Abstract
A generalized van der waals model is considered to study the thermodynamic properties of pure fluids. Analytical solution of the equivalent cubic equation of state is presented and the critical properties in the general form are derived. The fluctuations of number of particles are calculated in the grand canonical ensemble by using three quantities (scaled variance \(\omega (N)\), skewness \(S\sigma, \) and kurtosis \(k\sigma ^2\)). The critical behavior of these quantities is investigated in terms of the dimensionless particle number density and temperature for different models. It is found that the fluctuations have a singular behavior close to the critical point.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Equations of state (EoSs) are mathematical tools for describing the state of matters. Generalized EOSs are widely used to represent and predict thermodynamic properties and phase equilibria of pure fluids and mixtures. The two-parameter van der Waals (vdW) model (1873) was the first equation to predict vapor–liquid coexistence [1]. Later, numerous modifications to the vdW model have been presented (see for instance, [2,3,4,5,6,7,8,9,10,11,12,13,14]). The most commonly used equations of state are cubic equations, which have been extensively used over the last three decades. Cubic EoSs are a class of equations of state that may be represented by a polynomial when referencing the volume or compressibility factor, in such a way that the highest power in the polynomial is to the third degree [15, 16].
Here, we present a new generalization of the two-parameter vdW EoS and solve analytically the corresponding cubic EoS. Also, we reconsider the generalized vdW EoS in the grand canonical ensemble (GCE) formulation to calculate some measures of the particle number fluctuations such as the scaled variance, skewness, and kurtosis and investigate the critical behavior of their behavior in a vicinity of the critical point.
The paper is organized as follows: In Sect. 2, a new generalization of the vdW model and its corresponding cubic equation are presented. In Sect. 3, the cubic EoS is solved analytically and the critical properties are calculated in the general form. In Sect. 4, the GCE formulation of one family of the generalized vdW equation is derived. In Sect. 5, the particle number fluctuations for different models are studied. Three quantities measuring the particle number fluctuations are calculated and their behavior close to the critical point (CP) is then analyzed. The paper closes with a short discussion given in Sect. 6.
2 Generalized vdW EoS
Here, we consider a new generalization of the two-parameter vdW model [1] in the canonical ensemble (CE) formulation
where the first term represents the repulsive term and the the second represents the attractive term. The parameters \(r_1,r_2\) are two specific constants that vary depending on the EoS and the parameter k has the physical valid range of \(0\le k \le 2\). The characteristic parameters a and b describe, respectively, the attractive and repulsive interactions between N particles. With different values of k, \(r_1,\) and \(r_2\), most of the well-known EoS can be obtained. Table 1 presents the attractive term of some models, like vdW model [1], SRK model [3], PR model [6], PT model [9], and Nasrifar-Bolland (NB model) [17]. The repulsive term (not shown) is similar in all of these models as defined in Eq. 1.
Using the critical point conditions
EoS (1) can be transformed into the corresponding generalized cubic polynomial in the form
3 Analytical Solution of the Cubic EoS
The solution of the cubic EoS (5) depends on the value of k as follows:
I- When \(k=0\) and \(r_1=r_2=0\), one obtains the cubic equation of the original vdW model
In this case, we obtain the well-known critical point \(V_\mathrm{{c}}=3Nb\), \(k_BT_\mathrm{{c}}=\frac{8a}{27b},\) and \(P_\mathrm{{c}}=\frac{a}{27b^2}\).
II- When \(k=1\), the generalized cubic Eq. 5 reduces to the form
where
There are different analytical methods for solving the cubic EoS. Here, we consider Cardano method (see [18, 19]) which can be used to calculate all real and complex roots of cubic polynomials that have only real coefficients. In this method, the cubic polynomial (7) is reduced via the substitution
which leads to
with the coefficients
The numbers and types of roots of Eq. 10 depend on the sign of the the polynomial discriminant, \(\Delta \), defined as
There are three possibilities:
1. If \(\Delta >0,\) there is only one real root and two conjugate-complex solutions
where
The conjugate-complex solutions also may be ignored in the context of cubic EoS, because they do not describe any physical solution.
2. If \(\Delta < 0\), there are three real roots come from the expression
The angle \(\theta \) (in radius) is calculated as
Equation 15 can be rewritten in a way to provide the roots in ascending order, as follows
3. When \(\Delta =0\), we have \(q=\pm \sqrt{p^3}\), that is, \(\theta =\mathrm{arccos}(\pm 1)\), i.e., \(\theta \) ranges from 0 to \(\pi \). In this case, Eq. 17 can be simplified to obtain the special case of three real roots, where two of them are identical
Using the second critical condition (3), one can obtains the critical temperature
In this case, the critical compressibility factor \(Z_\mathrm{{c}}\) takes the form
III- When \(k=2\), we obtain the same cubic equation of \(k=1\) (7) and its solution but with \(r_1=2r_1\) and \(r_2=r_1^2+r_2\).
4 The GCE Formulation of the Generalized vdW Model
We consider one family of the generalized vdW model, by taking \(k=1, r_1=2\beta b,\) and \(r_2=\beta ^2 b^2\) in Eq. 1, we obtain
where \(n=N/V\) is the particle number density. Note that, for \(\beta =0\), the standard vdW model is obtained and for \(\beta =1/\sqrt{3}\), NB model is obtained.
The GCE formulation of (21) can be obtained as follows:
First, we find the free energy F(T, V, N) in the CE formulation, which can be obtained by integrating the thermodynamic identity
which for the model (21) yields
where \(F_{id}(T,V-Nb,N)\) is the free energy of the ideal gas,
and the quantum concentration \(n_Q\) is given by
where m is the mass of a particle and \(\hbar \) is Planck\(^{'}\)s constant. Using Eq. 23 for the free energy, we can find all the thermodynamic quantities, such as the chemical potential \(\mu \), the particle number density n, the entropy density s, and the energy density \(\varepsilon \). Differentiating the relation (23) with respect to T and N, one gets S and \(\mu \), respectively
where
are the entropy, the chemical potential, and the pressure of the ideal gas, respectively.
Second, we invert the relation (27) to get the particle number density \(n(T,\mu )\) which lies in the heart of the GCE formulation [20]
where \(n_{id}\) is the density of the ideal gas and \(\mu _{id}=\mu -bP_{id}+\frac{an}{1+\beta bn}\Big (1+\frac{1}{1+\beta bn}\Big )\).
Third, we put \(n(T,\mu )\) back into the CE pressure (21) to obtain the pressure in the GCE
5 Critical Behavior of the Particle Number Fluctuations
Our concentration in this section will be in the fluctuations of number of particles, which are absent in the CE. The particle number fluctuations in the GCE can be characterized by the following dimensionless cumulants (susceptibilities),
which are related to the moments of the particle number distribution by
In the GCE, the particle number N fluctuates around its average value \(<N>\) with the normalized probability distribution P(N). Let us introduce the k moment \(<N^k>\)
where symbol \(<\dots>\) denotes the GCE averaging, \(\Delta N\equiv N-<N>,\) and the variance \(\sigma ^2=<(\Delta N)^2>\).
Now, we consider three well-known measures of the particle number fluctuations: the scaled variance \(\omega (N)\), the skewness \(S\sigma, \) and the kurtosis \(k\sigma ^2\). The scaled variance \(\omega (N)\) is an intensive measure of N-fluctuations and is given by (Taking \(k_B=1\))
The skewness \(S\sigma \) measures the degree of asymmetry of the distribution P(N) around its mean value \(<N>\) and is defined as
The kurtosis \(\sigma ^2\) is the measure of “peakedness” of the probability distribution P(N),
Note that for \(\beta =0,\) the values of \(\omega (N)\), \(S\sigma, \) and \(k\sigma ^2\) of the model (21) coincide with the corresponding results obtained in [21].
To study the critical behavior of the particle number fluctuations close to the CP, we rewrite Eqs. (34), (35), and (36) in terms of the reduced parameters \(P_r=P/P_\mathrm{{c}}\), \(n_r=n/n_\mathrm{{c}},\) and \(T_r=T/T_\mathrm{{c}}\). By solving the cubic quation of Eq. 21 (using the method of Sect. 3), one obtains the critical point
The results obtained from Eq. 37 coincide with the critical properties of vdW EoS (\(\beta =0\)) [1], NB EoS (\(\beta =1/\sqrt{3}\)) [17], and \(\beta =1\) [22].
Using Eq. 37, we can rewrite Eqs. 34, 35, and 36 in the reduced form
and
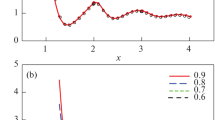
The scaled variance (38) as a function of \(n_r\) and different values of \(\beta \) (0, 0.5, and 1) is plotted in Fig. 1. We notice that \(\omega (N)\rightarrow 1\) as \(n_r\rightarrow 0\) (this corresponds to the ideal gas), \(\omega (N)\rightarrow 0\) as \(n_r\rightarrow 3+2\beta \) (this corresponds to the liquid with the highest possible density), and \(\omega (N)\rightarrow \infty \) at CP (\(T_r=n_r=1\)).
The scaled variance \(\omega (N)\) (38) as a function of \(n_r\) for \(\beta \)=0, 0.5, and 1 at (a) \(T_r=1.7\) and (b) \(T_r=1\)
For the skewness, it is clear from (39) that \(S\sigma >0\) at \(n_r<\frac{3+2\beta }{3}\) (the gas phase), \(S\sigma <0\) at \(n_r>\frac{3+2\beta }{3}\) (the liquid phase), and \(S\sigma =0\) at \(n_r=\frac{3+2\beta }{3}\) . We notice also, that \(S\sigma \rightarrow 1\) as \(n_r\rightarrow 0\) and \(S\sigma \rightarrow 0\) as \(n_r\rightarrow 3+2\beta \) (see Fig. 2).
The skewness \(S\sigma \) (39) as a function of \(n_r\) for \(\beta \)=0, 0.5, and 1 at (a) \(T_r=0.7\) and (b) \(T_r=1.7\)
Equation 40 shows that at \(T_r<1,\) the kurtosis has a large positive value (leptokurtic) for both \(n_r<\frac{3+2\beta }{3}\) (the gas phase) and \(n_r>\frac{3+2\beta }{3}\) (the liquid phases) (Fig. 3a). We notice also, that the kurtosis has a negative value (platykurtic) at \(T_r>1\) and \(n_r=\frac{3+2\beta }{3}\) (Fig. 3b).
The kurtosis \(k\sigma ^2\) (40) as a function of \(n_r\) for \(\beta \)=0, 0.5, and 1 at (a) \(T_r=0.7\) and (b) \(T_r=1.7\)
6 Conclusions
Here, a generalization of the two-parameter vdW model is presented by setting three parameters \(r_1,r_2,\) and k to the attractive term. The cubic equation is calculated exactly (see (5)) and solved analytically for \(0\le k\le 2\), through which the critical properties of the generalized vdW model are determined .
The Grand canonical ensemble formulation of one family of the generalized vdW equation is derived (see (30)). The particle fluctuations are characterized by three quantities scaled variance \(\omega (N)\), skewness \(S\sigma, \) and kurtosis \(k\sigma ^2\). An analytical expressions for these quantities are derived in terms of the reduced variables (\(n_r, T_r)\) for general value \(\beta \) (see (38)–(40)) and analyzed in a vicinity of the critical point . The results for \(\beta =0\) in the present work are consistent with the results obtained in [21].
As seen from Fig. 1, the scaled variance is a positive quantity, approaches the ideal gas in the limit of small densities, i.e., \(\omega (N)\cong 1\), as \(n\rightarrow 0\) and corresponds to the highest possible density (\(\omega (N)\rightarrow 0\)) as \(n_r\rightarrow 3+2\beta \). From Fig. 2, it is seen that the skewness is positive at \(n_r<\frac{3+2\beta }{3}\) (gas phase) and negative (liquid phase) at \(n_r>\frac{3+2\beta }{3}\) for all values of \(T_r\). Also, the skewness \(S\sigma \rightarrow 0\) as \(n_r\rightarrow \frac{3+2\beta }{3}\)(\(n=n_\mathrm{{c}}\) line), and this line is the transition line from gas to liquid phase. From Fig. 3a, it is noticed that the kurtosis is positive at \(T>T_\mathrm{{c}}\) for both \(n_r<\frac{3+2\beta }{3}\) and \(n_r>\frac{3+2\beta }{3}\) and close to the CP, the kurtosis changes rapidly from positive to negative values at \(T>T_\mathrm{{c}}\) (see Fig. 3b).
Finally, it is noticed from the calculations and the figures that the three quantities \(\omega (N)=S\sigma =k\sigma ^2=1\) when the reduced critical density \(n_r\rightarrow 0\), i.e., the vdW EoS corresponds to the ideal gas and the distribution approaches the Poisson distribution. Also, it is found that the fluctuations have a singular behavior close to the critical point where the three quantities (\(\omega (N)\), \(S\sigma, \) and \(k\sigma ^2\)) diverge at the critical point.
References
J.D. van der Waals, Ph.D. Thesis, Leiden Univ. (1873); English translation: J.D. van der Waals, On the Continuity of the Gaseous and Liquid States. Dover Publications Inc., New York (1988)
O. Redlich, J.N.S. Kwong, On the thermodynamics of solutions. V: an equation of state. Fugacities of gaseous solutions. Chem. Rev. 44, 233–244 (1949)
G. Soave, Equilibrium constants from a modified Redlich-Kwong equation of state. Chem. Eng. Sci. 27, 1197–1203 (1972)
C.H. Twu, J.E. Coon, J.R. Cunningham, A new generalized alpha function for a cubic equation of state Part 2. Redlich-Kwong equation. Fluid Phase Equilib. 105, 61–69 (1995)
F. Souahi, S. Sator, S.A. Albne, F.K. Kies, C.E. Chitour, Development of a New Form for the Alpha function of the Redlich–Kwong Cubic Equation of state. Fluid Phase Equilib. 153, 73–80 (1998)
D.Y. Peng, D.B. Robinson, A new two-constant equation of state. Ind. Eng. Chem. Fundam. 5, 59–64 (1976)
C.H. Twu, J.E. Coon, J.R. Cunningham, A new generalized Alpha function for a cubic equation of state part 1. Fluid Phase Equilib. 105, 49–59 (1995)
K.A.M. Gasem, W. Gao, Z. Pan, R.L. Robinson, A Modified temperature dependence for the Peng-robinson equation of State. Fluid Phase Equilib. 181, 113–125 (2001)
N.C. Patel, A.S. Teja, A new cubic Equation of state for Fluids and Fluid Mixtures. Chem. Eng. Sci. 37, 463–473 (1982)
J.O. Valderrama, A generalized Patel–Teja equation of state for polar and nonpolar fluids and their mixtures. J. Chem. Eng. Jpn. 23, 87–91 (1990)
M.A. Trebble, P.R. Bishnoi, Development of a new four-parameter cubic equation of state. Fluid Phase Equilib. 35, 1–18 (1987)
P.H. Salim, M.A. Trebble, A modified Trebble–Bishnoi equation of state: thermodynamic consistency revisited. Fluid Phase Equilib. 65, 59–71 (1991)
J.O. Valderrama, The state of the cubic equations of state. Ind. Eng. Chem. Res. 42, 1603–1618 (2003)
J.S. Lopez-Echeverry, S. Reif-Acherman, E. Araujo-Lopez, Peng-Robinson equation of state: 40 years through cubics. Fluid Phase Equilib. 447, 39–71 (2017)
L.A. Forero, J.A. Velasquez, A generalized cubic equation of state for non-polar and polar substances. Fluid Phase Equilib. 418, 74–87 (2016)
P.N.P. Ghoderao, V.H. Dalvi, M. Narayan, A five-parameter cubic equation of state for pure fluids and mixtures. Chem. Eng. Sci. 3, 100026 (2019)
K. Nasrifar, O. Bolland, Prediction of thermodynamic properties of natural gas mixtures using 10 equations of state including a new cubic two-constant equation of state. J. Petrol. Sci. Eng. 51, 253–266 (2006)
E. W. Weisstein, CRC Concise Encyclopedia of Mathematics, Chapter Cubic Equation, pp 362–365. CRC Press, (1999)
U.K. Deiters, Calculation of densities from cubic equations of state. AIChE J. 48, 882–886 (2002)
V. Vovchenko, D.V. Anchishkin, M.I. Gorenstein, Particle number fluctuations for the van der Waals equation of state. J. Phys. A 48, 305001 (2015)
V. Vovchenko, R.V. Poberezhnyuk, D.V. Anchishkin, M.I. Gorenstein, Non-Gaussian particle number fluctuations in vicinity of the critical point for van der Waals equation of state. J. Phys. A 49, 015003 (2016)
M.E. Amin, Analytical treatment of the critical properties of a generalized van der Waals equation. Z. Naturforsch 76, 625–632 (2021)
Funding
Open access funding provided by The Science, Technology & Innovation Funding Authority (STDF) in cooperation with The Egyptian Knowledge Bank (EKB).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interests
The author declares that he has no conflict of interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Amin, M.E. Further Results on a Generalized van der Waals Model. Int J Thermophys 43, 166 (2022). https://doi.org/10.1007/s10765-022-03090-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10765-022-03090-1