Abstract
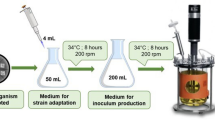
Dihydroxylation of dehydroepiandrosterone (DHEA) is an essential step in the synthesis of many important pharmaceutical intermediates. However, the solution to the problem of low biohydroxylation conversion in the biotransformation of DHEA has yet to be found. The effects of natural oils on the course of dihydroxylation of DHEA to 3β,7α,15α-trihydroxy-5-androsten-17-one (7α,15α-diOH-DHEA) were studied. With rapeseed oil (2 %, v/v) addition, the bioconversion efficiency was improved, and the 7α,15α-diOH-DHEA yield was increased by 40.8 % compared with that of the control at DHEA concentration of 8.0 g/L. Meantime, the ratio of 7α,15α-diOH-DHEA to 7α-OH-DHEA was also increased by 4.5 times in the rapeseed oil-containing system. To explain the mechanism underlying the increase of 7α,15α-diOH-DHEA yield, the effects of rapeseed oil on the pH of the bioconversion system, the cell growth and integrity of Gibberella intermedia CA3-1, as well as the membrane composition were systematically studied. The addition of rapeseed oil enhanced the substrate dispersion and maintained the pH of the system during bioconversion. Cells grew better with favorable integrity. The fatty acid profile of G. intermedia cells revealed that rapeseed oil changed the cell membrane composition and improved cell membrane permeability for lipophilic substrates.






Similar content being viewed by others
References
Angelova B, Schmauder HP (1999) Lipophilic compounds in biotechnology-interactions with cells and technological problems. J Biotechnol 67:13–32
Avramova T, Spassova D, Mutafov S, Momchilova S, Boyadjieva L, Damyanova B, Angelova B (2010) Effect of Tween 80 on 9α-steroid hydroxylating activity and ultrastructural characteristics of Rhodococcus sp. cells. World J Microbiol Biotechnol 26:1009–1014
Baumann MD, Daugulis AJ, Jessop PG (2005) Phosphonium ionic liquids for degradation of phenol in a two-phase partitioning bioreactor. Appl Microbiol Biotechnol 67:131–137
Cantone S, Hanefeld U, Basso A (2007) Biocatalysis in non-conventional media-ionic liquids, supercritical fluids and the gas phase. Green Chem 9:954–971
Choudhary MI, Sultan S, Tareq M, Khan H, Atta-ur-Rahman (2005) Microbial transformation of 17α-ethynyl- and 17α-ethylsteroids, and tyrosinase inhibitory activity of transformed products. Steroids 70:798–802
Davies JL, Baganz F, Ison AP, Lye GJ (2000) Studies on the interaction of fermentation and microfilteration operations: erythromycin recovery from Saccharopolyspora erythraea fermentation broths. Biotechnol Bioeng 69:429–439
Donova MV, Nikolayeva VM, Dovbnya DV, Gulevskaya SA, Suzina NE (2007) Methyl-β-cyclodextrin alters growth, activity and cell envelope features of sterol-transforming mycobacteria. Microbiology 153:1981–1992
El Refai HA, Abd-elslam IS (2010) Enhancement of β-sitosterol bioconversion by Fusarium solani using aqueous-organic solvent system. Aust J Basic Appl Sci 4:4107–4112
Gulla V, Banerjee T, Patil S (2010) Bioconversion of soysterols to androstenedione by Mycobacterium fortuitum subsp. fortuitum NCIM 5239, a mutant derived from total sterol degrader strain. J Chem Tech Biotechnol 85:1135–1141
Hamedi J, Malekzadeh F, Niknam V (2002) Improved production of erythromycin by Saccharopolyspora erythraea by various plant oils. Biotechnol Lett 24:697–700
Heipieper HJ, Neumann G, Cornelissen S, Meinhardt F (2007) Solvent-tolerant bacteria for biotransformations in two-phase fermentation systems. Appl Microbiol Biotechnol 74:961–973
Holland HL (1982) The mechanisms of the microbial hydroxylation of steroids. Chem Soc Rev 2:371
Huang LH, Li J, Xu G, Zhang XH, Wang YG, Yin YL, Liu HM (2010) Biotransformation of dehydroepiandrosterone (DHEA) with Penicillium griseopurpureum Smith and Penicillium glabrum (Wehmer)Westling. Steroids 75:1039–1046
Janeczko T, Dmochowska-Gładysz J, Kostrzewa-Susłow E, Białonska E, Ciunik Z (2009) Biotransformations of steroid compounds by Chaetomium sp. KCH 6651. Steroids 74:657–661
Kim PY, Pollard DJ, Woodley JM (2007) Substrate supply for effective biocatalysis. Biotechnol Prog 23:74–82
Kristana K, Rizner TL (2012) Steroid-transforming enzymes in fungi. J Steroid Biochem Mol Biol 129:79–91
Li H, Xu ZH, Fu ZZ, Shi JS, Zhang DD (2013) Synthesis of 3β,7α,15α-trihydroxy-5-androsten-17-one by Gibberella intermedia CA3-1. CHN Patent 2013/102965288A
Lobastova TG, Gulevskaya SA, Sukhodolskaya GV, Donova MV (2009) Dihydroxylation of dehydroepiandrosterone in positions 7α and 15α by mycelial fungi. Appl Biochem Microbiol 45:617–622
Lopez S, Bermudez B, Montserrat-de la Paz S, Jaramillo S, Varela LM, Ortega-Gomez A, Abia R, Muriana FJ (2014) Membrane composition and dynamics: A target of bioactive virgin olive oil constituents. Biochim Biophys Acta 1838:1638–1656
Lu WY, Du LX, Wang M, Wen JP, Sun B, Guo YW (2006) Effect of two-steps substrate addition on steroids 11α-hydroxylation by Curvularia lunata CL-114. Biochem Eng J 32:233–238
Machaia KM, Ziolkowski A, Galewicz AR, Lisowska K, Sedlaczek L (2001) Polycations increase the permeability of Mycobacterium vaccae cell envelopes to hydrophobic compounds. Microbiology 147:2769–2781
Malaviya A, Gomes J (2008) Enhanced biotransformation of sitosterol to androstenedione by Mycobacterium sp. using cell wall permeabilizing antibiotics. J Ind Microbiol Biotechnol 35:1235–1239
Peart PC, McCook KP, Russell FA, Reynolds WF, Reese PB (2011) Hydroxylation of steroids by Fusarium oxysporum, Exophiala jeanselmei and Ceratocystis paradoxa. Steroids 76:1317–1330
Phase N, Patil S (1994) Natural oils are better than organic solvents for the conversion of soybean sterols to 17-ketosteroids by Mycobacterium fortuitum. World J Microbiol Biotechnol 10:228–229
Quijano G, Revah S, Gutierrez-Rojas M, Flores-Cotera LB, Thalasso F (2009) Oxygen transfer in three-phase airlift and stirred tank reactors using silicone oil as transfer vector. Process Biochem 44:619–624
Romano A, Romano D, Ragg E, Costantino F, Lenna R, Gandolfi R, Molinari F (2006) Steroid hydroxylations with Botryodiplodia malorum and Colletotrichum lini. Steroids 71:429–434
Rumijowska-Galewicz A, Ziółkowski A, Korycka-Machała M, Sedlaczek L (2000) Alteration in lipid composition of Mycobacterium vaccae cell wall outer layer enhance β-sitosterol degradation. World J Microbiol Biotechnol 16:237–244
Shen YB, Wang M, Zhang LT, Ma YH, Ma B, Zheng Y, Liu H, Luo JM (2011) Effects of hydroxypropyl-β-cyclodextrin on cell growth, activity, and integrity of steroid-transforming Arthrobacter simplex and Mycobacterium sp. Appl Microbiol Biotechnol 90:1995–2003
Sharma P, Slathia PS, Somal P, Mehta P (2012) Biotransformation of cholesterol to 1,4-androstadiene-3,17-dione (ADD) by Nocardia species. Ann Microbial 62:1651–1659
Tong WY, Dong X (2009) Microbial biotransformation: recent developments on steroid drugs. Recent Patents Biotechnol 3:141–153
Woodley JM (2008) New opportunities for biocatalysis: making pharmaceutical processes greener. Trends Biotechnol 26:321–327
Wu DX, Guan YX, Wang HQ, Yao SJ (2011) 11α-Hydroxylation of 16α,17-epoxyprogesterone by Rhizopus nigricans in a biphasic ionic liquid aqueous system. Bioresour Technol 102:9368–9373
You KM, Rosenfield CL, Knipple DC (2003) Ethanol tolerance in the yeast Saccharomyces cerevisiae is dependent on cellular oleic acid content. Appl Environ Microbiol 69:1499–1503
Acknowledgments
This work is financially supported by the National Natural Science Foundation of China (No. 21206055), the National High-Tech R&D Program (No. 2011AA02A211) and the Natural Science Foundation of Jiangsu Province (No. BK2012117, No. BK2012127).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, H., Fu, Z., Li, H. et al. Enhanced biotransformation of dehydroepiandrosterone to 3β,7α,15α-trihydroxy-5-androsten-17-one with Gibberella intermedia CA3-1 by natural oils addition. J Ind Microbiol Biotechnol 41, 1497–1504 (2014). https://doi.org/10.1007/s10295-014-1498-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10295-014-1498-0