Abstract
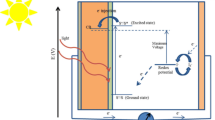
In this study, the newly designed organic dyes (CBS1-CBS3) were designed by the donor (D), π-linkers (π) and acceptor (A) to forming the D-π-A structure. Besides, the screened efficient spacer dye sensitizers were simulated by the density functional theory (DFT) and time-dependent DFT (TD-DFT) methods for dye-sensitized solar cell (DSSC) application. First, the optical absorption peak of CB1 dye was analyzed by the different exchange–correlation (XC) and long-range corrected (LC) functionals with 6-31G(d) basis set. As a result of functional, TD-CAM-B3LYP method nearly well matched with the literature data of CB1. The computational outcomes were shown that the highest occupied molecular orbitals (HOMOs) and lowest unoccupied MOs (LUMOs) of the CBS1-CBS3 dyes confirmed useful response on the electron injection (\(\Delta {G}_{\text{inject}}\)) and dye regeneration (\(\Delta {G}_{\text{reg}}\)). Therefore, the short-circuit current density (\({J}_{\text{SC}}\)) key factors of the light harvesting efficiency (LHE), \(\Delta {G}_{\text{inject}}\)and \(\Delta {G}_{\text{reg}}\) in CBS1-CBS3 dye derivatives were superior performance of the DSSCs. In addition to the highest vertical dipole moment (µnormal) and open circuit photovoltage (\(e{V}_{\text{OC}}\)) of the planned dyes were better performance for DSSCs. Hence, it benefits to higher efficiency. The present theoretical investigation results demonstrate that all the D-π-A dyes may be capable sensitizers for DSSC application.







Similar content being viewed by others
Availability of data and material
All the data and electronic materials are available for the Gaussian program.
Code availability
Chemdraw, Gaussian 09w, Gaussview, and Gausssum.
References
O’Regan B, Grätzel M (1991) A low-cost, high-efficiency solar cell based on dye-sensitized colloidal TiO2 films. Nature 353:737–740
Nazeeruddin MK, Kay A, Rodicio I, Humphry-Baker R, Müller E, Liska P, Vlachopoulos N, Grätzel M (1993) Conversion of light to electricity by cis-X2bis(2,20-bipyridyl4,40-dicarboxylate)ruthenium(II) charge transfer sensitizers (X = Cl−, Br−, I−, CN−, and SCN−) on nanocrystalline titanium dioxide electrodes. J Am Chem Soc 115:6382–6390
Nazeeruddin MK, Zakeeruddin SM, Humphry-Baker R, Jirousek M, Liska P, Vlachopoulos N, Shklover V, Fischer CH, Grätzel M (1999) Acid–base equilibria of (2,2’-bipyridyl-4,4′-dicarboxylic acid) ruthenium(II) complexes and the effect of protonation on charge transfer sensitization of nanocrystalline titania. Inorg Chem 38:6298–6305
Hagfeldt A, Grätzel M (2000) Molecular photovoltaics. Acc Chem Res 33:269–277
Nazeeruddin MK, Pechy P, Renouard T, Zakeeruddin SM, Humphry-Baker R, Comte P, Liska P, Cevey L, Costa E, Shklover V, Spiccia L (2001) Engineering of efficient panchromatic sensitizers for nanocrystalline TiO2-based solar cells. J Am Chem Soc 123:1613–1624
Nogueira AF, Longo C, De Paoli MA (2004) Polymers in dye sensitized solar cells: overview and perspectives. Coord Chem Res 248:1455–1468
Tsai MS, Hsu YC, Lin JT, Chen HC, Hsu CP (2007) Organic dyes containing 1H-phenanthro [9,10-d]imidazole conjugation for solar cells. J Phys Chem C 111:18785–18793
Chang YJ, Chow TJ (2009) Dye-sensitized solar cell utilizing organic dyads containing triarylene conjugates. Tetrahedron 65:4726–4734
Cai-Rong Z, Zi-Jiang L, Yu-Hong C, Hong-Shan C, You-Zhi W, Li-Hua Y (2009) DFT and TDDFT study on organic dye sensitizers D5, DST and DSS for solar cells. J Mol Struct Theochem 899:86–93
Hagberg DP, Edvinsson T, Marinado T, Boschloo G, Hagfeldt A, Sun L (2006) A novel organic chromophore for dye-sensitized nanostructured solar cells. Chem Commun 21:2245–2247
Mathew S, Yella A, Gao P, Humphry-Baker R, Curchod BF, Ashari-Astani N, Tavernelli I, Rothlisberger U, Nazeeruddin MK, Grätzel M (2014) Dye-sensitized solar cells with 13% efficiency achieved through the molecular engineering of porphyrin sensitizers. Nat Chem 6:242–247
Arunkumar A, Shanavas S, Acevedo R, Anbarasan PM (2020) Acceptor tuning effect on TPA-based organic efficient sensitizers for optoelectronic applications-quantum chemical investigation. Struct Chem 31:1029–1042
Xu H, Sun Q, An Z, Wei Y, Liu X (2015) Electroluminescence from europium(III) complexes. Coord Chem Rev 293:228–249
ou JH, Kumar S, Agrawal A, Li TH, Sahoo S, (2015) Approaches for fabricating high efficiency organic light emitting diodes. J Mater Chem C 3:2974–3002
Yang J, Huang J, Li QQ, Li Z (2016) Blue AIEgens: approaches to control the intramolecular conjugation and the optimized performance of OLED devices. J Mater Chem C 4:2663–2684
Zhang JL, Zhong S, Zhong JQ, Niu TC, Hu WP, Wee AT, Chen W (2015) rational design of two-dimensional molecular donor-acceptor nanostructure arrays. Nanoscale 7:4306–4324
Wu K, Zhang T, Zhan L, Zhong C, Gong S, Lu ZH, Yang C (2016) Tailoring optoelectronic properties of phenanthroline-based thermally activated delayed fluorescence. Adv Opt Mater 4:1558–1566
Turkoglu G, Cinar ME, Buyruk A, Tekin E, Mucur SP, Kaya K, Ozturk T (2016) Novel organoboron compounds derived from thieno[3,2-b]thiophene and triphenylamine units for OLED devices. J Mater Chem C 4:6045–6053
Vikramaditya T, Saisudhakar M, Sumithra K (2016) Computational study on thermally activated delayed fluorescence of donor-linker-acceptor network molecules. RSC Adv 6:37203–37211
Jiang J, Hu D, Hanif M, Li X, Su S, Xie Z, Liu L, Zhang S, Yang B, Ma Y (2016) Twist angle and rotation freedom effects on luminescent donor-acceptor materials: crystal structures, photophysical properties, and OLED application. Adv Opt Mater 4:2109–2118
Becke A (1993) Density-functional thermochemistry. III. The role of exact exchange. J Chem Phys 98:5648–5652
Fan W, Tan D, Deng WQ (2012) Acene-modified triphenylamine dyes for dye-sensitized solar cells: a computational study Chem Phys Chem 13:2051–2060
Haid S, Marszalek M, Mishra A, Wielopolski M, Teuscher J, Moser JE, Humphry-Baker R, Zakeeruddin SM, Grätzel M, Bäuerle P (2012) Significant improvement of dye-sensitized solar cell performance by small structural modification inp-conjugated donor-acceptor dyes. Adv Funct Mater 22:1291–1302
Hsu CP, Hirata S, Head-Gordon M (2000) Excitation energies from timedependent density functional theory for linear polyene oligomers:butadiene to decapentaene. J Phys Chem A 105:451–458
Meng S, Kaxiras E, Nazeeruddin MK, Grätzel M (2011) Design of dye acceptors for photovoltaics from first-principles calculations. J Phys Chem C 115:9276–9282
Pastore M, Mosconi E, De Angelis F (2012) Computational investigation of dye-iodine interactions in organic dye-sensitized solar cells. J Phys Chem C 116:5965–5973
Srinivas K, Kumar CR, Reddy MA, Bhanuprakash K, Rao VJ, Giribabu L (2011) D-π-A organic dyes with carbazole as donor for dye-sensitized solar cells. Synth Met 161:96–105
Zhang XH, Wang ZS, Cui Y, Koumura N, Furube A, Hara K (2009) Organic sensitizers based on hexylthiophene-functionalized indolo [3, 2-b] carbazole for efficient dye-sensitized solar cells. J Phys Chem C 113:13409–13415
Cai S, Tian G, Li X, Su J, Tian H (2013) Efficient and stable DSSC sensitizers based on substituted dihydroindolo [2, 3-b] carbazole donors with high molar extinction coefficients. J Mater Chem A 1:11295–11305
Klimeš J, Michaelides A (2012) Perspective: advances and challenges in treating van der Waals dispersion forces in density functional theory. J Chem Phys 137:120901–120912
Ruiz VG, Liu W, Zojer E, Scheffler M, Tkatchenko A (2012) Density-functional theory with screened van der Waals interactions for the modeling of hybrid inorganic-organic systems. Phys Rev Lett 108:46103–146105
Arunkumar A, Shanavas S, Anbarasan PM (2018) First-principles study of efficient phenothiazine-based D-π-A organic sensitizers with various spacers for DSSCs. J Comput Electron 17:1410–1420
Arunkumar A, Deepana M, Shanavas S, Acevedo R, Anbarasan PM (2019) Computational investigation on series of metal-free sensitizers in tetrahydroquinoline with different π-spacer groups for DSSCs. ChemistrySelect 4:4097–4104
Perdew JP, Wang Y (2018) Erratum: accurate and simple analytic representation of the electron-gas correlation energy [Phys. Rev. B 45, 13244 (1992)]. Phys Rev B 98:079904
Yanai T, Tew DP, Handy NC (2004) A new hybrid exchange-correlation functional using the Coulomb-attenuating method (CAM-B3LYP). Chem Phys Lett 393:51–7
Chai JD, Head-Gordon M (2008) Long-range corrected hybrid density functionals with damped atom-atom dispersion corrections. Phys Chem Chem Phys 10:6615–6620
Arunkumar A, Anbarasan PM (2019) Optoelectronic properties of a simple metal-free organic sensitizer with different spacer groups: quantum chemical assessments. J Electron Mater 48:1522–1530
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Mennucci B, Petersson GA, Nakatsuji H, Caricato M, Li X, Hratchian HP, Izmaylov AF, Bloino J, Zheng G, Sonnenberg JL, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Vreven T, Montgomery JA Jr, Peralta JE, Ogliaro F, Bearpark MJ, Heyd J, Brothers EN, Kudin KN, Staroverov VN, Kobayashi R, Normand J, Raghavachari K, Rendell AP, Burant JC, Iyengar SS, Tomasi J, Cossi M, Rega N, Millam NJ, Klene M, Knox JE, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RJ, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Martin RL, Morokuma K, Zakrzewski VG, Voth GA, Salvador P, Dannenberg JJ, Dapprich S, Daniels AD, Farkas Ö, Foresman JB, Ortiz JV, Cioslowski J, Fox DJ (2009) Gaussian 09. Gaussian Inc., Wallingford, CT, USA
Arunkumar A, Shanavas S, Acevedo R, Anbarasan PM (2020) Quantum chemical investigation of modified coumarin-based organic efficient sensitizers for optoelectronic applications. Eur Phys J D 74:1–8
Sang-aroon W, Laopha S, Chaiamornnugool P, Tontapha S, Saekow S, Amornkitbamrung V (2013) DFT and TDDFT study on the electronic structure and photoelectrochemical properties of dyes derived from cochineal and lac insects as photosensitizer for dye-sensitized solar cells. J Mol Model 19:1407–1415
Senthilkumar P, Nithya C, Anbarasan PM (2014) Quantum chemical investigations on the effect of dodecyloxy chromophore in 4-amino stilbene sensitizer for DSSCs. Spectrochim Acta A Mol Biomol Spectrosc 122:15–21
Peach MJ, Benfield P, Helgaker T, Tozer DJ (2008) Excitation energies in density functional theory: an evaluation and a diagnostic test. J Chem Phys 128:044118
Zhang L, Yang X, Wang W, Gurzadyan GG, Li J, Li X, An J, Yu Z, Wang H, Cai B, Hagfeldt A (2019) 13.6% efficient organic dye-sensitized solar cells by minimizing energy losses of the excited state. ACS Energy Lett 4:943–951
Chaitanya K, Ju XH, Heron BM (2014) Theoretical study on the light harvesting efficiency of zinc porphyrin sensitizers for DSSCs. RSC Adv 4:26621–26634
Narayan MR (2012) Dye sensitized solar cells based on natural photosensitizers. Renew Sustain Energy Rev 16:208–215
Zhang J, Li H-B, Sun S-L, Geng Y, Wu Y, Su Z-M (2012) Density functional theory characterization and design of high-performance diarylamine-fluorene dyes with different π spacers for dye-sensitized solar cells. J Mater Chem 22(2):568–576
Daeneke T, Mozer AJ, Uemura Y, Makuta S, Fekete M, Tachibana Y, Koumura N, Bach U, Spiccia L (2012) Dye regeneration kinetics in dye-sensitized solar cells. J Am Chem Soc 134:16925–16928
Arunkumar A, Anbarasan PM (2018) Highly efficient organic indolocarbazole dye in different acceptor units for optoelectronic applications-a first principle study. Struct Chem 29:967–976
Rühle S, Greenshtein M, Chen SG, Merson A, Pizem H, Sukenik CS, Cahen D, Zaban A (2005) Molecular adjustment of the electronic properties of nanoporous electrodes in dye-sensitized solar cells. J Phys Chem B 109:18907–18913
Sang-aroon W, Saekow S, Amornkitbamrung V (2012) Density functional theory study on the electronic structure of Monascus dyes as photosensitizer for dye-sensitized solar cells. J Photochem Photobiol A 236:35–40
Arunkumar A, Prakasam M, Anbarasan PM (2017) Influence of donor substitution at D-π-A architecture in efficient sensitizers for dye-sensitized solar cells: first-principle study. Bull Mater Sci 40:1389–1396
Ning Z, Zhang Q, Wu W, Pei H, Liu BO, Tian HE (2008) Starburst triarylamine based dyes for efficient dye-sensitized solar cells. J Org Chem 73:3791–3797
Sayama K, Tsukagoshi S, Hara K, Ohga Y, Shinpou A, Abe Y, Suga S, Arakawa H (2002) Photoelectrochemical properties of J aggregates of benzothiazole merocyanine dyes on a nanostructured TiO2 film. J Phys Chem B 106:1363–1371
Xu B, Li Y, Song P, Ma F, Yang Y (2021) Significant Improvements of near-IR absorption, electron injection, and oxidized regeneration on organic sensitizers for solar cells. J Phys Chem C
Islam A, Sugihara H, Arakawa H (2003) Molecular design of ruthenium (II) polypyridyl photosensitizers for efficient nanocrystalline TiO2 solar cells. J Photochem Photobiol A 158:131–138
Arunkumar A, Shanavas S, Acevedo R, Anbarasan PM (2020) Computational analysis on D-π-A based perylene organic efficient sensitizer in dye-sensitized solar cells. Opt Quant Electron 52:1–13
Roy JK, Kar S, Leszczynski J (2020) Revealing the photophysical mechanism of N, N′-diphenyl-aniline based sensitizers with the D-D-π-A framework: theoretical insights. ACS Sustain Chem Eng 8:13328–13341
Funding
The authors would like to express their gratitude to the Deanship of Scientifc Research at King Khalid University, Abha, Saudi Arabia for funding this work through the Research Groups Program under Grant No. R.G.P.2/103/41.
Author information
Authors and Affiliations
Contributions
The manuscript was written through contributions of all the authors. P. M. Anbarasan, A. Arunkumar, and Mohd Shkir have given approval to the final version of the revised manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ponnusamy Munusamy, A., Ammasi, A. & Shkir, M. Computational analysis of carbazole-based newly efficient D-π-A organic spacer dye derivatives for dye-sensitized solar cells. Struct Chem 33, 1097–1107 (2022). https://doi.org/10.1007/s11224-021-01853-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11224-021-01853-4