Abstract
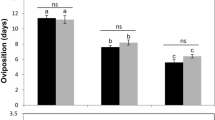
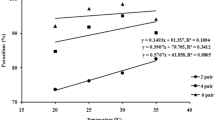
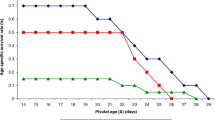
Trichogramma pretiosum Riley and Trichogramma atopovirilia Oatman & Platner are natural enemies of Plutella xylostella (L.) in Southern Brazil. Laboratory studies to evaluate parasitoids performance under different conditions, such as temperature regimes, are necessary to assess their potential as biocontrol agents of P. xylostella. In most studies involving Trichogramma, parasitism rate is the main parameter used to evaluate parasitoid performance, ignoring that parasitoids can cause egg mortality by feeding on the host content and/or to multiple drilling without laying eggs. This study was conducted to investigate three main issues: how temperature affects T. pretiosum and T.atopovirilia development on eggs of P. xylostella, whether or not these species respond differently to temperature, and how important is the mortality they cause besides parasitism on P. xylostella. Temperature effects (from 10 to 30°C) on development, survival, parasitism rate, mortality, and total mortality caused by T. pretiosum and T. atopovirilia on eggs of P. xylostella were evaluated. Temperature affected the development time, female longevity, parasitism rate, mortality not directly related to parasitoid larval development, and total mortality caused on the host. No significant differences were recorded for the estimated thermal requirements for T. pretiosum and T. atopovirilia. However, the higher mortality caused by T. pretiosum indicates that this parasitoid is the most suitable to be used against P. xylostella. Also, the results suggest that the use of parasitism rate as the only parameter to evaluate the performance of T. pretiosum and T. atopovirilia may underestimate the potential of these parasitoids in regulating pest populations.


Similar content being viewed by others
References
Abbot WS (1925) A method of computing the effectiveness of an insecticide. J Econ Entomol 18:265–267
Andrade GD, Pratissoli D, Dalpi LP, Desneux N, Santos HJG (2011) Performance of four Trichogramma species (Hymenoptera: Trichogrammatidae) as biocontrol agents of Heliothis virescens (Lepidoptera: Noctuidae) under various temperature regimes. J Pest Sci 84:313–320
Avanci MRF, Foerster LA, Cañete CL (2005) Natural parasitism in eggs of Anticarsia gemmatalis Hübner (Lepidoptera, Noctuidae) by Trichogramma spp. (Hymenoptera, Trichogrammatidae) in Brazil. Rev Bras Entomol 49:148–151
Ayres M, Ayres JRM, Ayres DL, Santos AS!(2007) BioEstat 5.0 - Aplicações Estatísticas nas Áreas das Ciências Biológicas e Médicas. Instituto de Desenvolvimento Sustentável Mamirauá, MCT-CNPq, Belém, 364 p
Bigler F (1994) Quality control in Trichogramma population. In: Wajnberg E, Hassan SA (eds) Biological control with egg parasitoids, Guildford & King’s Lynn, Biddles
Bueno RCOF, Parra JRP, Bueno AF (2012) Trichogramma pretiosum parasitism of Pseudoplusia includens and Anticarsia gemmatalis eggs at different temperatures. Biol Control 60:154–162
Campbell A, Frazer BD, Gilbert N, Gutierrez AP, Mackauer M (1974) Temperature requirements of some aphids and their parasites. J Appl Ecol 11:419–423
Colazza S, Peri E, Salerno G, Conti E (2014) Host searching by egg parasitoids: exploitation of host chemical cues. In: Côncoli FL, Parra JRP, Zucchi RA (eds) Egg parasitoids in agroecosystems with emphasis on Trichogramma. Springer, London, pp 97–148
DeBach P (1965) Weather and the success of parasites in population regulation. Can Entomol 97:848–863
Foerster MR, Foerster LA (2009) Effects of temperature on the immature development and emergence of five species of Trichogramma. BioControl 54:445–450
Grieshop MJ, Flinn PW, Nechols JR (2010) Effects of intra- and interpatch host density on egg parasitism by three species of Trichogramma. J Insect Sci 10:99
Hansen LS, Jensen KMV (2002) Effect of temperature on parasitism and host-feeding of Trichogramma turkestanica (Hymenoptera: Trichogrammatidae) on Ephestia kuehniella (Lepidoptera: Pyralidae). J Econ Entomol 95:50–56
Ikemoto T, Takai K (2000) A new linearized formula for the law of total effective temperature and the evaluation of line-fitting methods with both variables subject to error. Environ Entomol 29:671–682
Jervis MA, Kidd NAC (1986) Host-feeding strategies in Hymenopteran parasitoids. Biol Rev 61:395–434
Kalyebi A, Sithanantham S, Overholt WA, Hassan SA, Mueke JM (2005) Parasitism, longevity and progeny production of six indigenous Kenyan trichogrammatid egg parasitoids (Hymenoptera: Trichogrammatidae) at different temperature and relative humidity regimes. Biocontrol Sci Tech 15:255–270
Lactin DJ, Holliday NJ, Johnson DL, Craigen R (1995) Improved rate model of temperature-dependent development by arthropods. Environ Entomol 24:68–75
Makee H (2005) Factors influencing the parasitism of codling moth eggs by Trichogramma cacoeciae March. and T. principium Sug. et Sor. (Hymen. Trichogrammatidae). J Pest Sci 78:31–39
Marchioro CA, Foerster LA (2011) Development and survival of the diamondback moth, Plutella xylostella (L.) (Lepidoptera: Yponomeutidae) as a function of temperature: effect on the number of generations in tropical and subtropical regions. Neotrop Entomol 40:533–541
Miura K (2003) Supressive effect of the egg parasitoid Trichogramma chilonis Ishii (Hymenoptera: Trichogrammatidae) on the population density of the diamondback moth. Appl Entomol Zool 38:79–85
Pak GA (1990) Inundative release of Trichogramma for the control of cruciferous Lepidoptera: Preintrodutory selection of an effective parasitoid. In: Talekar NS (ed) Diamonback moth and other crucifer pests: Proceedings of the Second International Workshop. AVRDC, Shanhua, pp 297–308
Pak GA, Van Heiningen TG (1985) Behavioural variations among strains of Trichogramma spp.: adaptability to field-temperature conditions. Entomol Exp Appl 38:3–13
Pizzol J, Pintureau B, Khoualdia O, Desneux N (2010) Temperature-dependent diferences in biological traits between two strains of Trichogramma cacoeciae (Hymenoptera: Trichogrammatidae). J Pest Sci 83:447–452
Pratissoli D, Oliveira HN, Polanczyk RA, Holtz AM, Bueno RCOF, Bueno AF, Gonçalvez JR (2009) Adult feeding and mating effects on the biological potential and parasitism of Trichogramma pretiosum and T. acacioi (Hym.: Trichogrammatidae). Braz Arch Biol Technol 53:1057–1062
Reznik SY, Umarova TY (1991) Host population density influence on host acceptance in Trichogramma. Entomol Exp Appl 58:49–54
Samara R, Monje JC, Zebitz PW, Qubbaj T (2001) Comparative biology and life tables of Trichogramma aurosum on Cydia pomonella at constant temperatures. Phytoparasitica 39:109–119
Sarfraz M, Dosdall LM, Keddie BA (2006) Diamondback moth-host plant interactions: implications for pest management. Crop Prot 25:625–639
Shipp JL, Wang K (1998) Evaluation of commercially produced Trichogramma spp. (Hymenoptera: Trichogrammatidae) for control of tomato pinworm, Keiferia lycopersicella (Lepidoptera: Gelechiidae), on greenhouse tomatoes. Can Entomol 130:721–731
Smith SM (1996) Biological control with Trichogramma: advances, successes and potential for their use in biological control. Annu Rev Entomol 41:375–406
Inc S (2008) Statistica for Windows. Statsoft Inc, Tulsa
Inc S (2002) Table curve 2D. Systat Software Inc, Chicago
Tabone E, Bardon C, Desneux N, Wajnberg E (2010) Parasitism of different Trichogramma species and strains on Plutella xylostella L. on greenhouse cauliflower. J Pest Sci 83:251–256
Thomson L, Rundle B, Hoffmann AA (2001) Developing Trichogramma (Hymenoptera: Trichogrammatidae) as a pest management tool. In: Endersby N (ed) The management of diamondback moth and other crucifers pest: Proceedings of the fourth international workshop. The Regional Institute, Melbourne, pp 51–59
van Lenteren JC (1986) Evaluation, mass production, quality control and release of entomophagous insects. In: Franz JM (ed) Biological plant and health protection. Fischer, Stuttgart, pp 31–56
Vásquez LA, Shelton AM, Hoffmann MP, Roush RT (1997) Laboratory evaluation of commercial Trichogrammatid products for potential use against Plutella xylostella (L.) (Lepidoptera: Plutellidae). Biol Control 9:143–148
Voegele J, Pizzol J, Babi A (1988) The overwintering of some Trichogramma species. In: Trichogramma and other egg parasitoids. Les Colloques dÍNRA, Paris, San Antonio
Yuan XH, Song LW, Zhang JJ, Zang LS, Zhu L, Ruan CC, Sun GZ (2012) Performance of four Chinese Trichogramma species as biocontrol agents of the rice stem borer, Chilo suppressalis, under various temperature and humidity regimes. J Pest Sci 85:497–504
Zalucki MP, Shabbir A, Silva R, Adamson D, Shu-Sheng L, Furlong MJ (2012) Estimating the economic cost of one of the world’s major insect pests, Plutella xylostella (Lepidoptera: Plutellidae): just how long is a piece of string? J Econ Entomol 105:1115–1129
Acknowledgments
The authors are grateful to Dr. Robert M. Perrin, formerly at Syngenta Jealott’s Hill, for critically reviewing the manuscript and the two anonymous reviewers for their valuable comments to the manuscript. We also thank the Brazilian National Research Council (CNPq) and the Brazilian Federal Agency for Support and Evaluation of Graduate Education (CAPES) for the scholarships and financial support provided.
Author information
Authors and Affiliations
Corresponding author
Additional information
Edited by Fernando L Cônsoli – ESALQ/USP
Rights and permissions
About this article
Cite this article
Marchioro, C.A., Krechemer, F.S. & Foerster, L.A. Assessing the Total Mortality Caused by Two Species of Trichogramma on Its Natural Host Plutella xylostella (L.) at Different Temperatures. Neotrop Entomol 44, 270–277 (2015). https://doi.org/10.1007/s13744-014-0263-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13744-014-0263-4