Abstract
The world is experiencing an energy crisis and environmental issues due to the depletion of fossil fuels and the continuous increase in carbon dioxide concentrations. Microalgal biofuels are produced using sunlight, water, and simple salt minerals. Their high growth rate, photosynthesis, and carbon dioxide sequestration capacity make them one of the most important biorefinery platforms. Furthermore, microalgae's ability to alter their metabolism in response to environmental stresses to produce relatively high levels of high-value compounds makes them a promising alternative to fossil fuels. As a result, microalgae can significantly contribute to long-term solutions to critical global issues such as the energy crisis and climate change. The environmental benefits of algal biofuel have been demonstrated by significant reductions in carbon dioxide, nitrogen oxide, and sulfur oxide emissions. Microalgae-derived biomass has the potential to generate a wide range of commercially important high-value compounds, novel materials, and feedstock for a variety of industries, including cosmetics, food, and feed. This review evaluates the potential of using microalgal biomass to produce a variety of bioenergy carriers, including biodiesel from stored lipids, alcohols from reserved carbohydrate fermentation, and hydrogen, syngas, methane, biochar and bio-oils via anaerobic digestion, pyrolysis, and gasification. Furthermore, the potential use of microalgal biomass in carbon sequestration routes as an atmospheric carbon removal approach is being evaluated. The cost of algal biofuel production is primarily determined by culturing (77%), harvesting (12%), and lipid extraction (7.9%). As a result, the choice of microalgal species and cultivation mode (autotrophic, heterotrophic, and mixotrophic) are important factors in controlling biomass and bioenergy production, as well as fuel properties. The simultaneous production of microalgal biomass in agricultural, municipal, or industrial wastewater is a low-cost option that could significantly reduce economic and environmental costs while also providing a valuable remediation service. Microalgae have also been proposed as a viable candidate for carbon dioxide capture from the atmosphere or an industrial point source. Microalgae can sequester 1.3 kg of carbon dioxide to produce 1 kg of biomass. Using potent microalgal strains in efficient design bioreactors for carbon dioxide sequestration is thus a challenge. Microalgae can theoretically use up to 9% of light energy to capture and convert 513 tons of carbon dioxide into 280 tons of dry biomass per hectare per year in open and closed cultures. Using an integrated microalgal bio-refinery to recover high-value-added products could reduce waste and create efficient biomass processing into bioenergy. To design an efficient atmospheric carbon removal system, algal biomass cultivation should be coupled with thermochemical technologies, such as pyrolysis.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Developing alternative energy sources has become necessary due to the world's economy and population growth, as well as the resulting demand for oil and gas resources (Razeghifard 2013; Shuba and Kifle, 2018). This rising energy demand is related to the transportation sector, where the most demand is met by using fossil-based fuels (Asomaning et al. 2016); the transportation sector consumes approximately 28% of total global energy and is heavily reliant on fossil fuels. Nevertheless, 71% of crude oil contributes significantly to total greenhouse gas emissions (Leite et al. 2013). The use of fossil fuels has significantly contributed to air, water, and soil pollution, which has a negative impact on public health, an energy crisis caused by the irreversible depletion of global fossil-fuel reserves, and increased climate change (Fawzy et al. 2020). Biomass is the world's third-largest energy resource after coal and oil (Tumuluru et al. 2011; Osman et al. 2021a, b). Biomass for power generation is gaining popularity globally due to its long-term sustainability, potential, and environmental benefits. Furthermore, the produced biomass is almost carbon neutral and has the potential to significantly reduce net carbon emissions as well as hazardous emissions (Tumuluru et al. 2012); therefore, the need for sustainable and renewable energy sources has become an urgent demand (Silva et al. 2019).
Biofuels are typically derived from biomass and are composed of organic or biological components that can exist as solid, liquid, or vapors (Osman et al. 2021a). Over the last five decades, researchers have developed biomass as a feedstock for the first, second, third, and fourth generations of bio-based energy (Chowdhury et al. 2019). Third-generation biofuels, which are primarily based on microalgae, are thought to be a viable solution to the problems associated with first and second-generation biofuels. Algae (Fig. 1) are ubiquitous organisms in various habitats, but their commercialization as a substitute for fossil fuels is limited (Gajraj et al. 2018). Because of their rapid growth rate and high lipids, protein, minerals, and carbohydrate contents, microalgae are regarded as a viable feedstock for various bioproducts and bioenergy carriers (Hamed et al. 2020). On an industrial scale, the literature clearly demonstrates algae-based biofuels' positive economic and environmental impacts (Hamed et al. 2020a; Efroymson et al. 2021; El Shimi et al. 2018). After lipid extraction, the algal biomass residue can be converted into a variety of biofuels, including biomethane, bioethanol, and biohydrogen (Lam et al. 2019; Hamed et al. 2017).
Different types of algae cultivation, either in open or closed ponds. Algae growth is influenced by water supply and cultivation mode. Microalgae can be grown in two different ways. Ponds and lakes are examples of open systems, whereas photobioreactors are examples of closed systems. Every system has its benefits and drawbacks. Open farming (open pond) is regarded as the most basic and oldest method of producing and cultivating microalgae on a large scale
Microalgae are photosynthetic microorganisms that form the foundation of most aquatic food chains and contribute significantly to oxygen production (Chen et al. 2018). Numerous studies have examined how their mixotrophic nutrition on organic compounds during growth aided carbon transformation and storage (Hamed et al. 2021; Abomohra et al. 2014; Heredia-Arroyo et al. 2011). They have been shown to use nutrients from wastewater (Abou-Shanab et al. 2014; Dong et al. 2014; Pittman et al. 2011), carbon dioxide and exhaust gases emitted through industrial processes (Lizzul et al. 2014). As a result, producing microalgal biomass concurrently with existing industrial or municipal treatment activities could significantly reduce economic and environmental costs while also providing a valuable remediation service (Mishra et al. 2017; Karpagam et al. 2021; Rawat et al. 2011; Park et al. 2011).
Microalgae produce a variety of biotic substances with diverse applications in the chemical, food, pharmaceutical, carbon sequestration, and biofuels industries (Siddiki et al. 2022). Due to technical challenges, large-scale microalgae cultivation is limited, which is one of the major influences on its commercialization (Lam et al. 2019; Hamed et al. 2020). Therefore, this review aims to evaluate and critically describe the key factors that can be used to improve microalgae-based biofuels production by utilising potential microalgal species while highlighting related technologies and problematic issues in their production. Furthermore, evaluate the application of microalgae in atmospheric carbon removal as a carbon sequestration tool.
Applications
Biodiesel has recently received much attention as a renewable, biodegradable, and non-toxic fuel that emits fewer pollutants than regular diesel (Antolın et al. 2002; Lang et al. 2001; Vicente et al. 2004). Biodiesel has better chemical and physical properties than petro-diesel fuel, such as a higher cetane number, lower sulfur concentration, higher flash point, and better lubricating efficiency due to the contained oxygen (Goodrum et al. 2005; Anastopoulos et al. 2001; Nabi et al. 2006). Compared to diesel, direct use of biodiesel or biodiesel blends positively affects exhaust gases (Nabi et al. 2006, Knothe 2006, Schumacher et al. 1996). Biodiesel's relatively high oxygen content significantly reduced combustion gases and decreased carbon monoxide emissions.
Moreover, biodiesel is free of aromatic compounds and other chemical substances; thus, it has no negative environmental impact. In 2003, global biodiesel production was around 1.8 billion liters (Fulton 2004). In recent years, there has been a significant increase in biodiesel production. Biodiesel production is expected to increase in response to increased global demand for fuels and cleaner energy. Microalgae-derived biodiesel can completely replace petroleum; however, the cost of microalgal oil production must first fall from approximately $ 2.80/L to $ 0.48/L (Chisti 2007).
Compared to other feedstocks, microalgae-based biofuels are the most cost-effective; photoautotrophic microalgae, for example, convert sunlight into biomass more efficiently than higher plants (Demirbas et al. 2011). Whereas terrestrial plants have a photosynthetic efficiency of less than 4%, algae have a photosynthetic efficiency of 3–9% (Dismukes et al. 2008). The high growth rate of microalgae and consecutive biomass production reflect this light utilisation efficiency. Furthermore, algae are more tolerant to a wide range of light intensities than higher plants, allowing them to live autotrophically through photosynthesis. Meanwhile, some microalgal species can produce a relatively high content of energy-rich compounds by utilizing organic carbon sources such as glucose (Lee, 2001; Hamed and Klöck, 2014; Hassan et al. 2012).
Heterotrophs are algae that can grow in the absence of light energy but feed on organic carbon (Heredia-Arroyo et al. 2011; Joun et al. 2021; Feng et al. 2014; Shen et al. 2019). Heterotrophic microalgal growth is less efficient than phototrophic growth because the organic source required in heterotrophic growth is produced by another photosynthetic crop (Heredia-Arroyo et al. 2011; Patil et al. 2008). As a result, energy must first be used to grow the crop in the heterotrophic mode, whereas in the photoautotrophic mode, energy is used directly for algae growth.
Algae have adapted to live in various ecosystems ranging from hot springs to snow (Harwood et al. 2009). Some algal species live in terrestrial habitats, but the majority live in water bodies, including freshwater, brackish, marine, and hyper-saline waters (Hu et al. 2008). Algae are classified into nine main groups: green algae, cyanobacteria (blue-green microalgae), diatoms, yellow-green algae, golden algae, red algae, brown algae, dinoflagellates, and pico-plankton (Hu et al. 2008). Because algae are diverse and largely unexplored organisms, there is potential for further developments and applications. Their diverse genetic and biochemical composition could explain their ability to survive in many environments (Faramarzi et al. 2008).
To summarize, there is real potential for using algae in biodiesel production; however, the cost of microalgal oil production must first fall from around $ 2.80/L to $ 0.48/L. Algae's ability to live in various harsh conditions makes them ideal for various applications, with the potential to further explore undiscovered organisms within algae.
Algal biomass
Sources
Freshwater
Freshwater accounts for less than 3% of all water on the planet. Approximately 69% of the Earth's freshwater is inaccessible to humans, such as ice in glaciers and polar ice caps and groundwater. The world's available surface fresh water is not distributed evenly. The majority of the world's surface freshwater is located in Brazil, Russia, Canada, Indonesia, China, Columbia, and the United States. Lakes, rivers, wetlands, streams, and ponds are examples of freshwater habitats. Various types of algae and cyanobacteria can be found in low salt concentration ponds and freshwater lakes that can support diverse flora and fauna. For example, favorable conditions such as nutrient availability, adequate light, and temperature promote algal bloom biomass production (Gatamaneni et al. 2018). To reduce the negative environmental impact of algal blooms, researchers converted naturally occurring algal blooms into biofuel production (Kuo 2011; Ghosh et al. 2019). Streams and rivers have different environments, such as higher oxygen levels and faster flow. Because of agricultural runoff, streams have high nutrient content, such as nitrogen and phosphorus (Díez-Montero et al. 2020). Algae are usually abundant in the middle of the river due to the lower water flow in this section than in others. The river mouth is unsuitable for algal growth as the water becomes murky. Algal growth is also diminished due to limited light penetration and stagnant water conditions (Chew et al. 2018). Overall, using freshwater resources for algal biomass production is not economically feasible.
Saltwater
Saline or salt water constitutes the vast bulk of Earth's water. Saline water includes oceans, marginal seas, saline groundwater, and closed lakes. The availability of saltwater from the oceans introduces a low-cost option for microalgae growth and biomass production. The marine ecosystem is home to a diverse range of algae, including microalgae cyanobacteria as well as macroalgal species. Marine macroalgae are commonly known as seaweeds, which include three different classes red algae (Rhodophyta), brown (Phaeophyta) and green macroalgae (Chlorophyta). Marine macroalgae commonly occupy intertidal and sublittoral-to-littoral zones on rocks and other hard substrata. Marine algae are an important component of the marine ecosystem because they absorb sunlight energy, water and carbon dioxide to produce organic compounds and release oxygen to the ambient environment. This cycle contributes to the balance of the ocean's life cycle.
The unicellular microalgae, known as phytoplankton, constitute the base of the marine food chain. It is typically found near the water's surface, capturing sunlight. Marine phytoplankton plays a critical role in the biogeochemical cycling of the oceans. Their varied genome structure explains their adaptation to thrive in various conditions. Microalgae have become a dominant force within marine ecosystems. For example, coastal regions' microalgae are contended with high nutrients, low light and turbulence conditions, whereas open ocean microalgae are adapted with low nutrients and high irradiance. Polar microalgae are acclimatized to high nutrients with freezing temperatures and long periods of light and darkness.
Because salinity is an important factor in these environments, saltwater is a typically ideal condition for developing microalgae resistant species to high salinity. The average salinity of marine water is 35%, though this varies slightly according to the amount of runoff received from surrounding lands and rainfalls. Overall, marine water is an attractive resource for microalgae scales up, however cultivation in the marine environment requires careful monitoring to ensure optimal biomass productivity.
Wastewater
Wastewater is a rich nutrients source for microalgae growth. Phyco-remediation is a wastewater treatment method that employs microalgae for nutrient removal from wastewater, which can then be reused for multiple purposes (Lage et al. 2018). There are several types of wastewaters, including industrial, domestic, refinery, agricultural, and leachate. The organic composition of all wastewaters is comparable; however, the inorganic content varies among wastewater sources. Carbohydrates, proteins, lipids, amino acids, and volatile acids account for three-quarters of the organic carbon in sewage.
These wastewaters typically contain high nitrogen, phosphorus, and other inorganic compounds, which cause eutrophication to local water sources and impose ecological risk to aquatic life. Thus, employing wastewater for algal mass production could be a promising solution to replace synthetic culture media, which is currently prohibitively expensive to use on an algal scale. For instance, wastewaters of palm fertiliser, fruit bunches, and palm oil mill effluent are rich with high nitrogen and other nutrients content; they have been proposed as good culture media.
Although high nutrient levels in wastewater promote algal growth, the high levels of toxic compounds, emerging contaminants and pathogens can inhibit algal growth. Therefore, resistant microalgal species that adapt to the wastewater environment must be carefully selected for optimal growth and high biomass yields. For example, struvite is a wastewater nutritive medium that has been investigated as a microalgal supporting medium to reduce cultivation costs (Chew et al. 2018).
Types
Freshwater species
Freshwater microalgae could provide a promising feedstock for biofuels production due to their fast growth rates, high biomass yields, high carbon dioxide sequestration ability, and their strong potential to grow on marginal lands that do not compete with agricultural crops (Yun et al. 2014; Ramaraj et al. 2010; Saraf and Dutt, 2021; Al-Lwayzy et al. 2014). Every species has a distinct growth rate and innate metabolic profile. Consequently, selecting microalgal species with high biomass production and promising properties for large-scale production are important factors for sustainable biofuel technology.
Freshwater microalgae have been previously used for human and animal nutrition. These species can rapidly absorb nutrients from the liquid phase and thrive in the environment. Numerous studies showed the high ability of freshwater microalgae in biomass for bio-based energy production, such as Chlorella vulgaris (Al-Lwayzy et al. 2014), Chlorella pyrenoidosa (Yang et al. 2015), Muriellopsis sp. and Scenedesmus subpicatus (Gómez-Serrano et al. 2015), Ankistrodesmus falcatus (George et al. 2014), Coelastrella sp. (Narayanan et al. 2018), Asterarcys quadricellulare (Sangapillai and Marimuthu, 2019), Scenedesmus obliquus (Liu et al. 2013) and Tribonema sp. (Wang et al. 2014a).
Significant efforts have been made to use marine macroalgae and cyanobacteria for the biofuels industry; however, much less concern has been placed on using freshwater macroalgae (e.g. eukaryotic Chlorophyta) (Yun et al. 2014; Grayburn et al. 2013; Lawton et al. 2013; Demirbas 2010; Khola and Ghazala 2012). Nevertheless, freshwater macroalgae may have significant potential for liquid and solid biofuels that can be combusted directly or co-combusted with more traditional energy sources (Tumuluru et al. 2012; Grayburn et al. 2013). Moreover, biomass harvesting, represented as dense floating mats, is much easier and cheaper than dewatering equivalent biomass of suspended microalgae (Grayburn et al. 2013; Hillebrand 1983). For example, using an algal turf scrubber for large-scale freshwater macroalgae cultivation has been elucidated as a cost-effective and eco-friendly approach (Yun 2014). This technology combines nutrient removal in wastewater with bioenergy production. Several common freshwater macroalgal taxa, e.g., Oedogonium, Rhizoclonium, Ulothrix, and Microspora, have been reported (Adey et al. 2011, Kebede‐Westhead et al. 2003, Mulbry et al. 2008a, Pizarro et al. 2006).
Although the elemental biomass composition of common freshwater macroalgae has been analyzed, relatively few studies have evaluated the efficiency of freshwater macroalgae as a feedstock for biofuel (Tumuluru et al. 2012; Lawton et al. 2013). At the industrial scale, it is critical to investigate how variable environmental conditions such as temperature, salinity, light, and nutrient availability affect the growth of freshwater macroalgae, biomass productivity, and energy efficiency (Smith et al. 2010; Sturm et al. 2012; Shurin et al. 2013). In this context, nutrient limitation remarkably induced lipids accumulation in several eukaryotic algae; however, the yields and productivity of algal biomass were noticeably reduced (Shurin et al. 2013; Subramanian et al. 2013).
As a result, determining the net energy yield of freshwater macroalgal cultivation systems necessitates a thorough examination of biomass productivity and energy content. In this study, the exhaust emissions and fuel characteristics of the biodiesel produced from a mixture of freshwater macroalgae Cladophora and Rhizoclonium were nearly identical to petro-diesel (Grayburn et al. 2013). Furthermore, three common freshwater macroalgal species were found to have high bioenergy potential in terms of higher heating value and productivity (Lawton et al. 2013). Hence, more investigations on the effectiveness of freshwater macroalgae as a bio-based energy carrier are needed.
Marine or saltwater species
Marine microalgae are less diverse than freshwater microalgae due to their toxic and hazardous nature (Chew et al. 2018). Because of their large surface area, algae in seawater and oceans have evolved to outperform freshwater algae. The photoconversion rate of marine microalgae has increased to better use the abundant sunlight and synthesize biomass more quickly (Wei et al. 2013). Certain algal species can accelerate the production of valuable byproducts if exposed to harsh environmental conditions. The laboratory study of Rodolfi et al. (2009) on 21 different marine microalgal strains, e.g. Tetraselmis suecica, Phaeodactylum tricornutum, Chaetoceros calcitrans, Isochrysis galbana Nannochloropsis oculata, Pavlova lutheri, Skeletonema sp. revealed variable biomass productivities which ranged between 0.04 and 0.37 g/L/day and lipid productivity (17.4–61.0 mg/L/day) (Table 1). The marine diatom Chaetoceros muelleri exhibited moderate growth production (0.272 g/L/day) and high lipid productivity (51 mg/L/day) on synthetic saline water medium under phototrophic cultivation with high carbon dioxide aeration levels (10–20%) (Wang et al. 2014b) (Table 1). The marine microalgal strains Chlorella salina, Neochloris sp. and Nannochloropsis sp. were demonstrated for lipid-rich biomass production with lipid contents 28, 46 and 52% using synthetic medium and enriched seawater medium under phototrophic growth conditions (Surendhiran et al. 2014; Moazami et al. 2011). This implies the possibility of lowering the cost of biomass-derived biofuel production, which is currently not economically viable.
Wastewater species
The commercialization of algae-based biotechnology for the manufacturing of bio-based energy is restricted by the high production costs (Hamed et al. 2020). Thus, several ways have been proposed to make this technology economically feasible, such as using industrial effluent (Gómez-Serrano et al. 2015; Kim et al. 2015), municipal wastewater (Abou-Shanab et al. 2014; Dong et al. 2014; Sturm et al. 2012; Zhang et al. 2014; Han et al. 2016; Mahapatra et al. 2014), and dairy manure effluent (Kebede‐Westhead et al. 2003; Mulbry et al. 2008a; Pizarro et al. 2006; Wahal and Viamajala, 2016) as a culture media to reduce cultivation costs and freshwater requirements.
No specific species can grow in wastewater; instead, robust strains should be carefully chosen to survive under these harsh conditions (Lage et al. 2018). Oscillatoria, Euglena, Chlamydomonas, Chlorella, Scenedesmus, Navicula Nitzschia, and Stigeoclonium are the eighth-most tolerant genera (Randrianarison et al. 2017). Scenedesmus sp. (He et al. 2019) and Chlorella sp. (Makareviciene et al. 2011) are widespread in freshwater bodies of various types as primary producers cleansing the eutrophic waters (Abdel-Raouf et al. 2012).
Several laboratory studies on microalgae biomass production using wastewater have been investigated either in bioreactors, small semi-continuous culture systems, or batch cultures. Data in Table 1 showed that microalgae yielded differential lipid contents ranging from moderate such as Botryococcus braunii (17.85% dry wt) (Órpez et al. 2009), to high lipid content 23, 30 and 40% in Micractinium reisseri (Abou-Shanab et al. 2014), Chlorococcum sp. RAP13 (42%) (Ummalyma and Sukumaran, 2014), Chlorella sorokiniana (61.52%) (Ramanna et al. 2014). The study of Chinnasamy et al. (2010) revealed that Butyraceous Braunii, Chlorella saccharophila and Dunaliella tertiolecta grown in industrial wastewater (treated carpet mill) showed biomass productivity ranging from 0.016 to 0.038 g/L/day, and the estimated lipid productivity was ranged from 2.72 to 4.4 mg/L/day depending on species type. Implying that this kind of wastewater could be a sensible option for biomass-derived energy yield.
In this context, the green microalga Chlamydomonas reinhardtii grown in batch culture using 100% municipal wastewater showed reasonable lipid productivity (16.6% dry wt) coupled with high biomass productivity (Kong et al. 2010). Moreover, this green microalga had the potential to grow vigorously in the wastewater for 1 month when transferred to a biocoil with a relatively high lipid content (25.25% dry wt) and considerable biomass and lipid productivities of 2.0 g/L/day and 505 mg/ L/day in addition to high removal efficiency of nitrogen and phosphorous from the ambient medium, as shown in Table 1 (Kong et al. 2010). Botryococcus braunii grown in secondary treated municipal wastewater showed considerable levels of total lipid content (17.9% dry weight) and biomass productivity of 0.35 g/L/day, indicating that wastewater nutrient status influences biomass and lipid synthesis (Órpez et al. 2009).
The use of dairy manure as a microalgae nutritive supplement could increase the algae-based biofuel production potential. Where the lipid content of microalgal consortia of Chlorella sp. Micractinium sp. and Actinastrum sp. grown in anaerobically digested dairy manure wastewater in outdoor batch culture showed considerable lipid content of 14 to 29% dry wt depending on the used concentration of wastewater, giving estimated biomass and lipid productivity of 0.06 g/L/day and 17 mg/L/day (Woertz et al. 2009). A similar finding has been reported in Chlorella sp. using 20% diluted anaerobic digester effluent samples treated with dairy waste giving biomass and lipid productivity 0.34 g/L/day and 37.0 mg/L/day (Wahal and Viamajala, 2016). Remarkable increases in biomass and lipids productivity were observed in Rhizoclonium hieroglyphicum grown in dairy effluent enriched with carbon dioxide and manure (Mulbry et al. 2008b) and in Chlorococcum sp. RAP13 when grown in dairy effluent supplemented with 6 % waste glycerol (Ummalyma and Sukumaran, 2014).
To summarize, the high production costs limit the commercialization of algae-based biotechnology for the production of bio-based energy. Microalgal biomass production in agricultural, municipal, or industrial wastewater is a low-cost option that could significantly reduce economic and environmental costs while also providing a valuable remediation service.
Characterization
Microalgae have fast-growing cycles, more acceptable forms of stored carbon, and their ability to survive in sewage or saline water, making them a more appealing biofuel feedstock. The number of microalgae species ranges from 70,000 to one million. Only about 44,000 species are known, with new species and genera being discovered all the time. The continuous discovery of new species adds to the search for strains capable of high growth rates and lipid accumulation. A combination of microscopic and phylogenetic analysis is used to classify the performance of strains (Chew et al. 2018; Neofotis et al. 2016).
Potential as a fuel
Biofuel has been proposed as a potential future energy source. Liquid biofuels have experienced a rapid global expansion in recent years. The first-generation biofuels mainly depend on using sucrose producing plants (e.g. sugar cane, sugar beet and sweet sorghum) or starch-based crops such as wheat, corn and barely. This kind of biofuel is already commercially viable in the United States, Brazil, and the European Union. The impact of the first generation biofuels on the transportation industry is currently limited due to increased competition on agricultural land for food production. The second-generation biofuels are derived from agricultural lignocellulosic waste and non-edible crops, including agricultural residues, wood chips, straw and grass. Nevertheless, cereal and sugar crops have also been used as feedstocks for second generation processing technologies. Also, plant-based biodiesel production necessitates the utilization of cultivable lands for food production to grow oil producing crops such as palm soybean, oilseed rape and sunflower, which negatively affects food security. Therefore, governments limited the amount of these feedstocks for biofuel production (Shuba and Kifle 2018). At the economic scale, second generation biofuels have been reported to be not commercially viable due to extensive processing technologies, poor conversion rates and low net energy production (Milano et al. 2016, 2018).
Furthermore, vegetable and animal fats have been proposed to produce biodiesel using nonpolar solvents and catalyst, but there damaging effect on diesel engines, low oxidation stability and volatility, increased viscosity and density, fuel atomization, and a higher ratio of greenhouse gases emissions make them an unacceptable option. The third-generation biofuels are derived from microalgae and are considered to be a viable alternative energy resource. Microalgae have an ability to grow quickly and they can survive under harsh environments. Their metabolic profile can be easily engineered to produce high value added byproducts by selecting appropriate species and adjusting cultivation conditions (Milano et al. 2016).
Microalgae biomass is regarded as one of the most efficient renewable energy sources for bio-oil production with increased energy demands in addition to their role in mitigation of greenhouse gas emissions Lee et al. (2011). Algal oil (oilage) is a biodegradable compound that has gained popularity as a primary substrate for biodiesel manufacturing, due to its low emissions and adequate physio-chemical properties that positively affect efficiency of diesel engines. Algal biodiesel is produced through transesterification of algal lipids in a two steps process which, yields more fatty acid methyl esters than the direct transesterification process. Previous studies revealed that oilage feedstock surpasses the best seed crop oil in terms of productivity (Arvindnarayan et al. 2017). Algae-based fuels are environmentally friendly, non-toxic, and potentially reduce global carbon dioxide emissions (Pienkos et al. 2009). It has been stated that 1 kg of algal biomass can fix 1.83 kg of carbon dioxide. Interestingly some microalgal species can also fix sulfur and nitrogen oxides as nutrients source (Tu et al., 2019).
In conclusion, microalgae as a feedstock can be viewed as a potential alternative for balancing and compensating for the rising demands for biofuels. In terms of nutrient requirements and carbon dioxide sequestration capacity, wastewater combined with an inorganic carbon source (industrial flue gases) may be the most economically viable option for scale-up over freshwater resources. However, algal fuel technology is still in its early stages, and more work is required for commercialization.
Cultivation
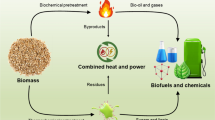
One of the most critical stages in the development of algal biomass is the design of affordable and efficient microalgae culture. The medium is considered a necessary component in cultivation because it regulates algae growth and reproduction. As a result, the medium must contain all necessary components for growth, including minerals such as phosphorus, nitrogen, magnesium, sulfur, calcium, manganese, silicon, and iron in sufficient quantities. Successful microalgae cultivation for sustainable biofuel production requires a high rate of productivity, a low cost of production, and a low cost of maintenance (Voloshin et al. 2016; Ullah et al. 2015). Microalgae can be grown in two different ways; indoor (closed system) such as tubular, flat-panel and vertical column photobioreactors and outdoor (open system) conditions such as ponds and lakes. Every system has its benefits and drawbacks. Open farming is regarded as the most basic and oldest method of producing and cultivating microalgae on a large scale (Fig. 3).
Influence of cultivation conditions on biomass and lipid productivity
Developing novel strategies for increasing microalgal biomass rich with lipid content would result in a low-cost, long-term biofuel production process. Several publications in the last decade investigated various strategies for inducing biomass production and lipid accumulation in microalgal biomass. This section will look at strategies that include various species types under various cultivation conditions. Cultivation conditions significantly impact microalgal growth characteristics and composition. There are four types of microalgal cultivation conditions: photoautotrophic, mixotrophic, heterotrophic and photoheterotrophic. The biomass and lipid productivity of different microalgal species under different cultivation conditions are summarized in Table 1, where the following sections discuss each type of cultivation in more detail.
Phototrophic cultivation
The most frequently used conditions for microalgae cultivation were reported in many publications (Illman et al. 2000; Mandal and Mallick, 2009; Liu et al. 2013; George et al. 2014; Mandotra et al. 2016; Narayanan et al. 2018; Sangapillai and Marimuthu, 2019; Saraf and Dutt, 2021). Microalgae use inorganic carbon sources (e.g., carbon dioxide) and light as the energy source to produce high energy organic compounds via the photosynthesis process. Overview of phototrophic cultivation conditions (Table 1) revealed a large variation in the biomass productivity of microalgae, ranging from 0.01 (Takagi and Yoshida 2006; Liang et al. 2009) to 0.7 g/L/day (Gómez-Serrano et al. 2015; An et al. 2003) and lipid productivity ranged between to 0.2 mg/L/day to (Illman et al. 2000) to 505 mg/L/day (Kong et al. 2010) depending on the type of microalgal species, cultivation system and nutrient status. Previous studies revealed significant increases in the lipid content of microalgae when nitrogen or nutrient deficiency was applied (Fernandes et al. 2013; Li et al. 2013; Hamed et al. 2020). However, achieving significant lipid productivity resulted in lower biomass production. Thus, lipid content is not the only factor influencing the energy value of microalgae. As a result, both biomass and lipid productivities should be considered simultaneously. Lipid productivity is a more accurate indicator of a microalga's ability to produce lipid because it combines the effects of lipid content and biomass production. Chlamydomonas reinhardtii recorded the highest lipid productivity by 505 mg/L/day under phototrophic cultivation. The microalga was grown in a biocoil photobioreactor using municipal wastewater (the centrate) (Kong et al. 2010) (Table 1). In this context, remarkably high lipid productivity was found in Chlorella sp. (121.3–178.8 mg/L/day) and Nannochloropsis oculata NCTU-3 (84.0‒142.0 mg/L/day) under phototrophic condition supplemented with different carbon dioxide concentration (2%, 5%, 10%, and 15%) (Chiu et al. 2008). The use of autotrophic cultivation for microalgal growth and lipid production has a significant benefit in terms of carbon dioxide sequestration as an essential carbon source. Accordingly, the microalgae's cultivation sites should be close to power plants or factories in order to provide a sustainable source of carbon dioxide for microalgal growth. Furthermore, autotrophic cultivation has a lower contamination rate than other types of cultivation. As a result, phototrophic cultivation conditions, such as open ponds and raceway ponds, are commonly used in microalgal outdoor scale-up.
Heterotrophic cultivation
Similar to bacteria, some microalgal species can grow in the absence of light by utilising organic carbon as a source of energy and carbon. This mode of nutrition is known as heterotrophic cultivation (Xu et al. 2006; Cheng et al. 2009; Ummalyma and Sukumaran, 2014). This cultivation method is suitable for large-scale photobioreactors as it avoids the hurdles of light limitation in the phototrophic conditions that could impede high cell density. Compared to other cultivation methods, heterotrophic cultivation recorded the highest biomass and lipid productivities (Xu et al. 2006; Li et al. 2007; Xiong et al. 2008; Cheng et al. 2009; Gao et al. 2010) (Table 1). Changing the cultivation conditions from phototrophic to heterotrophic induced lipid content in Chlorella protothecoides by 40% (Xu et al. 2006). Variable organic carbon sources, such as glycerol, glucose-fructose, sucrose, acetate, mannose, lactose and galactose, have been shown to promote microalgae growth under heterotrophic conditions (Xu et al. 2006; Liang et al. 2009; Heredia-Arroyo et al. 2011; Ummalyma and Sukumaran 2014). Many trials showed that using low-cost alternative organic carbon sources makes this process economically viable. For example, using corn powder hydrolysate and sweet sorghum hydrolysate in the culture of Chlorella protothecoides increased the biomass productivity to 2 g/L/day and 1.2 g/L/day, and lipid productivity was markedly induced to 932 mg/L/d and 586.8 mg/L/d as shown in Table 1 (Xu et al. 2006; Gao et al. 2010). The productivity of both lipids and biomass under heterotrophic cultivation is 20 times greater than that obtained under phototrophic cultivation. So far, Chlorella protothecoides achieved the highest lipid productivity (3700 mg/L/day) using a 5-L fermenter with an optimized fed-batch culture strategy of glucose and yeast extract (Xiong et al. 2008). Contamination is, however, a common issue in sugar-based heterotrophic cultivation; additionally, the economic feasibility of this system is still debatable.
Mixotrophic cultivation
In this process, microalgae assimilate inorganic carbon (carbon dioxide) through photosynthesis and utilize exogenous organic carbon sources for growth (Hamed and Klöck, 2014). Therefore, some microalgal species can live under different cultivation systems, heterotrophic or phototrophic conditions, or mixotrophic. Microalgae obtain their energy from the assimilation of organic carbon, and the released carbon dioxide during the respiration process is sequestered by the photosynthetic machinery. Compared to common propagation modes, microalgae experienced better growth and lipid productivity under mixotrophic conditions (Table 1). This mode has been proposed to combine the benefits of autotrophic and heterotrophic cultivation systems. For instance, Chlorella vulgaris 259 showed significant biomass (0.25 g/L/day) and lipid (54 mg/L/day) productivity under mixotrophic cultivation when compared with both autotrophic and heterotrophic (Liang et al. 2009). The highest productivity of biomass (0.39–1.87 g/L/day) and lipid (120–250 mg/L/day) was reported in a green microalgal species Chlorella vulgaris 2714 using mixotrophic cultivation with different initial glucose: glycerol concentrations (Heredia-Arroyo et al. 2011). Moreover, considerable biomass productivity (0.879 mg/L/day) was obtained by Parachlorella kessleri 211–11G under mixotrophic growth conditions using glucose in fermented brewery wastewater (O'Rourke et al. 2016). A similar finding was stated in different green microalgal species such as Chlorella pyrenoidosa using alcohol wastewater and anaerobically digested starch wastewater (Yang et al. 2015), Chlorella sp. using anaerobic digester effluent treated with dairy waste (Wahal and Viamajala 2016), and C. zofingiensis using artificial wastewater with repetitive injection with acetic acid (Zhu et al. 2014) as shown in Table 1.
Photoheterotrophic cultivation
This growth mode is like mixotrophic cultivation requires the addition of an organic carbon source. However, photoheterotrophic cultivation also needs light as the energy source. In contrast to the mixotrophic mode, photoheterotrophic microalgae cannot absorb and metabolise carbon dioxide. Whereas in a mixotrophic mode, algae can use organic compounds and replenish cell energy through photosynthesis. Therefore, the photoheterotrophic mode requires both carbon and light sources. Although the production of some light-dependent valuable metabolites by Euglena gracilis has been improved under photoheterotrophic cultivation, however using this cultivation mode for biofuel production is not economically feasible compared to mixotrophic cultivation.
In conclusion, this subsection covered the four main microalgal cultivation conditions: photoautotrophic, mixotrophic, heterotrophic, and photoheterotrophic. Open ponds and raceway ponds are common phototrophic cultivation conditions used in microalgal outdoor scale-up. While contamination is a common problem in sugar-based heterotrophic cultivation, the economic feasibility of this type is still being debated.
Economic evaluation of different cultivation modes
Table 1 revealed that taxonomic groups likely had different biomass and lipid productivities. According to the literature, the heterotrophic cultivation mode could provide higher oil and biomass productivity than other cultivation modes. As a result, this approach has sparked much interest. However, culture contamination problems impose technical hurdles in heterotrophic cultivation, especially in open systems. Additionally, from a commercial perspective, the cost of the organic carbon source is another major constrain. Phototrophic cultivation is the easiest mode for scale-up and the most frequently used in outdoor systems. In this cultivation mode, carbon dioxide and flue gas from power plants and factories could be employed in microalgae propagation and lipid production (Lizzul et al. 2014; Yadav et al. 2019).
Nevertheless, cell growth, biomass and lipid productivity are typically much lower than those of heterotrophic cultivation. At the economic scale, the low production cost of biomass by phototrophic mode suggests that this method is most attractive. So far, the available information on using photoheterotrophic and mixotrophic modes for microalgae biomass-derived biodiesel production is very limited.
Furthermore, the two cultivation modes' contamination risk and light requirements restricted their commercialization. Thus, scaling up under these conditions may require a special photobioreactor design which could increase the cost of operation. As a result, the use of autotrophic cultivation mode under well-adjusted conditions, e.g., light intensity, carbon dioxide levels, temperature, nutrient status of the culture, is a reliable and cost-efficient option.
Open ponds
Open ponds are large-scale outdoor algae cultivation systems in various sizes and shapes, including huge shallow ponds, circular ponds, close ponds, and raceway ponds. The pond's location is a critical factor because it affects the pond's type, used algal strain, and culture conditions (e.g. light availability, outdoor temperature, rain fall, time of day (Harun et al. 2010). The open pond system is associated with ease and low-cost design, high efficiency and low maintenance costs compared to closed-system bioreactors (Dahiya, 2015).
The open ponds system is more vulnerable to changing meteorological conditions, which primarily affect the temperature of the water and rate of evaporation. These factors can impact growth rates and biomass algal productivity (El Shenawy et al., 2020). Scenedesmus, Spirulina, and Chlorella are the most cultivated microalgal species in outdoor ponds system. This system is best suited for areas with high light irradiance and with open access to a water source. the Coastal zones are typically the most preferable sites. In this system, microalgae can be grown either in natural or artificial ponds, and microalgae mass production is maintained through a proper supply of necessary nutrients. For example, the biomass yield of a cyanobacterium Arthospira sp. has reached up to 40 tons per year near lake of Chad.
Circular ponds are another kind of artificial pond and are used for commercial-scale microalgae cultivation. Propagation in these ponds is usually restricted by size, as their huge diameters are not financially acceptable because of increased energy expenses required for rotor rotation (Voloshin et al., 2016). Raceway ponds are one of the best available options for cultivation due to their energy efficiency. A single paddle wheel is required to provide agitation in the area of 5 hectares (Siddiki et al., 2022).
Photobioreactors
Photobioreactors are classified as closed systems because they provide a controlled environment for the growth of photosynthetic algae in sterile environments (Dahiya 2015). Photobioreactors can provide increased photosynthetic efficiency, biomass production and cell density. Photobioreactor’s efficiency can be determined by integrating light capturing, transportation, distribution, and increased utilization by microalgae which increases biomass formation. In this system the composition of the nutrient medium can be controlled to maintain growth with optimal yields. Photobioreactors have been demonstrated to reduce capital and operational cost while minimizing energy consumption (Wang et al. 2012).
Productivity is one of the most important factors that determine the success of a bioreactor technology. Comparing productivity of bioreactors is difficult because of variable used microalgal strains and different operation settings. At the industrial scale, tubular photobioreactor (TPBR) and plate types photobioreactor are more appropriate for outdoor cultivation than conventional photobioreactors. The large illumination surface area of these bioreactors is achieved through utilizing translucent tubing and is considered the primary reason for its outdoor adaptability. The tubing system can be manipulated in various configurations to fit system design (Harun et al. 2010).
The low rate of water loss induced by evaporation, low risk of contamination, increased photosynthetic efficiency, increased cells density per unit area are considered the most important advantages of closed system bioreactors (Fig. 2) (Veerabadhran et al. 2021a). Continuous efforts are paid to improve the photobioreactor technologies to boost algal cultures and biomass productivity. For instance, maximizing the utilization capacity of solar radiation under outdoor conditions is a challenge in the design of high-efficiency photobioreactors. Commercial-scale outdoor photobioreactors must have large volumes and surfaces should be transparent with increased illuminating area with high mass transfer rates.
Rainwater affects the salinity and pH by diluting the culture medium. Erosion of the bank occurs due to water turbidity and leakage. Microalgal cultures can be easily contaminated, causing formation of a mixed biomass from various microbial species. Contamination can have adverse effects on microalgae yield due to growth inhibition or toxicity induced by other contaminants. Carbon source is the basic necessity for cell growth. A carbon source other than atmospheric carbon dioxide is required to fulfil optimum cell growth conditions; the carbon source can be organic or inorganic. Light intensity and temperature also vary with the time throughout the day in the case of open ponds (Siddiki et al. 2022; Voloshin et al. 2016). CO2 is referred to carbon dioxide
One of the major disadvantages of microalgae cultivation technology in developed countries is the requirement for wide land areas and cost pressures. As a result intensive efforts are being paid by scientists to increase the algal biomass production through looking for suitable cultivation sites and refining the biomass to multiple high value-added compounds to compensate the operation costs (Ugwu et al. 2008).
Photobioreactors and open ponds are used to produce a wide range of commercially important high-value compounds, novel materials, and feedstock for various industries, including cosmetics, food, and feed. Photobioreactors have high biomass productivity with the best use of the land area and a better ability to capture light irradiance (Fig. 3). Photobioreactors are particularly used to produce biomass-derived pharmaceutical products, food, and cosmetics due to low contamination risk with bacteria and fungi. In contrast, open ponds system is more cost-effective, however more vulnerable to contamination risk. Thus this system is used to produce biomass-based biofuels, biopolymers and chemicals. In addition few number of microalgal strains can grow in open pond system. Therefore, a cost-effective and efficient photobioreactor design is required to achieve high energy efficiency with positive net energy (Jorquera et al. 2010).
Photobioreactors are classified into three basic categories. Tubular, flat-panel and vertical column photobioreactors can be used for outdoor cultures as well. Biomass productivity is good due to high mass transfer opportunities and high surface illumination properties. All three photobioreactors are costly, limiting their use as they decline the whole process's cost efficiency (Siddiki et al. 2022; Voloshin et al. 2016; Harun et al. 2010; Wang et al. 2012; Ugwu et al. 2008). PBR is referring to a photobioreactor, while TPBR is a tubular photobioreactor. O2 is oxygen, and CO2 is carbon dioxide
In conclusion, the cost of algal biofuel production was primarily determined by culturing (77%), harvesting (12%), and lipid extraction (7.9%). As a result, the choice of microalgal species and cultivation modes such as autotrophic, heterotrophic, or mixotrophic are crucial factors in controlling biomass and bioenergy production, as well as fuel properties.
Conversion technologies
Following the biomass cultivation and preparation stages comes the stage of actual conversion into biofuels. There are numerous strategies for biofuel conversion, regardless of the nature of the biomass. They are generally classified into three types based on cost, end-product, and eco-friendliness (Voloshin et al. 2016).
Bioconversion
Anaerobic digestion, bioethanol and biogas production, and photobiological hydrogen or biohydrogen production are examples of how microalgae biomass can be converted (Osman et al. 2020; Osman et al. 2022a).
Fermentation
To reduce gas emissions, algal biomass and its derivatives can be used as gasoline, octane enhancers, or bioethanol diesel mixers (El-Mekkawi et al. 2019). Bioethanol can be used in place of gasoline due to its similar physicochemical properties. The biochemical composition of the biomass feedstock is used as a decisive factor for bioethanol yield. Carbohydrate-rich biomasses are typically used as a substrate in yeast-based fermentation to produce bioethanol (Fig. 4). (Kannah et al. 2021a; Abd El-Malek et al. 2021).
Finely ground biomass is converted to sugars with the help of enzymes or acids in a normal fomentation process. Then sugars are transformed into ethanol by yeasts. Ethanol is then separated using a distillation column. The high concentration ethanol stream is liquefied, making liquefied ethanol a substitute for gasoline in automobiles. The solid deposit can be used as cattle feed and gasification
Polysaccharides in the form of starch soluble and insoluble sugars and cellulose are abundant in algal biomass, for these reasons, algal biomass is regarded as an efficient feedstock for bioethanol synthesis. In a standard fermentation process, finely ground biomass is converted to sugars with the help of specific enzymes or acids, and sugars are then converted into ethanol by a yeast’s fermentation process. The ethanol is eventually separated using a distillation column. The highly concentrated ethanol stream is liquefied to be used as a gasoline alternative in automobiles. The residual solid deposits can be used for cattle feed and gasification process. This helps to reduce feedstock prices, which account for 55 to 80% of the total alcohol cost. Ethanol production from algal biomass requires an additional pretreatment phase in which content is mechanically or enzymatically hydrolyzed. Concentrated sulfuric acid is frequently used to decompose the biomass's intra and inter H-bonds, the acid is then neutralized and produced sugars are separated for fermentation process (Hossain et al. 2019).
Carbohydrate, crystalline cellulose, hemicellulose, and amorphous cellulose are generally hydrolyzed using various biological catalysts or enzymes. Specific functional enzymes such as cellulase have been extensively used to decompose cellulose polymer into simple glucose units, which could improve the process (Dayton and Foust, 2019).
The yeast-based biomass fermentation process does not require dewatering steps or the addition of toxic chemicals for extraction, which are considered important benefits of this technology. Combining sonication, enzyme and heat treatment during continuous bioethanol production from mixed microalgal biomass has been proposed by Hwang et al. (2016). In the microbial fermentation method, various pretreatments steps are required, resulting in varying degrees of cell lysis. Filamentous microalgae are usually hydrolyzed by sonication pretreatment, while a diatom species such as cyclotella, cells were destroyed by combining sonication and enzymatic hydrolysis. The sonication heat and enzymatic treatments increased the concentration of dissolved carbohydrates, which improved ethanol formation during microbial fermentation. It has been found that microbial species likely exhibited differential fermentation abilities. Thus, the selection of efficient microbial strains is pivotal factor in this technology. For instance, the increased activity of sugars fermenting enzymes produced by a yeast species Dekkera bruxellensis resulted in higher ethanol production when compared with mixed bacterial culture (Hwang et al. 2016). A study by El-Dalatony et al. (2019) revealed that three consecutive steps are required during microbial fermentation of microalgal biomass into biofuels. In phase I, carbohydrate is fermented to produce bioethanol and then residual proteins in biomass are fermented for increased alcohol production (phase II). Whereas, phase III involves in transesterification of remaining fatty acids into biodiesel. This approach allows for the most efficient recovery of energy careers from algal biomass.
Enzymatic and acidic hydrolysis of Chlorella vulgaris and Scenedesmus obliquus biomass were analyzed to evaluate the optimum yeast inoculum required for better hydrolysate's fermentation (Silva et al. 2018). Acidic hydrolysis with 3% sulfuric acid at 121 ℃ and 30 min of reaction time provided 90% of sugar recovery and are considered optimum conditions for biomass pretreatment. Ultrasonication is an efficient method for biomass pretreatment to increase enzyme availability. Whereas, combining ultrasonication with a cocktail of hydrolyzing enzymes (amylase, cellulase, hemicellulase, and pectinases) resulted in 90% sugar recovery in 8 h (Silva et al. 2018) (Table 2).
Anaerobic digestion
Anaerobic digestion is an effective strategy for converting organic wastes and biomass with high moisture content to high heating value gases. In this technology, microbes anaerobically degrade the organic matter through a series of biochemical processes into biogas containing carbon dioxide, methane, and other gases such as nitrogen and hydrogen sulfides. The composition and type of cell wall significantly influence the biodegradability of microalgae. In marine species, biodegradability is influenced by ammonia toxicity due to high cellular protein and sodium contents. Microalgae can be used as a renewable and sustainable substrate for biogas production through anaerobic digestion technology. The cost-efficiency of anaerobic digestion using microalgal biomass significantly impacts the prospect of energy sustainability. Thus, much work is needed to improve this technology(Veerabadhran et al. 2021b).
The biodegradable composition of microalgae biomass makes it an ideal substrate for anaerobic digestion and methane generation (Perazzoli et al. 2016). Anaerobic digestion comprises of a sequence of metabolic processes, including hydrolysis, acidification, acetogenesis and methanogenesis by diverse microbial groups. The first microbial group hydrolyzed complex chemical substances to monomers by using enzymes which are then transformed into volatile fatty acids, hydrogen and acetic acid. The volatile fatty acids, such as propionic and butyric acid, are converted to hydrogen, carbon dioxide, and acetic acid by acetogens. Hydrogen, carbon dioxide and acetate are eventually converted to methane and carbon dioxide by methanogenic bacteria (Kwietniewska et al. 2014). Various factors such as substrate status or co-digestion with other materials could reduce the anaerobic digestion. The methane yield, on the other hand, could be increased by adapting the microbial community to microalgal biomass digestion.
Nutrient-rich wastewater has been used as culture media for microalgae propagation (Perazzoli et al. 2016). This strategy contributes to wastewater bioremediation and provides multiple bioproducts and energy carriers from algal biomass (Gonzalez-Fernandez et al. 2015).
The study of Membere and Sallis (2018) showed that biogas and methane yields using Laminaria digitata, brown macroalgal species, are significantly influenced by temperature over 40 days experiments. Their results indicated that biogas could be produced at variable degrees of digestion temperatures and impacted biogas yield. For example the yields of methane gas were 318, 293, 271 and 352 mL methane/g volatile solids at 25, 35, 45 and 55 °C. According to their findings, the highest cumulative biogas output was achieved at 35 °C, while overall methane dual potential was optimum at 55 °C (Membere and Sallis 2018).
Biogas production efficiency could also be enhanced by using nanoparticle catalysts such as Fe3C nanoparticles 3 along with iron oxide nanoparticles (Fe3O4). The addition of Fe3O4 nanoparticles to an anaerobic waste digester resulted in a 180% increase in biogas content and a 234% increase in methane production at an ambient temperature of 37 °C for 60 days (Rahman et al. 2016).
Nickel and cobalt nanoparticles, as well as some metal oxide nanoparticles like Fe2O3 and MgO induced higher biogas and hydrogen production yields with variable degrees (Zaidi et al. 2018).
Anaerobic digestion has a number of advantages when compared to other biofuel industries, such as high energy yields of biogas compared to biodiesel, drying is not required, microalgal biomass mineral composition meets the requirements of anaerobic methanogens, the possibility of co-digestion, the culture used for biogas production can be reused for biogas upgrading through carbon dioxide sequestration, the offensive odor is reduced below the specified unprocessed waste odor levels, harmful gas emissions are reduced, reduces oxygen demand in wastewater, and production of valuable byproducts such as fertilizers and compost. The disadvantages of anaerobic digestion include a low carbon to nitrogen ratio due to the high nitrogen content of microalgal biomass, the existence of cell wall which reduces the bioavailability of intracellular compounds, a high capital cost, anaerobic digestion is only feasible for larger farms, a lengthy operational and maintenance period, and the use of a large piece of land (Hossain et al. 2019; Sakarika et al. 2019).
Biohydrogen production
Hydrogen is a high heating value gas (141.6 MJ/kg), clean, more versatile, efficient, and sustainable renewable energy carrier that can replace fossil fuels due to hydrogen’s high energy yield compared to typical hydrocarbon fuels. Biological hydrogen production is a method of producing hydrogen gas that involves the use of microbes (Khetkorn et al. 2017). Microalgae can produce hydrogen photobiologically due to their metabolic and enzymatic properties. Under anaerobic conditions, eukaryotic microalgae can produce H+ and oxygen while fixing carbon dioxide. In the presence of hydrogenases enzymes (Fe-hydrogenase and Ni-hydrogenase), hydrogen ions are reduced to produce hydrogen gas molecules. Various hydrogen production pathways are directed by either acetate or butyrate production using glucose as a model substrate.
It should be noted that both dark and photo fermentation are viable options for anaerobic hydrogen production. Acid fermentation consumes less energy than butyrate fermentation. Furthermore, the former approach produces hydrogen, which is theoretically superior to the latter (Hossain et al. 2019; Sambusiti et al. 2015).
The use of deoiled microalgal biomass as feedstock for anaerobic digestion is seen as a cost-effective and ecofriendly approach which resulted in zero waste, as shown in Fig. 5. The nature of deoiled microalgal biomass, including lignin-free cellulose, makes deoiled microalgal biomass an excellent substrate for dark fermentative hydrogen production. The efficiency of hydrogen production from a deoiled algal cake is influenced by various factors, including physical, biological, and operational factors. The conversion of deoiled microalgal biomass to hydrogen takes place in two stages. The first stage involves hydrolysis of the deoiled algal biomass to simple sugars. The second stage includes acidogenic bacteria fermentation, in which sugars are converted into hydrogen (Subhash et al. 2014).
Deoiled microalgal biomass nature, including lignin-free cellulose, makes it a good substrate for dark fermentative hydrogen production. In the process of dark fermentation two phases are involved in the conversion of deoiled microalgal biomass to hydrogen. The first stage comprises hydrolysis of the deoiled algal biomass while producing simple sugars. The second stage includes acidogenic bacteria fomentation where simple sugars are converted into hydrogen
In all bioconversion processes, the suitability of fermenting microbes to substrate feedstock and the values of product output are critical. Clostridium sp. is a common bacterial model used for hydrogen generation from organic substrates (Ferreira et al. 2013). Ding et al. (2016) stated that carbon-rich macroalgae produce hydrogen and methane when combined with nitrogen-rich microalgae in a two-stage process. Hydrolysis and acidogenesis were aided by the co-fermenting process resulting in 15.5–18.5% more hydrogen yield using Laminarial digitilia biomass. A considerable amount of energy left in hydrogenogenic effluents was recovered as biomethane in the second stage of methane co-fermentation, increasing energy efficiency from 4.6 to 6.6% during hydrogen fermentation from 57 to 70.9% in combined hydrogen and methane generation (Ding et al. 2016).
Strategies for increasing hydrogen production employ cutting-edge approaches such as genetic engineering, microalgae bacteria consortia, advanced biohydrogenation method, and nanomaterials for enzyme stability and hydrolytic efficiency. Dark fermentation is preferred over photo fermentation, biophotolysis, and microbial electrolysis because dark fermentation can produce hydrogen repeatedly and does not require sunlight.
Thermal conversion
Microalgae are the most suitable and promising organisms, possessing the morphological and physiological characteristics required for large-scale biomass production. Following the cultivation and preparation of biomass, microalgae are converted into biofuel using various techniques, the most common of which are thermochemical approaches. Thermal conversion methods subject the feedstock to higher temperatures and pressures to obtain compounds with low molecular weight and oxygen content through pyrolysis, gasification, and combustion processes.
Direct combustion
Direct combustion of biomass in the presence of excess air is used to convert chemical energy into heat while also producing carbon dioxide and sulfur dioxide/nitrogen dioxide. These emissions, as well as the energy yield generated throughout the process, are determined by the operating temperature and biomass content. Incomplete biomass combustion emits carbon monoxide, methane, and particulate matter. Notably, these emissions can be reduced to some extent by using certain catalysts such as copper chloride and magnesium oxide during the combustion of algal biomass. Emissions from direct or partial combustion cause secondary pollution. (Bhushan et al. 2020).
Direct combustion necessitates the use of microalgae with less than 50% moisture. As a result, the energy requirements and additional cost of drying and grinding microalgae for efficient combustion are rising. The efficient use of heat generated by direct combustion of microalgae can eliminate the need for additional drying and grinding. Co-firing coal with algae has the potential to reduce greenhouse gas emissions caused by combusting coal alone (Halder and Azad 2019).
Pyrolysis
Pyrolysis occurs at higher temperatures (typically from 500 to 800 °C) in the absence of oxygen, as well as in the absence or presence of a catalyst. Biomass is decomposed over a certain time at various temperatures to produce fuels with calorific values ranging from medium to low. Pyrolysis produces solid biochar, bio-oil, or gaseous materials (Fawzy et al. 2021, Osman et al. 2022b). The temperature is mostly between 300 and 600 °C during pyrolysis, while the temperature can exceed 800 or 900 °C in certain situations. Microalgae cells are generally small, considering other forms of biomass; therefore, no shredding is necessary. Microalgal biomass should include minimum moisture content for successful pyrolysis to reduce the cost of technology implementation (Chernova et al. 2020).
Chlorella protothecoides, Chlorella vulgaris, Scenedesmus sp., Microcystis aeruginosa, Nannochloropsis sp. and Chlorella vulgaris residues along with others, are among the most common algal species used for biofuel conversion. The conversion of algae to biofuel in the pyrolysis process is feasible due to its sustainability and economic viability for both industrial and residential uses. Heterotrophic cells produce more bio-oil than autotrophic cells. During rapid pyrolysis, bio-oil produced by heterotrophic cells has a high heating value, lower oxygen content, density, and viscosity. The increased storage stability is due to lower oxygen levels (Das et al. 2021a).
Parameters such as process temperature, heating rate, residence time or catalyst, along with others, affect the pyrolysis of microalgal biomass. Variation in these process parameters can lead to a change in the composition of the final product. For example, increasing temperature above 550 ℃ has decreased bio-oil yield, and a decrease in temperature increases biochar yield. Algal biomass is enriched in sodium, potassium, magnesium and calcium, which is suitable as a fertilizer when converted to biochar, while higher oil yield is obtained when the lipid content in algal biomass is high. The presence of carrier gas significantly increases the performance of the pyrolysis process. The type of catalyst that can be used to optimize the final product yield as acid catalysts improve the yield of biochar, while base catalyst improves the yield of bio-oil (Das et al. 2021b).
Gasification
Gasification produces syngas, which contains hydrogen, carbon monoxide, carbon dioxide, and methane due to partial oxidation of biomass. The composition of the selected algal biomass influences the final production and composition of syngas. Dried algal biomass reacts with water at high temperatures and 10 bar pressure. Algal biomass is converted into various gaseous products via combustion, dehydration, devolatilization, and gasification. The syngas can be used as fuel in engines or further processed into a liquid fluid. Catalyst improves gasification by increasing hydrogen yielding low-cost solid and gaseous products.
Several factors influence the gasification of microalgal biomass. Hydrogen production increases with increasing the temperature, while methane, carbon dioxide, char, and tar decrease. Temperature increases cause an increase in energy consumption. Carbon gasification becomes inefficient as operational pressure is increased. A catalyst such as ruthenium on titania (Ru/TiO2) or nickel-based improves process efficiency while increasing tar removal efficiency to 80–100%. A higher equivalence ratio improves fuel quality and conversion efficiency (Das et al. 2021b).
Moisture content and biomass availability are crucial elements for thermochemical conversion. Microalgae have 70 to 80% moisture content; therefore, bulk cultivation is a futuristic challenge. Low-cost drying techniques are required for thermochemical conversion (Bhushan et al. 2020). Because of the product's gaseous nature, storage and transportation are difficult, and thermal conversion of algal biomass has disadvantages (Aravind et al. 2020) (Table 3).
Chemical conversion
This route employs base, acid catalysts or enzyme catalysts, as well as supercritical transesterification. Because base catalysts cannot be recycled or reused, while acid catalysts are thought to be slower than base catalysts and are ideal for obtaining oils. The enzyme catalyst technique is time-consuming and hazardous to commercial biodiesel production. For oil extraction, supercritical transesterification requires a temperature of up to 250 °C and constant pressure. In this process, a catalyst is an option. (Chamola et al. 2020). Transesterification is the reaction of triglyceride with alcohol to form esters and glycerol. During this esterification process, in the presence of sodium or potassium hydroxide as a catalyst, triglyceride reacts with alcohol. Excess alcohol is utilized to ensure complete esterification. Mono alkyl ester is formed, an active ingredient of biodiesel with the reaction of alcohol and fats, whereas glycerol is also produced. At the end of the reaction, separating the oil phase from the rest of the esters and the glycerol layer is achieved (Demirbas 2009).
Biodiesel must undergo testing prior to commercialization. The absence of glycerol, catalyst, unreacted alcohol, and free fatty acids are all important properties of biodiesel. There is a need to introduce energy-efficient, low-cost liquid extraction and transesterification procedures for algal biodiesel production. After liquid extraction, biomass can be used efficiently to generate biogas via anaerobic digestion. Biogas can be produced as a byproduct without interfering with biodiesel production. Commercializing biogas will reduce overall costs while increasing biofuel production (Sialve et al. 2009; Wiley et al. 2011; Subhadra et al. 2011).
Acid catalysts are combined with the base catalyst in a two-stage process. High fatty acids containing low-cost feedstock, i.e., waste oil, are also processed through this method. In the first stage, methyl esters are formed by converting fatty acids, while the base catalyst transforms leftover triglycerides into methyl esters (Pragya et al. 2013).
Among all conversion processes, anaerobic digestion is an energy-efficient procedure for generating biogas as biomass with high moisture content is tolerable compared to thermal conversion processes. However, anaerobic digestion requires long periods. For biomass conversion to bio-oil, esterification (acid catalyst) is widely used.
Downstream processing
Sedimentation is considered an efficient method of harvesting microalgal mass because sedimentation involves biomass settling due to gravity forces. This approach is particularly appealing in terms of energy and cost because this method uses very little energy and has less expensive equipment and operational costs. Lamella settling tanks are simple settling tanks that are commonly used equipment. Lamella settling tanks are frequently chosen because the available settling surface is significantly increased in this form. The main disadvantage of this method is the time required for microalgal settling (Esteves et al. 2020). Downstream processing is an energy-efficient approach that is widely used to separate microalgae from water. The sedimentation technique has the potential to successfully separate microalgae such as Spirulina, which settle due to their increased density and size (Pragya et al. 2013). Before the sedimentation procedure, flocculation and coagulation occur.
Other harvesting processes, including coagulation-flocculation and flocculation-flocculation, are frequently employed in conjunction with flocculation. This process can enhance the microalgal particle size for suspension through aggregation while also boosting the settling rate. Bio-flocculation, auto-flocculation, physio-chemical flocculation and physical-flocculation are the most common forms of flocculation (Japar et al. 2017). Flocculants can be polyelectrolyte, organic or inorganic based on their chemical makeup. Multivalent cations, for example, ferric sulfate, ferric chloride, and aluminum sulphate can create polyhydroxy complexes at ideal pH, neutralizing and reducing negative surface charges on microalgal cells. Inorganic chemical flocculants are employed to flocculate microalgal cells. Cationic, anionic or nonionic organic flocculants or polyelectrolytes are examples of organic flocculants. Because cationic polymers physically bind cells together, they flocculate, but anionic and nonionic polymers fail to produce flocs of microalgae owing to electro-repulsion.
The flocculating power of the polyelectrolytes is affected by the charge and functional group on the surface of the microorganism, the pH of the growth media and the density of the algal culture. Cationic polyelectrolytes with high density are more effective flocculants for energy-efficient harvesting. The coagulant's efficacy decreases as the coagulant's molecular weight increases, whereas ionic polyelectrolytes do not show this behaviour (Barros et al. 2015).
The high-volume microalgal cultures that must be processed, as well as the need for a universal method that can be used for a wide range of species, contribute to the safety of these procedures. The suspension is concentrated 20 to 100 times during the harvesting process. The suspension improves the efficient particle size before dewatering, significantly lowering the energy requirement. For low-cost microalgae harvesting, coagulation and flocculation are usually followed by gravity sedimentation. Unlike coagulation, which requires a pH change or the addition of electrolytes, flocculation is based on the addition of cationic polymers to the broth. Chemical coagulation and flocculation are defined as the agglomerates of particles that are finally split in suspension or to the larger aggregates, followed by their agglomeration into the large flocks that settle at the bottom of the vessel, leaving a clear supernatant (Barros et al. 2015). Chemical coagulants should ideally be sustainable and renewable, resulting in no biomass contamination, allowing them to use culture media, inexpensive, nontoxic, and efficient at low doses while being derived from renewable resources.
Flotation is a gravity separation methodology in which gas or air bubbles lift suspended particles to the top of a liquid surface, where they can be collected using a skimming process. Because some microalgal organisms have low density and self-float properties, this approach can be faster and more successful than sedimentation. Freshwater and marine microalgae can be harvested through flotation separation. Many factors influence the attachment of suspended particles to wear on gas bubbles, including particle size, adhesion and collision chances. The key advantages are the short operating time, low volume requirements, large-scale harvesting, and rate adaptability at a low starting cost. This procedure typically requires flocculants and is followed by coagulation and flocculation (Singh et al. 2018). Electrolytic flotation, dissolved air flotation, and dispersed flotation are three major types of floatation based on bubble size (Pragya et al. 2013). Dispersed air, micro-flotation, foam floatation, vacuum gas, dissolved air, flocculation flotation, electro flotation, and ozone flotation are the methods used for flotation (Japar et al. 2017).
Electrical filtration techniques such as electrocoagulation filtration, magnetically induced membrane filtration, and electrochemical sacrificial electrode filtration have also been investigated. Due to the fact that this environmentally friendly method does not target specific species, this filtration technique is both safe and cost-effective. During an electrical field, the negative charge of microalgal cells can separate, allowing for culture growth. Precipitates can form on electrodes and collect on the vessel's bottom (electrophoresis) (electro-flocculation). As the bubbles form, electro-flotation is generated from dissolved air flotation. Water electrolysis can generate hydrogen bubbles at a lower cost than dissolved air flotation. These bubbles can be generated by the node and then combined with electrocoagulation that occurs at the cathode due to electrolytic oxidation. This is referred to as electrocoagulation flotation (Barros et al. 2015).
Electrocoagulation flotation is a traditional mechanical harvesting process in which substances are separated based on density variation, with heavier particles moving away from the axis and lighter particles moving towards the axis. Various types of centrifuges are introduced for algal cell harvesting at varying levels of efficiency, either as a one-step procedure or as a two-step procedure incorporating biomass pre-concentration, due to their speed and dependability. Tubular and multi-chamber centrifuges are two traditional methods for harvesting algal biomass. Despite the tubular’s efficiency, a tubular centrifuge cannot be used continuously due to its inability to release solids. As a result, the performance of disc centrifuges and bench-scale harvesting can be predicted. A multi-chamber centrifuge's closed bowl is divided into concentric, vertical chambers (Mathimani et al. 2018).
Upon separation through centrifugation, algal culture is exposed to shear stresses and significant gravitational forces that can damage the structure of cells. The following are the numerous centrifugation technique types for systems available: solid bowl decanter, nozzle type, hydro-cyclone and solid ejecting disk (Enamala et al. 2018).
To keep the algae in a thick paste state, the cultivation is allowed to flow through filters that can operate on gravity, pressure, and vacuum pressure. Because the cells are less disrupted and no chemicals are used, it is possible for the system to be continuous or batch membrane harvesting to improve the quality of collected by matching over the harvesting method. Several filter assemblies have been used to connect algae, but their effectiveness has been hampered by low throughput and rapid fouling; many different filter designs are dependent on solute–solvent properties, hydrodynamic conditions, and membrane features. A few types of filtrations include microfiltration (0.1–10 mm), microfiltration (10 mm), dead-end filtration, ultrafiltration (0.002–0.2 mm), vacuum filtration, tangential flow filtration and pressure filtration. Although ultrafiltration is a viable recovery method, ultrafiltration is not commonly used for microalgae harvesting due to high flux requirements, maintenance, and operating costs. This method is commonly used for large algae, such as filamentous species. Scenedesmus, Chlorella, Dunaliella, and other microalgae are not suitable for this technique. The main disadvantage of this filtering method is that filters and membranes must be replaced regularly, which makes the procedure time consuming (Enamala et al. 2018).
In summary, flotation, coagulation, and flocculation are common methods for harvesting microalgal biomass (Fig. 6). Microalgae with self-flotation characteristics benefit from flotation. Filtration techniques necessitate membranes, which have the drawback of fouling and corrosion. Sedimentation and centrifugation are strain-specific processes that necessitate longer periods that may not be feasible (Table 4).
Harvesting techniques are classified into three major categories based on their nature. Chemical harvesting techniques include flocculation/coagulation. The biological process includes bioflocculation. Physical processes include sedimentation, floatation, electrical harvesting techniques, filtration and centrifugation. They can also be categorized into two basic types based on the working principle or end result: either thickening or dewatering techniques
Extraction
Pretreatment
Microalgae have complex cell walls that contain intercalated proteins and polysaccharides. It is difficult to break down cell walls and extract lipids when there is insufficient energy. Prior to lipid extraction, the appropriate cell lysis on algal cell type is required. Mechanical such as sonication, homogenizer, microwave, electric pulse-field and chemical cell disruption techniques exist such as acid, surfactant, enzymes. Furthermore, there is a distinction between mechanical and chemical approaches, with chemical methods being more scalable than mechanical methods (Japar et al. 2017; Vasistha et al. 2021; Sirohi et al. 2021). Therefore, pretreatment is required to extract necessary biomolecules that improve the downstream processing of algal biomass.
Extraction of lipids
To achieve a high yield, efficient liquid extraction is required. There are two types of lipids in algae: polar lipids and nonpolar lipids. Nonpolar lipids such as mono, di, and triglycerides can be used to produce biodiesel. Polar lipids, such as glycolipids and phospholipids, are used for other purposes. Traditional lipid extraction from microalgae employs a combination of polar (chloroform, methanol) and nonpolar (hexane) solvents. Methanol can lyse cells, and chloroform is used as an eluting solvent, making liquid extraction easier (Vasistha et al. 2021). This is owing to the polarity and solubility of chloroform. Binary mixtures are efficient in this regard; their increased efficiency can be linked to the fact that using solvents other than binary combinations will be impossible for scaling and may even raise biodiesel production expenses (Ebhodaghe et al. 2021).
Microwave radiations are used to liberate intracellular components in nutraceuticals and medications. Microwaves can interact selectively with polar molecules such as water and create heat, disrupting the algal cell membrane and extracting lipids. This method is successful for both dry and wet algal biomass. Microwave radiation is one of the most effective methods for cells disruption and removing lipids. The culture medium receives sound waves with a frequency greater than 20 kHz (kilohertz). This method results in an alternative compression (high pressure) and rarefaction configuration (low pressure). In a low-pressure area, microbubbles can develop, causing a cytoplasmic rupture and the release of lipid biomolecules.
This method uses short electric pulses with a high electric field force to disrupt cells by creating micropores in the cell membrane. Electroporation or electric immobilization occurs as a result. With increasing exposure time, lipid production increases. However, due to the high energy requirements and costs, this is not a traditional mechanically aided cell rupture, and extraction approaches are not practical on a larger scale (Vasistha et al. 2021). Solvent extraction and ultrasonication are the most used techniques for cell disruption and lipid extraction, followed by microwave-assisted extraction.
Stress assessment on microalgae growth
Several factors influence the success of large-scale microalgae farming for biodiesel and other value-added products. Light, temperature, pH, carbon dioxide supply, and other macronutrients, such as nitrates, are required for microalgae cell growth. The amounts of these factors can be adjusted and optimized to produce desired biomass and concentrations (da Maia et al. 2020).
The growth rate of microalgae biomass increases as the intensity of appropriate light illumination increases; exposure above the ideal level inhibits photons, thus slowing the growth (Chhandama et al. 2021). Oversaturated light can cause the development of reactive oxygen species, which can damage microalgae cells by photoinhibition, thus lowering biomass output yield (Cheng et al. 2014).
The medium of development pH influences various cellular activities in microalgae, including enzymes, the structure and function of cell organelles, energy metabolism, and protein synthesis, with an ideal pH for growth ranging from 7.5 to 8.5. Microalgae are grown in an alkaline medium, sequester carbon dioxide more efficiently, and grow faster. It should also be noted that while cell development is taking place, the pH of the medium rises due to the constant increase in carbon dioxide. This increases the photosynthetic rate and promotes the formation of OH- ion aggregation.
Microalgae require various nutrients to multiply and produce biomass, including nitrogen, carbon, phosphorus, magnesium, potassium, calcium, and iron. These nutrient concentrations, particularly phosphorus and nitrogen, significantly impact mass cultivation and lipid production. Lack of nutrients can cause physiological and morphological changes in cells, resulting in changes in growth rate and biomass yield (Chhandama et al. 2021). Protein photosynthetic metabolism is shifted towards carbohydrates, and fat increases when nitrogen levels are low (De Carvalho Silvello et al. 2021).
In large-scale outdoor culture systems, temperature plays an important role. Frequent alterations and fluctuations in temperature can result in a considerable reduction in algal lipid production efficiency. Microalgae are adaptable in a wide range of environments and fluctuating temperatures. However, it has been observed that the ideal temperature for rapid and optimum production is between 20 to 30 °C. In general, biomass output relatively increases with increased temperature as low temperatures stifle the cell’s metabolic activity (Chhandama et al. 2021).
Carbon is hypothesized to affect nitrogenase activity and, as a result, affects nitrogenase dependent hydrogen generation. Furthermore, the quantity and composition of lipids in microalgae cells were demonstrated to be affected by varying quantities and sources of carbon. It has been observed that a high carbon dioxide concentration causes an increase in saturated fatty acids, while a low carbon dioxide concentration promotes the formation of unsaturated fatty acids (Cheng et al. 2014).
To summarize, all factors mentioned above influence the microalgal cell growth rate and productivity. However, these factors can increase the production of desired molecules, for example, nutrient starvation for accumulation of targeted molecules or increasing the pH of culture for efficient carbon dioxide sequestration.
Key drivers
Bioenergy feedstock should be high in biopolymers, widely available, low in cost, and easy to process, and microalgal biomass is among the renewable and potentially useful bioenergy feedstock. The three primary components of microalgae biomass are protein, glucose, and lipids. Green and blue microalgae species, such as Chlorella, Spirulina, Scenedesmus, and Nannochloropsis, are among the most promising for bioenergy generation (Kannah et al. 2021b).
Because of various factors, including increased feedstock availability and the ability to be generated globally while avoiding food competition, such as second and higher generation biofuels, biomass is a promising fossil substitute as a raw material (Silva et al. 2017). All microalgal ingredients are used to create a useful and sustainable fuel with the concept of circular economy and zero waste (El-Dalatony et al. 2019). Furthermore, unlike the transition to electric vehicles, the transition to liquid biofuels does not necessitate significant improvements in gasoline infrastructure. Biofuels can be used as a fuel blend to traditional engine fuels or as the primary fuel in engines.
In Brazil, gasoline is frequently blended with ethanol produced from sugar biomass. Plants are harvested only twice or four times per year, limiting biofuel production. Plant biomass production requires optimal conditions, and subsequent processing may necessitate the use of energy-intensive technologies. Algal biomass is free of these constraints while increasing the biofuel production ratio faster than plant biomass (Voloshin et al. 2016).
Various microalgae have higher lipid content, which benefits the use of microalgae instead of vascular plants for biodiesel production by more than 50%. Only the seeds of vascular plants are used to produce plant-derived biodiesel, while the rest are not used. Simultaneously, microalgae as a whole can be used to generate biofuel, making microalgae a viable alternative. Another critical factor to consider when using microalgae to produce biodiesel is the possibility of low nutritional requirements.
Furthermore, microalgae can be produced in fresh or saltwater, in remote areas, and even in conjunction with wastewater treatment facilities or industrial parks where bioremediation is advantageous. Following the extraction of hydrocarbons for biofuel production, methane is produced by processing biomass in an anaerobic digester, a secondary energy source, with the digester effluent being fed back into the algae growing system.
Even if microalgae production does not meet fuel consumption requirements, microalgae can significantly reduce imports, resulting in the decentralization of production while also benefiting local economies and the environment, establishing microalgae as a potential feedstock for future fuel supply ( Leite et al. 2013; Khan et al. 2018).
Global market and scale-up of bioenergy from microalgae
Several developed and developing countries have expressed a growing interest in identifying renewable feedstock for bioenergy production in order to meet global energy demand. Microalgae are being researched as viable sources that have traditionally contributed to producing various compounds and extracts, including carotenoids and proteins. The global market for keratin oils and proteins is expected to reach 2.0 and 35.54 billion USD in 2024, respectively (Kannah et al. 2021b).
Consumers are increasingly interested in reducing environmental pollution while extending their lives and avoiding the emergence of chronic diseases. This rising demand has resulted in a growing shift toward the production of microalgal biomass as a fossil fuel substitute in order to improve environmental conditions (García et al. 2017). Due to a lack of demand for petroleum-based fuels, the commercialization of bioenergy from microalgae has increased. It has resulted in economic growth benefits while being facilitated by a new technology directly involved in lowering production costs. However, there are some difficulties in producing microalgae and marketing them for commercial use. Microalgae biorefinery is ideal for overcoming these challenges and generating lucrative income (Camacho et al. 2019).
One of the most popular biorefinery techniques is the use of lipid extracted microalgae for bioenergy production. The extracted lipid is converted into biodiesel, and the residual is used in the anaerobic digestion process to produce biomethane. The global average market price for biodiesel and biomethane is 0.83 USD/L and 0.76 USD/L, respectively. In developing countries such as India, the market price of biomethane and biodiesel is around 0.59 and 0.89 USD/L, respectively (Kannah et al. 2021b). Increased production quantities and bulk synthesis of lipids, carbohydrates, and proteins using microalgae as cell factories are required in the near future. With significant fixed capital expenditures and labor expenses, economies of scale play a critical role in the process's capital and operational expenditures (Camacho et al. 2019). Even though commercial production and microalgal biofuels are still in their infancy due to cost inefficiency, algal cultivation for value-added product extraction and biofuels can strengthen the process due to the high likelihood of scale-up and profitability.
Outstanding issues
Over the last five decades, extensive research has been conducted on microalgae-based biofuel production. However, due to constraints such as strain selection for higher biomass production, microalgae culture system selection, quantity and quality of bio-based product recovery from microalgae, and operational and environmental variables, commercial microalgae production has yet to be recognised and implemented in the real world. For the successful implementation of large-scale microalgae production for bioenergy, it has been suggested that a few key elements, such as biomass composition and productivity, bioconversion platform selection, and other technical and administrative costs, be considered. A few phrases in microalgae processing for bioenergy, such as growth and harvesting, continue to be a major concern for cost-effective methodologies.
Microalgal harvesting loans account for 20–30% of total biogas production costs. Microalgae cultivation pond costs 10 to 20%. The commercialization of microalgal biofuels faces several challenges, including the inability to produce cost-effective fuels due to substrate composition, conversion platform, and technology. An expansion of large-scale examination and optimization of commercial microalgae production and cultivation was occurred. The research revealed that producing commercial-scale microalgae takes a long time. One of the challenges is the scaling-up process, which includes seed culture preparation for a large-scale manufacturing facility of approximately 300,000 L. Simultaneously, there is a scarcity of handling equipment and skilled labor for large-scale production. Several governments in developed countries are assisting this industry in developing a sustainable and environmentally friendly manufacturing system. By producing biomass, microalgae can help to mitigate global warming. Microalgae require 1.8 kg of carbon dioxide to produce 1 kg of biomass. This methodology has two advantages: treating wastewater while reducing environmental concerns and producing biomass at a low cost, resulting in the recovery of value-added products. The combination of bioenergy and value-added product recovery processes has the potential to increase biofuel market demand while decreasing production costs (Kannah et al. 2021b). For example, the cultivation and harvesting of microalgae for bioenergy remain a major concern for cost-effective methodologies.
Carbon sequestration
Carbon is identified as a vital component required to maintain ecological stability by providing a distinct cycle of capture and accumulation. Human intervention has significantly disrupted this equilibrium. The ecosystem has suffered negative consequences because of the disruption caused by increased industrialization (Osman et al. 2021b). Furthermore, unrestricted use of natural resources contributes to the implementation of this negative impact (Jaiswal et al. 2021).
The post-industrial era has resulted in increased atmospheric carbon dioxide and long-term viability, posing a threat to global ecosystems. Carbon dioxide contributes to global warming, accounting for 68% of total greenhouse gas emissions. Thus, the carbon capture storage and utilisation approach is extremely important, where carbon capture employs three major technologies: pre-combustion, post-combustion, and oxyfuel combustion routes. The first two routes accounted for 96.6% of the literature work until 2018, while oxy-reforming technology accounted for only 3.4% of total publications (Osman et al. 2021b). Carbon dioxide can be sequestered using three efficient strategies: chemical, physical and biological. Every methodology has advantages and disadvantages, whereas the earlier based on washing with alkaline solutions or carbon dioxide immobilization with the use of multi-walled carbon nanotubes, adsorption material, and amine coated activated carbon are examples of chemical approaches for carbon dioxide sequestration. Direct injection of pollutants into the ground, oceans, depleted oil/gas wells and aquifers are examples of physical techniques. The biological fixation of carbon dioxide involves photosynthetic microbes, algae, and plants (Shukla et al. 2017).
Physical methods involving direct injection are feasible for large-scale carbon dioxide sequestrations. This, however, necessitates the availability of geological and geomorphological structures, separation equipment, and carbon dioxide collection and compression technologies. This brings uncertainty and an increased risk of long-term leakage. Chemical neutralization methods are safer and provide long-term carbon dioxide fixation; however, the high cost of the reagents required for neutralization limits their use. Both physical and chemical methodologies face challenges in capturing carbon dioxide from diffused or nonpoint sources at low concentrations. Microalgae are photosynthetic organisms that use their photosynthetic machinery to sequester carbon dioxide from the environment with increased photosynthetic efficiency of 10 to 15 times that of traditional plants (Zhou et al. 2017).
Microalgae species absorb and sequester carbon dioxide, and their photosynthetic systems can harvest light photons and inorganic carbon. These microbes effectively capture and use carbon dioxide for biomass production, making them a viable source for the bioenergy and food industries (Jaiswal et al. 2021).
Microalgae can sequester 1.3 kg of carbon dioxide to produce 1 kg of biomass. Microalgae absorb light energy and convert light into adenosine diphosphate, nicotinamide adenine dinucleotide phosphate (NADP), adenosine triphosphate, and nicotinamide adenine dinucleotide phosphate reduced form (NADPH). This energy is then channeled into the dark cycle, which converts carbon dioxide into viable organic compounds via the Calvin Benson cycle. Carbon dioxide sequestration is possible in this case because this sequestration is global in scope, in contrast to other site-specific methods. This possibility has resulted in identifying potentially useful flora and fauna, such as microalgae. To achieve maximum carbon dioxide sequestration, optimal conditions such as temperature, pH, salinity, aeration, nutrition, and illumination should be maintained. To maximise production, the demand for a closed system, i.e., bioreactor, increases where the conditions can be adjusted for increased productivity. The point of focus is the selection of potential microalgae and design parameters for bioreactors in combination with carbon dioxide sequestration. Efficient design can be considered a breakthrough in sustainable sequestration by microalgae (Verma et al. 2018).
Mechanism and tolerance of carbon dioxide sequestration of microalgae
Microalgae are autotrophic photosynthetic microbes whose total metabolism exceeds higher plants of the same weight. Carbon, nitrogen, phosphorus, potassium, magnesium, calcium, and sulfur are the major nutrients for microalgae growth, with carbon being the most important. To adapt to changes in the concentration of inorganic carbon in water, various types of microalgae initiate a mechanism in which inorganic carbon is actively converted in their cells. This is referred to as carbon dioxide concentration mechanisms, which is a critical mechanism for microalgae because carbon dioxide concentration mechanisms are the only way to use carbon dioxide during their photosynthetic process. Organisms with concentration mechanisms have a high affinity for carbon dioxide, which is a major physiological characteristic that allows these organisms to efficiently utilize low carbon dioxide concentrations to fulfil their photosynthetic needs. Ribulose‒1,5‒bisphosphate carboxylase/oxygenase (RuBisCo) limiting enzyme is catalytically immobilized in vivo because RuBisCo has a low affinity for carbon dioxide and for normal reactions, requiring a high concentration of carbon dioxide (Xu et al. 2019).
The affinity and tolerance level of carbon dioxide varies between microalgae strains. Microalgae can survive in varying carbon dioxide environments. At carboxylated sites, microalgae have developed mechanisms like concentration mechanisms to survive in environments with low carbon dioxide concentrations. An increase in carbon dioxide concentration has an anesthetic effect on microalgal cells, inhibiting photosynthesis and algal growth. The initial concentration of carbon dioxide influences growth, which also influences lipid yield and composition. The synthesis of fatty acids is inhibited by low carbon dioxide concentration, whereas increased carbon dioxide concentration increases fatty acid accumulation regardless of the inhabited carbon change the saturation and elongation (Zhou et al. 2017). This cycle consists of 13 steps divided into three categories: fixation, reduction, and regeneration.
Carboxylation stage: 3-phosphoglyceric acid is produced by catalyzing carbon dioxide and ribose-1,5- diphosphate under the action of ribose-1,5- diphosphate carboxylase.
Reduction stage: Adenosine triphosphate is used for the acidification of 3-phosphoglyceric acids, which is then converted into 1,3-diphosphate glyceric acid under the action of the 3-phosphoglyceric acid kinase. This is then reduced into glyceraldehyde-3-phosphoric acid by NADPH under the action of phosphoglyceraldehyde dehydrogenase.
Regeneration stage: consisting of the regeneration of ribose 1,5-diphosphate. The glyceraldehyde-3-phosphate molecule is then acidified in ribonuclease 1,5-diphosphate under the action of Adenosine triphosphate and enzyme. Carbon dioxide fixation occurs due to the generation of ribose 1,5-diphosphate. This recycling of biochemical processes utilised carbon dioxide in the photosynthetic action (Xu et al. 2019).
Bicarbonate and gaseous carbon dioxide are used by microalgae as carbon sources. However, bicarbonate is considered a dominant carbon species in the most frequent pH range (6.5–10) in the medium of microalgae production. When an industrial flue gas stream is fed into microalgae cultures, the carbon dioxide content is generally greater than in ambient air, which results in better biomass production. The dissolved carbon dioxide in media is used as a buffer, increasing biomass productivity by enhancing carbon content. In microalgae, chloroplast produces lipids. Chloroplast fixes atmospheric carbon dioxide as an indigenous source of Acetyl-CoA and subsequently into carbon in the fatty acid chain. Chlamydomonas reinhardtii, Chlorella sp., Nannochloropsis sp., Ostreococcus tauri, Phaeodactylum tricornutum are considered the most promising microalgae for the synthesis of lipids and triacylglycerol.
Microalgae accumulate monosaccharide glucose through photosynthesis. This glucose serves as an energy source, as well as a source of proteins, lipids, and other carbohydrates. With increased irradiance or nutrient depletion, the cell's glucose production can exceed its consumption rate. Excess glucose can disrupt the cell's osmotic balance; thus, excess glucose is converted into stored products such as polysaccharides and lipids. In the future, these products will serve as an energy source and a carbon source (Choi et al. 2019).
Advantages of microalgae-based carbon sequestration
Cultivation of photosynthetic microalgae can provide a sustainable substitute for carbon sequestration compared to terrestrial plant systems, as illustrated in Fig. 7. Simple harvesting, quick production, low requirements, increased tolerance to environmental stress, increased carbon dioxide tolerance, high photosynthetic ability, and increased biomass production rates are important characteristics that make microalgae a promising solution. Several algal species grow at an exponential rate, doubling their biomass production. Solar energy is efficiently converted into biomass by the microalgae's ability to tolerate higher carbon dioxide concentrations, increasing their optimum growth through more efficient carbon dioxide fixation than higher plants. Carbon dioxide in the atmosphere and soluble carbonates can be used as a carbon source to cultivate microalgae. Microalgae can also fix the increased concentration of carbon dioxide in industrial flue gases. Because of the ability of microalgae to thrive in wastewater and the use of various trace elements, including heavy metals, microalgae are the most prominent substitute and alternative to dealing with rising environmental concerns. As a result, microalgae are recommended for bioremediation, particularly for wastewater treatment and heavy metal removal from water bodies (Banerjee et al. 2020).
Cultivation of photosynthetic microalgae can provide a sustainable substitute for carbon sequestration compared to terrestrial plant systems, where microalgae can grow at a rate ten times that of terrestrial plants. Simple harvesting, rapid production, low requirements, increased tolerance to environmental stress, increased carbon dioxide tolerance, high photosynthetic ability, and increased biomass production rates are all important features that make microalgae a promising solution. Microalgae can also fix increased carbon dioxide concentrations in industrial flue gases. Because of microalgae's ability to thrive in wastewater and use various trace elements, including heavy metals, microalgae are the most prominent substitute and alternative to dealing with rising environmental concerns. Microalgae can grow in agricultural and industrial wastewaters, especially those containing high levels of nitrogen and phosphorus. Microalgae biomass can also be converted into a carbon–neutral biofuel such as bioethanol, biodiesel, or biogas. CO2 is referred to carbon dioxide and O2 for oxygen
Yadav et al. (2019) used organic and inorganic nutrients derived from industrial wastewater and coal-fired flue gas to cultivate microalgae in closed photobioreactors for waste bioremediation and biomass production. In the industrial wastewater, Chlorella sp. and Chlorococcum sp. were grown with varying concentrations of coal-fired flue gas ranging from 1 to 10% carbon dioxide. The results showed a 1.7 fold increase in biomass production, while the microalgae cultivated with industrial wastewater with flue gas containing 5% carbon dioxide showed maximum growth and carbon dioxide fixation (Yadav et al. 2019).
Tu et al. (2019) studied the impacts of power plant tail gas to reduce carbon dioxide by using the tail gas as a carbon source and cultivated a freshwater microalga Chlorella pyrenoidosa. An increase in dry weight and lipid production by 84.9 and 74.4% was observed in the presence of power plant tail gas. Optimum carbon fixation sequestration of microalgae was 1.12 g/L having an average carbon fixation rate of 0.21 g/(Ld), which was 134.2% and 107.1% higher compared to the growth of microalgae in the open air. C. pyrenoidosa is tolerant to sulfur dioxide and nitric oxide, which is in accordance with the study mentioned above. The tolerance is 0.04%; however, pretreatment processes like desulfurization and denitrification are required (Tu et al. 2019) (Table 5).
Aghaalipour et al. (2020), in their research study, analyzed the assessment of carbon dioxide by fixation of two microalgal species, Scenedesmus obliquus and Chlorella vulgaris. Along with that, two new species, Monoraphidium contortum, and Psammothidium sp., were also studied for their capability of carbon dioxide inputs in two types of photobioreactors, including glass bottles and vertical columns. This study aimed to assess the carbon dioxide bioremediation rate, growth kinetics, and protein content of microalgal species of different types of photobioreactors with varying amounts of carbon dioxide ranging from 0.04% to 10%. According to the results, Chlorella vulgaris (3.35 g/L/day) was most significant as Chlorella vulgaris showed maximum carbon dioxide sequestration at 10% carbon dioxide in the vertical column photobioreactors, followed by Psammothidium sp. (3.24 g/L/day), Scenedesmus obliquus (2.40 g/L/day), and Monoraphidium contortum (1.40 g/L/day). Psammothidium sp. showed maximum carbon dioxide recovery (CR%), which was 41.70%. Chlorella vulgaris has also depicted maximum protein content during Chlorella vulgaris cultivation in a glass flask photobioreactor with 10% carbon dioxide (Aghaalipour et al. 2020).
Toxic pollutants in combustion flue gas
Sulphur dioxide
sulfur dioxide is considered a limiting factor for microalgal growth. An increase in 100 ppm sulfur dioxide concentration completely restricts microalgal growth. A few microalgal species have shown growth in high concentrations of sulfur dioxide; however, these species have depicted a longer lag phase. Increasing sulfur dioxide levels decreases carbon fixation and biomass production, ultimately inhibiting growth (Klinthong et al. 2015).
Sulphur dioxide is considered an important pollutant in flue gas and increases the acidification of microalgal culture, giving oxidative damage to the cells. Wang et al. (2020) utilize exogenous spermidine to reduce the negative impacts on Chlorella sp. and reduce the impacts of sulfur dioxide and 15% carbon dioxide impurity. Spermidine efficiently in habitat HSO3−/SO32- by penetrating through cell walls and protecting photosynthetic PSII structures and thylakoid membranes. It resulted in the recovery of chlorophyll from 0.5 mg/L to 8.5 mg/L while increasing the biomass yield recovery from 0.12 g/L to 1.52 g/L. The recovered lipid content of the biomass was improved from 5.28% to 19.12% (Wang et al. 2020).
Duarte et al. (2016) stated that sulfur dioxide and nitrogen dioxide injection until 400 part per million (ppm) do not affect the carbon dioxide bio-fixation in microalgae, although the optimum yield and results were obtained at low concentrations of industrial waste. This study examined that Chlorella fusca LEB 111 can grow under all conditions while showing resistance to sulfur dioxide and nitric oxide concentrations up to 400 ppm. Chlorella fusca LEB 111 can grow in a culture medium with 40 ppm ash and be unaffected by sulfur dioxide and nitric oxide concentrations of up to 400 ppm. The optimum carbon dioxide fixation efficiency was observed with 10% carbon dioxide, 200 ppm sulfur dioxide along with nitric oxide and 40 ppm ash (Duarte et al. 2016).
According to Song et al. (2021), the cultivation of microalgae with real flue gas has the probability of containing an impurity, reducing cost in the treatment of flue gas, and increasing the benefits of microalgal carbon sequestration. On the bubble dissolution characteristics of 15% carbon dioxide, the impact of sulfur dioxide impurity was observed in different sulfur dioxide concentrations, solution pH, different culture media, initial bubble sizes, and biomass concentrations in the microalgal genus Arthrospira. Arthrospira's photosynthetic growth and biomass yield were increased by 24% to 5.04 g/L with 200 mg/m3 sulfur dioxide in simulated flue gas containing 15% carbon dioxide, compared to without sulfur dioxide impurity. Arthrospira solution has an alkaline medium that nullifies sulfur dioxide's toxic effects (Song et al. 2021).
Nitrogen oxide emissions
The nitrogen oxide emission level in flue gas ranges from several hundred to several thousand parts per million with more than 90 to 95% nitric oxide and 5 to 10% nitrogen dioxide. After releasing flue gas, removing such emissions is still at the 50 to 200 ppm-level. Nitric oxide cannot directly impact microalgal growth through pH in the cultivation media. The concentration of nitric oxide poses a two-sided influence on microalgal growth. Low nitric oxide concentration can be absorbed by the cultivation media and converted into nitrogen dioxide, which acts as a source of nutrition for microalgae. Increased nitric oxide concentrations are observed to decrease microalgae growth for various species. More than 300 ppm of nitric oxide decreases microalgal growth. A selective catalytic reduction process and flue gas desulfurization can separately be used to treat oxides of sulfur and oxides of nitrogen. Sometimes, the combined treatment systems can be used simultaneously before entering the gas stream into a microalgae reactor ( Klinthong et al. 2015).
To summarize, microalgae can be used to efficiently capture carbon dioxide and convert microalgae to bio-fuel, simultaneously solving two major issues of the world. The point of focus is the selection of potential microalgae and design parameters for bioreactors in combination with carbon dioxide sequestration. Efficient design can be considered a breakthrough in sustainable sequestration by microalgae. To use inorganic sources for carbon dioxide, e.g., flue gas, a gas treatment system should be used to reduce or eliminate inhabitation factors. Furthermore, microalgae can sequester 1.3 kg of carbon dioxide for every kg of biomass produced. Using potent microalgal strains in efficient bioreactor designs for carbon dioxide sequestration is thus a challenge. In open and closed cultures, microalgae can theoretically use up to 9% of light energy to capture and convert 513 tons of carbon dioxide into 280 tons of dry biomass per hectare per year. Algal biomass cultivation should be coupled with thermochemical technologies, such as pyrolysis, to design an efficient atmospheric carbon removal system.
Conclusion
The value of biofuels extends beyond their use as a transportation fuel; the economic and environmental benefits of biofuel co-products should be considered. Material item development can play an important role in preventing future environmental damage. Various generations of biofuels reduce greenhouse gas emissions while increasing reliance on crude oil, encouraging energy diversification and the creation of a large number of rural jobs (Ahorsu et al. 2018). To accelerate implementation, the primary goal of integrated algae waste operations should be to maximise productivity and product accumulation while minimising energy, water, nutrients consumption, and land footprint, particularly for large-scale production and future research and development. Biorefinery technology capable of producing a wide range of high-value products will be required to fully implement algal biomass and enable commercially viable bioenergy co-production (Dayton and Foust 2019).
Microalgae are regarded as a viable biodiesel production option. The combination of microalgae and wastewater purification can reduce carbon dioxide emissions while also lowering biodiesel production costs, providing a way for practical application. Temperature, salinity, pH, light intensity, photobioreactor configuration, nutrient ratio, and carbon dioxide flow rate influence microalgae productivity and efficiency. One of the major constraints is the successful extraction of oil from microalgae biomass. Transesterification is a common procedure used in the production of biodiesel. (Li et al. 2021). While the use of microalgae in carbon sequestration as an effective carbon removal strategy should be considered in the near future.
Several methods are used to extract energy from algae, each with its own set of advantages and disadvantages. A few of these strategies are still in the early stages of development, and algae-based biofuel generation is deemed economically but commercially unfeasible. Given the early stage of research and the high cost, it is reasonable to conclude that there is still a long way to go in terms of influencing the process of algae biofuel production (Pragya et al. 2013). After liquid extraction, the residue algal biomass has improved economic viability for value-added products generation and biorefinery technology (Subhash et al. 2014). It is possible to use a single-step integration technology that combines cell destruction and liquid extraction procedures (Vasistha et al. 2021). While the use of algae in atmospheric carbon removal should have lower constraints than biofuel production (biodiesel), such as high oil content or oil extraction, what is required in terms of carbon sequestration is a species with a high growth rate and low oil content, as algae, in this case, will be converted into solid biochar.
References
Abd El-Malek F et al (2021) Microorganism-mediated algal biomass processing for clean products manufacturing: current status, challenges and future outlook. Fuel. https://doi.org/10.1016/j.fuel.2021.122612
Abdel-Raouf N et al (2012) Microalgae and wastewater treatment. Saudi Journal of Biological Sciences 19(3):257–275. https://doi.org/10.1016/j.sjbs.2012.04.005
Abomohra AE-F et al (2013) Lipid and total fatty acid productivity in photoautotrophic fresh water microalgae: screening studies towards biodiesel production. J Appl Phycol 25(4):931–936. https://doi.org/10.1007/s10811-012-9917-y
Abomohra AE-F et al (2014) Extracellular secretion of free fatty acids by the chrysophyte Ochromonas danica under photoautotrophic and mixotrophic growth. World J Microbiol Biotechnol 30(12):3111–3119. https://doi.org/10.1007/s11274-014-1738-5
Abou-Shanab RA et al (2014) Cultivation of a new microalga, Micractinium reisseri, in municipal wastewater for nutrient removal, biomass, lipid, and fatty acid production. Biotechnol Bioprocess Eng 19(3):510–518. https://doi.org/10.1007/s12257-013-0485-z
Adey WH et al (2011) Algal turf scrubbing: cleaning surface waters with solar energy while producing a biofuel. Bioscience 61(6):434–441. https://doi.org/10.1525/bio.2011.61.6.5
Aghaalipour E et al (2020) Carbon dioxide capture with microalgae species in continuous gas-supplied closed cultivation systems. Biochem Eng J 163:107741. https://doi.org/10.1016/j.bej.2020.107741
Ahorsu R et al (2018) Significance and challenges of biomass as a suitable feedstock for bioenergy and biochemical production: a review. Energies 11(12):3366. https://doi.org/10.3390/en11123366
Al-lwayzy SH et al (2014) Biofuels from the fresh water microalgae Chlorella vulgaris (FWM-CV) for diesel engines. Energies 7(3):1829–1851. https://doi.org/10.3390/en7031829
An J-Y et al (2003) Hydrocarbon production from secondarily treated piggery wastewater by the green alga Botryococcus braunii. J Appl Phycol 15(2):185–191. https://doi.org/10.1023/A:1023855710410
Anastopoulos G et al (2001) Influence of aceto acetic esters and di-carboxylic acid esters on diesel fuel lubricity. Tribol Int 34(11):749–755. https://doi.org/10.1016/S0301-679X(01)00067-6
Antolın G et al (2002) Optimisation of biodiesel production by sunflower oil transesterification. Bioresour Technol 83(2):111–114. https://doi.org/10.1016/S0960-8524(01)00200-0
Aravind S et al (2020) Conversion of green algal biomass into bioenergy by pyrolysis. A review. Environ Chem Lett 18(3):829–849. https://doi.org/10.1007/s10311-020-00990-2
Arvindnarayan S et al (2017) Algal biomass energy carriers as fuels: an alternative green source. Renew Sustain Energy Rev 90(2):300–315. https://doi.org/10.1016/j.rser.2015.12.150
Asomaning J et al (2016) Thermal processing of algal biomass for biofuel production. Curr Opin Green Sustain Chemi 2:1–5. https://doi.org/10.1016/j.cogsc.2016.08.005
Banerjee I et al (2020) Microalgae-based carbon sequestration to mitigate climate change and application of nanomaterials in algal biorefinery. Octa J Biosci 8:129–136
Barros AI et al (2015) Harvesting techniques applied to microalgae: a review. Renew Sustain Energy Rev 41:1489–1500. https://doi.org/10.1016/j.rser.2014.09.037
Bhushan S et al (2020) Current trends and prospects in microalgae-based bioenergy production. J Environ Chem Eng 8(5):104025. https://doi.org/10.1016/j.jece.2020.104025
Cai T et al (2013) Cultivation of Nannochloropsis salina using anaerobic digestion effluent as a nutrient source for biofuel production. Appl Energy 108:486–492. https://doi.org/10.1016/j.apenergy.2013.03.056
Camacho F et al (2019) Potential industrial applications and commercialization of microalgae in the functional food and feed industries: a short review. Mar Drugs 17(6):312. https://doi.org/10.3390/md17060312
Chamola R et al (2020) Direct transesterification for biodiesel extraction from micro-algal biomass: a review. Int J Appl Eng Res 14(9):180–184
Chen H et al (2018) Algal biofuel production coupled bioremediation of biomass power plant wastes based on Chlorella sp. C2 cultivation. Appl Energy 211:296–305. https://doi.org/10.1016/j.apenergy.2017.11.058
Cheng Y et al (2009) Biodiesel production from Jerusalem artichoke (Helianthus Tuberosus L.) tuber by heterotrophic microalgae Chlorella protothecoides. J Chem Technol Biotechnol Int Res Process Environ Clean Technol 84(5):777–781. https://doi.org/10.1002/jctb.2111
Cheng D et al (2014) Assessment of environmental stresses for enhanced microalgal biofuel production–an overview. Front Energy Res 2:26. https://doi.org/10.3389/fenrg.2014.00026
Chernova N et al (2020) Manufacturing gaseous products by pyrolysis of microalgal biomass. Int J Hydrogen Energy 45(3):1569–1577. https://doi.org/10.1016/j.ijhydene.2019.11.022
Chew KW et al (2018) Effects of water culture medium, cultivation systems and growth modes for microalgae cultivation: a review. J Taiwan Inst Chem Eng 91:332–344. https://doi.org/10.1016/j.jtice.2018.05.039
Chhandama MVL et al (2021) Microalgae as a feedstock for the production of biodiesel: a review. Bioresour Technol Reports 15:100771. https://doi.org/10.1016/j.biteb.2021.100771
Chinnasamy S et al (2010) Microalgae cultivation in a wastewater dominated by carpet mill effluents for biofuel applications. Bioresour Technol 101(9):3097–3105. https://doi.org/10.1016/j.biortech.2009.12.026
Chisti Y (2007) Biodiesel from microalgae. Biotechnol Adv 25(3):294–306. https://doi.org/10.1016/j.biotechadv.2007.02.001
Chiu S-Y et al (2008) Reduction of CO2 by a high-density culture of Chlorella sp. in a semicontinuous photobioreactor. Bioresour Technol 99(9):3389–3396. https://doi.org/10.1016/j.biortech.2007.08.013
Chiu S-Y et al (2009) Lipid accumulation and CO2 utilization of Nannochloropsis oculata in response to CO2 aeration. Biores Technol 100(2):833–838. https://doi.org/10.1016/j.biortech.2008.06.061
Choi YY et al (2019) Microalgae bioenergy with carbon capture and storage (BECCS): an emerging sustainable bioprocess for reduced CO2 emission and biofuel production. Bioresour Technol Rep 7:100270. https://doi.org/10.1016/j.biteb.2019.100270
Chowdhury H et al (2019) Third-generation biofuels from microalgae: a review. Curr Opin Green Sustain Chem 20:39–44. https://doi.org/10.1016/j.cogsc.2019.09.003
Dahiya A (2015) Algae biomass cultivation for advanced biofuel production. In: Bioenergy. Elsevier. https://doi.org/10.1016/B978-0-12-407909-0.00014-6
da Maia JL et al (2020) Microalgae starch: a promising raw material for the bioethanol production. Int J Biol Macromol. https://doi.org/10.1016/j.ijbiomac.2020.10.159
Das P et al (2021a) Recent advances in thermochemical methods for the conversion of algal biomass to energy. Sci Total Environ 766:144608. https://doi.org/10.1016/j.scitotenv.2020.144608
Das P et al (2021b) A comprehensive review on the factors affecting thermochemical conversion efficiency of algal biomass to energy. Sci Total Environ 766:144213. https://doi.org/10.1016/j.scitotenv.2020.144213
Dasgupta CN et al (2015) Dual uses of microalgal biomass: an integrative approach for biohydrogen and biodiesel production. Appl Energy 146:202–208. https://doi.org/10.1016/j.apenergy.2015.01.070
Dayton DC et al (2019) Analytical methods for biomass characterization and conversion. Elsevier, Amsterdam
Demirbas A (2009) Production of biodiesel from algae oils. Energy Sources, Part A; Recovery, Utilization and Environmental Effects. https://doi.org/10.1080/15567030701521775
de Carvalho Silvello MA et al (2021) Microalgae-based carbohydrates: A green innovative source of bioenergy. Biores Technol 344:126304. https://doi.org/10.1016/j.biortech.2021.126304
Demirbas A et al (2011) Importance of algae oil as a source of biodiesel. Energy Convers Manag 52(1):163–170. https://doi.org/10.1016/j.enconman.2010.06.055
Demirbas A (2010) Use of algae as biofuel sources. Energy Convers Manag 51(12):2738–2749. https://doi.org/10.1016/j.enconman.2010.06.010
Deng X et al (2019) Biomass production and biochemical profiles of a freshwater microalga Chlorella kessleri in mixotrophic culture: effects of light intensity and photoperiodicity. Bioresour Technol 273:358–367. https://doi.org/10.1016/j.biortech.2018.11.032
Díez-Montero R et al (2020) Evaluation of daily and seasonal variations in a semi-closed photobioreactor for microalgae-based bioremediation of agricultural runoff at full-scale. Algal Res 47:101859. https://doi.org/10.1016/j.algal.2020.101859
Ding L et al (2016) Co-generation of biohydrogen and biomethane through two-stage batch co-fermentation of macro-and micro-algal biomass. Biores Technol 218:224–231. https://doi.org/10.1016/j.biortech.2016.06.092
Dismukes GC et al (2008) Aquatic phototrophs: efficient alternatives to land-based crops for biofuels. Curr Opin Biotechnol 19(3):235–240. https://doi.org/10.1016/j.copbio.2008.05.007
Dong B et al (2014) Cultivation of Nannochloropsis salina in municipal wastewater or digester centrate. Ecotoxicol Environ Saf 103:45–53. https://doi.org/10.1016/j.ecoenv.2014.02.001
Duarte JH et al (2016) Utilization of simulated flue gas containing CO2, SO2, NO and ash for Chlorella fusca cultivation. Bioresour Technol 214:159–165. https://doi.org/10.1016/j.biortech.2016.04.078
Ebhodaghe SO et al (2021) Biofuels from microalgae biomass: a review of conversion processes and procedures. Arab J Chem 15(2):103591. https://doi.org/10.1016/j.arabjc.2021.103591
Efroymson RA et al (2021) Better management practices for environmentally sustainable production of microalgae and algal biofuels. J Clean Prod 289:125150. https://doi.org/10.1016/j.jclepro.2020.125150
El-Dalatony MM et al (2019) Whole conversion of microalgal biomass into biofuels through successive high-throughput fermentation. Chem Eng J 360:797–805. https://doi.org/10.1016/j.cej.2018.12.042
El-Mekkawi SA et al (2019) Optimization of some fermentation conditions for bioethanol production from microalgae using response surface method. Bull Natl Res Centre 43(1):1. https://doi.org/10.1186/s42269-019-0205-8
El-Sheekh M et al (2013) Optimization of biomass and fatty acid productivity of Scenedesmus obliquus as a promising microalga for biodiesel production. World J Microbiol Biotechnol 29(5):915–922. https://doi.org/10.1007/s11274-012-1248-2
El Shenawy E et al (2020) Effect of cultivation parameters and heat management on the algae species growth conditions and biomass production in a continuous feedstock photobioreactor. Renew Energy 148:807–815. https://doi.org/10.1016/j.renene.2019.10.166
El Shimi HI et al (2018) Biodiesel production from microalgae grown on domestic wastewater: feasibility and Egyptian case study. Renew Sustain Energy Rev 82:4238–4244. https://doi.org/10.1016/j.rser.2017.05.073
Enamala MK et al (2018) Production of biofuels from microalgae-a review on cultivation, harvesting, lipid extraction, and numerous applications of microalgae. Renew Sustain Energy Rev 94:49–68. https://doi.org/10.1016/j.rser.2018.05.012
Esteves AF et al. (2020) Microalgae harvesting techniques. In: Handbook of microalgae-based processes and products. Elsevier. pp 225–281. https://doi.org/10.1016/B978-0-12-818536-0.00010-5
Faramarzi MA et al (2008) Microalgal biotransformation of steroids. J Phycol 44(1):27–37. https://doi.org/10.1111/j.1529-8817.2007.00446.x
Fawzy S et al (2020) Strategies for mitigation of climate change: a review. Environ Chem Lett 18(6):2069–2094. https://doi.org/10.1007/s10311-020-01059-w
Fawzy S et al (2021) Industrial biochar systems for atmospheric carbon removal: a review. Environ Chem Lett 19(4):3023–3055. https://doi.org/10.1007/s10311-021-01210-1
Feng X et al (2014) Biomass and lipid production of Chlorella protothecoides under heterotrophic cultivation on a mixed waste substrate of brewer fermentation and crude glycerol. Bioresour Technol 166:17–23. https://doi.org/10.1016/j.biortech.2014.03.120
Fernandes B et al (2013) Relationship between starch and lipid accumulation induced by nutrient depletion and replenishment in the microalga Parachlorella kessleri. Biores Technol 144:268–274. https://doi.org/10.1016/j.biortech.2013.06.096
Ferreira AF et al (2013) Biohydrogen production from microalgal biomass: energy requirement, CO2 emissions and scale-up scenarios. Bioresour Technol 144:156–164. https://doi.org/10.1016/j.biortech.2013.06.079
Fulton L (2004) Biomass and agriculture sustainability, markets and policies. International Energy Agency (IEA) biofuels study–interim report: result and key messages so far. IEA, France. International Energy Agency, France, 105–112.
Gajraj RS et al (2018) Third-generation biofuel algal biofuels as a sustainable energy source. Biofuels Greenhouse Gas Mitig Global Warm. https://doi.org/10.1007/978-81-322-3763-1_17
Gao C et al (2010) Application of sweet sorghum for biodiesel production by heterotrophic microalga Chlorella Protothecoides. Appl Energy 87(3):756–761. https://doi.org/10.1016/j.apenergy.2009.09.006
García JL et al (2017) Microalgae, old sustainable food and fashion nutraceuticals. Microb Biotechnol 10(5):1017–1024. https://doi.org/10.1111/1751-7915.12800
Gatamaneni BL et al (2018) Factors affecting growth of various microalgal species. Environ Eng Sci 35(10):1037–1048. https://doi.org/10.1089/ees.2017.0521
George B et al (2014) Effects of different media composition, light intensity and photoperiod on morphology and physiology of freshwater microalgae Ankistrodesmus falcatus–A potential strain for bio-fuel production. Bioresour Technol 171:367–374. https://doi.org/10.1016/j.biortech.2014.08.086
Ghosh T et al (2019) Designing biofuel supply chains while mitigating harmful algal blooms with treatment wetlands. Comput Chem Eng 126:113–127. https://doi.org/10.1016/j.compchemeng.2019.03.041
Gómez-Serrano C et al (2015) Utilization of secondary-treated wastewater for the production of freshwater microalgae. Appl Microbiol Biotechnol 99(16):6931–6944. https://doi.org/10.1007/s00253-015-6694-y
Gonzalez-Fernandez C et al (2015) Anaerobic digestion of microalgal biomass: challenges, opportunities and research needs. Bioresour Technol 198:896–906. https://doi.org/10.1016/j.biortech.2015.09.095
Goodrum JW et al (2005) Influence of fatty acid methyl esters from hydroxylated vegetable oils on diesel fuel lubricity. Bioresour Technol 96(7):851–855. https://doi.org/10.1016/j.biortech.2004.07.006
Gouveia L et al (2009) Microalgae as a raw material for biofuels production. J Ind Microbiol Biotechnol 36(2):269–274. https://doi.org/10.1007/s10295-008-0495-6
Grayburn W et al (2013) Harvesting, oil extraction, and conversion of local filamentous algae growing in wastewater into biodiesel. Int J Energy Environ 4(2):185
Halder P, Azad AK (2019) Recent trends and challenges of algal biofuel conversion technologies. In: Advanced Biofuels, Elsevier. pp 167–179. https://doi.org/10.1016/B978-0-08-102791-2.00007-6
Hamed SM et al (2017) Evaluation of the effect of aerobic–anaerobic conditions on photohydrogen and chlorophyll a production by environmental Egyptian cyanobacterial and green algal species. Int J Hydrog Energy 42(10):6567–6577. https://doi.org/10.1016/j.ijhydene.2017.01.058
Hamed SM et al (2020) Influence of nutrient status on the biohydrogen and lipid productivity in Parachlorella kessleri: a biorefinery approach. Appl Microbiol Biotechnol 104(23):10293–10305. https://doi.org/10.1007/s00253-020-10930-3
Hamed SM et al (2021) Evaluation of the phycoremediation potential of microalgae for captan removal: Comprehensive analysis on toxicity, detoxification and antioxidants modulation. J Hazard Mater. https://doi.org/10.1016/j.jhazmat.2021.128177
Hamed SM, Klöck G (2014) Improvement of medium composition and utilization of mixotrophic cultivation for green and blue green microalgae towards biodiesel production. Adv Microbiol. https://doi.org/10.4236/aim.2014.43022
Han S-F et al (2016) Optimization of aeration for biodiesel production by Scenedesmus obliquus grown in municipal wastewater. Bioprocess Biosyst Eng 39(7):1073–1079. https://doi.org/10.1007/s00449-016-1585-x
Harun R et al (2010) Bioprocess engineering of microalgae to produce a variety of consumer products. Renew Sustain Energy Rev 14(3):1037–1047. https://doi.org/10.1016/j.rser.2009.11.004
Harwood JL et al (2009) The versatility of algae and their lipid metabolism. Biochimie 91(6):679–684. https://doi.org/10.1016/j.biochi.2008.11.004
Hassan SH et al (2012) Effect of different growth conditions on certain biochemical parameters of different cyanobacterial strains. Malays J Microbiol 8(4):266–272
He Y et al (2019) Influences of carbon and nitrogen sources and metal ions on the heterotrophic culture of Scenedesmus sp. LX1. Environ Sci Pollut Res 26(13):13381–13389. https://doi.org/10.1007/s11356-019-04807-w
Heredia-Arroyo T et al (2011) Mixotrophic cultivation of Chlorella Vulgaris and Its potential application for the oil accumulation from non-sugar materials. Biomass Bioenergy 35(5):2245–2253. https://doi.org/10.1016/j.biombioe.2011.02.036
Hillebrand H (1983) Development and dynamics of floating clusters of filamentous algae. In: Periphyton of freshwater ecosystems, Springer. pp 31–39. https://doi.org/10.1007/978-94-009-7293-3_7
Hossain SZJCE et al (2019) Biochemical conversion of microalgae biomass into biofuel. Chem Eng Technol 42(12):2594–2607. https://doi.org/10.1002/ceat.201800605
Hu Q et al (2008) Microalgal triacylglycerols as feedstocks for biofuel production: perspectives and advances. Plant J 54(4):621–639. https://doi.org/10.1111/j.1365-313X.2008.03492.x
Hultberg M et al (2016) Microalgal growth in municipal wastewater treated in an anaerobic moving bed biofilm reactor. Bioresour Technol 207:19–23. https://doi.org/10.1016/j.biortech.2016.02.001
Hwang J-H et al (2016) Enhancement of continuous fermentative bioethanol production using combined treatment of mixed microalgal biomass. Algal Res 17:14–20. https://doi.org/10.1016/j.algal.2016.03.029
Ibrahim FG et al (2020) Carbon dioxide capture from carbon dioxide–rich gases by microalgae. In: From biofiltration to promising options in gaseous fluxes biotreatment. Elsevier. pp 373–396.https://doi.org/10.1016/B978-0-12-819064-7.00018-2
Illman A et al (2000) Increase in Chlorella strains calorific values when grown in low nitrogen medium. Enzyme Microbial Technol 27(8):631–635. https://doi.org/10.1016/S0141-0229(00)00266-0
Jaiswal KK et al (2021) Photosynthetic microalgae–based carbon sequestration and generation of biomass in biorefinery approach for renewable biofuels for a cleaner environment. Biomass Convers Biorefinery. https://doi.org/10.1007/s13399-021-01504-y
Japar AS et al (2017) Harvesting microalgal biomass and lipid extraction for potential biofuel production: a review. J Environ Chem Eng 5(1):555–563. https://doi.org/10.1016/j.jece.2016.12.016
Ji F et al (2015) Fed-batch cultivation of Desmodesmus sp. in anaerobic digestion wastewater for improved nutrient removal and biodiesel production. Bioresour Technol 184:116–122. https://doi.org/10.1016/j.biortech.2014.09.144
Johnson MB et al (2010) Development of an attached microalgal growth system for biofuel production. Appl Microbiol Biotechnol 85(3):525–534. https://doi.org/10.1007/s00253-009-2133-2
Jorquera O et al (2010) Comparative energy life-cycle analyses of microalgal biomass production in open ponds and photobioreactors. Bioresour Technol 101(4):1406–1413. https://doi.org/10.1016/j.biortech.2009.09.038
Joun J et al (2021) Enhanced biomass production through a repeated sequential auto-and heterotrophic culture mode in Chlorella protothecoides. Biores Technol 338:125532. https://doi.org/10.1016/j.biortech.2021.125532
Kannah RY et al (2021a) A mini review of biochemical conversion of algal biorefinery. Energy Fuels 35(21):16995–17007. https://doi.org/10.1021/acs.energyfuels.1c02294
Kannah RY et al (2021b) A review on anaerobic digestion of energy and cost effective microalgae pretreatment for biogas production. Bioresour Technol 332:125055. https://doi.org/10.1016/j.biortech.2021.125055
Karpagam R et al (2021) Review on integrated biofuel production from microalgal biomass through the outset of transesterification route: a cascade approach for sustainable bioenergy. Sci Total Environ 766:144236. https://doi.org/10.1016/j.scitotenv.2020.144236
Kebede-westhead E et al (2003) Production and nutrient removal by periphyton grown under different loading rates of anaerobically digested flushed dairy manure. J Phycol 39(6):1275–1282. https://doi.org/10.1111/j.0022-3646.2003.02-159.x
Khan MI et al (2018) The promising future of microalgae: current status, challenges, and optimization of a sustainable and renewable industry for biofuels, feed, and other products. Microb Cell Fact 17(1):1–21. https://doi.org/10.1186/s12934-018-0879-x
Khetkorn W et al (2017) Microalgal hydrogen production–a review. Bioresour Technol 243:1194–1206. https://doi.org/10.1016/j.biortech.2017.07.085
Khola G et al (2012) Biodiesel production from algae. Pak J Bot 44(1):379–381
Kim G-Y et al (2015) Scenedesmus-based treatment of nitrogen and phosphorus from effluent of anaerobic digester and bio-oil production. Bioresour Technol 196:235–240. https://doi.org/10.1016/j.biortech.2015.07.091
Klinthong W et al (2015) A review: microalgae and their applications in CO2 capture and renewable energy. Aerosol and Air Qual Res 15(2):712–742. https://doi.org/10.4209/aaqr.2014.11.0299
Knothe G (2006) Analyzing biodiesel: standards and other methods. J Am Oil Chem Soc 83(10):823–833. https://doi.org/10.1007/s11746-006-5033-y
Kong Q-X et al (2010) Culture of microalgae Chlamydomonas Reinhardtii in wastewater for biomass feedstock production. Appl Biochem Biotechnol 160(1):9–18. https://doi.org/10.1007/s12010-009-8670-4
Kwietniewska E et al (2014) Process characteristics, inhibition factors and methane yields of anaerobic digestion process, with particular focus on microalgal biomass fermentation. Renew Sustain Energy Rev 34:491–500. https://doi.org/10.1016/j.rser.2014.03.041
Kuo C-T (2011) Harvesting natural algal blooms for concurrent biofuel production and hypoxia mitigation. M.S Thesis. Illinois Digital Environment for Access to Learning and Scholarship. http://hdl.handle.net/2142/18541
Lage S et al (2018) Algal biomass from wastewater and flue gases as a source of bioenergy. Energies 11(3):664. https://doi.org/10.3390/en11030664
Lam MK et al. (2019) Scale-up and commercialization of algal cultivation and biofuels production. In: Biofuels from algae, Elsevier. pp 475–506. https://doi.org/10.1590/1678-4324-2019160816
Lang X et al (2001) Preparation and characterization of bio-diesels from various bio-oils. Bioresour Technol 80(1):53–62. https://doi.org/10.1016/S0960-8524(01)00051-7
Lawton RJ et al (2013) Selecting reliable and robust freshwater macroalgae for biomass applications. PLoS ONE 8(5):e64168. https://doi.org/10.1371/journal.pone.0064168
Lee Y-K (2001) Microalgal mass culture systems and methods: their limitation and potential. J Appl Phycol 13(4):307–315. https://doi.org/10.1023/A:1017560006941
Lee S-J et al (2011) Oil production from five marine microalgae for the production of biodiesel. Biotechnol Bioprocess Eng 16(3):561–566. https://doi.org/10.1007/s12257-010-0360-0
Leite GB et al (2013) Algal biofuels: challenges and opportunities. Biores Technol 145:134–141. https://doi.org/10.1016/j.biortech.2013.02.007
Li X et al (2007) Large-scale biodiesel production from microalga Chlorella protothecoides through heterotrophic cultivation in bioreactors. Biotechnol Bioeng 98(4):764–771. https://doi.org/10.1002/bit.21489
Li X et al (2013) The microalga Parachlorella kessleri––a novel highly efficient lipid producer. Biotechnol Bioeng 110(1):97–107. https://doi.org/10.1002/bit.24595
Li S et al (2021) Microalgae as a solution of third world energy crisis for biofuels production from wastewater toward carbon neutrality: an updated review. Chemosphere. https://doi.org/10.1016/j.chemosphere.2021.132863
Liang Y et al (2009) Biomass and lipid productivities of Chlorella vulgaris under autotrophic, heterotrophic and mixotrophic growth conditions. Biotech Lett 31(7):1043–1049. https://doi.org/10.1007/s10529-009-9975-7
Liu T et al (2013) Attached cultivation technology of microalgae for efficient biomass feedstock production. Bioresour Technol 127:216–222. https://doi.org/10.1016/j.biortech.2012.09.100
Lizzul A et al (2014) Combined remediation and lipid production using Chlorella sorokiniana grown on wastewater and exhaust gases. Bioresour Technol 151:12–18. https://doi.org/10.1016/j.biortech.2013.10.040
Luo L et al (2016) Nutrient removal and lipid production by Coelastrella sp. in anaerobically and aerobically treated swine wastewater. Bioresour Technol 216:135–141. https://doi.org/10.1016/j.biortech.2016.05.059
Mahapatra DM et al (2014) Bioremediation and lipid synthesis through mixotrophic algal consortia in municipal wastewater. Bioresour Technol 168:142–150. https://doi.org/10.1016/j.biortech.2014.03.130
Makareviciene V et al (2011) Cultivation of microalgae Chlorella sp. and Scenedesmus sp. as a potentional biofuel feedstock. Environ Res Eng Manag 57(3):21–27
Mandal S et al (2009) Microalga Scenedesmus obliquus as a potential source for biodiesel production. Appl Microbiol Biotechnol 84(2):281–291. https://doi.org/10.1007/s00253-009-1935-6
Mandotra S et al (2016) Evaluation of fatty acid profile and biodiesel properties of microalga Scenedesmus abundans under the influence of phosphorus, pH and light intensities. Biores Technol 201:222–229. https://doi.org/10.1016/j.biortech.2015.11.042
Martınez M et al (2000) Nitrogen and phosphorus removal from urban wastewater by the microalga Scenedesmus Obliquus. Bioresour Technol 73(3):263–272. https://doi.org/10.1016/S0960-8524(99)00121-2
Mathimani T et al (2018) A comprehensive review on harvesting of microalgae for biodiesel–key challenges and future directions. Renew Sustain Energy Rev 91:1103–1120. https://doi.org/10.1016/j.rser.2018.04.083
Membere E, Sallis P (2018) Effect of temperature on kinetics of biogas production from macroalgae. Bioresour Technol 263:410–417. https://doi.org/10.1016/j.biortech.2018.05.023
Milano J et al (2016) Microalgae biofuels as an alternative to fossil fuel for power generation. Renew Sustain Energy Rev 58:180–197
Mishra V et al (2017) Algal biomass pretreatment for improved biofuel production. In: Algal biofuels, Springer. pp 259–280. https://doi.org/10.1007/978-3-319-51010-1_13
Moazami N et al (2011) Biomass and lipid productivities of marine microalgae isolated from the Persian Gulf and the Qeshm Island. Biomass Bioenergy 35(5):1935–1939. https://doi.org/10.1016/j.biombioe.2011.01.039
Mulbry W et al (2008a) Treatment of dairy manure effluent using freshwater algae: algal productivity and recovery of manure nutrients using pilot-scale algal turf scrubbers. Bioresour Technol 99(17):8137–8142. https://doi.org/10.1016/j.biortech.2008.03.073
Mulbry W et al (2008b) Treatment of dairy and swine manure effluents using freshwater algae: fatty acid content and composition of algal biomass at different manure loading rates. J Appl Phycol 20(6):1079–1085. https://doi.org/10.1007/s10811-008-9314-8
Nabi MN et al (2006) Improvement of engine emissions with conventional diesel fuel and diesel–biodiesel blends. Bioresour Technol 97(3):372–378. https://doi.org/10.1016/j.biortech.2005.03.013
Najjar YS et al (2020) Harvesting of microalgae by centrifugation for biodiesel production: a review. Algal Res 51:102046. https://doi.org/10.1016/j.algal.2020.102046
Narayanan GS et al (2018) Isolation, identification and outdoor cultivation of thermophilic freshwater microalgae Coelastrella sp. FI69 in bubble column reactor for the application of biofuel production. Biocatal Agric Biotechnol 14:357–365. https://doi.org/10.1016/j.bcab.2018.03.022
Neofotis P et al (2016) Characterization and classification of highly productive microalgae strains discovered for biofuel and bioproduct generation. Algal Res 15:164–178. https://doi.org/10.1016/j.algal.2016.01.007
O’Rourke R et al (2016) The effects of Parachlorella kessleri cultivation on brewery wastewater. Water Sci Technol 73(6):1401–1408. https://doi.org/10.2166/wst.2015.618
Órpez R et al (2009) Growth of the microalga Botryococcus Braunii in secondarily treated sewage. Desalination 246(1–3):625–630. https://doi.org/10.1016/j.desal.2008.07.016
Osman AI et al (2021a) Conversion of biomass to biofuels and life cycle assessment: a review. Environ Chem Lett 19(6):4075–4118. https://doi.org/10.1007/s10311-021-01273-0
Osman AI et al (2020) Critical challenges in biohydrogen production processes from the organic feedstocks. Biomass Convers Biorefinery. https://doi.org/10.1007/s13399-020-00965-x
Osman AI et al (2021b) Recent advances in carbon capture storage and utilisation technologies: a review. Environ Chem Lett 19(2):797–849. https://doi.org/10.1007/s10311-020-01133-3
Osman AI et al (2022a) Hydrogen production, storage, utilisation and environmental impacts: a review. Environ Chem Lett 20(1):153–188. https://doi.org/10.1007/s10311-021-01322-8
Osman AI et al (2022) Biochar for agronomy, animal farming, anaerobic digestion, composting, water treatment, soil remediation, construction, energy storage, and carbon sequestration: a review. Environ Chem Lett. https://doi.org/10.1007/s10311-022-01424-x
Park J et al (2011) Wastewater treatment high rate algal ponds for biofuel production. Bioresour Technol 102(1):35–42. https://doi.org/10.1016/j.biortech.2010.06.158
Patil V et al (2008) Towards sustainable production of biofuels from microalgae. Int J Mol Sci 9(7):1188–1195. https://doi.org/10.3390/ijms9071188
Paul S et al (2021) Review on the recent structural advances in open and closed systems for carbon capture through algae. Energy Nexus 4:100032. https://doi.org/10.1016/j.nexus.2021.100032
Perazzoli S et al (2016) Optimizing biomethane production from anaerobic degradation of Scenedesmus spp. biomass harvested from algae-based swine digestate treatment. Int Biodeterior Biodegrad 109:23–28. https://doi.org/10.1016/j.ibiod.2015.12.027
Pienkos PT et al (2009) The promise and challenges of microalgal-derived biofuels. Biofuels Bioproducts Biorefining Innov Sustain Econ 3(4):431–440. https://doi.org/10.1002/bbb.159
Pittman JK et al (2011) The potential of sustainable algal biofuel production using wastewater resources. Bioresour Technol 102(1):17–25. https://doi.org/10.1016/j.biortech.2010.06.035
Pizarro C et al (2006) An economic assessment of algal turf scrubber technology for treatment of dairy manure effluent. Ecol Eng 26(4):321–327. https://doi.org/10.1016/j.ecoleng.2005.12.009
Pragya N et al (2013) A review on harvesting, oil extraction and biofuels production technologies from microalgae. Renew Sustain Energy Rev 24:159–171. https://doi.org/10.1016/j.rser.2013.03.034
Pruvost J et al (2011) Systematic investigation of biomass and lipid productivity by microalgae in photobioreactors for biodiesel application. Bioresour Technol 102(1):150–158. https://doi.org/10.1016/j.biortech.2010.06.153
Rahman K et al (2016) Understanding bioenergy production and optimisation at the nanoscale–a review. J Exp Nanosci 11(10):762–775. https://doi.org/10.1080/17458080.2016.1157905
Ramanna L et al (2014) The optimization of biomass and lipid yields of Chlorella sorokiniana when using wastewater supplemented with different nitrogen sources. Bioresour Technol 168:127–135. https://doi.org/10.1016/j.biortech.2014.03.064
Ramaraj R et al (2010) Algae growth in natural water resources. J Soil Water Conserv 42(4):439–450
Randrianarison G et al (2017) Microalgae: a potential plant for energy production. Geol Ecol Landscapes 1(2):104–120. https://doi.org/10.1080/24749508.2017.1332853
Rawat I et al (2011) Dual role of microalgae: phycoremediation of domestic wastewater and biomass production for sustainable biofuels production. Appl Energy 88(10):3411–3424. https://doi.org/10.1016/j.apenergy.2010.11.025
Razeghifard R (2013) Algal biofuels. Photosynth Res 117(1):207–219. https://doi.org/10.1007/s11120-013-9828-z
Rodolfi L et al (2009) Microalgae for oil: strain selection, induction of lipid synthesis and outdoor mass cultivation in a low-cost photobioreactor. Biotechnol Bioeng 102(1):100–112. https://doi.org/10.1002/bit.22033
Ruiz-Ruiz, P et al. (2020) Carbon dioxide capture and utilization using microalgae. In: Handbook of Microalgae-Based Processes and Products. Elsevier. pp 185–206. https://doi.org/10.1016/B978-0-12-818536-0.00008-7
Sakarika M et al (2019) Chlorella vulgaris as a green biofuel factory: comparison between biodiesel, biogas and combustible biomass production. Bioresour Technol 273:237–243. https://doi.org/10.1016/j.biortech.2018.11.017
Sambusiti C et al (2015) Algae as promising feedstocks for fermentative biohydrogen production according to a biorefinery approach: a comprehensive review. Renew Sustain Energy Rev 44:20–36. https://doi.org/10.1016/j.rser.2014.12.013
Sangapillai K, Marimuthu T (2019) Isolation and selection of growth medium for freshwater microalgae Asterarcys quadricellulare for maximum biomass production. Water Sci Technol 80(11):2027–2036. https://doi.org/10.2166/wst.2020.015
Saraf C, Dutt K (2021) Oleaginous potential of indigenously isolated fresh water microalgae for biofuel production. Int J Sustain Energy. 40(4):326–343. https://doi.org/10.1080/14786451.2020.1806840
Schumacher LG et al (1996) Heavy-duty engine exhaust emission tests using methyl ester soybean oil/diesel fuel blends. Bioresour Technol 57(1):31–36. https://doi.org/10.1016/0960-8524(96)00043-0
Scragg A et al (2002) Growth of microalgae with increased calorific values in a tubular bioreactor. Biomass Bioenergy 23(1):67–73. https://doi.org/10.1016/S0961-9534(02)00028-4
Severo IA et al. (2019) Carbon dioxide capture and use by microalgae in photobioreactors. In Bioenergy with Carbon Capture and Storage, Elsevier. p 151–171. https://doi.org/10.1016/B978-0-12-816229-3.00008-9
Shen X-F et al (2019) Biodiesel production from Chlorella vulgaris under nitrogen starvation in autotrophic, heterotrophic, and mixotrophic cultures. J Appl Phycol 31(3):1589–1596. https://doi.org/10.1007/s10811-019-01765-1
Shukla S et al (2017) Atmospheric carbon sequestration through microalgae: status, prospects, and challenges. Agro-Environ Sustain. https://doi.org/10.1007/978-3-319-49724-2_10
Shurin JB et al (2013) Industrial-strength ecology: trade-offs and opportunities in algal biofuel production. Ecol Lett 16(11):1393–1404. https://doi.org/10.1111/ele.12176
Shuba ES, Kifle D (2018) Microalgae to biofuels: ‘promising’ alternative and renewable energy, review. Renew Sustain Energy Rev 81:743–755. https://doi.org/10.1016/j.rser.2017.08.042
Sialve B et al (2009) Anaerobic digestion of microalgae as a necessary step to make microalgal biodiesel sustainable. Biotechnol Adv 27(4):409–416. https://doi.org/10.1016/j.biotechadv.2009.03.001
Siddiki SYA et al (2022) Microalgae biomass as a sustainable source for biofuel, biochemical and biobased value-added products: an integrated biorefinery concept. Fuel 307:121782. https://doi.org/10.1016/j.fuel.2021.121782
Silva CE et al (2017) Dilute acid hydrolysis of microalgal biomass for bioethanol production: an accurate kinetic model of biomass solubilization, sugars hydrolysis and nitrogen/ash balance. React Kinet Mech Catal 122(2):1095–1114. https://doi.org/10.1007/s11144-017-1279-7
Silva CE et al (2018) A systematic study regarding hydrolysis and ethanol fermentation from microalgal biomass. Biocatal Agric Biotechnol 14:172–182. https://doi.org/10.1016/j.bcab.2018.02.016
Silva CEDF et al (2019) Bioethanol from microalgal biomass: a promising approach in biorefinery. Braz Arch Biol Technol 62:e19160816. https://doi.org/10.1590/1678-4324-2019160816
Singh J et al (2014) Characterization of endolithic cyanobacterial strain, Leptolyngbya sp. ISTCY101, for prospective recycling of CO2 and biodiesel production. Biores Technol 166:345–352. https://doi.org/10.1016/j.biortech.2014.05.055
Singh G et al (2018) Microalgae harvesting techniques: a review. J Environ Manag 217:499–508. https://doi.org/10.1016/j.jenvman.2018.04.010
Sirohi R et al (2021) Strategies and advances in the pretreatment of microalgal biomass. J Biotechnol 341:63–75. https://doi.org/10.1016/j.jbiotec.2021.09.010
Smith VH et al (2010) The ecology of algal biodiesel production. Trends Ecol Evol 25(5):301–309. https://doi.org/10.1016/j.tree.2009.11.007
Song Y et al (2021) SO2 Impurity in simulated flue gas with 15% CO2 affects dynamic bubble dissolution and arthrospira photosynthetic growth. ACS Sustain Chem Eng 9(16):5580–5589. https://doi.org/10.1021/acssuschemeng.0c09197
Sturm BS et al (2012) Controls of microalgal biomass and lipid production in municipal wastewater-fed bioreactors. Environ Prog Sustainable Energy 31(1):10–16. https://doi.org/10.1002/ep.10586
Subhadra BG et al (2011) Coproduct market analysis and water footprint of simulated commercial algal biorefineries. Appl Energy 88(10):3515–3523. https://doi.org/10.1016/j.apenergy.2010.12.051
Subhash GV et al (2014) Deoiled algal cake as feedstock for dark fermentative biohydrogen production: an integrated biorefinery approach. Int J Hydrogen Energy 39(18):9573–9579. https://doi.org/10.1016/j.ijhydene.2014.04.003
Subramanian S et al (2013) Comparative energetics and kinetics of autotrophic lipid and starch metabolism in chlorophytic microalgae: implications for biomass and biofuel production. Biotechnol Biofuels 6(1):1–12. https://doi.org/10.1186/1754-6834-6-150
Sun X et al (2014) Effect of nitrogen-starvation, light intensity and iron on triacylglyceride/carbohydrate production and fatty acid profile of Neochloris oleoabundans HK-129 by a two-stage process. Bioresour Technol 155:204–212. https://doi.org/10.1016/j.biortech.2013.12.109
Surendhiran D et al (2014) Biodiesel production from marine microalga Chlorella salina using whole cell yeast immobilized on sugarcane bagasse. J Environ Chem Eng 2(3):1294–1300. https://doi.org/10.1016/j.jece.2014.05.004
Taher H et al (2014) Effective extraction of microalgae lipids from wet biomass for biodiesel production. Biomass Bioenerg 66:159–167. https://doi.org/10.1016/j.biombioe.2014.02.034
Takagi M et al (2000) Limited feeding of potassium nitrate for intracellular lipid and triglyceride accumulation of Nannochloris sp. UTEX LB1999. Appl Microbiol Biotechnol 54(1):112–117. https://doi.org/10.1007/s002530000333
Takagi M, Yoshida T (2006) Effect of salt concentration on intracellular accumulation of lipids and triacylglyceride in marine microalgae Dunaliella cells. Jbiosci Bioeng 101(3):223–226. https://doi.org/10.1263/jbb.101.223
Tu R et al (2016) Improving of lipid productivity of the biodiesel promising green microalga Chlorella pyrenoidosa via low-energy ion implantation. J Appl Phycol 28(4):2159–2166. https://doi.org/10.1007/s10811-015-0783-2
Tu R et al (2019) Enhancement of microalgal lipid production in municipal wastewater: fixation of CO2 from the power plant tail gas. Biomass Bioenerg 131:105400. https://doi.org/10.1016/j.biombioe.2019.105400
Tumuluru JS et al (2011) A review of biomass densification systems to develop uniform feedstock commodities for bioenergy application. Biofuels Bioproducts Biorefining 5(6):683–707. https://doi.org/10.1002/bbb.324
Tumuluru JS et al (2012) Formulation, pretreatment, and densification options to improve biomass specifications for co-firing high percentages with coal. Ind Biotechnol 8(3):113–132. https://doi.org/10.1089/ind.2012.0004
Ugwu C et al (2008) Photobioreactors for mass cultivation of algae. Bioresour Technol 99(10):4021–4028. https://doi.org/10.1016/j.biortech.2007.01.046
Ullah K et al (2015) Assessing the potential of algal biomass opportunities for bioenergy industry: a review. Fuel 143:414–423. https://doi.org/10.1016/j.fuel.2014.10.064
Ummalyma SB, Sukumaran RK (2014) Cultivation of microalgae in dairy effluent for oil production and removal of organic pollution load. Bioresour Technol 165:295–301. https://doi.org/10.1016/j.biortech.2014.03.028
Vasistha S et al (2021) Current advances in microalgae harvesting and lipid extraction processes for improved biodiesel production: a review. Renew Sustain Energy Rev 137:110498. https://doi.org/10.1016/j.rser.2020.110498
Veerabadhran M et al (2021a) Using different cultivation strategies and methods for the production of microalgal biomass as a raw material for the generation of bioproducts. Chemosphere 285:131436. https://doi.org/10.1016/j.chemosphere.2021.131436
Veerabadhran M et al (2021b) Anaerobic digestion of microalgal biomass for bioenergy production, removal of nutrients and microcystin: Current status. J Appl Microbiol 131(4):1639–1651. https://doi.org/10.1111/jam.15000
Verma R et al (2018) Carbon dioxide sequestration and its enhanced utilization by photoautotroph microalgae. Environ Dev 27:95–106. https://doi.org/10.1016/j.envdev.2018.07.004
Vicente G et al (2004) Integrated biodiesel production: a comparison of different homogeneous catalysts systems. Bioresour Technol 92(3):297–305. https://doi.org/10.1016/j.biortech.2003.08.014
Voloshin RA et al (2016) Biofuel production from plant and algal biomass. Int J Hydrogen Energy 41(39):17257–17273. https://doi.org/10.1016/j.ijhydene.2016.07.084
Voltolina D et al (1999) Growth of Scenedesmus sp. in artificial wastewater. Bioresour Technol 68(3):265–268. https://doi.org/10.1016/S0960-8524(98)00150-3
Wahal S, Viamajala S (2016) Uptake of inorganic and organic nutrient species during cultivation of a Chlorella isolate in anaerobically digested dairy waste. Biotechnol Prog 32(5):1336–1342. https://doi.org/10.1002/btpr.2313
Wang B et al (2012) Closed photobioreactors for production of microalgal biomasses. Biotechnol Adv 30(4):904–912. https://doi.org/10.1016/j.biotechadv.2012.01.019
Wang H et al (2014a) Joint production of biodiesel and bioethanol from filamentous oleaginous microalgae Tribonema sp. Bioresour Technol 172:169–173. https://doi.org/10.1016/j.biortech.2014.09.032
Wang X-W et al (2014b) Biomass, total lipid production, and fatty acid composition of the marine diatom Chaetoceros muelleri in response to different CO2 levels. Bioresour Technol 161:124–130. https://doi.org/10.1016/j.biortech.2014.03.012
Wang Z et al (2020) Spermidine protects Chlorella sp. from oxidative damage caused by SO2 in flue gas from coal-fired power plants. ACS Sustain Chem Eng 8(40):15179–15188. https://doi.org/10.1021/acssuschemeng.0c04527
Wei N et al (2013) Marine macroalgae: an untapped resource for producing fuels and chemicals. Trends Biotechnol 31(2):70–77. https://doi.org/10.1016/j.tibtech.2012.10.009
Wiley PE et al (2011) Production of biodiesel and biogas from algae: a review of process train options. Water Environ Res 83(4):326–338. https://doi.org/10.2175/106143010X12780288628615
Woertz I et al (2009) Algae grown on dairy and municipal wastewater for simultaneous nutrient removal and lipid production for biofuel feedstock. J Environ Eng 135(11):1115–1122. https://doi.org/10.1061/(ASCE)EE.1943-7870.0000129
Xiong W et al (2008) High-density fermentation of microalga Chlorella protothecoides in bioreactor for microbio-diesel production. Appl Microbiol Biotechnol 78(1):29–36. https://doi.org/10.1007/s00253-007-1285-1
Xu H et al (2006) High Quality Biodiesel Production from a Microalga Chlorella Protothecoides by Heterotrophic Growth in Fermenters. J Biotechnol 126(4):499–507. https://doi.org/10.1016/j.jbiotec.2006.05.002
Xu X et al (2019) Progress, challenges and solutions of research on photosynthetic carbon sequestration efficiency of microalgae. Renew Sustain Energy Rev 110:65–82. https://doi.org/10.1016/j.rser.2019.04.050
Yadav G et al (2019) A biorefinery for valorization of industrial waste-water and flue gas by microalgae for waste mitigation, carbon-dioxide sequestration and algal biomass production. Sci Total Environ 688:129–135. https://doi.org/10.1016/j.scitotenv.2019.06.024
Yang L et al (2015) Nutrients removal and lipids production by Chlorella pyrenoidosa cultivation using anaerobic digested starch wastewater and alcohol wastewater. Bioresour Technol 181:54–61. https://doi.org/10.1016/j.biortech.2015.01.043
Yoo C et al (2010) Selection of microalgae for lipid production under high levels carbon dioxide. Bioresour Technol 101(1):S71–S74. https://doi.org/10.1016/j.biortech.2009.03.030
Yun J-H et al (2014) Freshwater macroalgae as a biofuels feedstock: mini-review and assessment of their bioenergy potential. Ind Biotechnol 10(3):212–220. https://doi.org/10.1089/ind.2013.0033
Yun J-H (2014b) The potential of freshwater macroalgae as a biofuels feedstock and the influence of nutrient availability on freshwater macroalgal biomass production. University of Kansas.
Zaidi AA et al (2018) Nanoparticles augmentation on biogas yield from microalgal biomass anaerobic digestion. Int J Hydrogen Energy 43(31):14202–14213. https://doi.org/10.1016/j.ijhydene.2018.05.132
Zhang C et al (2014) Strategic enhancement of algal biomass, nutrient uptake and lipid through statistical optimization of nutrient supplementation in coupling Scenedesmus obliquus-like microalgae cultivation and municipal wastewater treatment. Bioresour Technol 171:71–79. https://doi.org/10.1016/j.biortech.2014.07.060
Zhou W et al (2017) Bio-mitigation of carbon dioxide using microalgal systems: advances and perspectives. Renew Sustain Energy Rev 76:1163–1175. https://doi.org/10.1016/j.rser.2017.03.065
Zhu L et al (2014) Biodiesel production from algae cultivated in winter with artificial wastewater through pH regulation by acetic acid. Appl Energy 128:103–110. https://doi.org/10.1016/j.apenergy.2014.04.039
Acknowledgements
The authors wish to acknowledge the support of The Bryden Centre project (Project ID VA5048), which was awarded by The European Union’s INTERREG VA Programme, managed by the Special EU Programmes Body (SEUPB), with match funding provided by the Department for the Economy in Northern Ireland and the Department of Business, Enterprise and Innovation in the Republic of Ireland. The views and opinions expressed in this review do not necessarily reflect those of the European Commission or the Special EU Programmes Body (SEUPB).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Sarwer, A., Hamed, S.M., Osman, A.I. et al. Algal biomass valorization for biofuel production and carbon sequestration: a review. Environ Chem Lett 20, 2797–2851 (2022). https://doi.org/10.1007/s10311-022-01458-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10311-022-01458-1