Abstract
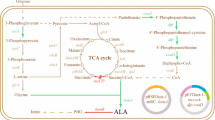
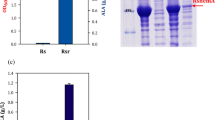
5-Aminolevulinic acid (ALA) is an important metabolic intermediate compound with high value and has recently been used in agriculture and medicine. In this study, we have constructed six recombinant Escherichia coli (E. coli) strains that are involved in pET system under the regulation of the T7 promoter and LacI to express codon-optimized hemA gene from Rhodobacter capsulatus (RchemA) for ALA production via the C4 pathway. Due to codon optimization, hemA has a high transcriptional level; however, most RcHemA proteins were formed as inclusion body. To improve expression in soluble form, the vector with TrxA fusion tag was successfully used and co-expressed with partner GroELS as chaperone in another vector. As a result, ALA production increased significantly from 1.21 to 3.67 g/L. In addition, optimal ALA production was developed through adjustment of induction time and isopropyl β-D-1-thiogalactopyranoside (IPTG) concentration, as well as substrate addition conditions. By adopting a two-stage induction strategy, the highest ALA reached 5.66 g/L when 0.1 mM of IPTG was added at early exponential phase (i.e., OD600 was equal to 0.7 to 0.8), while 6 g/L of glycine, 2 g/L of succinate, and 0.03 mM of pyridoxal 5′-phosphate (PLP) were provided in the mid-exponential phase in fermentation.






Similar content being viewed by others
References
Sasaki, K., Watanabe, M., & Tanaka, T. (2002). Biosynthesis, biotechnological production and applications of 5-aminolevulinic acid. Applied Microbiology and Biotechnology, 58(1), 23–29.
Liu, S., Zhang, G., Li, X., & Zhang, J. (2014). Applied Microbiology and Biotechnology, 98(17), 7349–7357.
Uehlinger, P., Zellweger, M., Wagnières, G., Juillerat-Jeanneret, L., et al. (2000). Journal of Photochemistry and Photobiology, B: Biology, 54(1), 72–80.
Cornelius, J. F., Slotty, P. J., El Khatib, M., Giannakis, A., Senger, B., & Steiger, H. J. (2014). Photodiagnosis and Photodynamic Therapy, 11(1), 1–6.
Noh, M. H., Lim, H. G., Park, S., Seo, S. W., & Jung, G. Y. (2017). Metabolic Engineering, 43, 1–8.
Li, T., Guo, Y. Y., Qiao, G. Q., & Chen, G. Q. (2016). Microbial synthesis of 5-aminolevulinic acid and its coproduction with polyhydroxybutyrate. ACS Synthetic Biology, 5(11), 1264–1274.
Shemin, D., & Russell, C. S. (1953). Journal of the American Chemical Society, 75(19), 4873–4874.
Hunter, G. A., & Ferreira, G. C. (1999). Biochemistry, 38(12), 3711–3718.
Ferreira, G. C., Neame, P. J., & Dailey, H. A. (1993). Heme biosynthesis in mammalian systems: evidence of a Schiff base linkage between the pyridoxal 5'-phosphate cofactor and a lysine residue in 5-aminolevulinate synthase. Protein Science, 2(11), 1959–1965.
Shoolingin-Jordan, P. M., Al-Daihan, S., Alexeev, D., et al. (2003). Biochimica et Biophysica Acta (BBA)-Proteins and Proteomics, 1647(1–2), 361–366.
Kang, Z., Wang, Y., Gu, P., Wang, Q., et al. (2011). Metabolic Engineering, 13(5), 492–498 21.
Schauer, S., Chaturvedi, S., Randau, L., Moser, J., Kitabatake, M., Lorenz, S., Verkamp, E., Schubert, W. D., Nakayashiki, T., Murai, M., Wall, K., Thomann, H. U., Heinz, D. W., Inokuchi, H., Söll, D., & Jahn, D. (2002). Escherichia coli glutamyl-tRNA reductase. Trapping the thioester intermediate. Journal of Biological Chemistry, 277(50), 48657–48663.
Chung, S. Y., Seo, K. H., & Rhee, J. I. (2005). Process Biochemistry, 40(1), 385–394 26.
Liu, X. X., Wang, L., Wang, Y. J., & Cai, L. L. (2010). D-glucose enhanced 5-aminolevulinic acid production in recombinant Escherichia coli culture. Applied Biochemistry and Biotechnology, 160(3), 822–830.
Zhang, J., Kang, Z., Ding, W., Chen, J., & du, G. (2016). Integrated optimization of the in vivo heme biosynthesis pathway and the in vitro iron concentration for 5-aminolevulinate production. Applied Biochemistry and Biotechnology, 178(6), 1252–1262.
Yasukawa, T., Kanei-Ishii, C., Maekawa, T., Fujimoto, J., Yamamoto, T., & Ishii, S. (1995). Increase of solubility of foreign proteins in Escherichia coli by coproduction of the bacterial thioredoxin. Journal of Biological Chemistry, 270(43), 25328–25331.
Quan, S., & Bardwell, J. C. (2012). Chaperone discovery. Bioessays, 34(11), 973–981.
Lou, J. W., Zhu, L., Wu, M. B., Yang, L. R. Lin, J. P., &, Cen, P. L. (2014) Journal of Zhejiang University. Science. B 15(5), 491–499.
Fanica-Gaignier, M., & Clement-Metral, J. (1973). 5-AMINOLEVULINIC-acid synthetase of Rhodopseudomonas spheroides Y. Purification and some properties. European Journal of Biochemistry, 40(1), 13–18.
Yubisui, T., & Yoneyama, Y. (1972). Aminolevulinic acid synthetase of Rhodopseudomonas spheroides: purification and properties of the enzyme. Archives of Biochemistry and Biophysics, 150(1), 77–85.
Jordan, P. M., & Laghai-Newton, A. (1986). Methods in enzymology, vol. 123 (pp. 435–443). Academic Press.
Nakakuki, M., Yamauchi, K., Hayashi, N., & Kikuchi, G. (1980). Purification and some properties of delta-aminolevulinate synthase from the rat liver cytosol fraction and immunochemical identity of the cytosolic enzyme and the mitochondrial enzyme. Journal of Biological Chemistry, 255(4), 1738–1745.
Volland, C., & Felix, F. (1984). Isolation and properties of 5-aminolevulinate synthase from the yeast Saccharomyces cerevisiae. European Journal of Biochemistry, 142(3), 551–557.
Ferreira, G. C., & Gong, J. (1995). 5-Aminolevulinate synthase and the first step of heme biosynthesis. Journal of Bioenergetics and Biomembranes, 27(2), 151–159.
Stojanovski, B. M., & Ferreira, G. C. (2015). Murine erythroid 5-aminolevulinate synthase: adenosyl-binding site Lys221 modulates substrate binding and catalysis. FEBS Open Bio, 5, 824–831.
Zhu, C., Chen, J., Wang, Y., Wang, L., Guo, X., Chen, N., Zheng, P., Sun, J., & Ma, Y. (2019). Enhancing 5-aminolevulinic acid tolerance and production by engineering the antioxidant defense system of Escherichia coli. Biotechnology and Bioengineering, 116(8), 2018–2028.
Hayer-Hartl, M., Bracher, A., & Hartl, F. U. (2016). Trends in Biochemical Sciences, 41(1), 62–76.
Xu, Z., Horwich, A. L., & Sigler, P. B. (1997). Nature, 388(6644), 741.
Collet, J. F., & Messens, J. (2010). Antioxidants & Redox Signaling, 13(8), 1205–1216.
Hayer-Hartl, M., Bracher, A., & Hartl, F. U. (2016). The GroEL-GroES chaperonin machine: a nano-cage for protein folding. Trends in Biochemical Sciences, 41(1), 62–76.
Fu, W., Lin, J., & Cen, P. (2010). Applied Biochemistry and Biotechnology, 160(2), 456–466.
Su, T., Guo, Q., Zheng, Y., Liang, Q., et al. (2019). Frontiers in Microbiology. https://doi.org/10.3389/fmicb.2019.01731.
Funding
This work received financial support from the Ministry of Science and Technology (MOST 108-2221-E-006-004-MY3, MOST 108-2218-E-006-006, and MOST-108-2621-M-006-015) in Taiwan.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics Approval and Consent to Participate
All the authors have read and agreed the ethics for publishing the manuscript.
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
ESM1
(DOCX 508 kb)
Rights and permissions
About this article
Cite this article
Yu, TH., Yi, YC., Shih, IT. et al. Enhanced 5-Aminolevulinic Acid Production by Co-expression of Codon-Optimized hemA Gene with Chaperone in Genetic Engineered Escherichia coli. Appl Biochem Biotechnol 191, 299–312 (2020). https://doi.org/10.1007/s12010-019-03178-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-019-03178-9