Abstract
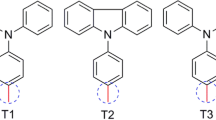
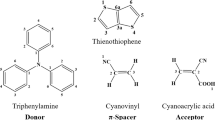
Using density functional theory and time-dependent density functional theory, we theoretically studied a new series of five novel metal-free organic dyes, namely D1–D5, for application in dye-sensitized solar cells based on donor–\(\uppi \)-spacer–acceptor (\(\mathrm{D}{-}\uppi {-}\mathrm{A}\)) groups. In this present study, five different donor groups have been designed based on triphenylamine–stilbene–cyanoacrylic acid (TPA–St–CA). The electronic structures, UV–visible absorption spectra and photovoltaic properties of these dyes were investigated. Different exchange-correlation functionals were used to establish a proper methodology procedure for calculation and comparison to experimental results of dye TPA–St–CA. The TD-WB97XD method, which gives the best correspondence to experimental values, is discussed. The calculated results reveal that the donor groups in D2 and D3 are promising functional groups for \(\mathrm{D}{-}\uppi {-}\mathrm{A}\). In particular, the D2 dye showed small energy levels and red-shift, negative \(\Delta {G}_{\mathrm{inject}}\), fastest regeneration and largest dipole moment and exciton binding energy when compared with TPA–St–CA.




Similar content being viewed by others
References
Grätzel M 2001 Nature 414 6861
Green M A, Emery K, Hishikawa Y and Warta W 2010 Prog. Photovolt. Res. Appl. 18 144
Ha E S, Yoo B, Baik H, Lee Y and Kim K J 2012 Chem. Asian J. 7 1624
Lu X, Zhou G, Wang H, Feng Q and Wang Z S 2012 Phys. Chem. Chem. Phys. 14 4802
Agrawal S, English N J, Thampi K R and MacElroy J M D 2012 Phys. Chem. Chem. Phys. 14 12044
Reagen B O and Gratzel M 1991 Nature 350 737
Irfan A 2013 Mater. Chem. Phys. 142 238
Tarsang R, Promarak V, Sudyoadsuk T, Namuangruk S and Jungsuttiwong S 2014 J. Photochem. Photobiol. A 273 8
Wu G, Kong F, Li J, Chen W, Fang X, Zhang C et al 2013 Dyes Pigm. 99 653
Ma X, Hua J, Wu W, Jin Y, Meng F, Zhan W et al 2008 Tetrahedron 64 345
Ehret A, Stuhl L and Spitler M T 2001 J. Phys. Chem. B 105 9960
Chen Y S, Chao L, Zeng Z H, Wang W B, Wang X S and Zhang B W 2005 J. Mater. Chem. 15 1654
Yao Q H, Meng F S, Li F Y, Tian H and Huang C H 2003 J. Mater. Chem. 13 1048
Tian H, Yang X, Chen R, Zhang R, Hagfeldt A and Sun L 2008 J. Phys. Chem. C 112 29
Zeng W D, Cao Y M, Bai Y, Wang Y H, Shi Y S and Wang P 2010 Chem. Mater. 22 5306
Prakasam M and Anbarasan P M 2016 RSC Adv. 6 75242
Hagberg D P, Yum J H, Lee H J, Angelis F D, Marinado T and Karlsson K M 2008 J. Am. Chem. Soc. 130 6259
Liang M, Xu W, Cai F, Chen P, Peng B and Chen J 2007 J. Phys. Chem. 111 11494
Choi H, Lee J K, Song K J, Song K, Kang S O and K O J 2007 Tetrahedron 63 1553
Koumura N, Wang Z S, Mori S, Miyashita M, Suzuki E and Hara K 2006 J. Am. Chem. Soc. 128 14256
Hara K, Sayama K, Ohga Y, Shinpo A, Suga S and Arakawa H 2001 Chem. Commun. 6 569
Wang Z S, Cui Y, Hara K, Dan-Oh Y, Kasada C and Shinpo A 2007 Adv. Mater. 19 1043
Hara K, Kurashige M, Danoh Y, Kasada C, Shinpo A, Suga S et al 2003 New J. Chem. 27 783
Balanay M P, Dipaling C V P, Lee S H, Kim D H and Lee K H 2007 Sol. Energy Mater. Sol. Cells 91 1775
Lin C Y, Lo C F, Luo L, Lu H P, Hung C S and Diau E W G 2008 J. Phys. Chem. C 113 2
Ito S, Zakeeruddin S M, Humphry-Baker R, Liska P, Charvet R and Comte P 2006 Adv. Mater. 18 1202
Schmidt-Mende L, Bach U, Humphry-Baker R, Horiuchi T, Miura H and Ito S 2005 Adv. Mater. 17 813
Horiuchi T, Miura H, Sumioka K and Uchida S 2004 J. Am. Chem. Soc. 126 12218
Cai N, Moon S J, Cevey-Ha L, Moehl T, Humphry-Baker R, Wang P et al 2011 Nano Lett. 11 1452
Xu M, Li R, Pootrakulchote N, Shi D, Guo J, Yi Z et al 2008 J. Phys. Chem. C 112 19268
Li G, Zhou Y F, Cao X B, Bao P, Jiang K J, Lin Y et al 2009 Chem. Commun. 16 2201
Yang J, Ganesan P, Teuscher J, Moehl T, Kim Y J, Yi C et al 2014 J. Am. Chem. Soc. 136 5722
Anderson S, Taylor P N and Verschoor G L B 2004 Chemistry 10 518
Duncan W R and Prezhdo O V 2007 Annu. Rev. Phys. Chem. 58 143
Garavelli M 2006 Theor. Chem. Acc. 116 87
Preat J, Jacquemin D, Wathelet V, André J M and Perpéte E A 2006 J. Phys. Chem. A 110 26477
Casanova D, Rotzinger F P and Gratzel M 2010 J. Chem. Theory Comput. 6 1219
Meng S, Kaxiras E, Nazeeruddin M K and Gratzel M 2011 J. Phys. Chem. C 115 9276
Becke A D 1993 J. Chem. Phys. 98 5648
Yanai T, Tew D P and Handy N C 2004 Chem. Phys. Lett. 393 51
Lin Y S, Li G D, Mao S P and Chai J D 2013 J. Chem. Theory Comput. 9 263
Rassolov V A, Ratner M A, Pople J A, Redfern P C and Curtiss L A 2001 J. Comput. Chem. 22 976
Barone V and Cossi M 1998 J. Phys. Chem. A 102 1995
Frisch M J, Trucks G W, Schlegel H B, Scuseria G E, Robb M A, Cheeseman J R et al 2009 Gaussian 09 (Wallingford, CT: Gaussian Inc.)
O’Boyle N M, Tenderholt A L and Langner K M 2008 J. Comput. Chem. 29 839
Yang Z, Shao C and Cao D 2015 RSC Adv. 5 22892
Jungsuttiwong S, Tarsang R, Sudyoadsuk T, Promarak V, Khongpracha P and Namuangruk S 2013 Org. Electron. 14 711
Asbury J B, Wang Y Q, Hao E, Ghosh H and Lian T 2001 Res. Chem. Intermed. 27 393
Cahen D, Hodes G, Grätzel M, Guillermoles J F and Riess I 2000 J. Phys. Chem. B 104 2053
Narayan M R 2012 Renew. Sustain. Energ. Rev. 16 208
Marinado T, Nonomura K, Nissfolk J, Karlsson M K, Hagberg D P, Sun L et al 2009 Langmuir 26 2592.
Rühle S, Greenshtein M, Chen S G, Merson A, Pizem H, Sukenik C S et al 2005 J. Phys. Chem. B 109 18907
Zhang J, Li H B, Sun S L, Geng Y, Wu Y and Su Z M 2012 J. Mater. Chem. 22 568
Preat J, Jacquemin D, Michaux C and Perpète E A 2010 Chem. Phys. 376 56
Katoh R, Furube A, Yoshihara T, Hara K, Fujihashi G, Takano S et al 2004 J. Phys. Chem. B 108 4818
Daeneke T, Mozer A J, Uemura Y, Makuta S, Fekete M, Tachibana Y et al 2012 J. Am. Chem. Soc. 134 16925
Scholes G D and Rumbles G 2006 Nat. Mater. 5 683
Li Y, Pullerits T, Zhao M and Sun M 2011 J. Phys. Chem. C 115 2156
Hwang S, Lee J H, Park C, Lee H, Kim C, Park C et al 2007 Chem. Commun. 46 4887
Acknowledgements
The authors are thankful to the learned referees for their useful and critical comments, which improved the quality of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Arunkumar, A., Prakasam, M. & Anbarasan, P.M. Influence of donor substitution at \(\mathrm{D}{-}\uppi {-}\mathrm{A}\) architecture in efficient sensitizers for dye-sensitized solar cells: first-principle study. Bull Mater Sci 40, 1389–1396 (2017). https://doi.org/10.1007/s12034-017-1497-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12034-017-1497-7