Abstract
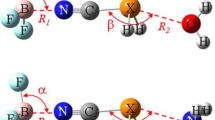
The complexes formed between TX3–ZX2 (T = C, Si, Ge; Z = P, As, Sb; X = F, Cl) and NH3 were studied at the MP2/aug-cc-pVTZ(PP) level. For each TX3–ZX2, two types of complex were obtained. For CX3-ZX2, NH3 is inclined to approach the σ-hole on the Z atom, forming a pnicogen bond. For TX3–ZX2 (T = Si and Ge), however, the base favors engaging in a tetrel bond with the σ-hole on the T atom although the corresponding pnicogen-bonded complex is also stable. When NH3 approaches the CX3 terminal of CX3–ZX2, weak interactions are observed that may be classified as van der Waals interactions. The relative stability of both types of complexes is not affected by the substituent X. The tetrel bond is very strong and the largest interaction energy is up to −144 kJ mol−1. Dispersion is dominant in the weak van der Waals complexes, while tetrel- and pnicogen-bonded complexes are dominated by electrostatic interactions, with comparable contributions from polarization.





Similar content being viewed by others
References
Schneider HJ (2009) Angew Chem Int Ed 48:3924–3977
Hunter CA, Sanders JKM (1990) J Am Chem Soc 112:5525–5534
Vickaryous WJ, Herges R, Jonhson DW (2004) Angew Chem Int Ed 43:5831–5833
Legon AC (2010) Phys Chem Chem Phys 12:7736–7747
Iwaoka M, Takemoto S, Tomoda S (2002) J Am Chem Soc 124:10613–10620
Murray JS, Lane P, Clark T, Politzer P (2007) J Mol Model 13:1033–1038
Scheiner S (2013) Acc Chem Res 46:280–288
Murray JS, Lane P, Politzer P (2007) Int J Quantum Chem 107:2286–2292
Bauzá A, Mooibroek TJ, Frontera A (2016) Chem Rec 16:473–487
Murray JS, Lane P, Politzer P (2009) J Mol Model 15:723–729
Alkorta I, Elguero J, Del Bene JE (2013) J Phys Chem A 117:10497–10503
Li QZ, Li R, Liu XF, Li WZ, Cheng JB (2012) ChemPhysChem 13:1205–1212
Li QZ, Li R, Liu XF, Li WZ, Cheng JB (2012) J Phys Chem A 116:2547–2553
Del Bene JE, Alkorta I, Elguero J (2015) Phys Chem Chem Phys 17:30729–30735
Li QZ, Guo X, Yang X, Li WZ, Cheng JB, Li HB (2014) Phys Chem Chem Phys 16:11617–11625
Guo X, Liu YW, Li QZ, Li WZ, Cheng JB (2015) Chem Phys Lett 620:7–12
Liu MX, Li QZ, Scheiner S (2017) Phys Chem Chem Phys 19:5550–5559
Bauzá A, Frontera A, Mooibroek TJ (2016) Phys Chem Chem Phys 18:1693–1698
Legon AC (2017) Phys Chem Chem Phys 19:14884–14896
Scheiner S (2017) J Phys Chem A 121:5561–5568
Martín-Fernández C, Montero-Campillo MM, Alkorta I, Elguero J (2017) J Phys Chem A 121:7424–7431
Clark T, Hennemann M, Murray JS, Politzer P (2007) J Mol Model 13:291–296
Murray JS, Lane P, Clark T, Riley KE, Politzer P (2012) J Mol Model 18:541–548
Grabowski SJ (2014) Phys Chem Chem Phys 16:1824–1834
Bauzá A, Mooibroek TJ, Frontera A (2016) ChemPhysChem 17:1608–1614
Scheiner S (2011) J Phys Chem A 115:11202–11209
Mani D, Arunan E (2013) Phys Chem Chem Phys 15:14377–14383
Gao L, Zeng YL, Zhang XY, Meng LP (2016) J Comput Chem 37:1321–1327
Bauzá A, Frontera A (2015) ChemPhysChem 16:3108–3113
Zhou PP, Yang X, Ye WC, Zhang LW, Yang F, Zhou DG, Liu SB (2016) New J Chem 40:9139–9147
Wei YX, Li QZ (2018) Mol Phys 116:222–230
Xu HL, Cheng JB, Yang X, Liu ZB, Li WZ, Li QZ (2017) ChemPhysChem 18:2442–2450
Dong WB, Yang X, Cheng JB, Li WZ, Li QZ (2018) J Fluor Chem 207:38–44
Solimannejad M, Ramezani V, Trujillo C, Alkorta I, Sánchez-Sanz G, Elguero J (2012) J Phys Chem A 116:5199–5206
Lang T, Li XY, Meng LP, Zheng SJ, Zeng YL (2015) Struct Chem 26:213–221
Nziko Vde PN, Scheiner S (2016) Phys Chem Chem Phys 18:3581–3590
Liu MX, Li QZ, Li WZ, Cheng JB, McDowell SAC (2016) RSC Adv 6:19136–19143
Wei QC, Li QZ, Cheng JB, Li WZ, Li HB (2016) RSC Adv 6:79245–79253
Scheiner S (2015) J Phys Chem A 119:9189–9199
Grabowski SJ (2013) Chem Eur J 19:14600–14611
Li QZ, Zhu HJ, Zhuo HY, Yang X, Li WZ, Cheng JB (2014) Spectrochim Acta A 132:271–277
Esrafili MD (2016) Mol Phys 114:1847–1855
Møller C, Plesset MS (1934) Phys Rev 46:618–622
Woon DE, Dunning Jr TH (1993) J Chem Phys 98:1358–1371
Boys SF, Bernardi F (1970) Mol Phys 19:553–566
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Montgomery Jr JA, Vreven T, Kudin KN, Burant JC, Millam JM, Iyengar SS, Tomasi J, Barone V, Mennucci B, Scalmani G, Cossi M, Rega N, Petersson GA, Nakatsuji H, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Klene M, Li X, Knox JE, Hratchian HP, Cross JB, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Ayala PY, Morokuma K, Voth GA, Salvador P, Dannenberg JJ, Zakrzewski VGDS, Daniels AD, Strain MC, Farkas O, Malick DK, Rabuck AD, Raghavachari K, Foresman JB, Ortiz JV, Cui Q, Baboul AG, Clifford S, Cioslowski J, Stefanov BB, Liu G, Liashenko A, Piskorz P, Komaromi I, Martin RL, Fox DJ, Keith T, Al-Laham MA, Peng CY, Nanayakkara A, Challacombe M, Gill PMW, Johnson B, Chen W, Gonzalez C, Wong MW, Pople JA (2009) Gaussian 09, revision A02. Gaussian Inc., Wallingford
Bulat FA, Toro-Labbé A, Brinck T, Murray JS, Politzer P (2010) J Mol Model 16:1679–1691
Bader RFW (1990) Atoms in Molecules: A Quantum Theory. Oxford University Press, Oxford
Bader RFW (2000) AIM2000 Program, Version 2.0. McMaster University, Hamilton Canada
Humphrey W, Dalke A, Schulten K (1996) J Mol Graph 14:33–38
Reed AE, Curtiss LA, Weinhold F (1988) Chem Rev 88:899–926
Su PF, Li H (2009) J Chem Phys 13:014102
Schmidt MW, Baldridge KK, Boatz JA, Elbert ST, Gordon MS, Jensen JH, Koseki S, Matsunaga N, Nguyen KA, Su SJ, Windus TL, Dupuis M, Montgomery JA (1993) J Comput Chem 14:1347–1363
Scheiner S (2011) Chem Phys 387:79–84
Keyvani ZA, Shahbazian S, Zahedi M (2016) Chem Eur J 22:5003–5009
Alkorta I, Sanchez-Sanz G, Elguero J (2014) J Phys Chem A 118:1527–1537
Arnold WD, Oldfield E (2000) J Am Chem Soc 122:12835–12841
Politzer P, Murray JS, Clark T (2015) J Mol Model 21:52
Hellmann H (1933) Z Phys 85:180–190
Feynman RP (1939) Phys Rev 56:340–343
Politzer P, Murray JS (2013) ChemPhysChem 14:278–294
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
ESM 1
(DOCX 169 kb)
Rights and permissions
About this article
Cite this article
Li, Y., Xu, Z. Competition between tetrel bond and pnicogen bond in complexes of TX3-ZX2 and NH3. J Mol Model 24, 247 (2018). https://doi.org/10.1007/s00894-018-3732-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00894-018-3732-6