Abstract
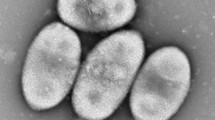
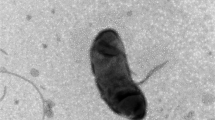
A novel alkaliphilic actinomycete, strain NCL716T, was isolated from a soil sample collected from the vicinity of Lonar Lake, an alkaline salt water meteorite lake in Buldhana district of Maharashtra State in India. The strain was characterised using a polyphasic taxonomic approach which confirmed that it belongs to the genus Streptomyces. Growth was observed over a pH range of 7–11 at 28 °C. The cell wall was found to contain ll-diaminopimelic acid and traces of meso-diaminopimelic acid. The major fatty acid components were identified as iso-C16:0 (46.8 %), C17:1 (12.4 %), anteiso-C15:0 (5.1 %) and anteiso-C17:1 (4.8 %). The major polar lipids were identified as diphosphatidylglycerol, phosphatidylethanolamine, phosphatidylglycerol and phosphatidylinositol. The major menaquinones were determined to be MK-9 (H6) (70.3 %), MK-9 (H4) (15.5 %) and MK-9 (H8) (7.2 %). The G+C content of the DNA of the type strain was determined to be 71.4 mol %. The 16S rRNA gene sequence has been deposited in GenBank with accession number FJ919811. Although the 16S rRNA gene sequence analysis revealed that strain NCL716T shares >99 % similarity with that of Streptomyces bohaiensis strain 11A07T, DNA–DNA hybridization revealed only 33.2 ± 3.0 % relatedness between them. Moreover, these two strains can be readily distinguished by some distinct phenotypic characteristics. Hence, on the basis of phenotypic and genetic analyses, it is proposed that strain NCL716T represents a novel species of the genus Streptomyces, for which the name Streptomyces lonarensis sp. nov., is proposed. The type strain is NCL 716T (=DSM 42084T = MTCC 11708T = KCTC 39684T).


Similar content being viewed by others
References
Acharyabhatta A, Kandula SK, Terli R (2013) Taxonomy and polyphasic characterization of alkaline amylase producing marine actinomycete Streptomyces rochei BTSS 1001. Int J Microbiol 276921(2013):1–8
Akhwale JK, Markus Goker M, Rohde M, Sproer C, Schumann P, Klenk HP, Boga HI (2015) Streptomyces alkaliphilus sp. nov., isolated from sediments of Lake Elmenteita in the Kenyan Rift Valley. Antonie Leeuwenhoek 107:1249–1259
Antony CP, Kumaresan D, Hunger S, Drake HL, Murrell JC, Shouche YS (2013) Microbiology of Lonar Lake and other soda lakes. ISME J 7:468–476
Antony-Babu S, Goodfellow M (2008) Biosystematics of alkaliphilic streptomycetes isolated from seven locations across a beach and dune sand system. Antonie Leeuwenhoek 94:581–591
Antony-Babu S, Okorafor LA, Stach JEM, Goodfellow M (2007) Alkaliphilic streptomycetes: prospective new candidates for bioprospecting. In: Proceedings of the 14th international symposium on the biology of actinomycetes, University of Newcastle, p 110
Bahn YS, Park JM, Bai DH, Takase S, Yu JH (1998) YUA001, a novel aldose reductase inhibitor isolated from alkalophilic Corynebacterium sp. YUA25. I. Taxonomy, fermentation, isolation and characterization. J Antibiot (Tokyo) 51:902–907
Becker B, Lechevalier MP, Lechevalier HA (1965) Chemical composition of cell-wall preparation from strains of various form-genera of aerobic actinomycetes. Appl Microbiol 13:236–243
Berdy J (2005) Bioactive microbial metabolites. J Antibiot (Tokyo) 58:1–26
Cha J, Kim YB, Seo BC, Park KH (1994) Characterisation of Streptomyces sp. KSM-35 and purification of its maltotetraose forming amylase. Korean J Food Sci Technol 26:633–637
Chakraborty S, Khopade A, Kokare C, Mahadik K, Chopade B (2009) Isolation and characterization of novel α-amylase from marine Streptomyces sp. D1. J Mol Catal B 58:17–23
Chakraborty S, Khopade RGA, Mahadik K, Kokare C (2012) Study on calcium ion independent -amylase from haloalkaliphilic marine Streptomyces strain A3”. Indian J Biotechnol 11:427–437
Chitte RR, Nalawade VK, Dey S (1999) Keratinolytic activity from the broth of a feather degrading S. thermoviolaceus strain SD8. Lett Appl Microbiol 28:131–136
Dastager SG, Li W-J, Agasar D, Sulochana MB, Tang S-K, Tian X-P, Zhi X-Y (2007) Streptomyces gulbargensis sp. nov., isolated from soil in Karnataka, India. Antonie Leeuwenhoek 91:99–104
Dastager SG, Kim C-J, Lee J-C, Agasar D, Park D-J, Li W-J (2008) Streptomyces deccanensis sp. nov., an alkaliphilic species isolated from soil. Int J Syst Evol Microbiol 58:1089–1093
Dastager SG, Agasar D, Pandey A (2009) Production and partial purification of alpha-amylase from a novel isolate Streptomyces gulbargensis. Ind J Microbiol Biotechnol 36:189–194
Deshmukh KB, Pathak AP, Karuppayil MS (2011) Bacterial diversity of Lonar soda lake of India. Ind J Microbiol 51(1):107–111
Ezaki T, Hashimoto Y, Yabuuchi E (1989) Fluorometric deoxyribonucleic acid-deoxyribonucleic acid hybridization in micro-dilution wells as an alternative to membrane filter hybridization in which radioisotopes are used to determine genetic relatedness among bacterial strains. Int J Syst Bacteriol 39:224–229
Felsenstein J (1985) Confidence limits on phylogenies: an approach using the bootstrap. Evolution 39:783–791
Goodfellow M (2012) Book Chapter : Phylum XXVI. Actinobacteria phy.nov. In : Goodfellow M, Kampfer P, Buss H-J, Trujillo ME, Suzuki K, Ludwig W and Whitman WB (eds.), Bergey’s manual of systematic bacteriology, vol. 5, The Actinobacteria, Part A and B. Springer, New York, pp 33–2028
Horikoshi K (1999) Alkaliphiles: some applications of their products for biotechnology. Microbiol Mol Biol Rev 63:735–750
Horikoshi K, Akiba T (1982) Alkalophilic microorganisms. Springer, Heidelberg
Hozzein WN, Ali MIA, Hammouda O, Mousa AS, Goodfellow M (2011) Streptomyces sannurensis sp. nov., a novel alkaliphilic streptomycete isolated from Wadi Sannur in Egypt. Afr J Microbiol Res 5(11):1329–1334
Joshi AA, Kanekar PP, Kelkar AS, Shouche YS, Vani AA, Borgave SB, Sarnaik SS (2008) Cultivable bacterial diversity of alkaline Lonar Lake, India. Microb Ecol 55:163–172
Kampfer P (2012).Genus streptomyces waksman and Henrici 1943, p 339
Kelly KL (1964) Inter-society color council-national bureau of standard color-name charts illustrated with centroid colors. US Government Printing Office, Washington
Kim OS, Cho YJ, Lee K, Yoon SH, Kim M, Na H, Park SC, Jeon YS, Lee JH, Yi H, Won S, Chun J (2012) Introducing EzTaxon: a prokaryotic 16S rRNA Gene sequence database with phylotypes that represent uncultured species. Int J Syst Evol Microbiol 62:716–721
Kloepper JW, Rodriguez KR, Mclnroy JA, Young RW (1992) Rhizosphere bacteria antagonistic to soybean cyst (Heterodera glycines) and root-knot (Meloidogyne incognita) nematodes: identification by fatty acid analysis and frequency of biological control activity. Plant Soil 139:75–84
Kroppenstedt RM (1982) Separation of bacterial menaquinones by HPLC using reverse phase (RP-18) and a silver-loaded ion exchanger. J Liq Chromatogr 5:2359–2367
Labeda DP, Goodfellow M, Brown R, Ward AC, Lanoot B, Vanncanneyt M, Swings J, Kim S-B, Liu Z et al (2012) Phylogenetic study of the species within the family Streptomycetaceae. Antonie Leeuwenhoek 101:73–104
Lechevalier MP, Lechevalier H (1970) Chemical composition as a criterion in the classification of aerobic actinomycetes. Int J Syst Bacteriol 20:435–443
Li WJ, Zhang YG, Zhang YQ, Tang SK, Xu P, Xu LH, Jiang CL (2005) Streptomyces sodiiphilus sp.nov., a novel alkaliphilic actinomycete. Int J Syst Evol Microbiol 55(3):1329–1333
Manfio GP, Zakrzewska-Czerwinska J, Atalan E, Goodfellow M (1995) Towards minimal standards for description of Streptomyces species. Biotechnologia 7:242–283
Mao J, Tang Q, Zhang Z, Wang W, Wei D, Huang Y et al (2007) Streptomyces radiopugnans sp. nov., a radiationresistant actinomycete isolated from radiation-polluted soil in China. Int J Syst Evol Microbiol 57:2578–2582
Marmur J (1961) A procedure for the isolation of deoxyribonucleic acid from micro-organisms. J Mol Biol 3(2):208–218
Meier-Kolthoff JP, Goker M, Sproer C, Klenk HP (2013) When should a DDH experiment be mandatory in microbial taxonomy? Arch Microbiol 195:413–418
Mesbah M, Premachandran U, Whitman WB (1989) Precise measurement of the G+C content of deoxyribonucleic acid by high-performance liquid chromatography. Int J Syst Bacteriol 39:159–167
Mikami Y, Miyashita K, Arai T (1982) Diaminopimelic acid profiles of alkalophilic and alkaline-resistant strains of actinomycetes. J Gen Microbiol 128:1709–1712
Miller L, Berger T (1985) Bacteria identification by gas chromatography of whole cell fatty acids. Hewlett-Packard application note. Hewlett-Packard Co., Avondale, pp 228–241
Minnikin DE, O’Donnell AG, Goodfellow M, Alderson G, Athaly M, Schaal KP, Parlett JH (1984) An integratedprocedure for the extraction of isoprenoid quinones and polar lipids. J Microbiol Methods 2:233–241
Moriyama Y, Mitsuo S, Morimatsu H, Shimizu Y, Sugiyama A, Kamiyama A (2000) Jpn. Kokai Tokkyo Koho JP 2000060546 A2 29 Feb 2000 (Japan)
Nakai R, Sato T, Okamoto K (1986). Japanese Kokai Koho patent 86, p 209, 588
Nei M, Kumar S (2000) Molecular evolution and phylogenetics. Oxford University Press, New York
Pan H-Q, Cheng J, Zhang D-F, Yu S-Y, Khieu T-N, Chu Ky Son C-K, Jiang Z, Hu J-C, Li W-J (2015) Streptomyces bohaiensis sp. nov., a novel actinomycete isolated from Scomberomorus niphonius in the Bohai Sea. J Antibiot 68:246–252
Patke D, Dey S (1998) Proteolytic activity from a thermophilic Streptomyces megaspores strain SDP4. Lett Appl Microbiol 26:171–174
Primarini D, Ohta Y (2000) Some enzyme properties of raw starch digesting amylases from Streptomyces sp. No. 4. Starch-Starke 52(1):28–32
Saitou N, Nei M (1987) The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evol 4:406–425
Sasser M (1990) Identification of bacteria by gas chromatography of cellular fatty acids, MIDI technical note 101.MIDI Inc, Newark
Sharma TK, Bhadane VA, Kumar LS, Rele MV, Bhawar G, Rahman I (2013) Optimization of the production of a maltooligosaccharides producing amylase from the alkalophilic Streptomyces lonarensis strain NCL 716 using SVR Modeling. Starch/Starke 65:179–185
Shirling EB, Gottlieb D (1966) Methods for characterization of Streptomyces species. Int J Syst Bacteriol 16:313–340
Srinivasan MC, Chidambaram P, Mathur SB, Neergaard P (1971) A simple method for inducing sporulation in seed-borne fungi. Trans Brit Mycol Soc 56:31–36
Stackebrandt E, Ebers J (2006) Taxonomic parameters revisited: tarnished gold standards. Microbiol Today 33:152–155
Sultanpuram VR, Mothe T (2015) Mohammed F (2015), Streptomyces alkalithermotolerans sp. nov., a novel alkaliphilic and thermotolerant actinomycete isolated from a soda lake. Antonie Leeuwenhoek 107:337–344
Tambekar D, Dhundale VR (2012) Studies on the physiological and cultural diversity of Bacilli characterized from Lonar Lake (MS) India. Biosci Discov 3(1):34–39
Tamura K, Nei M, Kumar S (2004) Prospects for inferring very large phylogenies by using the neighbor-joining method. Proc Natl Acad Sci USA 101:11030–11035
Tamura K, Peterson D, Peterson N, Stecher G, Nei M, Kumar S (2011) MEGA5: molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol Biol Evol 28:2731–2739
Thakker CD, Ranade DR (2002) Alkalophilic Methanosarcina isolated from Lonar Lake. Curr Sci 82:455–458
Touche L, Christie T (1912) The geology of Lonar Lake—with a note on the Lonar soda deposit. Rec Geol Surv India 41:266–285
Tsujibo H, Sato T, Inui M, Yamamoto H, Inamori Y (1988) Intracellular accumulation of phenazine antibiotics produced by an alkalophilic actinomycete. I. Taxonomy, isolation and identificationof the phenazine antibiotics. Agric Biol Chem 52:301–306
Tsujibo H, Sakamoto T, Miyamoto K, Kusano G, Ogura M, Hasegawa T, Inamori Y (1990) Isolation of cytotoxic substance, kalafungin from an alkalophilic actinomycete, Nocardiopsisdassonvillei subsp. prasina. Chem Pharm Bull (Tokyo) 38:2299–2300
Vukelic B, Ritonja A, Renko M, Pokornv M, Vitale L (1992) Extracellular alpha-amylase from Streptomyces rimosus. Appl Microbiol Biotechnol 37(2):202–204
Waksman SA, Henrici A (1943) The nomenclature and classification of the actinomycetes. J Bacteriol 46:337–341
Wani AA, Surakasi VP, Siddharth J, Raghavan RG, Patole MS, Ranade D, Shouche YS (2006) Molecular analyses of microbial diversity associated with the Lonar soda lake in India: an impact crater in a basalt area. Res Microbiol 157:928–937
Wellington EMH, Stackebrandt E, Sanders D, Wolstrup J, Jorgensen NOG (1992) Taxonomic status of Kitasatosporia, and proposed unification with. Streptomyces on the basis of phenotypic and 16srRNA analysis and emendation of Streptomyces Waksman and Henrici 1943, 339AL. Int J Syst Bacteriol 42(1):156–160
Williams ST, Goodfellow M, Alderson G, Wellington EMH, Sneath PHA, Sackin MJ (1983) Classification of Streptomyces and Related Genera. J Gen Microbiol 129(1743–1):813
Williams ST, Goodfellow M, Alderson G (1989) Genus Streptomyces waksman and henrici, 1943, 339AL. In: Williams ST, Sharpe ME, Holt JG (eds) Bergey’s manual of systematic bacteriology. Williams and Wilkins, Baltimore, pp 2452–2492
Zhang Y-G, Wang H-F, Liu Q, Hozzein WN, Wadaan MAM, Cheng J, Chen Y-J, Zhang Y-M, Li W-J (2013) Streptomyces fukangensis sp. nov., a novel alkaliphilic actinomycete isolated from a saline-alkaline soil. Antonie Leeuwenhoek 104:1227–1233
Acknowledgments
Trupti K. Sharma acknowledges the Council of Scientific and Industrial Research (CSIR), India, for the Senior Research Fellowship.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sharma, T.K., Mawlankar, R., Sonalkar, V.V. et al. Streptomyces lonarensis sp. nov., isolated from Lonar Lake, a meteorite salt water lake in India. Antonie van Leeuwenhoek 109, 225–235 (2016). https://doi.org/10.1007/s10482-015-0626-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10482-015-0626-9