Abstract
12-Tungstophosphoric acid (PW) supported on KSF montmorillonite, PW/KSF, was used as catalyst for deep oxidative desulfurization (ODS) of mixed thiophenic compounds in model oil and crude oil under mild conditions using hydrogen peroxide (H2O2) as an oxidizing agent. A one-factor-at-a-time method was applied for optimizing the parameters such as temperature, reaction time, amount of catalyst, type of extractant and oxidant-to-sulfur compounds (S-compounds) molar ratio. The corresponding products can be easily removed from the model oil by using ethanol as the best extractant. The results showed high catalytic activity of PW/KSF in the oxidative removal of dibenzothiophene (DBT) and mixed thiophenic model oil under atmospheric pressure at 75 °C in a biphasic system. To investigate the oxidation and adsorption effects of crude oil composition on ODS, the effects of cyclohexene, 1,7-octadiene and o-xylene with different concentrations were studied.
Similar content being viewed by others
Explore related subjects
Find the latest articles, discoveries, and news in related topics.Avoid common mistakes on your manuscript.
1 Introduction
A major cause of air pollution is the burning of fossil fuels and producing compounds such as NO x and SO x , which is released from factories and vehicle exhausts. Sulfur content in crude oil is highly significant due to the high toxicity, corrosivity and air pollution that results from the burning of sulfur-containing fuel products (Collins et al. 1997). In accordance with the increasingly stringent environmental regulations on sulfur concentration in transportation fuels and the demand for diesel fuel with low sulfur content, removal of sulfur-containing compounds is an important issue for the petroleum refining industry (Mei et al. 2003).
To achieve this goal, most sulfur-containing compounds are easily desulfurized by hydrodesulfurization (HDS), in which H2 gas is used to remove the S-compounds through the formation of hydrocarbons and H2S. It is difficult to decrease the sulfur content from several hundred ppm to a few ppm by the HDS method which is extremely efficient in removing thiols, sulfides and disulfides, but less effective for thiophenic compounds (García-Gutiérrez et al. 2008). Due to the high capital and operating costs of the HDS process due to factors such as reduced catalyst lifetime, higher hydrogen consumption, high temperature and pressure, oxidative desulfurization (ODS) is proposed as an appropriate substitute for the traditional HDS process and complementary method with ambient pressure, relatively low temperature, high selectivity and no consumption of hydrogen (Khenkin and Neumann 2011; Caero et al. 2005). The integration of an ODS unit with a conventional hydro-treating unit can improve the economics of diesel desulfurization process in comparison with the current HDS technology (Stanislaus et al. 2010).
Generally, an ODS process consists of two steps: The first step is the oxidation of sulfur; various oxidants such as hydrogen peroxide (H2O2) (Rafiee and Eavani 2013; Gao et al. 2010a, b), formic acid (Shojaei et al. 2014), acetic acid (Abdalla et al. 2009a, b, O2 (Dooley et al. 2013) and t-BuOOH (Hui-Peng et al. 2007; Ishihara et al. 2005) are used in ODS process. Among these oxidants, H2O2 is mostly chosen as an oxidant due to its innocuous side product (H2O). Thus, some studies of H2O2 have been conducted for investigation of its use in ODS reactions (Rafiee and Mirnezami 2014; Lei et al. 2013; Da Silva and Dos Santos 2013). The second step is the removal of products (sulfoxide or sulfone) by solvent extraction. These compounds are removed by polar solvents such as methanol (Abdalla and Li 2012), N,N-dimethylformamide (DMF) (Bakar et al. 2012), acetonitrile (MeCN) (Li et al. 2013) and DMSO (Hassan et al. 2013). In recent years, there has been an increasing interest in polyoxometalates (POMs) as catalysts due to their composition, size, shape, photochemical response, ionic charge, acidic properties and tunable redox properties (Zhang et al. 2013). Heteropoly acids (HPAs) and supported HPAs are widely used as catalysts in various acid-catalyzed reactions and partial oxidation reactions because of their strong acidity and oxidation activity (Chamack et al. 2014; Zhang et al. 2011a, b; Te et al. 2001). Among the different types of HPAs, Keggin-type HPAs and their salts have been widely used for acid–base and oxidation reactions (Zhang et al. 2011a, b). In general, H3PMo12O40 (PMo), H3PW12O40 (PW), H4SiMo12O40 (SiMo) and H4SiW12O40 (SiW) are employed in the first instance for many applications (Katsoulis 1998). The results show that Keggin-type HPA catalysts with H2O2 may be promising for ODS of model oils (Kozhevnikov 1998). Previous reports have demonstrated the effective catalytic performances of supported HPAs for the oxidation of thiophene (T) and its derivatives with different oxidants (Hui-Peng et al. 2007; Kukovecz et al. 2002; Yan et al. 2009b). However, most of the research up to now has investigated homogenous or amphiphilic catalysts and oxidants (Gao et al. 2006; Zhang et al. 2012a, b; Maity et al. 2009; Qiu et al. 2009; Xue et al. 2012; Bhutto et al. 2016). In these systems, the catalysts are usually dissolved in the continuous phase or inside the emulsion droplets; thus, it is difficult to separate the catalysts and recycle them from the emulsion systems.
The current study aims to investigate a kind of catalyst which is not soluble in the oil phase or extractant solvent. PW supported on KSF (PW/KSF) was prepared by impregnation and used as catalyst for the ODS of model oil. Thiophene (T), benzothiophene (BT) and dibenzothiophene (DBT) in n-hexane were the simulated model oils. Different extractants were EtOH, MeCN and DMF. H2O2 was chosen as an oxidant. The effect of various parameters such as temperature, oxidant-to-S-compounds molar ratio (O/S), type of extractant, reaction time and amount of the catalyst was investigated and optimal conditions of the process were evaluated for real oil. The catalytic activity of PW/KSF was investigated for crude oil before and after the HDS process. Additionally, for assessing the influence of crude oil composition, 5 vol%, 15 vol% and 25 vol% of o-xylene, cyclohexene and 1,7-octadiene were, respectively, added to 1000 ppm mixed model oil. For testing real catalytic activity of PW/KSF, ODS of a crude oil and HDS-treated crude oil was conducted under optimal conditions.
2 Experimental
2.1 Materials and methods
H3PW12O40 (PW) hydrate was purchased from Aldrich, and KSF montmorillonite was obtained from the Fluka Company. n-Hexane (98 %), ethanol (99 %), acetonitrile (98 %), N,N-dimethylformamide (DMF, 99.5 %), T (98 %), BT (98 %), DBT (98 %), H2O2 (30 %), cyclohexene (99 %), o-xylene (99 %) and 1,7-octadiene (97 %) were purchased from the Merck Company and used without further purification.
The ODS experiments were carried out at atmospheric pressure in a 50-mL glass batch reactor, equipped with a temperature controller, a condenser and a mechanical stirrer. Fourier transform infrared spectroscopy (FTIR) spectra were recorded with KBr pellets using a Rayleigh WQF-510 FTIR. The total sulfur content was analyzed with a Multi EA 3100 Element Analyzer (Analytik Jena AG Company). The Multi EA 3100 model can achieve detection limits of 10 ppb for sulfur determination by UV fluorescence and 50 ppb for nitrogen determination by chemiluminescence. All experiments were conducted and repeated twice more to ensure reproducibility of the results.
2.2 Catalyst preparation
The PW/KSF was prepared by the impregnation method, which was developed by our research group (Rafiee et al. 2009). For preparation of the PW/KSF, KSF montmorillonite was oven-dried at 120 °C for 2 h prior to its use as support. An appropriate amount of PW (to produce 40 wt% of PW to support) was dissolved in 5 mL of dry methanol and added dropwise to pre-dried KSF while stirring with a glass rod. Initially with addition of PW solution, the PW/KSF clay was in a powdery form, but with further addition of PW solution, the clay turned to a paste form. The paste changed to free flowing powder with further stirring as the methanol evaporated. Characterization of the catalyst was reported previously (Rafiee et al. 2009, 2011).
2.3 Activity test
2.3.1 Oxidation of DBT
In a typical experiment, the water bath was first heated to reach and maintain the reaction temperature (55, 65 and 75 °C), and then model oil was prepared by dissolving DBT in n-hexane/EtOH (50/50 vol. ratio) to get 1000 ppm, 500 ppm and 100 ppm solution of DBT. In addition, required amounts of 30 vol% aq. H2O2 solution (O/S molar ratio 8:1, 10:1, 12:1 and 14:1) and certain amounts of the catalyst (0.02 g (0.0028 mmol), 0.03 g (0.004 mmol) and 0.04 g (0.0056 mmol)) were added to the mixture and stirred vigorously while still in the water bath to start the reaction.
With the aid of thin-layer chromatography (TLC) analysis (with n-hexane/ethyl acetate, 2:1 volume ratio as eluent), the oxidized DBT (sulfones and sulfoxides) spots could be seen by illuminating the TLC plate with ultraviolet (UV) light. After completion of the reaction (80 min), the mixture was left to settle for 30 min at room temperature and the products were extracted by extractant solvent. The heterogeneous catalyst was separated by centrifuging at 1500 rpm. For quantification of ODS products, upper liquid layer (n-hexane) was withdrawn and analyzed for S by the Analytik Jena AG—Multi EA 3100 Element Analyzer.
2.3.2 Oxidation of mixed thiophenic model oil
A typical reaction procedure for ODS of mixed model oil was as follows: Mixed model oil was prepared by dissolving required amounts of DBT, BT and T (DBT/BT/T, 500:250:250 ppm) in 5 mL n-hexane to get mixed model oil with sulfur concentration of 1000 ppm. PW/KSF (0.03 g, 0.004 mmol), 5 mL extraction phase and required amount of 30 % aq. H2O2 solution (O/S molar ratio 10:1) were added to the model oil, and the resulting mixture was stirred at atmospheric pressure at 75 °C for 80 min. After completion of the reaction, the mixture was left to settle for 30 min at room temperature and the corresponding products were extracted by extractant solvent. TLC was performed with n-hexane/ethyl acetate, in 3:1 volume ratio, as eluent. The ODS of 500 ppm and 100 ppm (sulfur concentration) mixed model oil was performed in the same way for ODS of 1000 ppm (sulfur concentration) mixed model oil, with O/S molar ratio fixed at 10:1 in the presence of PW/KSF (0.03 g) at 75 °C.
In order to test the catalytic recyclability of PW/KSF in the ODS of 1000 ppm (sulfur concentration) mixed model oil, the heterogeneous catalyst was separated by centrifuge with 1500 rpm, washed three times with 2 mL MeCN and then dried at room temperature for 24 h. The recovered catalyst was charged into the next run with fresh reactants.
2.3.3 Oxidation of crude oil
In the same manner as the oxidation of the mixed thiophenic model oil, PW/KSF (0.03 g) catalyst, 50 μL 30 % aq. H2O2 and 5 mL extractant (EtOH, DMF or MeCN) were added to 5 mL crude oil (with sulfur concentration of 1000 ppm, API 41.67). The mixture was stirred vigorously at atmospheric pressure at 75 °C. After completion of the reaction, the polar phase was analyzed for S. For crude oil after HDS, 5 mL of HDS-treated crude oil (with sulfur concentration of 300 ppm), PW/KSF (0.03 g) catalyst and 15 μL 30 % aq. H2O2 (under the same conditions as above mentioned) were added and the mixture stirred at 75 °C for 80 min. The upper phase was withdrawn and analyzed by the total sulfur analyzer.
3 Results and discussion
3.1 DBT oxidative desulfurization (ODS)
In order to investigate the activity of PW/KSF and optimize the reaction conditions, deep desulfurization of 1000 ppm DBT in n-hexane was carried out, with EtOH as extractant, at different temperatures, O/S molar ratios and amounts of the catalyst (Table 1). As the temperature increased from 55 °C to 75 °C, the conversion of DBT increased greatly from 31 % to 99 % at an O/S molar ratio of 10:1. The corresponding TON = Nproduct/NPW was calculated (where TON is the mole number of converted DBT per mole of PW supported on KSF). The TON value increased with an increase in reaction time at each reaction temperature (Table 1, Nos. 5, 6 and 8). With an increase in temperature, the TON value for ODS increased at each reaction time. Above an O/S molar ratio 10:1, TON value increased very little.
Table 1 shows that at 75 °C, the conversion of DBT and corresponding reaction time changed very little when the amount of the catalyst increased from 0.03 g to 0.04 g. In addition, the ODS activity of PW/KSF increased only slightly when the O/S molar ratio was higher than 10:1. Based on the above experimental results, it was found that optimum conditions for ODS were O/S (10:1), temperature (75 °C) and amount of the catalyst (0.03 g). These optimum conditions were used in the next experiments. Prior to investigating the effect of the catalyst (PW/KSF) loading on ODS, two more experiments were conducted for comparison: one with O/S molar ratio 10:1 in the presence of 0.03 g of KSF and another without using any catalyst at optimum reaction conditions. The results showed that the conversion of DBT was 46 % and 28 %, respectively, which did not meet the requirement of deep desulfurization. It was due to the lack of a suitable reaction medium, and DBT could not effectively contact the H2O2 (Zhang et al. 2013). In both cases, noticeable transformation of DBT into sulfone was not observed on the TLC plate.
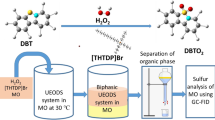
During the reaction, it was found that the mixture (EtOH, n-hexane and DBT) tended to form two phases. The active polyoxoperoxo (peroxo-metallate complexes) species converted DBT to DBTO2 and the oxidized DBT moved into the EtOH phase. The catalyst was a separate phase during the entire reaction process (Scheme 1).
The experiment was also conducted with 500 ppm and 100 ppm DBT in n-hexane with EtOH as extractant. Figure 1 shows the catalytic performance of PW/KSF for 1000, 500 and 100 ppm of DBT in n-hexane at optimum reaction conditions. The results showed that the catalyst was active for all the concentrations of DBT in n-hexane with EtOH as extractant. It can be seen that DBT was almost fully converted by PW/KSF. Table 2 shows the comparison of catalytic activity of PW/KSF with that of other reported catalysts. The results indicated that PW/KSF was quite reliable for ODS in comparison with other catalysts. There are some disadvantages of these reported systems (compared with our system) such as long reaction time, high reaction temperature or large amount of catalyst and using ionic liquids as additives.
The mechanism of the catalytic desulfurization process (Scheme 2) is assumed to include the following steps: (1) transformation of [PW12O40]3− (denoted as PW) into its peroxide form [PO4{WO(O2)2}4]3− (denoted as PW4) in the presence of H2O2; (2) oxidation of the extracted DBT to DBTO (corresponding sulfoxide) and DBTO2 (corresponding sulfone) by PW4, and reduction of PW4 to [PO4{WO(O2)}4]3−; (3) regeneration of PW4 via oxidizing its reduced form [PO4{WO(O2)}4]3− by H2O2; (4) transformation of PW4 into PW12 species with free tungsten species, after the desulfurization (Zhang et al. 2012a, b).
3.2 Desulfurization of mixed thiophenic model oil
Another model oil was prepared by dissolving required amounts of DBT, BT and T in n-hexane to get mixed model oil with sulfur concentration of 1000, 500 and 100 ppm, respectively. The experiments were performed under the conditions of O/S molar ratio 10:1, 0.03 g of PW/KSF, 75 °C and reaction time of 80 min (Figure 2). The result showed that the catalyst was effective for mixed thiophenic compounds models. It was clear that the conversion rate of 1000, 500 and 100 ppm mixed model could reach 97 %, 95 % and 93 %, respectively. As shown in Figs. 1 and 2, the initial S-compounds concentration did not have much more effect on ODS after 80 min.
It is instructive to compare the catalytic activity of PW/KSF with different extractants (EtOH, DMF and MeCN) in the oxidation process (Fig. 3). In general, the performance of the extractant solvent depends considerably on the solubility of the sulfur oxidation products in the reaction mixture. During ODS process, the oxidized S-compounds are transferred to the extractant solvent, and the solvent can influence the mass transport and subsequently have diffusional problems, especially with porous catalysts (Caero et al. 2005). As shown in Fig. 3, EtOH was the best extractant for removing oxidized thiophenic compounds in the model oil. It can be found that the catalytic activity of PW/KSF depended on the kind of the extractant (Fig. 3). To study the recyclability of the catalyst, 1000 ppm mixed model oil was used successively seven times and the catalytic activity decreased a little (Fig. 4). The recycled catalyst showed 83 % ODS removal after seventh recycling. The average recovery weight was ~93 % and ~92 % for PW/KSF and model oil, respectively. The FTIR spectrum of the recovered PW/KSF after fourth run confirmed no significant change in the Keggin structure of the catalyst (Fig. 5). The PW12O −340 Keggin ion structure consists of a PO4 tetrahedron surround by four W3O13 groups formed by edge-sharing octahedral. These groups are connected with each other by corner-sharing oxygens. This structure consists of four types of oxygens, being responsible for the fingerprint bands of the Keggin ion between 700 cm−1 and 1200 cm−1. Bulk PW shows the typical bands for absorptions of P–O (1080 cm−1), W = Ot (985 cm−1), W–Oc–W (890 cm−1) and W–Oe–W (814 cm−1). For PW supported on KSF, the bands at 1078, 986, 899 and 802 cm−1 are attributed to stretching vibrations of P–O, W=Ot, W–Oc–W corner-shared bonds and W–Oe–W edge-shared bonds, respectively, which indicates the encapsulation of PW in the KSF frameworks, respectively (Rafiee et al. 2009; Zhang et al. 2011a, b). After catalytic experiments on 1000 ppm mixed model oil using ethanol as extractant solvent, the PW/KSF exhibits a TON = 8.69 at optimum conditions.
3.3 ODS desulfurization of crude oil
For investigating the industrial performance of PW/KSF, the ODS was tested using 1000 ppm crude oil as a real model oil with different extractants (MeCN, DMF and EtOH). The results showed that the extractability of S-compounds decreased in the order of MeCN > DMF > EtOH (Fig. 6). The conversion of S-compounds of 1000 ppm crude oil only reached 60 %, 53 % and 47 % by employing MeCN, DMF and EtOH, respectively, as extractant.
Desulfurization of crude oil has been a challenge for a long time, and ODS can be a promising complementary method for hydro-treated crude oil. So, the HDS-treated crude oil was selected for testing performance of the catalyst. By using EtOH, MeCN and DMF as extractants at optimum conditions, the conversion of S-compounds of 300 ppm HDS crude oil only reached 41 %, 38 % and 39 %, respectively (Fig. 6). Indeed, when crude oil was first desulfurized with HDS process, thiols, sulfides and disulfides were removed but aromatic S-compounds (thiophenic compounds) were remained in crude oil. It should be noted that EtOH is the most effective extractant for crude oil after HDS process but it is not as efficient for crude oil without HDS.
3.4 Effect of composition of mixed model oil on the ODS conversion
With respect to the considerable decrease in ODS conversion of crude oil in comparison with simulated model oil (see Fig. 6), the effect of crude oil composition on ODS conversion was investigated. Crude oil is the most important source of aromatics and olefins, and they may have a significant effect on ODS (Abdalla et al. 2009a, b; Yan et al. 2013; Xiao et al. 2014). In these systems (PW catalysts), the active sites of the catalyst with H2O2 exhibited considerable activity in epoxidation of alkenes (Wenjia et al. 2013; Aoto et al. 2014). For investigation of the effects of composition of crude oil and epoxidation of alkenes, 5 vol%, 15 vol% and 25 vol% (according to crude oil properties of west Iranian oil wells) of o-xylene, cyclohexene and 1,7-octadiene were, respectively, added to the 1000 ppm model oil (DBT/BT/T, 500:250:250 ppm) to examine their effects on ODS using the PW/KSF catalyst with H2O2 at optimum reaction conditions. Figure 7 shows the ODS conversion of the model oil with addition of o-xylene at various concentrations. The ODS conversion (97 %) of model oil decreased by 10 %, 24 % and 28 %, respectively, after 80 min of reaction time when 5 vol%, 15 vol% and 25 vol% of o-xylene were added to the model oil.
Figure 8 shows that when 5 vol%, 15 vol% and 25 vol% of cyclohexene were added to the model oil, the ODS conversion decreased to 63 %, 52 % and 47 %, respectively, compared to that of model oil. Under the same reaction conditions, the ODS conversion of model oil decreased to 84 %, 72 % and 66 % by the addition of 5 vol%, 15 vol% and 25 vol% of 1,7-octadiene (Fig. 9). The results of olefins and aromatics addition showed strong negative effects on ODS. Therefore, the presence of aromatics and olefins in crude oil will decrease ODS conversion.
The high electron-donating ability of the olefins and aromatics double bonds is considered to be the problem factor in ODS of crude oil (Te et al. 2001; Xiao et al. 2014). Figures 7, 8 and 9 show that the inhibiting effect on ODS conversion increased in the order of o-xylene < 1,7-octadiene < cyclohexene. To explain this trend, the electronic and steric effects should be taken into consideration. The partial electron charge on the alkenes and aromatics plays a detrimental role for oxidation reactivity of the catalyst (Xiao et al. 2014; Yan et al. 2009a).
4 Conclusions
Catalytic activity of PW/KSF was investigated for ODS of DBT and mixed thiophenic model oils and crude oil. First, effects of temperature, O/S molar ratio, amount of the catalyst and type of the extractant solvent on ODS of DBT were investigated. The results of the experiments show that DBT conversion in model oil was excellent for 100 ppm, 500 ppm and even 1000 ppm and conversion for 1000, 500 and 100 ppm mixed thiophenic compounds was 97 %, 95 % and 93 %, respectively. For 1000 ppm crude oil and 300 ppm HDS-treated crude oil, catalytic activities were 47 % and 41 % by using EtOH as extractant. It was discovered that EtOH is the best extractant solvent for removing oxidized S-compounds of mixed model oil. In the second part of the study, effects of cyclohexene, 1,7-octadiene and o-xylene were investigated for mixed model oil and the ODS decreases in order of: o-xylene < 1,7-octadiene < cyclohexene.
References
Abdalla ZEA, Li B, Tufail A. Preparation of phosphate promoted Na2WO4/Al2O3 catalyst and its application for oxidative desulfurization. J Ind Eng Chem. 2009a;15:780–3.
Abdalla ZEA, Li B, Tufail A. Direct synthesis of mesoporous (C19H42N)4H3(PW11O39)/SiO2 and its catalytic performance in oxidative desulfurization. Coll Surf A. 2009b;341:86–92.
Abdalla ZEA, Li B. Preparation of MCM-41 supported (Bu4N)4H3(PW11O39) catalyst and its performance in oxidative desulfurization. Chem Eng J. 2012;200–202:113–21.
Aoto H, Matsui K, Sakai Y, et al. Zirconium(IV)- and hafnium(IV)- containing polyoxometalates as oxidation precatalysts: homogeneous catalytic epoxidation of cyclooctene by hydrogen peroxide. J Mol Catal A: Chem. 2014;394:224–31.
Bakar WAWA, Ali R, Kadir AAA, Mokhtar WNAW. Effect of transition metal oxides catalysts on oxidative desulfurization of model diesel. Fuel Process Technol. 2012;101:78–84.
Bhutto AW, Abro R, Gao S, et al. Oxidative desulfurization of fuel oils using ionic liquids: a review. J Taiwan Inst Chem Eng. 2016;62:84–97.
Caero LC, Hernández E, Pedraza F, Murrieta F. Oxidative desulfurization of synthetic diesel using supported catalysts: part I. Study of the operation conditions with a vanadium oxide based catalyst. Catal Today. 2005;107:564–9.
Chamack M, Mahjoub AR, Aghayan H. Cesium salts of tungsten-substituted molybdophosphoric acid immobilized onto platelet mesoporous silica: efficient catalysts for oxidative desulfurization of dibenzothiophene. Chem Eng J. 2014;255:686–94.
Collins FM, Lucy AR, Sharp C. Oxidative desulphurisation of oils via hydrogen peroxide and heteropolyanion catalysis. J Mol Catal A: Chem. 1997;117:397–403.
Da Silva MJ, Dos Santos LF. Novel oxidative desulfurization of a model fuel with H2O2 catalyzed by AlPMo12O40 under phase transfer catalyst-free conditions. J Appl Chem. 2013;2013:1–8.
Dooley KM, Liu D, Madrid AM, Knopf FC. Oxidative desulfurization of diesel with oxygen: reaction pathways on supported metal and metal oxide catalysts. Appl Catal A: Gen. 2013;468:143–9.
Gao J, Wang S, Jiang Z, et al. Deep desulfurization from fuel oil via selective oxidation using an amphiphilic peroxotungsten catalyst assembled in emulsion droplets. J Mol Catal A: Chem. 2006;258:261–6.
Gao G, Cheng S, An Y, et al. Oxidative desulfurization of aromatic sulfur compounds over titanosilicates. ChemCatChem. 2010a;2:459–66.
Gao H, Guo C, Xing J, et al. Extraction and oxidative desulfurization of diesel fuel catalyzed by a brønsted acidic ionic liquid at room temperature. Green Chem. 2010b;12:1220–4.
García-Gutiérrez JL, Fuentes GA, Hernández-Terán ME, García P, Murrieta-Guevara F, Jiménez-Cruz F. Ultra-deep oxidative desulfurization of diesel fuel by the Mo/Al2O3–H2O2 system: the effect of system parameters on catalytic activity. Appl Catal A: Gen. 2008;334:366–73.
García-Gutiérrez JL, Laredo GC, García-Gutiérrez P, et al. Oxidative desulfurization of diesel using promising heterogeneous tungsten catalysts and hydrogen peroxide. Fuel. 2014;138:118–25.
Hassan SI, Sif El-Din OI, Tawfik SM, Abd El-Aty DM. Solvent extraction of oxidized diesel fuel: phase equilibrium. Fuel Process Technol. 2013;106:127–32.
Hui-Peng L, Jian S, Hua Z. Desulfurization using an oxidation/catalysis/adsorption scheme over H3PW12O40/SiO2–Al2O3. Pet Chem. 2007;47:452–6.
Ishihara A, Wang D, Dumeignil F, Amano H, Qian EW, Kabe T. Oxidative desulfurization and denitrogenation of a light gas oil using an oxidation/adsorption continuous flow process. Appl Catal A: Gen. 2005;279:279–87.
Katsoulis DE. A survey of applications of polyoxometalates. Chem Rev. 1998;98:359–88.
Khenkin AM, Neumann R. Desulfurization of hydrocarbons by electron transfer oxidative polymerization of heteroaromatic sulfides catalyzed by H5PV2Mo10O40 polyoxometalate. ChemSusChem. 2011;4:346–8.
Kozhevnikov IV. Catalysis by heteropoly acids and multicomponent polyoxometalates in liquid-phase reactions. Chem Rev. 1998;98:171–98.
Kukovecz Á, Balogi Z, Kónya Z, et al. Synthesis, characterisation and catalytic applications of sol–gel derived silica–phosphotungstic acid composites. Appl Catal A. Gen. 2002;228:83–94.
Lei J, Chen L, Yang P, Du X, Yan X. Oxidative desulfurization of diesel fuel by mesoporous phosphotungstic acid/SiO2: the effect of preparation methods on catalytic performance. J Porous Mater. 2013;20:1379–85.
Li B, Liu Z, Liu J, et al. Preparation, characterization and application in deep catalytic ODS of the mesoporous silica pillared clay incorporated with phosphotungstic acid. J Coll Interface Sci. 2011;362:450–6.
Li J, Hu B, Tan J, Zhuang J. Deep oxidative desulfurization of fuels catalyzed by molybdovanadophosphoric acid on amino-functionalized SBA-15 using hydrogen peroxide as oxidant. Transit Metal Chem. 2013;38:495–501.
Lo W-H, Yang H-Y, Wei G-T. One-pot desulfurization of light oils by chemical oxidation and solvent extraction with room temperature ionic liquids. Green Chem. 2003;5:639–42.
Maity P, Mukesh D, Bhaduri S, et al. A water soluble heteropolyoxotungstate as a selective, efficient and environment friendly oxidation catalyst. J Chem Sci. 2009;121:377–85.
Mei H, Mei BW, Yen TF. A new method for obtaining ultra-low sulfur diesel fuel via ultrasound assisted oxidative desulfurization. Fuel. 2003;82:405–14.
Qiu J, Wang G, Zeng D, et al. Oxidative desulfurization of diesel fuel using amphiphilic quaternary ammonium phosphomolybdate catalysts. Fuel Process Technol. 2009;90:1538–42.
Rafiee E, Eavani S. Organic–inorganic polyoxometalate based salts as thermoregulated phase-separable catalysts for selective oxidation of thioethers and thiophenes and deep desulfurization of model fuels. J Mol Catal A: Chem. 2013;380:18–27.
Rafiee E, Eavani S, Rashidzadeh S, et al. Silica supported 12-tungstophosphoric acid catalysts for synthesis of 1, 4-dihydropyridines under solvent-free conditions. Inorg Chim Acta. 2009;362:3555–62.
Rafiee E, Mahdavi H, Joshaghani M. Supported heteropoly acids offering strong option for efficient and cleaner processing for the synthesis of imidazole derivatives under solvent-free condition. Mol Divers. 2011;15:125–34.
Rafiee E, Mirnezami F. Keggin-structured polyoxometalate-based ionic liquid salts: thermoregulated catalysts for rapid oxidation of sulfur-based compounds using H2O2 and extractive oxidation desulfurization of sulfur-containing model oil. J Mol Liq. 2014;199:156–61.
Sampanthar JT, Xiao H, Dou J, et al. A novel oxidative desulfurization process to remove refractory sulfur compounds from diesel fuel. Appl Catal B Environ. 2006;63:85–93.
Shojaei AF, Rezvani MA, Loghmani MH. Comparative study on oxidation desulphurization of actual gas oil and model sulfur compounds with hydrogen peroxide promoted by formic acid: synthesis and characterization of vanadium containing polyoxometalate supported on anatase crushed nanoleaf. Fuel Process Technol. 2014;118:1–6.
Stanislaus A, Marafi A, Rana MS. Recent advances in the science and technology of ultra low sulfur diesel (ULSD) production. Catal Today. 2010;153:1–68.
Te M, Fairbridge C, Ring Z. Oxidation reactivities of dibenzothiophenes in polyoxometalate/H2O2 and formic acid/H2O2 systems. Appl Catal A: Gen. 2001;219:267–80.
Wenjia C, Yan Z, Renlie B, et al. Catalytic epoxidation of cyclohexene over mesoporous-silica immobilized Keggin-type tungstophosphoric acid. Chin J Chem. 2013;34:193–9.
Xiao J, Wu L, Wu Y, et al. Effect of gasoline composition on oxidative desulfurization using a phosphotungstic acid/activated carbon catalyst with hydrogen peroxide. Appl Energy. 2014;113:78–85.
Xue X, Zhao W, Ma B, et al. Efficient oxidation of sulfides catalyzed by a temperature-responsive phase transfer catalyst [(C18H37)2(CH3)2N]7PW11O39 with hydrogen peroxide. Catal Commun. 2012;29:73–6.
Yan X-M, Mei P, Lei J, et al. Synthesis and characterization of mesoporous phosphotungstic acid/TiO2 nanocomposite as a novel oxidative desulfurization catalyst. J Mol Catal A: Chem. 2009a;304:52–7.
Yan X-M. Su G-S, Xiong L. Oxidative desulfurization of diesel oil over Ag-modified mesoporous HPW/SiO2 catalyst. J Fuel Chem Technol. 2009b;37:318–23.
Yan X-M, Mei P, Xiong L, et al. Mesoporous titania–silica–polyoxometalate nanocomposite materials for catalytic oxidation desulfurization of fuel oil. Catal Sci Technol. 2013;3:1985–92.
Zhang B, Zhou M-D, Cokoja M, et al. Oxidation of sulfides to sulfoxides mediated by ionic liquids. RSC Adv. 2012a;2:8416–20.
Zhang H, Gao J, Meng H, et al. Removal of thiophenic sulfurs using an extractive oxidative desulfurization process with three new phosphotungstate catalysts. Ind Eng Chem Res. 2012b;51:6658–65.
Zhang J, Wang A, Li X, et al. Oxidative desulfurization of dibenzothiophene and diesel over [Bmim]3PMo12O40. J Catal. 2011a;279:269–75.
Zhang M, Zhu W, Xun S, Li H, Gu Q, Zhao Z, Wang Q. Deep oxidative desulfurization of dibenzothiophene with POM-based hybrid materials in ionic liquids. Chem Eng J. 2013;220:328–36.
Zhang Z, Zhang F, Zhu Q, et al. Magnetically separable polyoxometalate catalyst for the oxidation of dibenzothiophene with H2O2. J Coll Interface Sci. 2011b;360:189–94.
Acknowledgments
The authors thank the Razi University Research Council for support of this work.
Author information
Authors and Affiliations
Corresponding author
Additional information
Edited by Xiu-Qin Zhu
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Rafiee, E., Sahraei, S. & Moradi, G.R. Extractive oxidative desulfurization of model oil/crude oil using KSF montmorillonite-supported 12-tungstophosphoric acid. Pet. Sci. 13, 760–769 (2016). https://doi.org/10.1007/s12182-016-0127-0
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12182-016-0127-0