Abstract
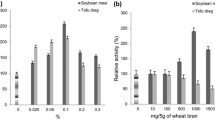
Lentinus tigrinus SSB_W2, isolated from Mahabaleshwar in the Western Ghats of Maharashtra, India, was employed to enhance laccase production in solid-state fermentation (SSF). The spectral analysis indicated that the laccase produced by L. tigrinus is a typical yellow laccase, exhibiting no absorption at 600 nm. Notably, this yellow laccase demonstrated exceptional catalytic activity, as confirmed by electrochemical analysis. Four agricultural processing wastes were evaluated as substrates for SSF, and the results showed that L. tigrinus effectively utilized wheat bran. Initial testing by one-factor-at-a-time method showed 3.79-fold increase in yellow laccase production, which subsequently increased to 6.51-fold after Plackett–Burman design. Moreover, employing response surface methodology resulted in 11.87-fold increase (108,472 IU gds−1) in laccase production. The utilization of yellow laccase for the biotransformation of various textile dyes was investigated, and it exhibited the highest degradation efficiency toward Reactive blue 4, a recalcitrant anthraquinone dye, with a rate of 18.36 mg L−1 h−1, for an initial concentration of 1000 mg L−1.








Similar content being viewed by others
References
Adekunle A, Zhang C, Guo C, Liu C-Z (2017) Laccase production from Trametes versicolor in solid-state fermentation of steam-exploded pretreated cornstalk. Waste Biomass Valoriz. https://doi.org/10.1007/s12649-016-9562-9
Ademakinwa AN, Agboola FK (2016) Biochemical characterization and kinetic studies on a purified yellow laccase from newly isolated Aureobasidium pullulans NAC8 obtained from soil containing decayed plant matter. J Genet Eng Biotechnol 14:143–151. https://doi.org/10.1016/j.jgeb.2016.05.004
Afreen S, Shamsi TN, Baig MA, Ahmad N, Fatima S, Qureshi MI, Hassan MI, Fatma T (2017) A novel multicopper oxidase (laccase) from cyanobacteria: Purification, characterization with potential in the decolorization of anthraquinonic dye. PLoS ONE 12:e0175144
Agrawal K, Chaturvedi V, Verma P (2018) Fungal laccase discovered but yet undiscovered. Bioresour Bioprocess 5:4. https://doi.org/10.1186/s40643-018-0190-z
Akpinar M, Ozturk Urek R (2017) Induction of fungal laccase production under solid state bioprocessing of new agroindustrial waste and its application on dye decolorization. 3 Biotech 7:98. https://doi.org/10.1007/s13205-017-0742-5
Asther M, Lesage L, Drapron R, Corrieu G, Odier E (1988) Phospholipid and fatty acid enrichment of Phanerochaete chrysosporium INA-12 in relation to ligninase production. Appl Microbiol Biotechnol 27:393–398. https://doi.org/10.1007/BF00251775
Aydınoğlu T, Sargın S (2013) Production of laccase from Trametes versicolor by solid-state fermentation using olive leaves as a phenolic substrate. Bioprocess Biosyst Eng 36:215–222. https://doi.org/10.1007/s00449-012-0777-2
Ben Younes S, Sayadi S (2011) Purification and characterization of a novel trimeric and thermotolerant laccase produced from the ascomycete Scytalidium thermophilum strain. J Mol Catal B Enzym 73:35–42. https://doi.org/10.1016/j.molcatb.2011.07.014
Bertrand B, Martínez-Morales F, Trejo-Hernández MR (2017) Upgrading laccase production and biochemical properties: strategies and challenges. Biotechnol Prog 33:1015–1034. https://doi.org/10.1002/btpr.2482
Castro R, Sato H (2015) Enzyme production by solid state fermentation: general aspects and an analysis of the physicochemical characteristics of substrates for agro-industrial wastes valorization. Waste Biomass Valorization 6:1085–1093. https://doi.org/10.1007/s12649-015-9396-x
Cen Q, Wu X, Cao L, Lu Y, Lu X, Chen J, Fu G, Liu Y, Ruan R (2022) Green production of a yellow laccase by Coriolopsis gallica for phenolic pollutants removal. AMB Express 12:96. https://doi.org/10.1186/s13568-022-01434-6
Chaudhari AU, Paul D, Dhotre D, Kodam KM (2017) Effective biotransformation and detoxification of anthraquinone dye reactive blue 4 by using aerobic bacterial granules. Water Res 122:603–613. https://doi.org/10.1016/j.watres.2017.06.005
Chaudhari A, Paul D, Thamke V, Bagade A, Bapat V, Kodam K (2022) Concurrent removal of reactive blue HERD dye and Cr(VI) by aerobic bacterial granules. J Clean Prod 367:133075. https://doi.org/10.1016/j.jclepro.2022.133075
Chaudhari AU, Tapase SR, Markad VL, Kodam KM (2013) Simultaneous decolorization of reactive orange M2R dye and reduction of chromate by Lysinibacillus sp. KMK-A. J Hazard Mater 262:580–588. https://doi.org/10.1016/j.jhazmat.2013.09.006
Chauhan R (2019) Nitrogen sources and trace elements influence Laccase and peroxidase enzymes activity of Grammothele fuligo. Vegetos 32:316–323. https://doi.org/10.1007/s42535-019-00049-w
Chauhan RS, Singh BM, Develash RK (1997) Effect of toxic compounds of exserohilum turcicum on chlorophyll content, callus growth and cell viability of susceptible and resistant inbred lines of maize. J Phytopathol 145:435–440. https://doi.org/10.1111/j.1439-0434.1997.tb00346.x
Chhaya U, Gupte A (2013) Effect of different cultivation conditions and inducers on the production of laccase by the litter-dwelling fungal isolate Fusarium incarnatum LD-3 under solid substrate fermentation. Ann Microbiol 63:215–223. https://doi.org/10.1007/s13213-012-0464-1
Daroch M, Houghton CA, Moore JK, Wilkinson MC, Carnell AJ, Bates AD, Iwanejko LA (2014) Glycosylated yellow laccases of the basidiomycete Stropharia aeruginosa. Enzyme Microb Technol 58–59:1–7. https://doi.org/10.1016/j.enzmictec.2014.02.003
Durán-Sequeda D, Suspes D, Maestre E, Alfaro M, Perez G, Ramírez L, Pisabarro AG, Sierra R (2021) Effect of nutritional factors and copper on the regulation of laccase enzyme production in Pleurotus ostreatus. J Fungi (basel, Switzerland). https://doi.org/10.3390/jof8010007
El-Batal AI, ElKenawy NM, Yassin AS, Amin MA (2015) Laccase production by Pleurotus ostreatus and its application in synthesis of gold nanoparticles. Biotechnol Rep 5:31–39. https://doi.org/10.1016/j.btre.2014.11.001
Elisashvili V, Kachlishvili E, Khardziani T, Agathos SN (2010) Effect of aromatic compounds on the production of laccase and manganese peroxidase by white-rot basidiomycetes. J Ind Microbiol Biotechnol 37:1091–1096. https://doi.org/10.1007/s10295-010-0757-y
Ferraroni M, Myasoedova NM, Schmatchenko V, Leontievsky AA, Golovleva LA, Scozzafava A, Briganti F (2007) Crystal structure of a blue laccase from Lentinus tigrinus: evidences for intermediates in the molecular oxygen reductive splitting by multicopper oxidases. BMC Struct Biol 7:60. https://doi.org/10.1186/1472-6807-7-60
Garg N, Bieler N, Kenzom T, Chhabra M, Ansorge-Schumacher M, Mishra S (2012) Cloning, sequence analysis, expression of Cyathus bulleri laccase in Pichia pastoris and characterization of recombinant laccase. BMC Biotechnol 12:75. https://doi.org/10.1186/1472-6750-12-75
Gonzalez JC, Medina SC, Rodriguez A, Osma JF, Alméciga-Díaz CJ, Sánchez OF (2013) Production of Trametes pubescens laccase under submerged and semi-solid culture conditions on agro-industrial wastes. PLoS ONE 8:e73721
Hadibarata T, Kristanti RA (2012) Fate and cometabolic degradation of benzo[a]pyrene by white-rot fungus Armillaria sp. F022. Bioresour Technol 107:314–318. https://doi.org/10.1016/j.biortech.2011.12.046
Hsu C-A, Wen T-N, Su Y-C, Jiang Z-B, Chen C-W, Shyur L-F (2012) Biological degradation of anthroquinone and azo dyes by a novel laccase from Lentinus sp. Environ Sci Technol 46:5109–5117. https://doi.org/10.1021/es2047014
Ike PTL, Moreira AC, de Almeida FG, Ferreira D, Birolli WG, Porto ALM, Souza DHF (2015) Functional characterization of a yellow laccase from Leucoagaricus gongylophorus. Springerplus 4:654. https://doi.org/10.1186/s40064-015-1464-y
Jegatheesan M, Eyini M (2015) Response surface methodology mediated modulation of laccase production by Polyporus arcularius. Arab J Sci Eng 40:1809–1818. https://doi.org/10.1007/s13369-014-1499-3
Kumar A, Chandra R (2020) Ligninolytic enzymes and its mechanisms for degradation of lignocellulosic waste in environment. Heliyon 6:e03170. https://doi.org/10.1016/j.heliyon.2020.e03170
Lechner BE, Papinutti VL (2006) Production of lignocellulosic enzymes during growth and fruiting of the edible fungus Lentinus tigrinus on wheat straw. Process Biochem 41:594–598. https://doi.org/10.1016/j.procbio.2005.08.004
Leontievsky AA, Vares T, Lankinen P, Shergill JK, Pozdnyakova NN, Myasoedova NM, Kalkkinen N, Golovleva LA, Cammack R, Thurston CF, Hatakka A (1997a) Blue and yellow laccases of ligninolytic fungi. FEMS Microbiol Lett 156:9–14. https://doi.org/10.1111/j.1574-6968.1997.tb12698.x
Leontievsky A, Myasoedova N, Pozdnyakova N, Golovleva L (1997b) “Yellow” laccase of Panus tigrinus oxidizes non-phenolic substrates without electron-transfer mediators. FEBS Lett 413:446–448. https://doi.org/10.1016/s0014-5793(97)00953-8
Liers C, Ullrich R, Pecyna M, Schlosser D, Hofrichter M (2007) Production, purification and partial enzymatic and molecular characterization of a laccase from the wood-rotting ascomycete Xylaria polymorpha. Enzyme Microb Technol 41:785–793. https://doi.org/10.1016/j.enzmictec.2007.07.002
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Mayolo-Deloisa K, González-González M, Rito-Palomares M (2020) Laccases in food industry: bioprocessing, potential industrial and biotechnological applications. Front Bioeng Biotechnol. https://doi.org/10.3389/fbioe.2020.00222
Meehnian H, Jana AK, Jana MM (2016) Effect of particle size, moisture content, and supplements on selective pretreatment of cotton stalks by Daedalea flavida and enzymatic saccharification. 3 Biotech 6:235. https://doi.org/10.1007/s13205-016-0548-x
Mot AC, Coman C, Hadade N, Damian G, Silaghi-Dumitrescu R, Heering H (2020) “Yellow” laccase from Sclerotinia sclerotiorum is a blue laccase that enhances its substrate affinity by forming a reversible tyrosyl-product adduct. PLoS ONE 15:e0225530
Mukhopadhyay M, Banerjee R (2015) Purification and biochemical characterization of a newly produced yellow laccase from Lentinus squarrosulus MR13. 3 Biotech 5:227–236. https://doi.org/10.1007/s13205-014-0219-8
Nayak B, Choudhary R (2022) Optimization, purification and characterization of laccase from lignocellulolytic fungi Dichotomopilus funicola NFCCI 4534 and Alternaria padwickii NFCCI 4535. Biocatal Agric Biotechnol 42:102344. https://doi.org/10.1016/j.bcab.2022.102344
Niku-Paavola M-L, Karhunen E, Kantelinen A, Viikari L, Lundell T, Hatakka A (1990) The effect of culture conditions on the production of lignin modifying enzymes by the white-rot fungus Phlebia radiata. J Biotechnol 13:211–221. https://doi.org/10.1016/0168-1656(90)90106-L
Omeje K, Nnolim N, Ezema B, Ozioko J, EZE, S.O., (2020) Synthetic dyes decolorization potential of agroindustrial waste-derived thermo-active laccase from Aspergillus species. Biocatal Agric Biotechnol 29:101800. https://doi.org/10.1016/j.bcab.2020.101800
Patel H, Gupte A (2016) Optimization of different culture conditions for enhanced laccase production and its purification from Tricholoma giganteum AGHP. Bioresour Bioprocess 3:11. https://doi.org/10.1186/s40643-016-0088-6
Pinheiro VE, Michelin M, Vici AC, de Almeida PZ, Teixeira de Moraes Polizeli MdeL (2020) Trametes versicolor laccase production using agricultural wastes: a comparative study in Erlenmeyer flasks, bioreactor and tray. Bioprocess Biosyst Eng 43:507–514. https://doi.org/10.1007/s00449-019-02245-z
Piscitelli A, Giardina P, Lettera V, Pezzella C, Sannia G, Faraco V (2011) Induction and transcriptional regulation of laccases in fungi. Curr Genomics 12:104–112. https://doi.org/10.2174/138920211795564331
Pointing S, Jones E, Vrijmoed L (2000) Optimization of laccase production by Pycnoporus sanguineus in submerged liquid culture. Mycologia 92:139. https://doi.org/10.2307/3761458
Pozdnyakova NN, Rodakiewicz-Nowak J, Turkovskaya OV (2004) Catalytic properties of yellow laccase from Pleurotus ostreatus D1. J Mol Catal B Enzym 30:19–24. https://doi.org/10.1016/j.molcatb.2004.03.005
Radveikienė I, Vidžiūnaitė R, Meškienė R, Meškys R, Časaitė V (2021) Characterization of a yellow laccase from Botrytis cinerea 241. J Fungi (basel, Switzerland). https://doi.org/10.3390/jof7020143
Reddy MS, Kanwal HK (2022) Influence of carbon, nitrogen sources, inducers, and substrates on lignocellulolytic enzyme activities of Morchella spongiola. J Agric Food Res 7:100271. https://doi.org/10.1016/j.jafr.2022.100271
Revankar MS, Desai KM, Lele SS (2007) Solid-state fermentation for enhanced production of laccase using indigenously isolated Ganoderma sp. Appl Biochem Biotechnol 143:16–26. https://doi.org/10.1007/s12010-007-0029-0
Sadeghian-Abadi S, Rezaei S, Yousefi-Mokri M, Faramarzi MA (2019) Enhanced production, one-step affinity purification, and characterization of laccase from solid-state culture of Lentinus tigrinus and delignification of pistachio shell by free and immobilized enzyme. J Environ Manag 244:235–246. https://doi.org/10.1016/j.jenvman.2019.05.058
Sadh PK, Duhan S, Duhan JS (2018) Agro-industrial wastes and their utilization using solid state fermentation: a review. Bioresour Bioprocess 5:1. https://doi.org/10.1186/s40643-017-0187-z
Saha R, Mukhopadhyay M (2022) Time-dependent electrochemical characteristics of a phenolic and non-phenolic compound in the presence of laccase/ABTS system. PLoS ONE 17:e0275338
Santana TT, Linde GA, Colauto NB, do Valle JS (2018) Metallic-aromatic compounds synergistically induce Lentinus crinitus laccase production. Biocatal Agric Biotechnol 16:625–630. https://doi.org/10.1016/j.bcab.2018.10.018
Savino S, Bulgari D, Monti E, Gobbi E (2021) Agro-industrial wastes: a substrate for multi-enzymes production by Cryphonectria parasitica. Fermentation. https://doi.org/10.3390/fermentation7040279
Singh AK, Singh RKP (2017) Effect of surfactants on lignin modifying enzyme activity of Ganoderma species KX879638 and Lentinus species KY006984 under solid state fermentation. Afr J Microbiol Res 11:1520–1527
Slagman S, Zuilhof H, Franssen MCR (2018) Laccase-mediated grafting on biopolymers and synthetic polymers: a critical review. ChemBioChem 19:288–311. https://doi.org/10.1002/cbic.201700518
Sondhi S, Saini K (2019) Response surface based optimization of laccase production from Bacillus sp. MSK-01 using fruit juice waste as an effective substrate. Heliyon 5:e01718. https://doi.org/10.1016/j.heliyon.2019.e01718
Tamura K, Stecher G, Peterson D, Filipski A, Kumar S (2013) MEGA6: molecular evolutionary genetics analysis version 6.0. Mol Biol Evol 30:2725–2729. https://doi.org/10.1093/molbev/mst197
Teodoro T, Oliveira F, Poffo C, Braga L, Arbigaus A, Rampinelli J, Elisabeth W, Bonatti-Chaves M, Furlan S (2018) The influence of Tween 80 on laccase production by Pleurotus sajor-caju and the efficiency of crude enzyme broth in the removal of bisphenol-A. Arq Inst Biol (sao Paulo). https://doi.org/10.1590/1808-1657001022017
Walde P, Kashima K, Ćirić-Marjanović G (2019) Synthesizing polyaniline with laccase/O2 as catalyst. Front Bioeng Biotechnol. https://doi.org/10.3389/fbioe.2019.00165
Wang X, Zhang G, Dang Y (2022) Enhanced extraction of flavonoids from licorice residues by solid-state mixed fermentation. Waste Biomass Valorization. https://doi.org/10.1007/s12649-022-01803-z
Xin F, Geng A (2011) Utilization of horticultural waste for laccase production by Trametes versicolor under solid-state fermentation. Appl Biochem Biotechnol 163:235–246. https://doi.org/10.1007/s12010-010-9033-x
Yuan T, Zhang S, Chen Y, Zhang R, Chen L, Ruan X, Zhang S, Zhang F (2021) Enhanced reactive blue 4 biodegradation performance of newly isolated white rot fungus Antrodia p5 by the synergistic effect of herbal extraction residue. Front Microbiol. https://doi.org/10.3389/fmicb.2021.644679
Zhao D, Zhang X, Cui D, Zhao M (2012) Characterisation of a novel white laccase from the deuteromycete fungus Myrothecium verrucaria NF-05 and its decolourisation of dyes. PLoS ONE 7:e38817
Zhou Q, Wu W, Xing T (2022) Study on the mechanism of laccase-catalyzed polydopamine rapid dyeing and modification of silk. RSC Adv 12:3763–3773. https://doi.org/10.1039/D1RA08807F
Acknowledgements
The authors Seema Bhoyar (Ref. No: 20/12/2015(ii)EU-V) and Ashvini Chaudhari (F.4-2/2006(BSR)/BL/19-20/0128) would like to thank University Grants Commission (UGC), New Delhi, India for providing UGC-JRF fellowship and Dr. D. S. Kothari Postdoctoral Fellowship, respectively. The authors would like to thank National Fungal Culture Collection of India Agharkar Research Institute, Pune for their help in fungal identification. We would like to express our sincere gratitude to the experienced editor from EditPi (https://sites.google.com/editpi.com/editpi/home) for their invaluable assistance in editing and enhancing the proficiency of our research paper.
Author information
Authors and Affiliations
Contributions
SSB: Plan of work, experimentation, analysis of results and draft manuscript writing. RVL: Carried out the statistical analysis of Response surface methodology. AUC: Planned and carried out phytotoxicity study, data analysis and manuscript writing. MAD: Helped in electrochemical characterization of enzyme. SDS: Providing the infrastructure for electrochemical characterization and data analysis. KMK: Supervision, designing of work plan and experiments, article editing and submission.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they do not have any competing financial or personal interests.
Ethical approval
The study does not involve human participants or use of animals.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Bhoyar, S.S., Chaudhari, A.U., Desai, M.A. et al. Wheat bran as an efficient agro-process waste for enhanced yellow laccase production by Lentinus tigrinus SSB_W2 and its application in anthraquinone dye degradation. 3 Biotech 14, 33 (2024). https://doi.org/10.1007/s13205-023-03881-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-023-03881-9