Abstract
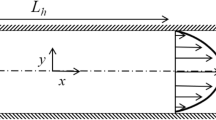
A rheometric method based on velocity profiling by optical coherence tomography (OCT) was used in the analysis of rheological and boundary layer flow properties of a 0.5% microfibrillated cellulose (MFC) suspension. The suspension showed typical shear thinning behaviour of MFC in the interior part of the tube, but the measured shear viscosities followed interestingly two successive power laws with an identical flow index (exponent) and a different consistency index. This kind of viscous behaviour, which has not been reported earlier for MFC, is likely related to a sudden structural change of the suspension. The near-wall flow showed existence of a slip layer of 2–12 μm thickness depending on the flow rate. Both the velocity profile measurement and the amplitude data obtained with OCT indicated that the slip layer was related to a concentration gradient appearing near the tube wall. Close to the wall the fluid appeared nearly Newtonian with high shear rates, and the viscosity approached almost that of pure water with decreasing distance from the wall. The flow rates given by a simple model that included the measured yield stress, viscous behavior, and slip behavior, was found to give the measured flow rates with a good accuracy.












Similar content being viewed by others
References
Agoda-Tandjawa G, Durand S, Berot S, Blassel C, Gaillard C, Garnier C, Doublier J (2010) Rheological characterization of microfibrillated cellulose suspensions after freezing. Carbohydr Polym 80:677–686. doi:10.1016/j.carbpol.2009.11.045
Barnes HA (1995) A review of the slip (wall depletion) of polymer solutions, emulsions and particle suspensions in viscometers: its cause, character, and cure. J Non Newtonian Fluid Mech 56:221–251. doi:10.1016/0377-0257(94)01282-M
Barnes HA, Hutton JF, Walters K (1989) An introduction to rheology. Elsevier, Amsterdam
Bergman TL, Incropera FP (2011) Fundamentals of heat and mass transfer. Wiley, Hoboken
Bonesi M, Churmakov D, Meglinski I (2007) Study of flow dynamics in complex vessels using doppler optical coherence tomography. Meas Sci Technol 18:3279–3286. doi:10.1088/0957-0233/18/11/003
Bounoua S, Lemaire E, Férec J, Ausias G, Kuzhir P (2016) Shear-thinning in concentrated rigid fiber suspensions: aggregation induced by adhesive interactions. J Rheol 60:1279–1300. doi:10.1122/1.4965431
Bukowska DM, Derzsi L, Tamborski S, Szkulmowski M, Garstecki P, Wojtkowski M (2013) Assessment of the flow velocity of blood cells in a microfluidic device using joint spectral and time domain optical coherence tomography. Opt Express 21:24025–24038. doi:10.1364/OE.21.024025
Chen P, Yu H, Liu Y, Chen W, Wang X, Ouyang M (2013) Concentration effects on the isolation and dynamic rheological behavior of cellulose nanofibers via ultrasonic processing. Cellulose 20:149–157. doi:10.1007/s10570-012-9829-7
Cheng DC (1986) Yield stress: a time-dependent property and how to measure it. Rheol Acta 25:542–554. doi:10.1007/BF01774406
Cloitre M, Bonnecaze R (2017) A review on wall slip in high solid dispersions. Rheol Acta 56:283–305. doi:10.1007/s00397-017-1002-7
Czajkowski J, Vilmi P, Lauri J, Sliz R, Fabritius T, Myllyla R (2012) Characterization of ink-jet printed RGB color filters with spectral domain optical coherence tomography. In Proc SPIE 8493:849308. doi:10.1117/12.929681
Derakhshandeh B, Hatzikiriakos SG, Bennington CPJ (2010) The apparent yield stress of pulp fiber suspensions. J Rheol 54:1137–1154. doi:10.1122/1.3473923
Dimic-Misic K, Gane PAC, Paltakari J (2013) Micro- and nanofibrillated cellulose as a rheology modifier additive in CMC-containing pigment-coating formulations. Ind Eng Chem Res 52:16066–16083. doi:10.1021/ie4028878
Dimic-Misic K, Rantanen J, Maloney TP, Gane P (2016) Gel structure phase behavior in micro nanofibrillated cellulose containing in situ precipitated calcium carbonate. J Appl Polym Sci 133:43486. doi:10.1002/app.43486
Haavisto S, Liukkonen J, Jäsberg A, Koponen A, Lille M, Salmela J (2011) Laboratory-scale pipe rheometry: a study of a microfibrillated cellulose suspension. In: Proceedings of papercon, pp 704–717
Haavisto S, Salmela J, Jäsberg A, Saarinen T, Karppinen A, Koponen A (2015) Rheological characterization of microfibrillated cellulose suspension using optical coherence tomography. Tappi J 14:291–302
Harvey M, Waigh TA (2011) Optical coherence tomography velocimetry in controlled shear flow. Phys Rev E 83:031502. doi:10.1103/PhysRevE.83.031502
Hoeng F, Denneulin A, Bras J (2016) Use of nanocellulose in printed electronics: a review. Nanoscale 8:13131–13154. doi:10.1039/C6NR03054H
Huang D, Swanson E, Lin C, Schuman J, Stinson W, Chang W, Hee M, Flotte T, Gregory K, Puliafito C, Fujimoto J (1991) Optical coherence tomography. Science 254:1178–1181. doi:10.1126/science.1957169
Iotti M, Gregersen ØW, Moe S, Lenes M (2011) Rheological studies of microfibrillar cellulose water dispersions. J Polym Environ 19:137–145. doi:10.1007/s10924-010-0248-2
Jaradat S, Harvey M, Waigh TA (2012) Shear-banding in polyacrylamide solutions revealed via optical coherence tomography velocimetry. Soft Matter 8:11677–11686. doi:10.1039/C2SM26395E
Jia Y, Bagnaninchi PO, Yang Y, Haj AE, Hinds MT, Kirkpatrick SJ, Wang RK (2009) Doppler optical coherence tomography imaging of local fluid flow and shear stress within microporous scaffolds. J Biomed Opt 14:034014–034014. doi:10.1117/1.3130345
Jonoobi M, Oladi R, Davoudpour Y, Oksman K, Dufresne A, Hamzeh Y, Davoodi R (2015) Different preparation methods and properties of nanostructured cellulose from various natural resources and residues: a review. Cellulose 22:935–969. doi:10.1007/s10570-015-0551-0
Kalia S, Boufi S, Celli A, Kango S (2014) Nanofibrillated cellulose: surface modification and potential applications. Colloid Polym Sci 292:5–31. doi:10.1007/s00396-013-3112-9
Karppinen A, Saarinen T, Salmela J, Laukkanen A, Nuopponen M, Seppälä J (2012) Flocculation of microfibrillated cellulose in shear flow. Cellulose 19:1807–1819. doi:10.1007/s10570-012-9766-5
Kumar V, Nazari B, Bousfield D, Toivakka M (2016) Rheology of microfibrillated cellulose suspensions in pressure-driven flow. Appl Rheol 26:43534. doi:10.3933/ApplRheol-26-43534
Lasseuguette E, Roux D, Nishiyama Y (2008) Rheological properties of microfibrillar suspension of TEMPO-oxidized pulp. Cellulose 15:425–433. doi:10.1007/s10570-007-9184-2
Lauri J, Bykov AV, Myllyla R (2011a) Determination of suspension viscosity from the flow velocity profile measured by Doppler Optical Coherence Tomography. Photon Lett Poland 3:82–84. doi:10.4302/plp.2011.2.13
Lauri J, Bykov AV, Priezzhev AV, Myllylä R (2011b) Experimental study of the multiple scattering effect on the flow velocity profiles measured in Intralipid phantoms by DOCT. Laser Phys 21:813–817. doi:10.1134/S1054660X11070164
Lauri J, Czajkowski J, Myllylä R, Fabritius T (2015) Measuring flow dynamics in a microfluidic chip using optical coherence tomography with 1 µm axial resolution. Flow Meas Instrum 43:1–5. doi:10.1016/j.flowmeasinst.2015.02.001
Lavoine N, Desloges I, Dufresne A, Bras J (2012) Microfibrillated cellulose–its barrier properties and applications in cellulosic materials: a review. Carbohydr Polym 90:735–764. doi:10.1016/j.carbpol.2012.05.026
Markstedt K, Mantas A, Tournier I, Ávila H, Hägg D, Gatenholm P (2015) 3D Bioprinting human chondrocytes with nanocellulose–alginate bioink for cartilage tissue engineering applications. Biomacromolecules 16:1489–1496. doi:10.1021/acs.biomac.5b00188
Martoïa F, Perge C, Dumont PJJ, Orgeas L, Fardin MA, Manneville S, Belgacem MN (2015) Heterogeneous flow kinematics of cellulose nanofibril suspensions under shear. Soft Matter 11:4742–4755. doi:10.1039/C5SM00530B
Metzner AB, Reed JC (1955) Flow of non-Newtonian fluids—correlation of the laminar, transition, and turbulent-flow regions. AIChE J 1:434–440. doi:10.1002/aic.690010409
Missoum K, Belgacem NM, Bras J (2013) Nanofibrillated cellulose surface modification: a review. Materials 6:1745–1766. doi:10.3390/ma6051745
Moberg T, Sahlin K, Yao K, Geng S, Westman G, Zhou Q, Oksman K, Rigdahl M (2017) Rheological properties of nanocellulose suspensions: effects of fibril/particle dimensions and surface characteristics. Cellulose 24:2499–2510. doi:10.1007/s10570-017-1283-0
Moger J, Matcher SJ, Winlove CP, Shore A (2004) Measuring red blood cell flow dynamics in a glass capillary using Doppler optical coherence tomography and Doppler amplitude optical coherence tomography. J Biomed Optics 9:982–994. doi:10.1117/1.1781163
Mykhaylyk OO, Warren NJ, Parnell AJ, Pfeifer G, Laeuger J (2016) Applications of shear-induced polarized light imaging (SIPLI) technique for mechano-optical rheology of polymers and soft matter materials. J Polym Sci, Part B: Polym Phys 54:2151–2170. doi:10.1002/polb.24111
Naderi A, Lindström T (2015) Rheological measurements on nanofibrillated cellulose systems: a science in progress. In: Mondal MIH (ed) Cellulose and cellulose derivatives: synthesis, modification and applications. Nova Science Publishers, New York
Nazari B, Kumar V, Bousfield D, Toivakka M (2016) Rheology of cellulose nanofibers suspensions: boundary driven flow. J Rheol 60:1151–1159. doi:10.1122/1.4960336
Nechyporchuk O, Belgacem MN, Pignon F (2014) Rheological properties of micro-/nanofibrillated cellulose suspensions: wall-slip and shear banding phenomena. Carbohydr Polym 112:432–439. doi:10.1016/j.carbpol.2014.05.092
Olmsted PD (2008) Perspectives on shear banding in complex fluids. Rheol Acta 47:283–300. doi:10.1007/s00397-008-0260-9
Osong SH, Norgren S, Engstrand P (2016) Processing of wood-based microfibrillated cellulose and nanofibrillated cellulose, and applications relating to papermaking: a review. Cellulose 23:93–123. doi:10.1007/s10570-015-0798-5
Pääkkö M, Ankerfors M, Kosonen H, Nykänen A, Ahola S, Österberg M, Ruokolainen J, Laine J, Larsson PT, Ikkala O, Lindström T (2007) Enzymatic hydrolysis combined with mechanical shearing and high-pressure homogenization for nanoscale cellulose fibrils and strong gels. Biomacromolecules 8:1934–1941. doi:10.1021/bm061215p
Petrich MP, Koch DL, Cohen C (2000) An experimental determination of the stress–microstructure relationship in semi-concentrated fiber suspensions. J Non Newtonian Fluid Mech 95:101–133. doi:10.1016/S0377-0257(00)00172-5
Raiskinmäki P, Kataja M (2005) Disruptive shear stress measurements of fibre suspension using ultrasound Doppler techniques. Ann Trans The Nordic Rheol Soc 13:207–211
Robles FE, Wilson C, Grant G, Wax A (2011) Molecular imaging true-colour spectroscopic optical coherence tomography. Nat Photon, 5:744–747. doi: http://www.nature.com/nphoton/journal/v5/n12/abs/nphoton.2011.257.html#supplementary-information
Saarinen T, Haavisto S, Sorvari A, Salmela J, Seppälä J (2014) The effect of wall depletion on the rheology of microfibrillated cellulose water suspensions by optical coherence tomography. Cellulose 21:1261–1275. doi:10.1007/s10570-014-0187-5
Salmela J, Haavisto S, Koponen A, Jäsberg A, Kataja M (2013) Rheological characterization of micro-fibrillated cellulose fibre suspension using multi scale velocity profile measurements. In: Proceedings of 15th fundamental research symphosium
Shao Y, Chaussy D, Grosseau P, Beneventi D (2015) Use of microfibrillated cellulose/lignosulfonate blends as carbon precursors: impact of hydrogel rheology on 3D printing. Ind Eng Chem Res 54:10575–10582. doi:10.1021/acs.iecr.5b02763
Siró I, Plackett D (2010) Microfibrillated cellulose and new nanocomposite materials: a review. Cellulose 17:459–494. doi:10.1007/s10570-010-9405-y
Sorvari A, Saarinen T, Haavisto S, Salmela J, Vuoriluoto M, Seppälä J (2014) Modifying the flocculation of microfibrillated cellulose suspensions by soluble polysaccharides under conditions unfavorable to adsorption. Carbohydr Polym 106:283–292. doi:10.1016/j.carbpol.2014.02.032
Swerin A, Ödberg L, Lindström T (1990) Deswelling of hardwood kraft pulp fibers by cationic polymers. Nord Pulp Pap Res J 5:188–196. doi:10.3183/NPPRJ-1990-05-04-p188-196
Vesterinen A, Myllytie P, Laine J, Seppälä J (2010) The effect of water-soluble polymers on rheology of microfibrillar cellulose suspension and dynamic mechanical properties of paper sheet. J Appl Polym Sci 116:2990–2997. doi:10.1002/app.31832
Wang RK (2004) High-resolution visualization of fluid dynamics with Doppler optical coherence tomography. Meas Sci Technol 15:725–733. doi:10.1088/0957-0233/15/4/016
Wang XJ, Milner TE, Nelson JS (1995) Characterization of fluid flow velocity by optical Doppler tomography. Opt Lett 20:1337–1339. doi:10.1364/OL.20.001337
Wang L, Wang Y, Guo S, Zhang J, Bachman M, Li GP, Chen Z (2004a) Frequency domain phase-resolved optical Doppler and Doppler variance tomography. Opt Commun 242:345–350. doi:10.1016/j.optcom.2004.08.035
Wang L, Xu W, Bachman M, Li G, Chen Z (2004b) Phase-resolved optical Doppler tomography for imaging flow dynamics in microfluidic channels. Appl Phys Lett 85:1855–1857. doi:10.1063/1.1785854
Wiklund JA, Stading M, Pettersson AJ, Rasmuson A (2006) A comparative study of UVP and LDA techniques for pulp suspensions in pipe flow. AIChE J 52:484–495. doi:10.1002/aic.10653
Yadav R, Lee K, Rolland JP, Zavislan JM, Aquavella JV, Yoon G (2011) Micrometer axial resolution OCT for corneal imaging. Biomed Opt Express 2:3037–3046. doi:10.1364/BOE.2.003037
Yang VXD, Gordon ML, Seng-Yue E, Lo S, Qi B, Pekar J, Mok A, Wilson BC, Vitkin IA (2003) High speed, wide velocity dynamic range Doppler optical coherence tomography (Part II): imaging in vivo cardiac dynamics of Xenopus laevis. Opt Express 11:1650–1658. doi:10.1364/OE.11.001650
Zhao Y, Chen Z, Saxer C, Xiang S, de Boer JF, Nelson JS (2000) Phase-resolved optical coherence tomography and optical Doppler tomography for imaging blood flow in human skin with fast scanning speed and high velocity sensitivity. Opt Lett 25:114–116. doi:10.1364/OL.25.000114
Acknowledgments
Academy of Finland (project Rheological Properties of Complex Fluids) is gratefully acknowledged for supporting this work. We also want to thank senior research technician Ulla Salonen for the photograph of the MFC fibers (Fig. 1), and senior scientist Panu Lahtinen for preparing the MFC.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lauri, J., Koponen, A., Haavisto, S. et al. Analysis of rheology and wall depletion of microfibrillated cellulose suspension using optical coherence tomography. Cellulose 24, 4715–4728 (2017). https://doi.org/10.1007/s10570-017-1493-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-017-1493-5