Abstract
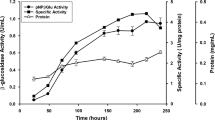
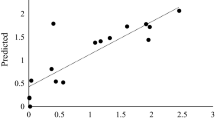
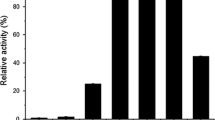
Penicillium funiculosum NCL1, a filamentous fungus, produced significantly higher levels of β-glucosidase. The effect of initial pH, incubation temperature, and different carbon sources on extracellular β-glucosidase production was studied in submerged fermentation. At 30 °C with initial pH 5.0, enzyme production was increased by 48-fold upon induction with paper mill waste, as compared to commercial cellulose powder. In zymogram analysis, four isoforms of β-glucosidases were observed with wheat bran whereas a minimum of one isoform was observed with other carbon sources. A major β-glucosidase (Bgl3A) with the apparent molecular weight of ~120 kDa, induced by paper mill waste, was purified 19-fold to homogeneity, with a specific activity of 1,796 U/mg. Bgl3A was a monomeric glycoprotein with 29% of neutral carbohydrate content. It showed optimum activity at pH 4.0 and 5.0, optimum temperature at 60 °C, and exhibited a half-life of 1 h at 60 °C. K m of Bgl3A was found to be 0.057 mM with p-nitrophenyl β-d-glucoside and V max was 1,920 U/mg. The purified enzyme exhibited glucose tolerance with a K i of 1.5 mM. Bgl3A readily hydrolyzed glucosides with β-linkage. Bgl3A activity was enhanced (156%) by Zn2+ and was not affected by other metal cations and reagents. The supplementation of Bgl3A (5 U/mg) with Trichoderma reesei cellulase complex (5 FPU/mg) resulted in about 70% of enhanced glucose production, which emphasizes the industrial importance of Bgl3A.






Similar content being viewed by others
References
Lynd, L. R., Weimer, P. J., Van Zyl, W. H., & Pretorius, I. S. (2002). Fundamentals and biotechnology. Microbiology and Molecular Biology Reviews, 66, 506–577.
Saha, B. C., Freer, S. N., & Bothast, R. J. (1994). Applied and Environmental Microbiology, 60, 3774–3780.
Ahmed, S., Riaz, S., & Jamil, A. (2009). Applied Microbiology and Biotechnology, 84, 19–35.
Gao, J., Weng, H., Zhu, D., Yuan, M., Guan, F., & Xi, Y. (2008). Bioresource Technology, 99, 7623–7629.
Lis, H., & Sharon, N. (1993). European Journal of Biochemistry, 218, 1–27.
Bhiri, F., Chaabouni, S. E., Limam, F., Ghrir, R., & Marzouki, N. (2008). Applied Biochemistry and Biotechnology, 149, 169–182.
Shi, Q. Q., Sun, J., Yu, H. L., Li, C. X., Bao, J., & Xu, J. H. (2011). Applied Biochemistry and Biotechnology, 164, 819–830.
Szijarto, N., Szengyel, Z., Liden, G., & Reczey, K. (2004). Applied Biochemistry and Biotechnology, 113–116, 115–124.
Kantham, L., & Jagannathan, V. (1985). Biotechnology and Bioengineering, 27, 786–791.
Mandels, M., & Reese, E. T. (1957). Journal of Bacteriology, 73, 269–278.
Mandels, M., & Weber, J. (1969). Advances in Chemsitry Series, 95, 394–404.
Singhania, R. R., Sukumaran, R. K., Rajasree, K. P., Mathew, A., Gottumukkala, L., & Pandey, A. (2011). Process Biochemistry, 46, 1521–1524.
Elyas, K. K., Mathew, A., Sukumaran, R. K., Ali, P. P., Sapna, K., Kumar, S. R., & Mol, K. R. (2010). New Biotechnology, 27, 347–351.
Menon, K., Rao, K. K., & Pushalkar, S. (1994). Indian Journal of Experimental Biology, 32, 706–709.
Ng, I. S., Li, C. W., Chan, S. P., Chir, J. L., Chen, P. T., Tong, C. G., Yu, S. M., & Ho, T. H. D. (2010). Bioresource Technology, 101, 1310–1317.
Prasetyo, J., Sumitha, S., Okuda, N., & Park, E. Y. (2010). Applied Biochemistry and Biotechnology, 162, 52–61.
Lee, K. M., Joo, A. R., Jeya, M., Lee, K. M., Moon, H. J., & Lee, J. K. (2011). Applied Biochemistry and Biotechnology, 163, 25–39.
Jeya, M., Joo, A. R., Lee, K. M., Tiwari, M. K., Lee, K. M., Kim, S. H., & Lee, L. K. (2009). Applied Microbiology and Biotechnology, 86, 1473–1484.
Kumar, R., Singh, S., & Singh, O. V. (2008). Indian Journal of Microbial Biotechnology, 35, 377–391.
Kang, L., Wang, W., & Lee, Y. Y. (2010). Applied Biochemistry and Biotechnology, 161, 53–66.
Rho, D., Desrochers, J., Jurasek, L., Driguez, H., & Defaye, J. (1982). Journal of Bacteriology, 149, 47–53.
Weil, J., Westgate, P., Kohlman, K., & Ladisch, M. R. (1994). Enzyme Microbiology and Technology, 16, 1002–1004.
Saibi, W., Amouri, B., & Gargouri, A. (2007). Applied Microbiology and Biotechnology, 77, 293–300.
Decker, C. H., Visser, J., & Schreier, P. (2001). Applied Microbiology and Biotechnology, 55, 157–163.
Takashima, S., Nakamura, A., Hidaka, M., Masaki, H., & Uozumi, T. (1999). Journal of Biochemistry, 125, 728–736.
Sun, X., Liu, Z., Qu, Y., & Li, X. (2008). Applied Biochemistry and Biotechnology, 146, 119–128.
Bhatia, Y., Mishra, S., & Bisaria, V. S. (2002). Critical Reviews in Biotechnology, 22, 375–407.
Hong, J., Tamaki, H., & Kumagai, H. (2006). Applied Microbiology and Biotechnology, 73, 80–88.
Tsukada, T., Igarashi, K., Yoshida, M., & Samejima, M. (2006). Applied Microbiology and Biotechnology, 73, 807–814.
Bhatia, Y., Mishra, S., & Bisaria, V. S. (2002). Applied Biochemistry and Biotechnology, 102–103, 367–369.
Wallecha, A., & Mishra, S. (2003). Biochimica et Biophysica Acta, 1649, 74–84.
Korotkova, O. G., Semenova, M. V., Morozova, V. V., Zorov, I. N., Sokolova, L. M., Bubnova, T. M., Okunev, O. N., & Sinitsyn, A. P. (2009). Biochemistry (Mosc), 74, 569–577.
Acknowledgments
The authors thank Sheik Asraf and Manjula for their valuable technical advice and discussion. We acknowledge the Council of Scientific and Industrial Research, India for financial support through a research grant (no. 5/258/51/2006-NMITLI). The Centre for Advanced Studies in Functional Genomics, The Centre for Excellence in Genomic Sciences, and the Networking Resource Centre in Biological Sciences are gratefully acknowledged for their support facilities.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ramani, G., Meera, B., Vanitha, C. et al. Production, Purification, and Characterization of a β-Glucosidase of Penicillium funiculosum NCL1. Appl Biochem Biotechnol 167, 959–972 (2012). https://doi.org/10.1007/s12010-012-9645-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-012-9645-4