and thyroid cancer/benign nodule risk. Environ Pollut 326:121462. https://doi.org/10.1016/j.envpol.2023.121462. (Epub 2023/03/24)
Barrea L, Pugliese G, Frias-Toral E, Laudisio D, Rodriguez D, Vitale G et al (2021) Diet as a possible influencing factor in thyroid cancer incidence: the point of view of the nutritionist. Panminerva Med 63(3):349–360. https://doi.org/10.23736/S0031-0808.21.04213-0. (Epub 2021/04/22)
Kochman J, Jakubczyk K, Bargiel P, Janda-Milczarek K (2021) The influence of oxidative stress on thyroid diseases. Antioxidants (Basel). https://doi.org/10.3390/antiox10091442. (Epub 2021/09/29)
Li Z, Huang Y, Chen X, Wei C, Yang P, Xu W (2020) The effect of inflammation on the formation of thyroid nodules. Int J Endocrinol 2020:9827349. https://doi.org/10.1155/2020/9827349. (Epub 2020/07/23)
Mancini A, Di Segni C, Raimondo S, Olivieri G, Silvestrini A, Meucci E et al (2016) Thyroid hormones, oxidative stress, and inflammation. Mediators Inflamm 2016:6757154. https://doi.org/10.1155/2016/6757154. (Epub 2016/04/07)
Weetman AP (2021) An update on the pathogenesis of Hashimoto’s thyroiditis. J Endocrinol Invest 44(5):883–890. https://doi.org/10.1007/s40618-020-01477-1. (Epub 2020/12/18)
Buczynska A, Sidorkiewicz I, Rogucki M, Siewko K, Adamska A, Kosciuszko M et al (2021) Oxidative stress and radioiodine treatment of differentiated thyroid cancer. Sci Rep 11(1):17126. https://doi.org/10.1038/s41598-021-96637-5. (Epub 2021/08/26)
Liotti F, Visciano C, Melillo RM (2012) Inflammation in thyroid oncogenesis. Am J Cancer Res 2(3):286–297 (Epub 2012/06/09)
Muzza M, Pogliaghi G, Colombo C, Carbone E, Cirello V, Palazzo S et al (2022) Oxidative stress correlates with more aggressive features in thyroid cancer. Cancers (Basel). https://doi.org/10.3390/cancers14235857. (Epub 2022/12/12)
Lumachi F, Basso SM, Orlando R (2010) Cytokines, thyroid diseases and thyroid cancer. Cytokine 50(3):229–233. https://doi.org/10.1016/j.cyto.2010.03.005. (Epub 2010/04/13)
Song Y, Driessens N, Costa M, De Deken X, Detours V, Corvilain B et al (2007) Roles of hydrogen peroxide in thyroid physiology and disease. J Clin Endocrinol Metab 92(10):3764–3773. https://doi.org/10.1210/jc.2007-0660. (Epub 2007/08/02)
Cao LZ, Peng XD, Xie JP, Yang FH, Wen HL, Li S (2017) The relationship between iodine intake and the risk of thyroid cancer: a meta-analysis. Medicine (Baltimore) 96(20):e6734. https://doi.org/10.1097/MD.0000000000006734. (Epub 2017/05/18)
Serra-Majem L, Bes-Rastrollo M, Roman-Vinas B, Pfrimer K, Sanchez-Villegas A, Martinez-Gonzalez MA (2009) Dietary patterns and nutritional adequacy in a Mediterranean country. Br J Nutr 101(Suppl 2):S21–S28. https://doi.org/10.1017/S0007114509990559. (Epub 2009/07/15)
Franceschi S, Levi F, Negri E, Fassina A, La Vecchia C (1991) Diet and thyroid cancer: a pooled analysis of four European case–control studies. Int J Cancer 48(3):395–398. https://doi.org/10.1002/ijc.2910480315. (Epub 1991/05/30)
Haselkorn T, Stewart SL, Horn-Ross PL (2003) Why are thyroid cancer rates so high in southeast asian women living in the United States? The bay area thyroid cancer study. Cancer Epidemiol Biomark Prev 12(2):144–150 (Epub 2003/02/13)
Steinmetz KA, Potter JD (1991) Vegetables, fruit, and cancer. II. Mechanisms. Cancer Causes Control 2(6):427–442. https://doi.org/10.1007/BF00054304. (Epub 1991/11/01)
Liu ZT, Lin AH (2014) Dietary factors and thyroid cancer risk: a meta-analysis of observational studies. Nutr Cancer 66(7):1165–1178. https://doi.org/10.1080/01635581.2014.951734. (Epub 2014/09/27)
Bosetti C, Negri E, Kolonel L, Ron E, Franceschi S, Preston-Martin S et al (2002) A pooled analysis of case–control studies of thyroid cancer. VII. Cruciferous and other vegetables (International). Cancer Causes Control 13(8):765–775. https://doi.org/10.1023/a:1020243527152. (Epub 2002/11/08)
Kanno J, Matsuoka C, Furuta K, Onodera H, Miyajima H, Maekawa A et al (1990) Tumor promoting effect of goitrogens on the rat thyroid. Toxicol Pathol 18(2):239–246. https://doi.org/10.1177/019262339001800202. (Epub 1990/01/01)
Peterson E, De P, Nuttall R (2012) BMI, diet and female reproductive factors as risks for thyroid cancer: a systematic review. PLoS ONE 7(1):e29177. https://doi.org/10.1371/journal.pone.0029177. (Epub 2012/01/26)
de Souza Dos Santos MC, Goncalves CF, Vaisman M, Ferreira AC, de Carvalho DP (2011) Impact of flavonoids on thyroid function. Food Chem Toxicol 49(10):2495–2502. https://doi.org/10.1016/j.fct.2011.06.074. (Epub 2011/07/13)
Kang HJ, Youn YK, Hong MK, Kim LS (2011) Antiproliferation and redifferentiation in thyroid cancer cell lines by polyphenol phytochemicals. J Korean Med Sci 26(7):893–899. https://doi.org/10.3346/jkms.2011.26.7.893. (Epub 2011/07/09)
Schroder-van der Elst JP, van der Heide D, Romijn JA, Smit JW (2004) Differential effects of natural flavonoids on growth and iodide content in a human Na*/I-symporter-transfected follicular thyroid carcinoma cell line. Eur J Endocrinol 150(4):557–564. https://doi.org/10.1530/eje.0.1500557. (Epub 2004/04/15)
Yin F, Giuliano AE, Van Herle AJ (1999) Growth inhibitory effects of flavonoids in human thyroid cancer cell lines. Thyroid 9(4):369–376. https://doi.org/10.1089/thy.1999.9.369. (Epub 1999/05/13)
Goncalves CF, Santos MC, Ginabreda MG, Fortunato RS, Carvalho DP, Freitas Ferreira AC (2013) Flavonoid rutin increases thyroid iodide uptake in rats. PLoS ONE 8(9):e73908. https://doi.org/10.1371/journal.pone.0073908. (Epub 2013/09/12)
Thoma ME, McLain AC, Louis JF, King RB, Trumble AC, Sundaram R et al (2013) Prevalence of infertility in the United States as estimated by the current duration approach and a traditional constructed approach. Fertil Steril 99(5):1324–31 e1. https://doi.org/10.1016/j.fertnstert.2012.11.037. (Epub 2013/01/08)
Agarwal A, Mulgund A, Hamada A, Chyatte MR (2015) A unique view on male infertility around the globe. Reprod Biol Endocrinol 13:37. https://doi.org/10.1186/s12958-015-0032-1. (Epub 2015/05/01)
Levine H, Jorgensen N, Martino-Andrade A, Mendiola J, Weksler-Derri D, Mindlis I et al (2017) Temporal trends in sperm count: a systematic review and meta-regression analysis. Hum Reprod Update 23(6):646–659. https://doi.org/10.1093/humupd/dmx022. (Epub 2017/10/06)
Cannarella R, Caruso M, Condorelli RA, Timpanaro TA, Caruso MA, La Vignera S et al (2023) Testicular volume in 268 children and adolescents followed-up for childhood obesity—a retrospective cross-sectional study. Eur J Endocrinol 188(4):331–342. https://doi.org/10.1093/ejendo/lvad033. (Epub 2023/05/02)
Busch AS, Hojgaard B, Hagen CP, Teilmann G (2020) Obesity is associated with earlier pubertal onset in boys. J Clin Endocrinol Metab. https://doi.org/10.1210/clinem/dgz222. (Epub 2019/11/26)
Carrageta DF, Oliveira PF, Alves MG, Monteiro MP (2019) Obesity and male hypogonadism: tales of a vicious cycle. Obes Rev 20(8):1148–1158. https://doi.org/10.1111/obr.12863. (Epub 2019/04/30)
Alamo A, Condorelli RA, Mongioi LM, Cannarella R, Giacone F, Calabrese V et al (2019) Environment and male fertility: effects of benzo-alpha-pyrene and resveratrol on human sperm function in vitro. J Clin Med. https://doi.org/10.3390/jcm8040561. (Epub 2019/04/28)
Christou MA, Christou PA, Markozannes G, Tsatsoulis A, Mastorakos G, Tigas S (2017) Effects of anabolic androgenic steroids on the reproductive system of athletes and recreational users: a systematic review and meta-analysis. Sports Med 47(9):1869–1883. https://doi.org/10.1007/s40279-017-0709-z. (Epub 2017/03/05)
Gabrielsen JS, Tanrikut C (2016) Chronic exposures and male fertility: the impacts of environment, diet, and drug use on spermatogenesis. Andrology 4(4):648–661. https://doi.org/10.1111/andr.12198. (Epub 2016/05/28)
Salas-Huetos A, Bullo M, Salas-Salvado J (2017) Dietary patterns, foods and nutrients in male fertility parameters and fecundability: a systematic review of observational studies. Hum Reprod Update 23(4):371–389. https://doi.org/10.1093/humupd/dmx006. (Epub 2017/03/24)
Sansone A, Di Dato C, de Angelis C, Menafra D, Pozza C, Pivonello R et al (2018) Smoke, alcohol and drug addiction and male fertility. Reprod Biol Endocrinol 16(1):3. https://doi.org/10.1186/s12958-018-0320-7. (Epub 2018/01/18)
Esteves SC, Zini A, Coward RM (2021) Best urological practices on testing and management of infertile men with abnormal sperm DNA fragmentation levels: the SFRAG guidelines. Int Braz J Urol 47(6):1250–1258. https://doi.org/10.1590/S1677-5538.IBJU.2020.1004. (Epub 2021/02/11)
Walczak-Jedrzejowska R, Wolski JK, Slowikowska-Hilczer J (2013) The role of oxidative stress and antioxidants in male fertility. Cent European J Urol 66(1):60–67. https://doi.org/10.5173/ceju.2013.01.art19. (Epub 2014/03/01)
Giahi L, Mohammadmoradi S, Javidan A, Sadeghi MR (2016) Nutritional modifications in male infertility: a systematic review covering 2 decades. Nutr Rev 74(2):118–130. https://doi.org/10.1093/nutrit/nuv059. (Epub 2015/12/26)
Jensen TK, Andersson AM, Jorgensen N, Andersen AG, Carlsen E, Petersen JH et al (2004) Body mass index in relation to semen quality and reproductive hormones among 1,558 Danish men. Fertil Steril 82(4):863–870. https://doi.org/10.1016/j.fertnstert.2004.03.056. (Epub 2004/10/16)
Ricci E, Al-Beitawi S, Cipriani S, Alteri A, Chiaffarino F, Candiani M et al (2018) Dietary habits and semen parameters: a systematic narrative review. Andrology 6(1):104–116. https://doi.org/10.1111/andr.12452. (Epub 2017/12/22)
Jedrzejczak P, Fraczek M, Szumala-Kakol A, Taszarek-Hauke G, Pawelczyk L, Kurpisz M (2005) Consequences of semen inflammation and lipid peroxidation on fertilization capacity of spermatozoa in in vitro conditions. Int J Androl 28(5):275–283. https://doi.org/10.1111/j.1365-2605.2005.00547.x. (Epub 2005/09/01)
Liu Y, Ding Z (2017) Obesity, a serious etiologic factor for male subfertility in modern society. Reproduction 154(4):R123–R131. https://doi.org/10.1530/REP-17-0161. (Epub 2017/07/2)
Gonzalez-Marin C, Gosalvez J, Roy R (2012) Types, causes, detection and repair of DNA fragmentation in animal and human sperm cells. Int J Mol Sci 13(11):14026–14052. https://doi.org/10.3390/ijms131114026. (Epub 2012/12/04)
Mihalca R, Fica S (2014) The impact of obesity on the male reproductive axis. J Med Life 7(2):296–300 (Epub 2014/11/20)
El Salam MAA (2018) Obesity, an enemy of male fertility: a mini review. Oman Med J 33(1):3–6. https://doi.org/10.5001/omj.2018.02. (Epub 2018/02/23)
Dias TR, Alves MG, Silva BM, Oliveira PF (2014) Sperm glucose transport and metabolism in diabetic individuals. Mol Cell Endocrinol 396(1–2):37–45. https://doi.org/10.1016/j.mce.2014.08.005. (Epub 2014/08/17)
Martins AD, Majzoub A, Agawal A (2019) Metabolic syndrome and male fertility. World J Mens Health 37(2):113–127. https://doi.org/10.5534/wjmh.180055. (Epub 2018/10/24)
Cabler S, Agarwal A, Flint M, du Plessis SS (2010) Obesity: modern man’s fertility nemesis. Asian J Androl 12(4):480–489. https://doi.org/10.1038/aja.2010.38. (Epub 2010/06/10)
Hagiuda J, Ishikawa H, Furuuchi T, Hanawa Y, Marumo K (2014) Relationship between dyslipidaemia and semen quality and serum sex hormone levels: an infertility study of 167 Japanese patients. Andrologia 46(2):131–135. https://doi.org/10.1111/and.12057. (Epub 2013/01/03)
Smit M, Romijn JC, Wildhagen MF, Weber RF, Dohle GR (2010) Sperm chromatin structure is associated with the quality of spermatogenesis in infertile patients. Fertil Steril 94(5):1748–1752. https://doi.org/10.1016/j.fertnstert.2009.10.030. (Epub 2009/12/17)
Wright C, Milne S, Leeson H (2014) Sperm DNA damage caused by oxidative stress: modifiable clinical, lifestyle and nutritional factors in male infertility. Reprod Biomed Online 28(6):684–703. https://doi.org/10.1016/j.rbmo.2014.02.004. (Epub 2014/04/22)
Cutillas-Tolin A, Minguez-Alarcon L, Mendiola J, Lopez-Espin JJ, Jorgensen N, Navarrete-Munoz EM et al (2015) Mediterranean and western dietary patterns are related to markers of testicular function among healthy men. Hum Reprod 30(12):2945–2955. https://doi.org/10.1093/humrep/dev236. (Epub 2015/09/27)
Braga DP, Halpern G, Figueira Rde C, Setti AS, Iaconelli A Jr, Borges E Jr (2012) Food intake and social habits in male patients and its relationship to intracytoplasmic sperm injection outcomes. Fertil Steril 97(1):53–59. https://doi.org/10.1016/j.fertnstert.2011.10.011. (Epub 2011/11/15)
Eslamian G, Amirjannati N, Rashidkhani B, Sadeghi MR, Hekmatdoost A (2012) Intake of food groups and idiopathic asthenozoospermia: a case–control study. Hum Reprod 27(11):3328–3336. https://doi.org/10.1093/humrep/des311. (Epub 2012/09/04)
Di Giacomo M, Zara V, Bergamo P, Ferramosca A (2020) Crosstalk between mitochondrial metabolism and oxidoreductive homeostasis: a new perspective for understanding the effects of bioactive dietary compounds. Nutr Res Rev 33(1):90–101. https://doi.org/10.1017/S0954422419000210. (Epub 2019/10/17)
Forbes-Hernandez TY, Giampieri F, Gasparrini M, Mazzoni L, Quiles JL, Alvarez-Suarez JM et al (2014) The effects of bioactive compounds from plant foods on mitochondrial function: a focus on apoptotic mechanisms. Food Chem Toxicol 68:154–182. https://doi.org/10.1016/j.fct.2014.03.017. (Epub 2014/04/01)
Montano L, Maugeri A, Volpe MG, Micali S, Mirone V, Mantovani A et al (2022) Mediterranean diet as a shield against male infertility and cancer risk induced by environmental pollutants: a focus on flavonoids. Int J Mol Sci. https://doi.org/10.3390/ijms23031568. (Epub 2022/02/16)
Clinton SK, Giovannucci EL, Hursting SD (2020) The World Cancer Research Fund/American Institute for Cancer Research third expert report on diet, nutrition, physical activity, and cancer: impact and future directions. J Nutr 150(4):663–671. https://doi.org/10.1093/jn/nxz268. (Epub 2019/11/24)
McGlynn KA, Trabert B (2012) Adolescent and adult risk factors for testicular cancer. Nat Rev Urol 9(6):339–349. https://doi.org/10.1038/nrurol.2012.61. (Epub 2012/04/18)
Arab A, Rafie N, Mansourian M, Miraghajani M, Hajianfar H (2018) Dietary patterns and semen quality: a systematic review and meta-analysis of observational studies. Andrology 6(1):20–28. https://doi.org/10.1111/andr.12430. (Epub 2017/10/13)
Estruch R, Ros E, Salas-Salvado J, Covas MI, Corella D, Aros F et al (2018) Primary prevention of cardiovascular disease with a mediterranean diet supplemented with extra-virgin olive oil or nuts. N Engl J Med 378(25):e34. https://doi.org/10.1056/NEJMoa1800389. (Epub 2018/06/14)
Karayiannis D, Kontogianni MD, Mendorou C, Douka L, Mastrominas M, Yiannakouris N (2017) Association between adherence to the Mediterranean diet and semen quality parameters in male partners of couples attempting fertility. Hum Reprod 32(1):215–222. https://doi.org/10.1093/humrep/dew288. (Epub 2016/12/21)
Ricci E, Bravi F, Noli S, Ferrari S, De Cosmi V, La Vecchia I et al (2019) Mediterranean diet and the risk of poor semen quality: cross-sectional analysis of men referring to an Italian Fertility Clinic. Andrology 7(2):156–162. https://doi.org/10.1111/andr.12587. (Epub 2019/01/22)
Salas-Huetos A, Babio N, Carrell DT, Bullo M, Salas-Salvado J (2019) Adherence to the Mediterranean diet is positively associated with sperm motility: a cross-sectional analysis. Sci Rep 9(1):3389. https://doi.org/10.1038/s41598-019-39826-7. (Epub 2019/03/06)
Caruso P, Caputo M, Cirillo P, Scappaticcio L, Longo M, Maiorino MI et al (2020) Effects of Mediterranean diet on semen parameters in healthy young adults: a randomized controlled trial. Minerva Endocrinol 45(4):280–287. https://doi.org/10.23736/S0391-1977.20.03362-3. (Epub 2021/01/23)
Montano L, Ceretti E, Donato F, Bergamo P, Zani C, Viola GCV et al (2022) Effects of a lifestyle change intervention on semen quality in healthy young men living in highly polluted areas in italy: the FASt randomized controlled trial. Eur Urol Focus 8(1):351–359. https://doi.org/10.1016/j.euf.2021.01.017. (Epub 2021/02/14)
Alesi S, Villani A, Mantzioris E, Takele WW, Cowan S, Moran LJ et al (2022) Anti-inflammatory diets in fertility: an evidence review. Nutrients. https://doi.org/10.3390/nu14193914. (Epub 2022/10/15)
Cao LL, Chang JJ, Wang SJ, Li YH, Yuan MY, Wang GF et al (2022) The effect of healthy dietary patterns on male semen quality: a systematic review and meta-analysis. Asian J Androl 24(5):549–557. https://doi.org/10.4103/aja202252. (Epub 2022/08/02)
Ferramosca A, Conte A, Moscatelli N, Zara V (2016) A high-fat diet negatively affects rat sperm mitochondrial respiration. Andrology 4(3):520–525. https://doi.org/10.1111/andr.12182. (Epub 2016/04/12)
Ferramosca A, Moscatelli N, Di Giacomo M, Zara V (2017) Dietary fatty acids influence sperm quality and function. Andrology 5(3):423–430. https://doi.org/10.1111/andr.12348. (Epub 2017/03/24)
Molaie S, Shahverdi A, Sharafi M, Shahhoseini M, Rashki Ghaleno L, Esmaeili V et al (2019) Dietary trans and saturated fatty acids effects on semen quality, hormonal levels and expression of genes related to steroid metabolism in mouse adipose tissue. Andrologia 51(5):e13259. https://doi.org/10.1111/and.13259. (Epub 2019/03/16)
Willett WC, Sacks F, Trichopoulou A, Drescher G, Ferro-Luzzi A, Helsing E et al (1995) Mediterranean diet pyramid: a cultural model for healthy eating. Am J Clin Nutr 61(6 Suppl):1402S-S1406. https://doi.org/10.1093/ajcn/61.6.1402S. (Epub 1995/06/01)
Tremellen K (2008) Oxidative stress and male infertility—a clinical perspective. Hum Reprod Update 14(3):243–258. https://doi.org/10.1093/humupd/dmn004. (Epub 2008/02/19)
Vanden Berghe W (2012) Epigenetic impact of dietary polyphenols in cancer chemoprevention: lifelong remodeling of our epigenomes. Pharmacol Res 65(6):565–576. https://doi.org/10.1016/j.phrs.2012.03.007. (Epub 2012/04/03)
Ricci E, Noli S, Ferrari S, La Vecchia I, Castiglioni M, Cipriani S et al (2020) Fatty acids, food groups and semen variables in men referring to an Italian Fertility Clinic: cross-sectional analysis of a prospective cohort study. Andrologia 52(3):e13505. https://doi.org/10.1111/and.13505. (Epub 2020/01/09)
Ferramosca A, Conte A, Guerra F, Felline S, Rimoli MG, Mollo E et al (2016) Metabolites from invasive pests inhibit mitochondrial complex II: a potential strategy for the treatment of human ovarian carcinoma? Biochem Biophys Res Commun 473(4):1133–1138. https://doi.org/10.1016/j.bbrc.2016.04.028. (Epub 2016/04/20)
Calder PC (2010) Omega-3 fatty acids and inflammatory processes. Nutrients 2(3):355–374. https://doi.org/10.3390/nu2030355. (Epub 2010/03/01)
Safarinejad MR, Hosseini SY, Dadkhah F, Asgari MA (2010) Relationship of omega-3 and omega-6 fatty acids with semen characteristics, and anti-oxidant status of seminal plasma: a comparison between fertile and infertile men. Clin Nutr 29(1):100–105. https://doi.org/10.1016/j.clnu.2009.07.008. (Epub 2009/08/12)
Retterstol K, Haugen TB, Tran TN, Christophersen BO (2001) Studies on the metabolism of essential fatty acids in isolated human testicular cells. Reproduction 121(6):881–887. https://doi.org/10.1530/rep.0.1210881. (Epub 2001/05/25)
Ferramosca A, Zara V (2014) Bioenergetics of mammalian sperm capacitation. Biomed Res Int 2014:902953. https://doi.org/10.1155/2014/902953. (Epub 2014/05/03)
Piomboni P, Focarelli R, Stendardi A, Ferramosca A, Zara V (2012) The role of mitochondria in energy production for human sperm motility. Int J Androl 35(2):109–124. https://doi.org/10.1111/j.1365-2605.2011.01218.x. (Epub 2011/09/29)
Salas-Huetos A, Moraleda R, Giardina S, Anton E, Blanco J, Salas-Salvado J et al (2018) Effect of nut consumption on semen quality and functionality in healthy men consuming a Western-style diet: a randomized controlled trial. Am J Clin Nutr 108(5):953–962. https://doi.org/10.1093/ajcn/nqy181. (Epub 2018/11/27)
Afeiche MC, Bridges ND, Williams PL, Gaskins AJ, Tanrikut C, Petrozza JC et al (2014) Dairy intake and semen quality among men attending a fertility clinic. Fertil Steril 101(5):1280–1287. https://doi.org/10.1016/j.fertnstert.2014.02.003. (Epub 2014/03/19)
Tsilidis KK, Travis RC, Appleby PN, Allen NE, Lindstrom S, Albanes D et al (2013) Insulin-like growth factor pathway genes and blood concentrations, dietary protein and risk of prostate cancer in the NCI Breast and Prostate Cancer Cohort Consortium (BPC3). Int J Cancer 133(2):495–504. https://doi.org/10.1002/ijc.28042. (Epub 2013/01/24)
Holly JM, Perks CM (2012) Insulin-like growth factor physiology: what we have learned from human studies. Endocrinol Metab Clin North Am 41(2):249–263. https://doi.org/10.1016/j.ecl.2012.04.009. (Epub 2012/06/12)
Sigurdson AJ, Chang S, Annegers JF, Duphorne CM, Pillow PC, Amato RJ et al (1999) A case–control study of diet and testicular carcinoma. Nutr Cancer 34(1):20–26. https://doi.org/10.1207/S15327914NC340103. (Epub 1999/08/24)
Darko G, Acquaah SO (2008) Levels of organochlorine pesticides residues in dairy products in Kumasi, Ghana. Chemosphere 71(2):294–298. https://doi.org/10.1016/j.chemosphere.2007.09.005. (Epub 2007/10/19)
Jia Q, Qiu J, Zhang L, Liao G, Jia Y, Qian Y (2022) Multiclass comparative analysis of veterinary drugs, mycotoxins, and pesticides in bovine milk by ultrahigh-performance liquid chromatography-hybrid quadrupole-linear ion trap mass spectrometry. Foods. https://doi.org/10.3390/foods11030331. (Epub 2022/02/16)
Tian H (2011) Determination of chloramphenicol, enrofloxacin and 29 pesticides residues in bovine milk by liquid chromatography–tandem mass spectrometry. Chemosphere 83(3):349–355. https://doi.org/10.1016/j.chemosphere.2010.12.016. (Epub 2011/01/05)
Sharpe RM (2003) The ’oestrogen hypothesis’—where do we stand now? Int J Androl 26(1):2–15. https://doi.org/10.1046/j.1365-2605.2003.00367.x. (Epub 2003/01/22)
Signal V, Huang S, Sarfati D, Stanley J, McGlynn KA, Gurney JK (2018) Dairy consumption and risk of testicular cancer: a systematic review. Nutr Cancer 70(5):710–736. https://doi.org/10.1080/01635581.2018.1470655. (Epub 2018/05/22)
Cyrus A, Kabir A, Goodarzi D, Moghimi M (2015) The effect of adjuvant vitamin C after varicocele surgery on sperm quality and quantity in infertile men: a double blind placebo controlled clinical trial. Int Braz J Urol 41(2):230–238. https://doi.org/10.1590/S1677-5538.IBJU.2015.02.07. (Epub 2015/05/26)
Shabanian S, Farahbod F, Rafieian M, Ganji F, Adib A (2017) The effects of Vitamin C on sperm quality parameters in laboratory rats following long-term exposure to cyclophosphamide. J Adv Pharm Technol Res 8(2):73–79. https://doi.org/10.4103/japtr.JAPTR_153_16. (Epub 2017/05/19)
Yousef MI, Abdallah GA, Kamel KI (2003) Effect of ascorbic acid and Vitamin E supplementation on semen quality and biochemical parameters of male rabbits. Anim Reprod Sci 76(1–2):99–111. https://doi.org/10.1016/s0378-4320(02)00226-9. (Epub 2003/02/01)
Yue D, Yan L, Luo H, Xu X, Jin X (2010) Effect of Vitamin E supplementation on semen quality and the testicular cell membranal and mitochondrial antioxidant abilities in Aohan fine-wool sheep. Anim Reprod Sci 118(2–4):217–222. https://doi.org/10.1016/j.anireprosci.2009.08.004. (Epub 2009/09/08)
Accardi G, Aiello A, Gambino CM, Virruso C, Caruso C, Candore G (2016) Mediterranean nutraceutical foods: Strategy to improve vascular ageing. Mech Ageing Dev 159:63–70. https://doi.org/10.1016/j.mad.2016.02.007. (Epub 2016/10/22)
Amaral A, Lourenco B, Marques M, Ramalho-Santos J (2013) Mitochondria functionality and sperm quality. Reproduction 146(5):R163–R174. https://doi.org/10.1530/REP-13-0178. (Epub 2013/08/01)
Nowicka-Bauer K, Lepczynski A, Ozgo M, Kamieniczna M, Fraczek M, Stanski L et al (2018) Sperm mitochondrial dysfunction and oxidative stress as possible reasons for isolated asthenozoospermia. J Physiol Pharmacol. https://doi.org/10.26402/jpp.2018.3.05. (Epub 2018/08/28)
Ferramosca A, Lorenzetti S, Di Giacomo M, Lunetti P, Murrieri F, Capobianco L et al (2021) Modulation of human sperm mitochondrial respiration efficiency by plant polyphenols. Antioxidants (Basel). https://doi.org/10.3390/antiox10020217. (Epub 2021/02/06)
de Oliveira MR, Nabavi SM, Braidy N, Setzer WN, Ahmed T, Nabavi SF (2016) Quercetin and the mitochondria: a mechanistic view. Biotechnol Adv 34(5):532–549. https://doi.org/10.1016/j.biotechadv.2015.12.014. (Epub 2016/01/08)
Houghton MJ, Kerimi A, Tumova S, Boyle JP, Williamson G (2018) Quercetin preserves redox status and stimulates mitochondrial function in metabolically-stressed HepG2 cells. Free Radic Biol Med 129:296–309. https://doi.org/10.1016/j.freeradbiomed.2018.09.037. (Epub 2018/09/30)
Hendrich AB (2006) Flavonoid-membrane interactions: possible consequences for biological effects of some polyphenolic compounds. Acta Pharmacol Sin 27(1):27–40. https://doi.org/10.1111/j.1745-7254.2006.00238.x. (Epub 2005/12/21)
Annunziata G, Jimenez-Garcia M, Capo X, Moranta D, Arnone A, Tenore GC et al (2020) Microencapsulation as a tool to counteract the typical low bioavailability of polyphenols in the management of diabetes. Food Chem Toxicol 139:111248. https://doi.org/10.1016/j.fct.2020.111248. (Epub 2020/03/12)
Zamora-Ros R, Knaze V, Lujan-Barroso L, Kuhnle GG, Mulligan AA, Touillaud M et al (2012) Dietary intakes and food sources of phytoestrogens in the European Prospective Investigation into Cancer and Nutrition (EPIC) 24-hour dietary recall cohort. Eur J Clin Nutr 66(8):932–941. https://doi.org/10.1038/ejcn.2012.36. (Epub 2012/04/19)
Nordeen SK, Bona BJ, Jones DN, Lambert JR, Jackson TA (2013) Endocrine disrupting activities of the flavonoid nutraceuticals luteolin and quercetin. Horm Cancer 4(5):293–300. https://doi.org/10.1007/s12672-013-0150-1. (Epub 2013/07/10)
Goldin BR, Adlercreutz H, Gorbach SL, Warram JH, Dwyer JT, Swenson L et al (1982) Estrogen excretion patterns and plasma levels in vegetarian and omnivorous women. N Engl J Med 307(25):1542–1547. https://doi.org/10.1056/NEJM198212163072502. (Epub 1982/12/16)
Sevilla y Ruiz A, Moya Gordillo C, Torres Lili G, Canales Perez ES (1991) Serum concentrations of estradiol and testosterone in patients with oligoasthenozoospermia and asthenozoospermia. Ginecol Obstet Mex 59:313–315 (Epub 1991/10/01)
Vander Borght M, Wyns C (2018) Fertility and infertility: definition and epidemiology. Clin Biochem 62:2–10. https://doi.org/10.1016/j.clinbiochem.2018.03.012. (Epub 2018/03/21)
Skoracka K, Ratajczak AE, Rychter AM, Dobrowolska A, Krela-Kazmierczak I (2021) Female fertility and the nutritional approach: the most essential aspects. Adv Nutr 12(6):2372–2386. https://doi.org/10.1093/advances/nmab068. (Epub 2021/06/18)
Ma X, Wu L, Wang Y, Han S, El-Dalatony MM, Feng F et al (2022) Diet and human reproductive system: insight of omics approaches. Food Sci Nutr 10(5):1368–1384. https://doi.org/10.1002/fsn3.2708. (Epub 2022/05/21)
Caprio M, Fabbrini E, Isidori AM, Aversa A, Fabbri A (2001) Leptin in reproduction. Trends Endocrinol Metab 12(2):65–72. https://doi.org/10.1016/s1043-2760(00)00352-0. (Epub 2001/02/13)
Negron AL, Radovick S (2020) High-fat diet alters LH secretion and pulse frequency in female mice in an estrous cycle-dependent manner. Endocrinology. https://doi.org/10.1210/endocr/bqaa146. (Epub 2020/08/26)
Weiss G, Goldsmith LT, Taylor RN, Bellet D, Taylor HS (2009) Inflammation in reproductive disorders. Reprod Sci 16(2):216–229. https://doi.org/10.1177/1933719108330087. (Epub 2009/02/12)
Barrea L, Frias-Toral E, Verde L, Ceriani F, Cucalon G, Garcia-Velasquez E et al (2021) PCOS and nutritional approaches: differences between lean and obese phenotype. Metabol Open 12:100123. https://doi.org/10.1016/j.metop.2021.100123. (Epub 2021/10/09)
Lizneva D, Suturina L, Walker W, Brakta S, Gavrilova-Jordan L, Azziz R (2016) Criteria, prevalence, and phenotypes of polycystic ovary syndrome. Fertil Steril 106(1):6–15. https://doi.org/10.1016/j.fertnstert.2016.05.003. (Epub 2016/05/29)
Moghetti P, Tosi F (2021) Insulin resistance and PCOS: chicken or egg? J Endocrinol Invest 44(2):233–244. https://doi.org/10.1007/s40618-020-01351-0. (Epub 2020/07/11)
Teede HJ, Misso ML, Costello MF, Dokras A, Laven J, Moran L et al (2018) Recommendations from the international evidence-based guideline for the assessment and management of polycystic ovary syndrome. Fertil Steril 110(3):364–379. https://doi.org/10.1016/j.fertnstert.2018.05.004. (Epub 2018/07/24)
Frias-Toral E, Garcia-Velasquez E, de Los Angeles Carignano M, Rodriguez-Veintimilla D, Alvarado-Aguilera I, Bautista-Litardo N (2022) Polycystic ovary syndrome and obesity: clinical aspects and nutritional management. Minerva Endocrinol (Torino) 47(2):215–241. https://doi.org/10.23736/S2724-6507.21.03349-6. (Epub 2021/04/02)
Pugliese G, de Alteriis G, Muscogiuri G, Barrea L, Verde L, Zumbolo F et al (2023) Liraglutide and polycystic ovary syndrome: is it only a matter of body weight? J Endocrinol Invest. https://doi.org/10.1007/s40618-023-02084-6. (Epub 2023/04/24)
Afrin S, AlAshqar A, El Sabeh M, Miyashita-Ishiwata M, Reschke L, Brennan JT et al (2021) Diet and nutrition in gynecological disorders: a focus on clinical studies. Nutrients. https://doi.org/10.3390/nu13061747. (Epub 2021/06/03)
Gaskins AJ, Chavarro JE (2018) Diet and fertility: a review. Am J Obstet Gynecol 218(4):379–389. https://doi.org/10.1016/j.ajog.2017.08.010. (Epub 2017/08/29)
Grieger JA (2020) Preconception diet, fertility, and later health in pregnancy. Curr Opin Obstet Gynecol 32(3):227–232. https://doi.org/10.1097/GCO.0000000000000629. (Epub 2020/04/24)
Mirabelli M, Chiefari E, Arcidiacono B, Corigliano DM, Brunetti FS, Maggisano V et al (2020) Mediterranean diet nutrients to turn the tide against insulin resistance and related diseases. Nutrients. https://doi.org/10.3390/nu12041066. (Epub 2020/04/16)
Korre M, Tsoukas MA, Frantzeskou E, Yang J, Kales SN (2014) Mediterranean diet and workplace health promotion. Curr Cardiovasc Risk Rep 8(12):416. https://doi.org/10.1007/s12170-014-0416-3. (Epub 2014/10/21)
Hoek A, Wang Z, van Oers AM, Groen H, Cantineau AEP (2022) Effects of preconception weight loss after lifestyle intervention on fertility outcomes and pregnancy complications. Fertil Steril 118(3):456–462. https://doi.org/10.1016/j.fertnstert.2022.07.020. (Epub 2022/09/19)
Cincione IR, Graziadio C, Marino F, Vetrani C, Losavio F, Savastano S et al (2023) Short-time effects of ketogenic diet or modestly hypocaloric Mediterranean diet on overweight and obese women with polycystic ovary syndrome. J Endocrinol Invest 46(4):769–777. https://doi.org/10.1007/s40618-022-01943-y. (Epub 2022/11/20)
Chavarro JE, Rich-Edwards JW, Rosner BA, Willett WC (2007) Diet and lifestyle in the prevention of ovulatory disorder infertility. Obstet Gynecol 110(5):1050–1058. https://doi.org/10.1097/01.AOG.0000287293.25465.e1. (Epub 2007/11/06)
Toledo E, Lopez-del Burgo C, Ruiz-Zambrana A, Donazar M, Navarro-Blasco I, Martinez-Gonzalez MA et al (2011) Dietary patterns and difficulty conceiving: a nested case–control study. Fertil Steril 96(5):1149–1153. https://doi.org/10.1016/j.fertnstert.2011.08.034. (Epub 2011/09/29)
Vujkovic M, de Vries JH, Lindemans J, Macklon NS, van der Spek PJ, Steegers EA et al (2010) The preconception Mediterranean dietary pattern in couples undergoing in vitro fertilization/intracytoplasmic sperm injection treatment increases the chance of pregnancy. Fertil Steril 94(6):2096–2101. https://doi.org/10.1016/j.fertnstert.2009.12.079. (Epub 2010/03/02)
Vitale G, Carra S, Ferrau F, Guadagno E, Faggiano A, Colao A et al (2020) Gastroenteropancreatic neuroendocrine neoplasms and inflammation: a complex cross-talk with relevant clinical implications. Crit Rev Oncol Hematol 146:102840. https://doi.org/10.1016/j.critrevonc.2019.102840. (Epub 2020/01/10)
Cigrovski Berkovic M, Cacev T, Catela Ivkovic T, Zjacic-Rotkvic V, Kapitanovic S (2014) New insights into the role of chronic inflammation and cytokines in the etiopathogenesis of gastroenteropancreatic neuroendocrine tumors. Neuroendocrinology 99(2):75–84. https://doi.org/10.1159/000362339. (Epub 2014/04/02)
Lin WW, Karin M (2007) A cytokine-mediated link between innate immunity, inflammation, and cancer. J Clin Invest 117(5):1175–1183. https://doi.org/10.1172/JCI31537. (Epub 2007/05/04)
Zhang WH, Wang WQ, Gao HL, Yu XJ, Liu L (2019) The tumor immune microenvironment in gastroenteropancreatic neuroendocrine neoplasms. Biochim Biophys Acta Rev Cancer 1872(2):188311. https://doi.org/10.1016/j.bbcan.2019.188311. (Epub 2019/08/24)
Wang M, Zhao J, Zhang L, Wei F, Lian Y, Wu Y et al (2017) Role of tumor microenvironment in tumorigenesis. J Cancer 8(5):761–773. https://doi.org/10.7150/jca.17648. (Epub 2017/04/07)
Berkovic MC, Jokic M, Marout J, Radosevic S, Zjacic-Rotkvic V, Kapitanovic S (2010) IL-2-330 T/G SNP and serum values-potential new tumor markers in neuroendocrine tumors of the gastrointestinal tract and pancreas (GEP-NETs). J Mol Med (Berl) 88(4):423–429. https://doi.org/10.1007/s00109-009-0581-x. (Epub 2010/01/06)
Cai L, Michelakos T, Deshpande V, Arora KS, Yamada T, Ting DT et al (2019) Role of tumor-associated macrophages in the clinical course of pancreatic neuroendocrine tumors (PanNETs). Clin Cancer Res 25(8):2644–2655. https://doi.org/10.1158/1078-0432.CCR-18-1401. (Epub 2019/01/24)
Tran Janco JM, Lamichhane P, Karyampudi L, Knutson KL (2015) Tumor-infiltrating dendritic cells in cancer pathogenesis. J Immunol 194(7):2985–2991. https://doi.org/10.4049/jimmunol.1403134. (Epub 2015/03/22)
Herman Mahecic D, Cigrovski Berkovic M, Zjacic-Rotkvic V, Cacev T, Kapitanovic S, Ulamec M (2020) Inflammation-related cytokines and their roles in gastroenteropancreatic neuroendocrine neoplasms. Bosn J Basic Med Sci 20(4):445–450. https://doi.org/10.17305/bjbms.2020.4471. (Epub 2020/03/12)
Huang S (2007) Regulation of metastases by signal transducer and activator of transcription 3 signaling pathway: clinical implications. Clin Cancer Res 13(5):1362–1366. https://doi.org/10.1158/1078-0432.CCR-06-2313. (Epub 2007/03/03)
Altieri B, Barrea L, Modica R, Muscogiuri G, Savastano S, Colao A et al (2018) Nutrition and neuroendocrine tumors: an update of the literature. Rev Endocr Metab Disord 19(2):159–167. https://doi.org/10.1007/s11154-018-9466-z. (Epub 2018/09/30)
Gallo M, Muscogiuri G, Pizza G, Ruggeri RM, Barrea L, Faggiano A et al (2019) The management of neuroendocrine tumours: a nutritional viewpoint. Crit Rev Food Sci Nutr 59(7):1046–1057. https://doi.org/10.1080/10408398.2017.1390729. (Epub 2017/10/12)
Parsons BN, Ijaz UZ, D’Amore R, Burkitt MD, Eccles R, Lenzi L et al (2017) Comparison of the human gastric microbiota in hypochlorhydric states arising as a result of Helicobacter pylori-induced atrophic gastritis, autoimmune atrophic gastritis and proton pump inhibitor use. PLoS Pathog 13(11):e1006653. https://doi.org/10.1371/journal.ppat.1006653. (Epub 2017/11/03)
Bansal P, Sonnenberg A (1995) Pancreatitis is a risk factor for pancreatic cancer. Gastroenterology 109(1):247–251. https://doi.org/10.1016/0016-5085(95)90291-0. (Epub 1995/07/01)
Capurso G, Falconi M, Panzuto F, Rinzivillo M, Boninsegna L, Bettini R et al (2009) Risk factors for sporadic pancreatic endocrine tumors: a case–control study of prospectively evaluated patients. Am J Gastroenterol 104(12):3034–3041. https://doi.org/10.1038/ajg.2009.466. (Epub 2009/08/20)
Haugvik SP, Hedenstrom P, Korsaeth E, Valente R, Hayes A, Siuka D et al (2015) Diabetes, smoking, alcohol use, and family history of cancer as risk factors for pancreatic neuroendocrine tumors: a systematic review and meta-analysis. Neuroendocrinology 101(2):133–142. https://doi.org/10.1159/000375164. (Epub 2015/01/24)
Solcia E, Vanoli A (2014) Histogenesis and natural history of gut neuroendocrine tumors: present status. Endocr Pathol 25(2):165–170. https://doi.org/10.1007/s12022-014-9312-0. (Epub 2014/04/05)
Bojesen RD, Riis LB, Hogdall E, Nielsen OH, Jess T (2017) Inflammatory bowel disease and small bowel cancer risk, clinical characteristics, and histopathology: a population-based study. Clin Gastroenterol Hepatol 15(12):1900–1907. https://doi.org/10.1016/j.cgh.2017.06.051. (Epub 2017/07/12)
Szabo GG, Barta Z, Kerekes L, Szakall S (1999) Association of carcinoid tumor of the appendix and Crohn disease (case report and review of the literature). Orv Hetil 140(29):1635–1639 (Epub 1999/08/12)
Pellino G, Marcellinaro R, Candilio G, De Fatico GS, Guadagno E, Campione S et al (2016) The experience of a referral centre and literature overview of GIST and carcinoid tumours in inflammatory bowel diseases. Int J Surg 28(Suppl 1):S133–S141. https://doi.org/10.1016/j.ijsu.2015.12.051. (Epub 2015/12/29)
Budek M, Nuszkiewicz J, Piorkowska A, Czuczejko J, Szewczyk-Golec K (2022) Inflammation related to obesity in the etiopathogenesis of gastroenteropancreatic neuroendocrine neoplasms. Biomedicines. https://doi.org/10.3390/biomedicines10102660. (Epub 2022/10/28)
Kolb R, Sutterwala FS, Zhang W (2016) Obesity and cancer: inflammation bridges the two. Curr Opin Pharmacol 29:77–89. https://doi.org/10.1016/j.coph.2016.07.005. (Epub 2016/07/19)
Mentella MC, Scaldaferri F, Ricci C, Gasbarrini A, Miggiano GAD (2019) Cancer and Mediterranean diet: a review. Nutrients. https://doi.org/10.3390/nu11092059. (Epub 2019/09/05)
Harris IS, DeNicola GM (2020) The complex interplay between antioxidants and ROS in cancer. Trends Cell Biol 30(6):440–451. https://doi.org/10.1016/j.tcb.2020.03.002. (Epub 2020/04/19)
Li G, Ding K, Qiao Y, Zhang L, Zheng L, Pan T et al (2020) Flavonoids regulate inflammation and oxidative stress in cancer. Molecules. https://doi.org/10.3390/molecules25235628. (Epub 2020/12/04)
Spencer CN, McQuade JL, Gopalakrishnan V, McCulloch JA, Vetizou M, Cogdill AP et al (2021) Dietary fiber and probiotics influence the gut microbiome and melanoma immunotherapy response. Science 374(6575):1632–1640. https://doi.org/10.1126/science.aaz7015. (Epub 2021/12/24)
Marion-Letellier R, Savoye G, Ghosh S (2015) Polyunsaturated fatty acids and inflammation. IUBMB Life 67(9):659–667. https://doi.org/10.1002/iub.1428. (Epub 2015/09/24)
Kraft TE, Parisotto D, Schempp C, Efferth T (2009) Fighting cancer with red wine? Molecular mechanisms of resveratrol. Crit Rev Food Sci Nutr 49(9):782–799. https://doi.org/10.1080/10408390802248627. (Epub 2010/05/06)
Santos-Buelga C, Gonzalez-Manzano S, Gonzalez-Paramas AM (2021) Wine, polyphenols, and Mediterranean diets. What else is there to say? Molecules. https://doi.org/10.3390/molecules26185537. (Epub 2021/09/29)
Barrea L, Altieri B, Muscogiuri G, Laudisio D, Annunziata G, Colao A et al (2018) Impact of nutritional status on gastroenteropancreatic neuroendocrine tumors (GEP-NET) aggressiveness. Nutrients. https://doi.org/10.3390/nu10121854. (Epub 2018/12/06)
Funding
Open access funding provided by Università degli Studi di Napoli Federico II within the CRUI-CARE Agreement. The authors did not receive support from any organization for the submitted work.
Author information
Authors and Affiliations
Contributions
LB and GM had the idea for the article, LV, GA, EC, MC, VG, ASS, LVM, performed the literature search and drafted the manuscript. LB, AC and GM critically revised the work.
Corresponding author
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest. The authors have no competing interests to declare that are relevant to the content of this article.
Research involving human participants and/or animals
Not applicable.
Informed consent
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Barrea, L., Verde, L., Annunziata, G. et al. Role of Mediterranean diet in endocrine diseases: a joint overview by the endocrinologist and the nutritionist. J Endocrinol Invest 47, 17–33 (2024). https://doi.org/10.1007/s40618-023-02169-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40618-023-02169-2