Abstract
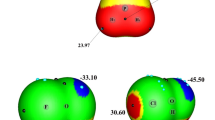
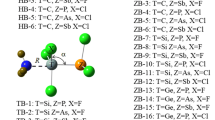
Quantum calculations at the MP2/cc-pVTZ have been used to examine 1:1 and 1:2 complexes between O2NX (X = Cl, Br) with HCN and HNC moieties. The interaction of the lone pair of the HCN(HNC) with the σ-hole and π-hole of O2NX molecules and hydrogen bonding between lone pairs of X and O of O2NX with H of HCN and HNC have been considered in 1:1 complexes. The 1:1 complexes can easily be differentiated using the stretching frequency of the N–X bond. Thus, those complexes with σ-hole and H···O2NX interactions show a blue shift of the N-X bond stretching while a red shift is observed in the complexes along the π-hole and H···XNO2 interactions. In the 1:2 complexes, the cooperative and diminutive energetic effects have been analyzed using the many-body interaction energies. The nature of the interactions has been characterized with the Atoms in Molecules (AIM) and Natural Bond Orbital (NBO) methodologies. Stabilization energies of 1:1 and 1:2 complexes including the variation of the zero-point vibrational energy (ΔZPVE) are in the range 3–9 kJ mol−1 and 21–40 kJ mol−1, respectively.




Similar content being viewed by others
References
Müller-Dethlefs K, Hobza P (1999) Chem Rev 100:143
Clark T, Hennemann M, Murray J, Politzer P (2007) J Mol Model 13:291
Auffinger P, Hays FA, Westhof E, Ho PS (2004) Proc Natl Acad Sci USA 101:16789
Politzer P, Lane P, Concha M, Ma Y, Murray J (2007) J Mol Model 13:305
Murray JS, Lane P, Politzer P (2007) Int J Quantum Chem 107:2286
Murray J, Concha M, Lane P, Hobza P, Politzer P (2008) J Mol Model 14:699
Murray J, Lane P, Clark T, Riley K, Politzer P (2012) J Mol Model 18:541
Molina MJ, Rowland FS (1974) Nature 249:810
Wayne RP (1991) Chemistry of atmospheres. Oxford University Press, Oxford
Fickert S, Helleis F, Adams JW, Moortgat GK, Crowley JN (1998) J Phys Chem A 102:10689
Lesclaux R, Caralp F, Dognon AM, Cariolle D (1986) Geophys Res Lett 13:933
Møller C, Plesset MS (1934) Phys Rev 46:618
Dunning TH (1989) J Chem Phys 90:1007
Riley KE, Pitoňák M, Černý Ji, Hobza P (2009) J Chem Theory Comput 6:66
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Montgomery JA, Vreven JT, Kudin KN, Burant JC, Millam JM, Iyengar SS, Tomasi J, Barone V, Mennucci B, Cossi M, Scalmani G, Rega N, Petersson GA, Nakatsuji H, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Klene M, Li X, Knox JE, Hratchian HP, Cross JB, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Ayala PY, Morokuma K, Voth GA, Salvador P, Dannenberg JJ, Zakrzewski VG, Dapprich S, Daniels AD, Strain MC, Farkas O, Malick DK, Rabuck AD, Raghavachari K, Foresman JB, Ortiz JV, Cui Q, Baboul AG, Clifford S, Cioslowski J, Stefanov BB, Liu G, Liashenko A, Piskorz P, Komaromi I, Martin RL, Fox DJ, Keith T, Al-Laham MA, Peng CY, Nanayakkara A, Challacombe M, Gill PMW, Johnson B, Chen W, Wong MW, Gonzales C, Pople JA (2003) Gaussian 03 Rev B02. Gaussian Inc, Pittsburgh
Boys SF, Bernardi F (1970) Mol Phys 19:553
Bader RFW (1990) Atoms in molecules: a quantum theory. Clarendon Press, Oxford
Popelier PLA (2000) Atoms in molecules. An introduction. Prentice Hall, Harlow
Keith TA (2011) AIMAll, 11.10.16. TK Gristmill Software, Overland Park
Bulat F, Toro Labbé A, Brinck T, Murray J, Politzer P (2010) J Mol Model 16:1679
Francis SG, Harvey JN, Walker NR, Legon AC (2008) J Chem Phys 128:204305
Kwabia Tchana F, Orphal J, Kleiner I, Rudolph HD, Willner H, Garcia P, Bouba O, Demaison J, Redlich B (2004) Mol Phys 102:1509
Zou WS, Han J, Jin WJ (2009) J Phys Chem A 113:10125
Zhang Y, Li AY, Cao LJ (2012) Struct Chem 23:627
Zhou ZJ, Liu HL, Huang XR, Li QZ, Sun CC (2010) Mol Phys 108:2021
Wang W, Zhang Y, Ji B (2010) J Phys Chem A 114:7257
Solimannead M, Ramezani V, Trujillo C, Alkorta I, Sanchez-Sanz G, Elguro J (2012) J Phys Chem A 116:5199
Rozas I, Alkorta I, Elguero J (2000) J Am Chem Soc 122:11154
Ziółkowski M, Grabowski SJ, Leszczynski J (2006) J Phys Chem A 110:6514
Domagała M, Matczak P, Palusiak M, Comp Theor Chem.http://dx.doi.org/10.1016/j.comptc.2012.05.012
Scheiner S (2011) Chem Phys Lett 514:32
Valiron P, Mayer I (1997) Chem Phys Lett 275:46
White JC, Davidson ER (1990) J Chem Phys 93:8029
Hankins D, Moskowitz JW, Stillinger FH (1970) J Chem Phys 53:4544
Xantheas SS, Burnham CJ, Harrison RJ (2002) J Chem Phys 116:1493
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Solimannejad, M., Nassirinia, N. & Amani, S. A computational study of 1:1 and 1:2 complexes of nitryl halides (O2NX) with HCN and HNC. Struct Chem 24, 651–659 (2013). https://doi.org/10.1007/s11224-012-0116-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11224-012-0116-x