Abstract
The enrichment with antioxidants (glutathione or ascorbate) or prooxidants (alloxan, methylviologen, hydrogen peroxide) of root inducing medium significantly decreased rhizogenesis frequency (alloxan, hydrogen peroxide) or inhibited roots regeneration (ascorbate, methylviologen) during the in vitro culture of Mesembryanthemum crystallinum L. hypocotyls. The adventitious roots morphology, root hairs length and density, was also influenced. Changes in the rhizogenesis course were related to the differences in hydrogen peroxide concentration during following days of culture between explants exhibiting morphogenic potential and those without the ability to form adventitious roots. In explants with morphogenic potential, rhizogenesis induction was always accompanied by a high level of hydrogen peroxide followed by the decrease in H2O2 content in following days. In contrast, in the explants without regeneration potential, the level of hydrogen peroxide was increasing during the culture period. Activity patterns of superoxide dismutase (SOD) and guaiacol peroxidase (POX) in the following days of culture were similar in the explants exhibiting regeneration potential cultured on different media. Total activity of SOD decreased during initial days of culture and then increased due to the activation of additional SOD isoform described as MnSODII. The activity of POX was low during the rhizogenesis induction, and then increased during following days of culture; the increase was correlated with the decrease in hydrogen peroxide content. In the explants without the ability to regenerate roots, the total activity of SOD was low throughout the whole culture period, whereas the POX activity was significantly higher than in hypocotyls with regeneration potential. It might be concluded that the increase in hydrogen peroxide during initial stages of rhizogenesis and the induction of MnSODII are prerequisites for adventitious roots formation from hypocotyls of M. crystallinum, independently in the presence of anti- or prooxidant in the culture medium.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Many factors influence induction and course of in vitro plants regeneration. They include the explants properties, the chemical composition of the medium as well as physical culture conditions. To improve the efficiency of regeneration, selected stress factors that cause overproduction of reactive oxygen species (ROS) have been applied with a positive effect (Papadakis et al. 2001; Tian et al. 2003; Mitrovic et al. 2012). Among ROS, hydrogen peroxide (H2O2) has been the most efficiently studied signaling molecule in plants (van Breusegem et al. 2008; Schmidt and Schippers 2015). It was found that organogenesis in vitro is accompanied by changes in the endogenous H2O2 content and this corresponds to the coordinated action of cellular antioxidant machinery (Millar et al. 1998; Gaspar et al. 2002; Balen et al. 2009; Bienert et al. 2007). Superoxide dismutases (SODs; EC 1.15.1.1)—the H2O2-generating enzymes and catalase (CAT; EC 1.11.1.6.) or peroxidases (POXs; EC 1.11.1.7)—the H2O2-scavenging enzymes as well as low molecular weight antioxidants like reduced ascorbate (AA) and reduced glutathione (GSH) have been noted as important regulatory factors in morphogenesis (Racchi et al. 2001; Li et al. 2009a, b; Tyburski et al. 2006, 2009; Tyburski and Tretyn 2010a, b; Konieczny et al. 2014; Libik-Konieczny et al. 2015).
Within the last decade, the knowledge concerning the mechanisms by which ROS affect plant regeneration processes were significantly improved. ROS were proved to be engaged in the stimulated by auxin cell cycle entry. It was described that ROS affect the activity of cell cycle system in in vitro cultured mesophyll protoplasts from alfalfa leaves (Feher et al. 2008). ROS and antioxidants were described as important factors involved in the cell cycle regulation within the meristems and especially in the formation of quiescence center (QC) within root apical meristem. A complex mechanism for the regulation of QC differentiation relies in general on of the induction of ascorbate oxidase activity stimulated by auxin, which oxidizes AA to dehydroascorbic acid (DHA). The oxidized environment is necessary to slow down the cell cycle in future cells of QC. In addition to DHA, glutathione in its oxidized form as glutathione disulfide (GSSG) is predominantly present in QC cells (Jiang et al. 2003).
Transition from cell proliferation phase to elongation phase is also in part regulated by redox cell status. It was found that class III heme peroxidases (POXs) in roots of Arabidopsis (Tsukagoshi et al. 2010) control the balance of ROS between the cell proliferation and the cell elongation phase during induction of differentiation process. This regulatory pathway functions independently of phytohormone signaling. POXs are known to exhibit ability to produce H2O2 or to scavenge this molecule depending on the chemical properties of environment (Passardi et al. 2004). During differentiation phase, the increased level of H2O2 accompany the events consisting discontinuation of divisions and beginning of cells elongation.
The growth of newly formed organ requires loosening of the cell wall matrix to allow cells elongation. Recently it was shown that two homologous cell wall-located enzymes POX33 and POX34 are involved in growth-promoting local redox changes in Arabidopsis roots (Passardi et al. 2006). Moreover, it was shown that NADPH oxidases are engaged in ROS generation participating in process of the microtubule depolymerization and organization of the cytoskeleton during root hairs growth (Yao et al. 2011). Interestingly, the balance between H2O2 and O •−2 regulates root hairs differentiation and decreased expression of POX genes correlated with a lower level of H2O2, while increased expression of NADPH oxidases coincided with a higher level of O •−2 . The POX-dependent increase in the level of H2O2 is required for the differentiation of root hairs cells (Sundaravelpandian et al. 2013).
Mesembryanthemum crystallinum L. (ice plant) turned out to be a useful model for studying organogenesis and the role of oxidative stress in morphogenesis in vitro (Libik et al. 2005a, b; Libik-Konieczny et al. 2012; Konieczny et al. 2014). Different SODs and CAT activities, as well as the corresponding changes in the content of H2O2 in calluses exhibiting rhizogenic or embryogenic potential were suggested to be crucial in the induction of specific developmental pathway. Studies carried out on M. crystallinum hypocotyls with the use of tissue transfer-experiment (Christianson and Warnick 1983, 1985) pointed out that explants became competent to react on auxin via rhizogenesis induction on the 3rd day of culture when hydrogen peroxide content in cultured tissue gained a high concentration (Konieczny et al. 2014). At the ultrastructural level, the highest level of hydrogen peroxide was found in the apoplast of rhizogenesis competent cells (Libik-Konieczny et al. 2015). Present studies constitute the continuation of previous experiments concerning the engagement of ROS in the regulation of rhizogenesis from hypocotyls of M. crystallinum. We hypothesize that the shift of redox balance into the direction of ROS scavenging activity or ROS production activity might significantly influence the rhizogenesis course. To verify this hypothesis we compared the activity of some antioxidant enzymes, endogenous H2O2 content, and we visualized the distribution of H2O2 in the hypocotyl explants cultured on the media containing auxin alone or in combination with different antioxidants (reduced glutathione, reduced ascorbate) or prooxidants (methylviologen, alloxan, hydrogen peroxide).
Materials and methods
Plant material
Seeds of M. crystallinum L. were surface sterilized in 70% (v/v) ethanol for 2 min, followed by immersion in a commercial bleach solution (Domestos) diluted with water (1:2; v/v) for 10 min, and next in a more diluted bleach solution (1:10; v/v) for 5 min. Then, seeds were rinsed three times with sterile, distilled water and placed onto 9 cm Petri dishes (30–50 seeds per dish) containing 20 ml of solid MS (Murashige and Skoog 1962) basal medium consisting of MS salts and vitamins (Sigma, Germany), 30 g l−1 sucrose, 7 g l−1 agar (Difco Bacto, USA). Dishes with seeds were placed in a growth chamber for germination at 25/20 °C under a 16/8 h light/dark with light provided by cool fluorescent light, 150–200 mmol m−2 s−1. Hypocotyls (5–7 mm in length) were excised from 10-day-old seedlings and placed on the culture media.
Induction of rhizogenesis in in vitro culture conditions
Excised hypocotyls were put horizontally on root inducing medium (RIM) composed of solid MS basal medium supplemented with 1-naphthaleneacetic acid (NAA) 1 mg l−1 (Sigma, Germany), pH 5.7. To determine the engagement of modified ROS level in the process of rhizogenesis some antioxidants, such as reduced glutathione (5 µmol l−1 GSH) or reduced ascorbate (5 µmol l−1 AA), as well as some prooxidants such as alloxan (1,5 µmol l−1 ALX) or methylviologen (1,5 µmol l−1 MV) and hydrogen peroxide (5 µmol l−1 H2O2) were added into RIM. Hypocotyls cultured for 3, 7, and 14 days on RIM and RIM supplemented with above mentioned anti-or prooxidants, were stripped of the developed roots and used as the studied material. Hypocotyls excised from 10-day-old seedlings were treated as a control (0 days of culture). For each analysis, 30 hypocotyls were collected from 3 Petri dishes containing 10 hypocotyls each.
Macroscopic studies
Macroscopic studies were performed under stereomicroscope Nikon SMZ1000. Canon EOS 450D device camera was used to take photos. The images were stored and processed using the Precoptic co. Cool View software. All measurements were taken at 14th day of culture. The frequency of rhizogenesis was calculated according to the formula: hypocotyls producing roots/total number of explants × 100. Root thickness of the five longest adventitious roots produced by three independent explants was measured at a distance of 3 mm from the root tip. Root hairs length of ten fully-elongated root hairs and root hairs density of the five longest adventitious roots produced from three hypocotyls were measured at a distance of 3 mm from the root tip. Root hairs density was expressed as the number of hairs in the 1-mm segment.
Histochemical localization of hydrogen peroxide using DAB staining
The method used in the experiment was developed by Thordal-Christensen et al. 1997 and it is based on the formation of brown polymerization product upon reaction of DAB with hydrogen peroxide in the presence of peroxidases.Hypocotyls were incubated in a solution of 2 mg ml−1 3,3′-diaminobenzidine (DAB) prepared in water, pH 5.5. Incubation was carried out for 1 h in the dark at room temperature. Then, the samples were rinsed with distilled water and submerged in a glycerin–water (1:1; v/v) solution and mounted on slides for light microscopy (Nikon Eclipse E200) observations. Photos were taken using a Canon EOS 450D device camera and images were stored and processed with the use of Precoptic co. Cool View software.
Biochemical studies
Protein isolation
To extract soluble proteins, plant material (1 g fresh weight) was homogenized at 4 °C with a mortar in 2.5 ml homogenization buffer (17.9 g l−1 Tricine, 0.74 g l−1 MgSO4, 0.155 g l−1 DTT, 1.14 g l−1 EGTA, adjusted with 1 mol l−1 Tris to pH 8.0). The insoluble material was removed by centrifugation for 3 min at 3000g. The Bradford protein assay kit (Bio-Rad) with BSA as standard was used to assess the protein concentration in the protein extracts (Bradford 1976) which were stored at −80 °C until further use.
Measurement of H2O2 content
Endogenous concentration of H2O2 was determined according to the modified method described previously by Brennan and Frenkel (1977). For H2O2 extraction 0.1 g of investigated material was homogenized in 0.25 ml cold acetone. Homogenate was centrifuged for 5 min at 12,000g. The pellet was discarded and a 0.2 ml of supernatant was collected. Then, a titanium reagent (20 μl 20% titanium tetrachloride in concentrated HCl, v/v, Sigma-Aldrich, Poland) was added to the 0.2 ml extract, followed by the addition of 0.025 ml NH3 (25%; v/v). In the next step, the peroxide–titanium complex was separated by 5 min centrifugation at 10,000g, the supernatant was discarded and the precipitate was repeatedly washed in 0.2 ml acetone and centrifuged again for 5 min at 10,000g. The precipitate was solubilized in 0.2 ml 1 N H2SO4 and brought to a final volume of 0.2 ml. The absorbance of the obtained solution was assessed at 415 nm against a water blank. The concentration of hydrogen peroxide was determined by comparing the absorbance against a standard curve of titanium–H2O2 complex over a range from 0 to 15 μmol ml−1. H2O2 concentration was calculated for 1 g of tissue fresh weight.
Measurement of POX activity
Guaiacol peroxidase (POX) activity was determined according to the method described by Pütter (1974). Frozen tissue (0.1 g) was homogenized in 1 ml 300 mmol l−1 potassium phosphate extraction buffer, pH 7.0, containing 1 mmol l−1 EDTA. Homogenate was centrifuged for 3 min at 10,000g in 4 °C and the supernatant was used for spectrophotometric analyses. The reaction was run for 5 min, at room temperature in a 1-ml cuvette filled with 50 µl of supernatant in 300 mM potassium phosphate buffer, pH 6.1, in the presence of 8.42 mmol l−1 guaiacol and 2.10 mmol l−1 H2O2. Conversion of guaiacol to tetraguaiacol was monitored at 470 nm, and POX activity was calculated using the absorbance coefficient 26,600 l M−1 cm−1.
Visualization of SOD activity
Separation of protein fractions was performed by native PAGE at 4 °C and 180 V in the buffer system (Laemmli 1970) without sodium dodecyl sulfate (SDS). Bands corresponding with SOD activity were visualized on 12% polyacrylamide gels using the activity-staining procedure according to Beauchamp and Fridovich (1971). Gels with separated proteins were incubated in staining solution composed of 50 mmol l−1 potassium phosphate buffer, pH 7.8, with the addition of 0.372 g l−1 EDTA, 31% (w/v) Temed, 7.5 mg l−1 riboflavin and 0.2 g l−1 NBT. Gels were incubated for 30 min in the dark at room temperature and then exposed to white light until SOD activity bands became visible.
Densitometric analysis
Densitometric analysis of SOD activity was performed on three gels from three independent experiments. ImageJ program was used for the gel images analyses. The activities of all isoforms were expressed in arbitrary units corresponding to the area under the densitometric curve.
Statistical analysis
The experiments were repeated three times. For each experiment, the means of replicates were calculated. Statistically significant differences between means (p ≤ 0.05) were determined by a two-way ANOVA followed by Tukey’s multiple range test using STATISTICA for Windows ver. 8.0 (StatSoft, Inc., Tulsa, OK, USA).
Results
The addition of antioxidants or prooxidants into the culture medium affected the frequency of rhizogenesis as well as root morphology (Table 1; Figs. 1, 2). Hypocotyls grown on RIM produced roots with the frequency of 90%. The addition of ALX or H2O2 to RIM reduced the frequency of rhizogenesis, whilst MV and AA totally inhibited root formation. In contrast, GSH had no effect on the number of rhizogenic hypocotyls which was comparable to those found on RIM without addition (Table 1).
Differences in the morphology between roots produced on the control medium (RIM) and roots produced on the medium containing reduced glutathione (RIM + GSH) was based on the production of slimmer roots (on average 0.2 mm) characterized with less abundant root hairs (Table 1; Fig. 1). The length of hairy roots produced on RIM + GSH was similar to the length of hairy roots produced on the control medium (Table 1; Fig. 1). The addition of ALX or H2O2 led to the changes in adventitious roots morphology. Explants cultured on RIM + ALX produced slimmer adventitious roots (on average 0.3 mm) with shorter root hairs (on average 0.2 mm length), while adventitious roots from RIM + H2O2 grew thicker (on average 0.8 mm) and possessed much longer root hairs (on average 1.3 mm length) than explants cultured on the control medium (RIM).
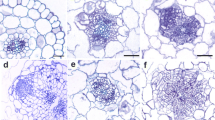
DAB staining of freshly cut explants (0 day) revealed that H2O2 was localized mainly in vascular cylinder along the entire explants, but single dark spots of DAB precipitates were also observed throughout the cortex (Fig. 2a, arrowheads). A similar pattern of H2O2 localization, mainly in the vascular cylinder, was confirmed in the hypocotyls cultured for 3 days on different media, however, no single dark spots were visualized in the cortex of the analyzed explants. In addition, products of DAB polimerization were accumulated at the cut ends of explants with the exception of samples cultured on RIM + AA and RIM + MV (Fig. 2b, arrowheads). After 7 days of explantation, cone-shaped root initials were produced on RIM, RIM + GSH, RIM + ALX and RIM + H2O2 (Fig. 2c, arrowheads) and this was accompanied by the increase in H2O2 concentration in the explants exhibited rhizogenesis (Fig. 3a). In the hypocotyls cultured on RIM + MV, the root initials were not confirmed. Instead, irregular swellings apparently developed from cells of the vascular cylinder at the cut ends of explants were produced. These swellings stained intensively with DAB indicating high H2O2 accumulation but never formed roots (Fig. 2c, d, arrowheads). No differences in H2O2 distribution were noted in the explants cultured on RIM + AA in comparison to previous days but the intensity of DAB staining was stronger than in previous days indicating higher H2O2 accumulation (Fig. 2b, c, arrowheads). After 14 days of culture, fully developed roots were observed on RIM, RIM + GSH, RIM + ALX, and RIM + H2O2 (Fig. 2 c). As revealed by DAB staining, H2O2 was the most abundant at the tip of newly formed roots as well as in their vascular cylinder (Fig. 2c, arrowheads). No differences in H2O2 localization were confirmed between the explants exposed to AA and MV for 7 and 14 days (Fig. 2c, d). Analysis of the endogenous content of hydrogen peroxide revealed that culture of hypocotyls for 3 days showed a significant increase in the concentration of this molecule in most measured samples when compared to the level of H202 in the hypocotyls from 0 day. The exceptions were samples from RIM + AA and RIM + MV media where the concentration of H2O2 was not changed in comparison to the hypocotyls from 0 day (Fig. 3a). After 7 days of culture, the endogenous level of H2O2 decreased with the exception of explants exposed to AA and MV which showed an increased level of H2O2 (Fig. 3b). Further culture did not show changes in endogenous H2O2 content in the studied samples. Hydrogen peroxide level remained lower in the explants exhibiting adventitious roots formation (RIM, RIM + GSH, RIM + ALX, RIM + H2O2) than in non-rhizogenic ones (RIM + AA, RIM + MV) (Fig. 3c).
Endogenous level of H2O2 in hypocotyls cultured on RIM and on RIM with the addition of anti- or prooxidants: a 3 day of culture, b 7 days of culture, c 14 days of culture. Values on the graph represent mean of three replicates and the error bars show standard error (SE). Values sharing the same letter are not significantly different (p ≤ 0.05) according to Tukey’s multiple test
At the 3rd day of culture, the activity of POX was relatively low on the most media used when compared to the one found in the freshly cut explants (0 day) (Fig. 4a). The exception was noted for the hypocotyls cultured on RIM + AA and RIM + MV where the level of POX activity was about 4-times higher (Fig. 4a) and comparable to the level observed in the samples from freshly cut explants (0 day). POX activity increased significantly by day 7 in the hypocotyls cultured on RIM, RIM + GSH, RIM + ALX and RIM + H2O2, but it remained unchanged in the explants exposed to AA and MV (Fig. 4b). The further culture had no effect on POX activity which was found at the level similar to the one determined at day 7 (Fig. 4c).
Spectrophotometric analysis of POX activity in hypocotyls cultured on RIM and on RIM with the addition of anti- or prooxidants: a 3 day of culture, b 7 days of culture, c 14 days of culture. Values on the graph represent mean of three replicates and the error bars show standard error (SE). Values sharing the same letter are not significantly different (p ≤ 0.05) according to Tukey’s multiple test
Three isoforms of superoxide dismutase (SOD): MnSODI, FeSOD, and Cu/ZnSOD were visualized on polyacrylamide gels after activity-staining of electrophoretically separated proteins isolated from explants cut off from hypocotyls (0 day) as well as from hypocotyls cultured for 3 days on RIM (Fig. 5a). The presence of antioxidants or prooxidants in the culture medium caused a significant decrease in the activity of all SOD isoforms leading to the decrease in the total SOD activity. After 7 days of culture, total activity of SOD increased in the hypocotyls cultured on RIM, RIM + GSH, RIM + ALX and RIM + H2O2. It was due to the activation of additional isoform visualized as MnSODII, as well as the intensification of other isoforms activity in case of RIM, RIM + GSH, RIM + ALX and RIM + H2O2. A different pattern of SOD isoforms activity was found in the samples cultured on RIM + AA and RIM + MV where a decrease in FeSOD and Cu/ZnSOD was visualized and the total enzyme activity was even lower than after 3 days of culture (Fig. 5b). The pattern of total SOD activity in the samples cultured for 14 days on different media was similar as it was described for hypocotyls cultured for 7 days. No changes in the activity of particular isoforms were noted in relation to the previously analyzed day of culture (Fig. 5c).
Visualization of SOD activity on polyacrylamide gels after the activity-staining of electrophoretically separated proteins from hypocotyls cultured on RIM and on RIM with the addition of anti- or prooxidants: a 3 day of culture, b 7 days of culture, c 14 days of culture. Lanes separated with gaps were combined from representative gels. Equal amount of protein (15 µg) were loaded on each lane. Numbers below the gel indicate total SOD activity calculated on the basis of the densitometric analysis and expressed in arbitrary units (area under the densitometric curve). Values in rows were statistically compared, and treatments sharing the same letter are not statistically different (p ≤ 0.05) according to Tukey’s multiple test
Discussion
GSH and AA affect rhizogenesis in opposite manner
According to the data presented here, the addition of antioxidants (GSH or AA) or prooxidants (ALX, MV, H2O2) to the medium containing auxin strongly affected the course of rhizogenesis in hypocotyls of M. crystallinum. Previously it was shown that non-enzymatic antioxidants such as GSH and AA are engaged in a sophisticated interactions between auxin and various components of cellular redox system during root formation (Tyburski and Tretyn 2010a; Li et al. 2009a) reported that exposure of mung bean seedlings to AA decreased or blocked root differentiation depending on the concentration used. In our study, the addition of AA to auxin-containing medium resulted in total inhibition of rhizogenesis (Table 1; Fig. 2). Observations under stereomicroscope revealed that vascular cylinder of ice plant hypocotyl cultured on medium with AA was thin with no visible signs of root initials. In contrast, cone-shaped root apices were regularly observed in the explants cultured in the presence of GSH (Fig. 2). Both GSH and AA are considered as strong antioxidants (Noctor and Foyer 1998). The results obtained in our study showed, however, a conspicuous increase in the endogenous H2O2 content in explants maintained on media with AA for 7 and 14 days (Fig. 3). Conversely, when hypocotyls were exposed to GSH, hydrogen peroxide content was significantly reduced after 7 and 14 days of culture. Thus, ascorbate in applied concentration acts rather as a prooxidant than an antioxidant. AA has been shown to exert either antioxidant and prooxidant effect depending upon the dose; it is suggested that at higher concentrations the prooxidant effect predominates (Seo and Lee 2002). Roots formed on RIM + GSH were slimmer and characterized by root hairs growing with lower density than those developed on RIM medium containing auxin alone (Fig. 1; Table 1). Previously Tyburski and Tretyn (2010b) have indicated that GSH enhances roots formation. They have stated, however, that only an optimal concentration of GSH in meristematic parts of growing roots is effective in stimulation of root growth. Root hairs growth is also dependent on the amount of GSH and the maintenance of its redox state. Sánchez-Fernández et al. 1997 have found a link between the GSH level and root hair tip growth in Arabidopsis roots. Optimal ROS level is important for sustaining the polar growth of root hair cells since it can activate the opening of calcium channels and regulate cell wall properties allowing cell growth and expansion (Foreman et al. 2003; Perilli et al. 2012; Monshausen et al. 2007). Since GSH is involved in ROS detoxification (Noctor and Foyer 1998) it can be considered as an important factor regulating an optimal ROS level in growing root hairs. It has to be noted that auxin treatment might increase endogenous GSH concentration to the supraoptimal level (Tyburski and Tretyn 2010b) leading to the deceleration of root hairs growth.
Prooxidants strongly affect adventitious roots formation and root hair morphology
The signaling role of ROS in root formation was explored in numerous studies (Foreman et al. 2003; Rentel et al. 2004; Pasternak et al. 2005; Su et al. 2006; Dunand et al. 2007; Li et al. 2007; Konieczny et al. 2014; Takáč et al. 2016). In our experimental system, exogenous application of H2O2, as well as other prooxidants such as ALX and MV, either decreased the frequency of root formation (50% ALX and 70% H2O2 when compared to RIM) or completely inhibited rhizogenesis (MV application) (Table 1; Fig. 2). Alloxan is a toxic glucose analog that can generate ROS in cyclic redox reactions in the presence of intracellular thiols like, for example, glutathione (Liptáková et al. 2012). Thus, oxidative stress induced by alloxan is also linked to the decrease in GSH level in the cells. This kind of redox balance disturbance led to the production of slimmer adventitious roots exhibiting shorter root hairs than those found during hypocotyls culture on RIM (Table 1; Fig. 1). Methylviologen is known to overproduce ROS in the light due to the disturbances in the photosynthetic electron transport chain (Mano et al. 2001), as well as in the dark, but by an unknown mechanism (Oracz et al. 2007). Reduced methylviologen interacts with molecular oxygen resulting in superoxide production which oxidizes the plastids. Previously it was stated that treatment with minimal concentrations of methylviologen leads to the broad oxidation of the whole cell due to the cross talk among subcellular compartments (Stonebloom et al. 2012). Moreover, plastid redox state has been found as a central part in the intracellular signaling pathway coordinating stress responses (Foyer and Noctor 2005). In the case of our studies, treatment of hypocotyls with methylviologen led to the induction of different than organized growth stress-induced morphogenic response (Fig. 2d). The addition of H2O2 into the culture medium caused the decrease in rhizogenic frequency but the adventitious root produced in this type of culture were thicker and characterized by much longer roots hairs growing with a higher density when compared with those cultured on RIM (Fig. 1; Table 1). This is in line with the previous reports showing that roots of Arabidopsis seedlings grown in the presence of an efficient scavenger of H2O2 were significantly longer and had no root hairs. Moreover, an inhibition of root elongation was observed in the conditions that promotes H2O2 formation in vivo and in vitro, by exogenous application of H2O2 and under the effect of umbelliferone application (Dunand et al. 2007). Taking into the consideration described above findings, we hypothesize that the differences in the length and the thickness between adventitious roots grown out from M. crystallinum hypocotyls cultured on the media containing different anti- or prooxidants might be caused by the disturbances in redox status of cell walls during growth and development of adventitious roots. ROS, as well as growth regulators are engaged in the processes of cell wall relaxation and cell wall stiffening necessary for cells growth (Wolf et al. 2016). ROS might influence a chemical structure of polysaccharides chains that build up cell walls. Lately, it was stated that differences in roots anatomy of mung bean, reflected in root diameter resulting from variations in the dimension of cortex, is affected by the different chemical structure of polysaccharides and combination with certain auxin (Kollárová et al. 2012). In the experimental system we used in our studies it was previously described that two ROS species as superoxide anion and hydrogen peroxide are differently localized in the hypocotyls during subsequent stages of rhizogenesis induced on the medium containing NAA. This might indicate a difference between the function of ROS types during developmental processes (Libik-Konieczny et al. 2015). Moreover, it was found out that also different elements of antioxidant system are engaged in the regulation of ROS level appropriate for rhizogenesis induction and adventitious roots development. The disruption of appropriate balance between ROS and antioxidants due to exogenous application of anti- or prooxidants can significantly change the processes of cell wall remodeling leading to the inhibition in roots elongation due to decrease in cells proliferation caused by cell wall stiffening or to enlargement of cortical cells due to cell wall loosening.
Fluctuations in H2O2 content do not correspond with a composition of rhizogenesis inducing media but rather with rhizogenesis per se
Measurement of the endogenous level of H2O2 in rhizogenic explants of ice plant revealed the increase of H2O2 content during early stages of rhizogenesis followed by a conspicuous decrease at days 7 and 14 of culture. These observations correspond with previous studies by Siminis et al. (1994) and Agrawal and Purohit (2012), who reported that early stages of regeneration were accompanied by a high content of endogenous H2O2. The results presented here stay in agreement with previous findings confirming the hypothesis that auxin together with the optimal oxidative status act together to set the asymmetry of mother cells, which is a necessary condition for generation of daughter cells and beginning of new developmental pathway (Livanos et al. 2016; Libik-Konieczny et al. 2014; Nick 2016). Previously we reported that addition of AA to the medium supplemented with an auxin analog 2,4-dichlorophenoxyacetic acid (2,4-d) resulted in irregular cell division within hypocotyl stele, which gave rise to callus instead of roots (Konieczny et al. 2014). In our study, roots became visible macroscopically around 14th day of culture whilst the cone-shaped root apices could be detected inside the hypocotyls as early as after 7 days of explantation. Interestingly, in explants maintained on medium with NAA and AA or MV the vascular cylinder of hypocotyl was knobby and root primordia have never been confirmed by microscopic observations within the cultured hypocotyls. These observations together with previous studies (Konieczny et al. 2014) suggests the crosstalk between auxins (2,4-d or NAA) and H2O2 during early stages of root initiation. The question whether the one of the possible role of H2O2 during very early stages of root initiation is to direct the divisions of competent cells in a specific plane requires further studies. It has to be also considered that during adventitious root formation an adequate balance between H2O2 level and auxin concentration is necessary to achieve a positive effect of ROS on rhizogenesis. Takác et al. (2016) have found that maintaining the proper intracellular pool of active (non-oxidized) auxin level is a crucial factor positively affecting adventitious root formation in flax.
Whilst transient increase of H2O2 seems to be necessary for initiation of in vitro morphogenesis (Siminis et al. 1994; Kairong et al. 1999; Agrawal and Purohit 2012; data presented here), the long-lasting overproduction of H2O2 was shown to cause severe oxidative stress which inhibits root formation (Li et al. 2009a; Pal Singh et al. 2009). In our study, the non-rhizogenic hypocotyls were characterized by low H2O2 content at day 3 of culture and relatively high steady-state level at day 7 and 14.
Specific pattern of SOD and POX activities is related with the course of rhizogenesis
Similarities in the fluctuations of hydrogen peroxide content during the explants culture on the media inducing rhizogenesis indicated the importance of redox balance system. It most probably mediated in maintaining the imbalance between anti- and pro-oxidants and avoiding negative influence of possible oxidative stress generation. In our experimental system, measurements of enzymatic antioxidants activity such as superoxide dismutase (SOD) isoforms and peroxidases (POX), revealed that these enzymes could be important in the regulation of hydrogen peroxide content during root primordia formation, as well as roots growth and development. From all described isoforms of SOD that have been previously identified in M. crystallinum by in-gel activity assay with selective inhibitors (Ślesak and Miszalski 2003), MnSOD II deserved special attention. Previously, the induction of this specific isoform of SOD, described as MnSOD-2 or MnSODII as well as MnSOD-like protein, was found in roots of both in vitro cultured plants (Ślesak and Miszalski 2003) and plants growing in vivo (Libik et al. 2005a, 2005b). Moreover, MnSODII was also earlier identified as a rhizogenesis marker in the in vitro culture of M. crystallinym hypocotyls on the medium containing 2,4-d (Konieczny et al. 2014). On the basis of the results presented here, we could state that rhizogenesis induced on the medium containing another auxin—NAA is also accompanied by the induction of MnSODII. This protein might be considered as a component of antioxidant system regulating ROS level in the hypocotyls exhibiting adventitious roots formation independently on the presence of antioxidants or prooxidants in the medium. It is commonly known that the success in in vitro plant regeneration via rhizogenesis depends on the maintenance and function of quiescent center (QC). It acts as a storage of cells for regeneration and secures the apex meristem functioning. The main biochemical property of the QC is its oxidized redox status, maintained by the low concentrations of the reduced forms of glutathione and ascorbic acid in the QC. It is suggested that highly oxidized state of QC is a consequence of polar auxin transport (Jiang et al. 2003). It has been lately proposed that the changes in mitochondrial function as an altered mitochondrial membrane potential and disturbances in tricarboxylic acid cycle, may determine the establishment and maintenance of the QC as well as can be a bond between auxin and oxidative stress (Jiang et al. 2006). Since MnSOD isoform is mainly localized in mitochondria of plant cells (Fridovich 1986), our results concerning an induction of MnSOD II activity might indicate indirectly the changes in mitochondrial metabolism accompanying rhizogenesis process, however, to give a precise information about MnSODII function, further studies have to be performed.
There are already many results of studies concerning the changes in peroxidase activity in correlation with phases of adventitious roots growth and development. According to these results, it can be stated that root formation took place after the explants have attained and went through a peak of maximum enzyme activity (Rout et al. 2000; Metaxas et al. 2004; Syros et al. 2004; Hatzilazarou et al. 2006; da Costa et al. 2013). In mung bean cuttings peroxidase activity has been described as a possible candidate involved in H2O2 level regulation during adventitious roots formation (Li et al. 2009b). In our studies, the significant increase in POX activity was noted after 7 days of hypocotyls culture on the media inducing rhizogenesis. In contrast, the level of POX activity in the hypocotyls without the ability to regenerate adventitious roots was high from the early days of culture. It is already known that POX activity regulates auxin catabolism and that the induction of rhizogenesis is characterized by a decrease in POX activity, while root development phase exhibits an increase in POD activity (Haissig and Davis 1994; Gaspar et al. 1994). Since high endogenous auxin concentration is necessary for the induction phase of rhizogenesis, whereas decrease in auxin content is a crucial event during the roots formation and growth (de Klerk et al. 1999), it can be stated that POX activity pattern during the course of adventitious roots formation from hypocotyls of M. crystallinum cultured on the media containing different anti- or prooxidants reflected the auxin level necessary to induce and proceed rhizogenesis.
Redox-dependent modulation of plants growth and development resulting from redox sensing of the environment may be a basic component in the strategy that plants have developed to survive in a changing environment (Sánchez-Fernández et al. 1997). Root is not only the main organ for water and nutrient absorption, and for anchoring the plant in the soil but it is also a crucial plant organ that often first come across many abiotic and biotic stress factors. Artificial manipulation of redox balance by the addition of anti- or pro-oxidants into the media causes a strong response of cultured hypocotyls forcing them to adapt to new circumstances. This fact probably causes the necessity to regulate pro-antioxidative balance resulting in the increase of H2O2 level in plant tissue. This stress compound may induce many adaptive processes, among other, new roots formation, which allows surviving in the new circumstances due to roots extraordinary plasticity. Such sequence of events can occur also in physiological conditions when stress situations can be solved with the development of new roots capable of soil exploration under detrimental conditions with short and less dense root hairs production or benefit from the advantageous environment by the generation of numerous long root hairs.
Change history
26 April 2019
The original version of this article unfortunately contained a mistake. The Acknowledgement section of the article should read as:
References
Agrawal M, Purohit S (2012) Changes in antioxidant enzymes activity during in vitro morphogenesis of carnation and the effect of antioxidants on plant regeneration. World J Sci Tech 2:87–92
Balen B, Tkalec M, Pavokovič D, Pevalek-Kozlina B, Krsnik-Raso M (2009) Growth conditions in in vitro culture can induce oxidative stress in Mammillaria gracilis tissues. J Plant Growth Regul 28:36–45
Beauchamp CO, Fridovich I (1971) Superoxide dismutase: improved assays and an assay applicable to acrylamide gels. Anal Biochem 44:276–287
Bienert GP, Moller AL, Kristiansen KA, Schulz A, Moller IM, Schjoerring JK, Jahn TP (2007) Specific aquaporins facilitate the diffusion of hydrogen peroxide across membranes. J Biol Chem 282:1183–1192
Bradford MM (1976) A rapid and sensitive method for quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Brennan T, Frenkel C (1977) Involvement of hydrogen peroxide in the regulation of senescence in pear. Plant Physiol 59:411–416
Christianson ML, Warnick DA (1983) Competence and determination in the process of in vitro shoot organogenesis. Dev Biol 95:288–293
Christianson ML, Warnick DA (1985) Temporal requirement for phytohormone balance in the control of organogenesis in vitro. Dev Biol 12:494–497
da Costa CT, de Almeida MR, Ruedell CM, Schwambach J, Maraschin FS, Fett-Neto AG (2013) When stress and development go hand in hand: main hormonal controls of adventitious rooting in cuttings. Front Plant Sci 4:1–19
de Klerk GJ, Van der Krieken W, De Jong JC (1999) The formation of adventitious roots: new concepts, new possibilities. In Vitro Cell Dev Biol Plant 35:189–199
Dunand C, Crèvecoeur M, Penel C (2007) Distribution of superoxide and hydrogen peroxide in Arabidopsis root and their influence on root development: possible interaction with peroxidases. New Phytol 174:332–341
Fehér A, Ötvös K, Pasternak TP, Szandtner AP (2008) The involvement of reactive oxygen species (ROS) in the cell cycle activation (G0-to-G1 transition) of plant cells. Plant Signal Behav 3:823–826
Foreman J, Demidchik V, Bothwell JH, Mylona P, Miedema H, Torres MA, Linstead P, Costa S, Brownlee C, Jones JD, Davies JM, Dolan L (2003) Reactive oxygen species produced by NADPH oxidase regulate plant cell growth. Nature 422:442–446
Foyer CH, Noctor G (2005) Oxidant and antioxidant signalling in plants: a re-evaluation of the concept of oxidative stress in a physiological context. Plant Cell Environ 28:1056–1071
Fridovich I (1986) Superoxide dismutases. Adv Enzymol 41:35
Gaspar T, Kevers C, Hausman JF, Ripetti V (1994) Peroxidase activity and endogenous free auxin during adventitious root formation. Physiology, Growth and Development of Plants in Culture. In: Lumdsen PJ, Nicholas JR, Davies WJ (eds) Kluwer Academic Publishers, Dordrecht, pp 289–298
Gaspar T, Frank T, Bisbis B, Kevers C, Jouve L, Hausman JF, Dommes J (2002) Concepts in plant stress physiology. Application to plant tissue cultures. Plant Growth Regul 37:263–285
Haissig BE, Davis TD (1994) A historical evaluation of adventitious rooting research to 1993. Biology of root formation. In: Davis TD, Haissing BE (eds) Plenum Press, New York, pp 275–331
Hatzilazarou SP, Syros TD, Yupsanis TA, Bosabalidis AM, Economou AS (2006) Peroxidases, lignin and anatomy during in vitro and ex vitro rooting of gardenia (Gardenia jasminoides Ellis) microshoots. J Plant Physiol 163:827–836
Jiang K, Meng YL, Feldman LJ (2003) Quiescent center formation in maize roots is associated with an auxin-regulated oxidizing environment. Development 130:1429–1438
Jiang K, Ballinger T, Li D, Zhang S, Feldman L (2006) A role for mitochondria in the establishment and maintenance of the maize root quiescent center. Plant Physiol 140:1118–1125
Kairong C, Gengsheng X, Xinmin L, Gengmei X, Wang Gafu W (1999) Effect of hydrogen peroxide on somatic embryogenesis of Lycium barbarum L. Plant Sci 146:9–16
Kollárová K, Zelko I, Henselová M, Capek P, Lišková D (2012) Growth and anatomical parameters of adventitious roots formed on mung bean hypocotyls are correlated with galactoglucomannan oligosaccharides structure. Sci World J 7:1–7
Konieczny R, Banaś AK, Surówka E, Michalec Ż, Miszalski Z, Libik-Konieczny M (2014) Pattern of antioxidant enzyme activities and hydrogen peroxide content during developmental stages of rhizogenesis from hypocotyl explants of Mesembryanthemum crystallinum L. Plant Cell Rep 33:165–177
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Li S, Xue L, Xu S, Feng H, An L (2007) Hydrogen peroxide involvement in formation and development of adventitious roots in cucumber. Plant Growth Regul 52:173–180
Li S, Xue L, Xu S, Feng H, An L (2009a) Hydrogen peroxide acts as a signal molecule in the adventitious root formation of mung bean seedlings. Environ Exp Bot 65:63–71
Li S, Xue L, Xu S, Feng H, An L (2009b) IBA-induced changes in antioxidant enzymes during adventitious rooting in mung bean seedlings: the role of H2O2. Environ Exp Bot 66:442–445
Libik M, Konieczny R, Pater B, Ślesak I, Miszalski Z (2005a) Differences in the activities of some antioxidant enzymes and H2O2 content during rhizogenesis and somatic embryogenesis in callus cultures of the ice plant. Plant Cell Rep 23:834–841
Libik M, Konieczny R, Surówka E, Miszalski Z (2005b) Superoxide dismutase activity in organs of Mesembryanthemum crystallinum L. at different stages of CAM development. Acta Biol Crac Ser Bot 47:199–204
Libik-Konieczny M, Konieczny R, Surówka E, Ślesak I, Michalec Ż, Rozpądek P, Miszalski Z (2012) Pathways of ROS homeostasis regulation in Mesembryanthemum crystallinum L. calli exhibiting differences in rhizogenesis. Plant Cell Tissue Organ Cult 110:123–131
Libik-Konieczny M, Kozieradzka-Kiszkurno M, Desel Ch, Michalec-Warzecha Ż, Miszalski Z, Konieczny R (2015) The localization of NADPH oxidase and reactive oxygen species in in vitro-cultured Mesembryanthemum crystallinum L. hypocotyls discloses their differing roles in rhizogenesis. Protoplasma 252:477–487
Liptáková L, Bočová B, Huttová J, Igor Mistrίk I, Tamás L (2012) Superoxide production induced by short-term exposure of barley roots to cadmium, auxin, alloxan and sodium dodecyl sulfate. Plant Cell Rep 31:2189–2197
Livanos P, Galatis B, Apostolakos P (2016) Deliberate ROS production and auxin synergistically trigger the asymmetrical division generating the subsidiary cells in Zea mays stomatal complexes. Protoplasma 253:1081–1099
Mano J, Ohno C, Domae Y, Asada K (2001) Chloroplastic ascorbate peroxidase is the primary target of methylviologen-induced photooxidative stress in spinach leaves: its relevance to monodehydroascorbate radical detected with in vivo ESR. Biochem Biophys Acta 1504:275–287
Metaxas DJ, Syros TD, Yupsanis T, Economou AS (2004) Peroxidases during adventitious rooting in cuttings of Arbutus unedo and Taxus baccata as affected by plant genotype and growth regulator treatment. Plant Growth Regul 44:257–266
Millar TM, Stevens CR, Benjamin N, Eisenthal R, Harrison R, Blake DR (1998) Xanthine oxidoreductase catalyses the reduction of nitrates and nitrite to nitric oxide under hypoxic conditions. FEBS Lett 427:225–228
Mitrović A, Janošević D, Budimir S, Bogdanović PJ (2012) Changes in antioxidative enzymes activities during Tacitus bellus direct shoot organogenesis. Biol Plant 56:357–361
Monshausen GB, Bibikova TN, Messerli MA, Shi C, Gilroy S (2007) Oscillations in extracellular pH and reactive oxygen species modulate tip growth of Arabidopsis root hairs. Proc Natl Acad Sci USA 104:20996–21001
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio-assays with tobacco tissue cultures. Physiol Plant 15:473–497
Nick P (2016) Life breaks symmetry. Protoplasma 253:965–966
Noctor G, Foyer Ch (1998) Ascorbate and glutathione: keeping active oxygen under control. Annu Rev Plant Biol 49:249–279
Oracz K, Bouteau HE-L, Farrant JM, Cooper K, Belghazi M, Job C, Job D, Corbineau F, Bailly Ch (2007) ROS production and protein oxidation as a novel mechanism for seed dormancy alleviation. Plant J 50:452–465
Pal Singh H, Kaur S, Batish DR, Kohli RK (2009) Caffeic acid inhibits in vitro rooting in mung bean [Vigna radiata (L.) Wilczek] hypocotyls by inducing oxidative stress. Plant Growth Regul 57:21–30
Papadakis AK, Siminis C, Roubelakis-Angelakis KA (2001) Reduced activity of antioxidant machinery is correlated with suppression of totipotency in plant protoplasts. Plant Physiol 126:434–444
Passardi F, Penel C, Dunand C (2004) Performing the paradoxical: how plant peroxidases modify the cell wall. Trends Plant Sci 9:534–540
Passardi F, Tognolli M, De Meyer M, Penel C, Dunand C (2006) Two cell wall associated peroxidases from Arabidopsis influence root elongation. Planta 223:965–974
Pasternak T, Potters G, Caubergs R, Jansen MAK (2005) Complementary interactions between oxidative stress and auxins control plant growth responses at plant, organ, and cellular level. J Exp Bot 56:1991–2001
Perilli S, Mambro R, Sabatini S (2012) Growth and development of the root apical meristem. Curr Opin Plant Biol 15:17–23
Pütter J (1974) Peroxidase. In: Bergmeyer HU (ed) Methods of enzymatic analysis. Academic Press, NewYork, pp 685–690
Racchi ML, Bagnoli F, Balla I, Daut S (2001) Differential activity of catalase and superoxide dismutase in seedlings and in vitro micropropagated oak (Quercus robur L.). Plant Cell Rep 20:169–174
Rentel MC, Lecourieux D, Ouaked F, Usher SL, Petersen L, Okamoto H, Knight H, Peck SC, Grierson CS, Hirt H, Knight MR (2004) OXI1 kinase is necessary for oxidative burst-mediated signalling in Arabidopsis. Nature 427:858–861
Rout GR, Samantaray S, Das P (2000) In vitro rooting of Psoralea corylifolia Linn.: peroxidase activity as a marker. Plant Growth Regul 30:215–219
Sánchez-Fernández R, Fricker M, Corben LB, White NS, Sheard N, Leaver ChJ, van Montagnu M, Inzé D, May MJ (1997) Cell proliferation and hair tip growth in the Arabidopsis root are under mechanistically different forms of redox control. Proc Natl Acad Sci USA 94:2745–2750
Schmidt R, Schippers JHM (2015) ROS-mediated redox signaling during cell differentiation in plants. Biochem Biophys Acta 1850:1497–1508
Seo MY, Lee SM (2002) Protective effect of low dose of ascorbic acid on hepatobiliary function in hepatic ischemia/reperfusion in rats. J Hepatol 36:72–77
Siminis CJ, Kanellis AK, Roubelakis-Angelakis KA (1994) Catalase is differentially expressed in dividing and nondividing protoplasts. Plant Physiol 105:1375–1383
Ślesak I, Miszalski Z (2003) Superoxide dismutase-like protein from roots of the intermediate C3-CAM plant Mesembryanthemum crystallinum L. in in vitro culture. Plant Sci 164:497–505
Stonebloom S, Brunkard JO, Cheung AC, Jiang K, Feldman L, Zambryski P (2012) Redox states of plastids and mitochondria differentially regulate intercellular transport via plasmodesmata. Plant Physiol 158:190–199
Su GX, Zhang WH, Liu YL (2006) Involvement of hydrogen peroxide generated by polyamine oxidative degradation in the development of lateral roots in soybean. J Integr Plant Biol 48:426–432
Sundaravelpandian K, Chandrika N, Schmidt W (2013) PFT1, a transcriptional Mediator complex subunit, controls root hair differentiation through reactive oxygen species (ROS) distribution in Arabidopsis. New Phytol 197:151–161
Syros TD, Yupsanis T, Zafiriadis H, Economou AS (2004) Activity and isoforms of peroxidases, lignins and anatomy, during adventitious rooting in cuttings of Ebenus cretica L. J Plant Physiol 161:69–77
Takáč T, Obert B, Rolčik J, Šamaj J (2016) Improvement of adventitious root formation in flax using hydrogen peroxide. New Biotechnol 33:728–734
Thordal-Christensen H, Zhang Z, Wei Y, Collinge DB (1997) Subcellular localization of H2O2 in plants. H2O2 accumulation in papillae and hypersensitive response during the barley powdery mildew interaction. Plant J 11:1187–1194
Tian M, Gu Q, Zhu M (2003) The involvement of hydrogen peroxide and antioxidant enzymes in the process of shoot organogenesis of strawberry callus. Plant Sci 165:701–707
Tsukagoshi H, Busch W, Benfey PN (2010) Transcriptional regulation of ROS controls transition from proliferation to differentiation in the root. Cell 143:606–616
Tyburski J, Tretyn A (2010a) Ascorbate and glutathione in organogenesis, regeneration and differentiation in plant in vitro cultures. In: ©Anjum NA et al (eds) Ascorbate-glutathione pathway and stress tolerance in plants. Springer, Berlin. doi:10.1007/978-90-481-9404-9_2
Tyburski J, Tretyn A (2010b) Glutathione and glutathione disulfide affect adventitious root formation and growth in tomato seedling cuttings. Acta Physiol Plant 32:411–417
Tyburski J, Jasionowicz P, Tretyn A (2006) The effects of ascorbate on root regeneration in seedling cuttings of tomato. Plant Growth Regul 48:157–173
Tyburski J, Dunajska K, Tretyn A (2009) Reactive oxygen species localization in roots of Arabidopsis thaliana seedlings grown under phosphate deficiency. Plant Growth Regul 59:27–36
van Breusegem F, Bailey-Serres J, Mittler R (2008) Unraveling the tapestry of networks involving reactive oxygen species in plants. Plant Physiol 147:978–984
Wolf S, Hématy K, Höfte H (2016) Growth control and cell wall signaling in plants. Annu Rev Plant Biol 63:381–407
Yao LL, Zhou Q, Pei BL, Li YZ (2011) Hydrogen peroxide modulates the dynamic microtubule cytoskeleton during the defence responses to Verticillium dahliae toxins in Arabidopsis. Plant Cell Environ 34:1586–15898
Acknowledgements
This work was partially financed by projects funded by Polish Ministry of Science and Higher Education (no. 303356935) and National Science Centre (Nos. 2012/05/B/NZ9/01035 and 2013/09/N/NZ9/01650). We would like to thank Botanical Garden of the University of Vienna for providing seeds of Mesembryanthemum crystallinum.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by J Van Huylenbroeck.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Libik-Konieczny, M., Kozieradzka-Kiszkurno, M., Michalec-Warzecha, Ż. et al. Influence of anti- and prooxidants on rhizogenesis from hypocotyls of Mesembryanthemum crystallinum L. cultured in vitro. Acta Physiol Plant 39, 166 (2017). https://doi.org/10.1007/s11738-017-2466-x
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11738-017-2466-x