Abstract
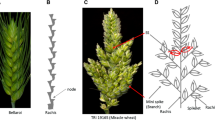
The “false glume” is an extra glume between the glume and lemma of the outer florets of each spikelet. The “false glume” phenotype was found in tetraploid wheat Triticum durum Desf. (2n = 4x = 28, genome BBAuAu) ‘Sharik’ and a Chernobyl radio mutant (‘UKR-1’) of hexaploid wheat T. aestivum L. (2n = 6x = 42, genome BBAuAuDD). The spikes of Sharik and UKR-1 were semi-compact; suggesting homoeoallelism or allelism at the false glume and semi-compact spike loci, which are specific to diploid wheat T. sinskajae A. Filat. et Kurkiev (2n = 2x = 14, genome AmAm). The objective of this study was to map the novel mutant genes for false glume and semi-compact spike in T. aestivum, T. durum and T. sinskajae, and to assess homoeologous relationship in genus Triticum L. by using molecular markers. It was evident that the gene false glume (fg) and semi-compact spike genes were linked in the F2 of UKR-1/T. aestivum ‘Novosibirskaya 67’, F2 of Sharik/T. durum ‘LD222’ and F2 of T. sinskajae k-48993/T. monococcum L. (2n = 2x = 14, genome AmAm) CItr 13962. The segregation analyses confirmed that the semi-compact spike locus (sc1) in Sharik was allelic to the sc1 locus in UKR-1, whereas neither loci were allelic to the C locus on chromosome 2D or the Cp locus on chromosome 5A. The sc1 gene and fg gene were bracketed by Xgwm372 and Xgwm312 in the F2 of Sharik/LD222, and by Xgwm372 and Xwmc644 in the F2 of UKR-1/N67. The sog gene for semi-compact spike of T. sinskajae, and fg genes were bracketed by Xgwm558 and Xwmc644 in the F2 of k-48993/CItr 13962. It was concluded that the semi-compact and spike false glume genes in T. sinskajae, T. durum and T. aestivum were determined by homoeoalleles in the short arm of chromosome 2A.



Similar content being viewed by others
References
Amagai Y, Martinek P, Watanabe N, Kuboyama T (2014) Microsatellite mapping of genes for branched spike and soft glumes in Triticum monococcum L. Genet Resour Crop Evol 61:465–471
Amagai Y, Takayama A, Kuboyama T, Watanabe N (2016) Microsatellite mapping of the gene conferring compact spike in Japanese landraces “Gumbai” of common wheat (Triticum aestivum L.). Genet Resour Crop Evol 209:709–714
Burdenyuk-Tarasevych LA (2006) Formation of systemic mutations of winter wheat as a consequence of radioactive irradiation of Triticum aestivum in the Chernobyl Power Station Exclusion Zone. In: Factors of experimental evolution of organisms. Ukrainian Society of Genetics and Plant Breeders named after N. I. Vavilov. Kiev, pp 339–344 (in Ukrainian)
Burdenyuk-Tarasevych LA (2009) Results of utilization of Chernobyl radio mutant in breeding programs of Triticum aestivum L. In: Shu QY (ed) Induced plant mutations in the genomics era. Food and Agriculture Organization of the United Nations, Rome, pp 80–82
Dellaporta SL, Wood J, Hicks JB (1983) A plant DNA minipreparation: version II. Plant Mol Biol Rep 1:19–21
Dorofeev VF, Filatenko AA, Migushova EF, Udaczin RA, Jakubziner MM (1979) Wheat. In: Dorofeev VF, Korovina ON (eds) Flora of cultivated plants, vol 1. Kolos, Leningrad, p 346
Faris JD, Zhang Z, Garvin DF, Xu SS (2014a) Molecular and comparative mapping of genes governing spike compactness from wild emmer wheat. Mol Genet Genomics 289:641–651
Faris JD, Zhang Z, Chao S (2014b) Map-based analysis of the tenacious glume gene Tg-B1 of wild emmer and its role in wheat domestication. Gene 542:198–208
Goncharov NP (1997) Comparative genetic study of tetraploid forms of common wheat without D genome. Genetika 33:660–663
Goncharov NP, Kondratenko EJ, Bannikova SV, Konovalov AA, Golovnina KA (2007) Comparative genetic analysis of diploid naked wheat Triticum sinskajae and the progenitor T. monococcum accession. Russ J Genet 43:1248–1256
Grodzinsky D, Kolomiets O, Burdenyuk L (1999) Collection of Chernobyl mutants of winter wheat. Mimeograph (Natl Acad Sci, Ukraine), Kiev, pp 1–29 (in Russian)
Hammer K, Filatenko AA, Pistrick K (2011) Taxonomic remarks on Triticum L. and× Triticosecale Wittm. Genet Resour Crop Evol 58:3–10
Johnson EB, Nalam VJ, Zemetra RS, Riera-Lizarazu O (2008) Mapping the compactum locus in wheat (Triticum aestivum L.) and its relationship to other spike morphology genes of the Triticeae. Euphytica 163:193–201
Kaltsikes PJ, Evans LE, Larter EN (1969) Morphological and meiotic characteristics of the extracted AABB tetraploid component of three varieties of common wheat. Can J Genet Cytol 11:65–71
Kerber ER (1964) Wheat: reconstitution of the tetraploid component (AABB) of hexaploids. Science 143:253–255
Kerber ER, Rowland GG (1974) Origin of the free threshing character in hexaploid wheat. Can J Genet Cytol 16:145–154
Kosambi DD (1943) The estimation of map distances from recombination values. Ann Eugen 12:172–175
Kosuge K, Watanabe N, Kuboyama T, Melnik VM, Yanchenko VI, Rosova MA, Goncharov NP (2008) Cytological and microsatellite mapping of mutant genes for spherical grain and compact spikes in durum wheat. Euphytica 159:289–296
Kosuge K, Watanabe N, Melnik VM, Laikova LI, Goncharov NP (2012) New sources of compact spikes morphology determined by genes on chromosome 5A in hexaploid wheat. Genet Resour Crop Evol 59:1115–1124
Kuspira J, MacLagen J, Bhambhani RN, Sadasivaiah RS, Kim N-S (1989) Genetic and cytogenetic analyses of the A genome of Triticum monococcum L. V. Inheritance and linkage relationships of genes determining the expression of 12 qualitative characters. Genome 32:869–881
Maan SS, Gordon J (1988) Compendium of alloplasmic lines and amphiploids in the Triticeae. In: Miller TE, Koebner RMD (eds) Proceedings of 7th international wheat genet symposium, Cambridge Lab, Inst Plant Sci Res, Trumpington, Camridge, pp 1325–1371
Malinowski E (1914) Les hybrids du formet. Bull Int Acad Sci Cravovie 3:410–450
Manly KF, Cudmore RH Jr, Meer JM (2001) Map manager QTX, cross-platform software for genetic mapping. Mam Genome 12:930–932
Reinhardt D, Kuhlemeier C (2002) Plant architecture. EMBO Rep 3:846–851
Röder MS, Korzun V, Wendehake K, Plaschke J, Tixier MH, Leroy P, Ganal MW (1998) A microsatellite map of wheat. Genetics 149:2007–2023
Rowland GG, Kerber ER (1974) Telocentric mapping in hexaploid wheat of genes for leaf rust resistance and other characters derived from Aegilops squarrosa. Can J Genet Cytol 16:137–144
Song QJ, Shi JR, Singh S, Fickus EW, Costa JM, Lewis J, Gill BS, Ward R, Cregan PB (2005) Development and mapping of microsatellite (SSR) markers in wheat. Theor Appl Genet 110:550–560
Sood S, Kuraparthy V, Bai G, Gill BS (2009) The major threshability genes soft glume (Sog) and tenacious glume (Tg) of diploid and polyploidy wheat, trace their origin to independent mutations at non-orthologous loci. Theor Appl Genet 119:341–351
Taenzler B, Esposti RF, Vaccino P, Brandolini A, Effgen S, Heun M, Schäfer-Pregl, Borghi B, Salamini F (2002) Molecular linkage map of einkorn wheat: mapping of storage-protein and soft-glume genes and bread-making quality QTLs. Genet Res Camb 80:131–143
Torada A, Koike M, Mochida K, Ogihara Y (2006) SSR-based linkage map with new markers using an intraspecific population of common wheat. Theor Appl Genet 112:1042–1051
Voss-Fels K, Frisch M, Qian L, Kontowski S, Friedt W, Gottwald S, Snowdon RJ (2015) Subgenomic diversity patterns caused by directional selection in bread wheat gene pools. Plant Genome 8:1–13
Zhang H, Zhu B, Qi B, Gou X, Dong Y, Xu C, Zhang B, Huang W, Liu C, Wang X, Yang C, Zhou H, Kashkush K, Feldman M, Wendel JF, Liu B (2014) Evolution of the BBAA component of bread wheat during its history at the allohexaploid level. Plant Cell 26:2761–2776
Acknowledgements
We thank Dr. D. L. Klindworth, USDA-ARS, Northern Crop Science Laboratory, Fargo, for helpful comments on the manuscript. N. W. acknowledges the finacial support by the Elizabeth Arnold Fuji Foundation, Nagoya, Japan. N. P. G. acknowledges the financial support by the Russian Science Foundation (Grant Number: 16-16-10021).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Amagai, Y., Burdenyuk-Tarasevych, L.A., Goncharov, N.P. et al. Microsatellite mapping of the loci for false glume and semi-compact spike in Triticum L.. Genet Resour Crop Evol 64, 2105–2113 (2017). https://doi.org/10.1007/s10722-017-0500-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10722-017-0500-x