Abstract
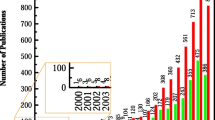
The electrochemistry research team activity from Poland is marked by significant increase in the last 20 years. The joining of European Community in 2004 gives an impulse for the development of Polish science. The development of electrochemistry has been stimulated by cooperation with industry and the establishment of technology transfer centers, technology parks, business incubators, etc. and the mostly by simplified international collaborations. Five research institutions from Krakow reports work in the field of electrochemistry. The achievements of all teams are briefly described.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The political transformation in 1989 strongly influences the development of Polish science, through its connection with industry and the establishment of technology transfer centers, technology parks, business incubators, etc. [1]. Then, joining the European Union by Poland in 2004 significantly simplified international collaborations. These days, there are many opportunities for Polish scientists to receive funds for projects in cooperation with foreign centers. Researchers can apply for a scientific stay abroad. It is possible thanks to the Polish Science Centre (NCN), the National Centre for Research and Development (NCBR), and the Polish National Agency for Academic Exchange (NAWA). Nowadays, there are almost 380 public and private universities in Poland divided into academic and vocational institutions [2]. Additionally, in 2010, due to the reform of science, research and development units were transformed into research institutes. Sources from 2015 reported that there are 115 research institutes, 58% of which work in the field of science and engineering and 34% in life sciences [3]. There are 70 research institutes of the Polish Academy of Sciences (PAN). Political transformation connected with opening to collaboration with foreign research groups and injection of funds to build or buy advanced research instrumentation results in blooming of research activity in a field of electrochemistry.
Only in Krakow, five research institutions report work in the field of electrochemistry. The achievements of all teams, over the last 20 years, are briefly described below. The research area of mentioned laboratory groups is strongly connected with the field of electrochemistry. The units are divided by the main research centers.
Electrochemistry at Krakowian research facilities
AGH university of science and technology
The AGH University of Science and Technology (the AGH UST) is a public university strongly involved in many national and international research projects. It is composed of sixteen faculties and one research institute. The research in the field of electrochemistry is performed at six of them. The biggest group works at the Department of Physiochemistry and Metallurgy of Non-Ferrous Metals at the Faculty of Non-Ferrous Metals. In the first years of the 2000s, the research teams guided by professors Lidia Burzyńska, Wanda Gumowska, and Krzysztof Fitzner performed intensive experiments in the field of, e.g., anodic dissolution [4,5,6,7], metal recovery [8, 9], and electrodeposition of transition group composites [10,11,12] and metal alloys [13, 14]. Then, with the growth of the research group, many new directions of interest have appeared, such as catalytic properties of transition group metals and alloys [15,16,17,18,19], tellurium thin films [20, 21], and synthesis of nanostructures in the anodic alumina oxide templates [22,23,24]. The influence of the magnetic field on the properties of the electrodeposited coatings has been investigated [25,26,27]. Nowadays, 3 groups guided by professors Ewa Rudnik, Remigiusz Kowalik, and Piotr Żabiński can be distinguished. Professor Żabiński is also a head of the department. Under their supervision, young scientists and Ph.D. students are working on the co-deposition of chalcogenides with noble [28] and transition metals [29, 30]. The synthesis of noble and transition metal alloys [31, 32] has been still developing. The synthesis of Zn–Ni [33], Zn–Mg [34], and Zn–Ni–Mg [35] from gluconate baths has been also investigated. Besides that, the metallization of 3D prints [36,37,38] and electrochemical synthesis of 1D nanostructures by the one-step method [39,40,41,42] have been studied. The superhydrophobic properties of coatings are also in interest of scientists [43]. Research groups have been collaborating with international groups from Germany [44,45,46,47], Japan [48,49,50,51,52], France [53], Algeria [54], Bulgaria [55, 56], Italy [57], Serbia [58, 59], and Turkey [32]. In 2015, one of the members of the Department, Professor Krzysztof Mech, moved to the Academic Centre for Materials and Nanotechnology to perform his own research. In his experiments, he mostly focuses on the electrodeposition of Ni coatings [60] and composites [61]. He took under the consideration also the influence of the magnetic field on, interalia, the deposition rate, composition, current efficiency, structure, surface states, and morphology of the synthesized materials [62,63,64].
At the Faculty of Metals Engineering and Industrial Computer Science, Professor Tomasz Moskalewicz has been the head of the project entitled “Development of electrophoretic co-deposition of bioactive and antibacterial ceramics with biodegradable polymers to produce novel composite coatings for biomedical applications.” The project has been funded by NCN within the Beethoven competition. The research is focused on the development and characterization of novel composite coatings exhibiting bioactive and antibacterial functionalities for orthopedic and dental applications employing electrophoretic deposition (EPD) [65]. The experiments are performed in the collaboration with Professor Aldo R. Boccaccini from the Institute of Biomaterials at the University of Erlangen-Nuremberg.
Several research groups work at the Faculty of Materials Science and Ceramics in three different Departments. Professor Tomasz Brylewski, from the Department of Physical Chemistry and Modelling, is the co-author of the reviews about the hydrogen market, research, and development progress in central and Eastern European countries [66, 67]. With other researchers, he also performed experiments strongly connected with the solid oxide fuel cells (SOFC) operating in the higher temperatures [68,69,70]. The influence of applied solid electrolyte on the properties of obtained composites was investigated [71]. With the groups from German and Italian institutes, the first prototypes of dual membrane cell were electrochemically tested [72]. Another person, at the Department of Physical Chemistry and Modelling, performing some experiments connected with the field of the electrochemistry is Professor Robert Filipek. With other co-authors, he modeled corrosion of steel [73] and electrodiffusion processes for ion-selective electrodes [74]. Generally, in his works, he focuses mostly on simulations of diffusion in various materials at higher temperatures [75,76,77]. At the Department of Analytical Chemistry and Biochemistry, the research teams were guided and formed by professors, inter alia, Bogusław Józef Baś, Władysław Wojciech Kubiak, Andrzej Lewenstam, Jan Migdalski, and Mieczysłąw Rękas. They work, e.g., on the design and calibration of measuring equipment for electrochemistry and applications of electrochemical sensors [78,79,80,81], including conducting polymers [82, 83] and ion-selective membrane electrodes [84]. The investigation of voltammetric methods and their improvement was performed [85, 86]. The development of new signal processing algorithms for analytical chemistry research methodologies [87,88,89,90,91] is also an important issue for scientists. The topic of SOFC [92] is explored as well. There are three research groups at the Department of Inorganic Chemistry related to electrochemistry. Scientists perform research in synthesizing of ceramic, metallic, and composite layers, as well as materials based on intermetallic phases [93, 94]. They determine the physical and chemical properties of the coatings, including the resistance to high-temperature oxidation [95, 96] and corrosion in electrolyte solutions [97]. They study the kinetics and mechanism of diffusion [98, 99]. Additionally, the research related to the development of materials such as compounds with the perovskite structure [100, 101], composite, including carbon–metal oxide systems [102], thermoelectric materials [103], and materials with catalytic properties [101], is performed. Also, the topics related to the materials, including transition metal oxides in the form of micro- and nanomaterials and thin layers, for the various applications, such as anodes for photoelectrochemical cells (PEC) [104,105,106], and semiconductor gas sensors [107, 108] are explored.
Researchers from the Faculty of Energy and Fuels at the AGH UST focus on the currently developed issues of Li-ion [109, 110] and Na-ion batteries [111, 112], SOFC [113, 114], oxygen storage [115, 116], etc. Professor Janina Molenda is the head of the Department of Hydrogen at this faculty. Besides the mentioned topics expanded at the faculty, her research interests relate to, among other things, catalytic properties of perovskite oxides [117, 118] and structural defects in solid electrolytes [119, 120]. Professor Konrad Świerczek, the Deputy Dean for Science at the Faculty of Energy and Fuels, also pays attention to oxide materials, their characterization, and measurements of their catalytic activity [121,122,123]. He works with ceramic membranes possessing mixed ionic-electronic conductivity [124]. He has collaborated with scientists from, inter alia, China [125, 126], Japan [127], and the USA [128].
The last unit at the AGH UST where the research connected with the electrochemistry is performed is the Faculty of Foundry Engineering represented by the group from the Department of Metal Chemistry and Corrosion governed now by Professor Maria Starowicz. The research is carried out by great specialists in the field of corrosion on the micro- [129,130,131] and nanoscale [132], as well as in Krakow water supply network [133]. Professor Halina Krawiec was awarded inter alia by DCNS-French Embassy-French Institute for her scientific achievements in the field of corrosion. Professor Urszula Lelek-Borkowska focuses on, inter alia, the electrochemistry of metals and semiconductors in alcohol solutions [134]. The scientists at the department focus on synthesis of nanomaterials [135,136,137] and electrochemical behavior of metals [138, 139] and semiconductors [140, 141] in aqueous and organic electrolyte solutions. The research group also works on modern directions such as green chemistry and biomaterials [142, 143]. The team has collaborated with the group from, inter alia, France [144,145,146], Germany [147], UK [148], Austria [149], and Spain [150].
Faculty of chemistry jagiellonian university
Jagiellonian University is the oldest university in Poland and one of the oldest in Europe [151]. It consists of 16 faculties. The team specialized in Electrochemistry, previously guided by Professor Marian Jaskuła, and now by Professor Grzegorz Sulka, and has been working at the Faculty of Chemistry. The group has impressive achievements in fields of anodization of Al [152,153,154,155]; electrochemical synthesis of nanostructured oxides of, e.g., Sn [156, 157] and Zn [158, 159]; and nanoporous titanium dioxide [160,161,162]. The obtained anodic alumina oxide (AAO) templates are widely used for the fabrication of nanostructured electrodes of metals [163], alloys [164, 165], and polymers [166, 167]. Produced nanostructures and oxides can be applied in catalysis [168], photoelectrochemistry, and photocatalysis [169,170,171,172,173,174], as H2O2 sensors [175,176,177,178], semiconductors [158, 179], and in biomedical applications [180,181,182,183,184]. The research group worked also on the synthesis of Au [185, 186] and Au–Ag [187] for electrochemical epinephrine sensing. Furthermore, the team works on bioelectrodes [188] and materials for energy storage [189]. The scientists have attended many projects funded by NCN, NAWA, the Polish Ministry of Science and Higher Education (MNiSW), and the European Cooperation in Science and Technology (COST). They have collaborated with groups from, e.g., the Czech Republic [190], the USA [191], Portugal [192], and Ukraine [193].
Jerzy haber institute of catalysis and surface chemistry of the polish academy of sciences
The research aim for scientists from the Jerzy Haber Institute of Catalysis and Surface Chemistry is the application of obtained results in fields of protection of health, environment, and cultural heritage, as well as to improve technological processes [194]. One of the scientists working in the field of electrochemistry was Professor Alicja Drelinkiewicz. With other researchers, she performed experiments, inter alia, on the synthesis and characterization of mono- and bimetallic-supported catalysts [195,196,197]. Professor Paweł Nowak and his research group have worked on the synthesis of metal alloys and metal/ceramic composites by electrodeposition [198,199,200,201]. They have investigated the prevention of corrosion of metal and its alloys by using anti-corrosion coatings [202]. The research group is also a specialist in surface doping of titanium dioxide with transition metals and transition metal ions and their photocatalytic properties [203,204,205,206]. The group tests also materials for SOFC [207,208,209]. In 2000, the American Electroplaters and Surface Finishers Society awarded Professor Nowak for an outstanding work published in 1999 in the Plating and Surface Finishing magazine published by this society. He has collaborated with research groups from, inter alia, Germany [210, 211], the USA [212,213,214], Finland [215], and Switzerland [216].
Institute of metallurgy and materials science of the polish academy of sciences
In the Institute of Metallurgy and Materials, two research groups, under the supervision of Professors Ewa Bełtowska-Lehman and Piotr Ozga, can be recognized. Professor Bełtowska-Lehman is a specialist in the kinetics and mechanism of the processes occurring in complex electrolytes during electrocrystallization of metal, alloy, and composite coatings with increased tribological and anti-corrosion properties [217, 218]. The group performed research in electrochemical synthesis and micromechanical properties of nanocomposite coatings with a metal matrix containing the addition of a refractory metal, reinforced with a nanodisperse ceramic phase [219,220,221]. Professor Bełtowska-Lehman works also on photovoltaic silicon cells [222, 223]. In 2006, she was nominated by the MNiSW for the position of Polish representative in the Mirror Group of the European Union Technology Platform in the field of photovoltaics. Professor Ozga works on, inter alia, the electrochemical deposition of materials for different applications, such as lead-free solders (e.g., Sn–Ag and Sn–Ag–Cu [224], In–Sn [225], Sn–Zn–Cu [226]), and solder nanocomposites, alloys, and protective layers of high corrosion resistance, e.g., Zn–Ni [227, 228], are investigated by Professor and his team. He collaborates with researchers from Israel [229] and Ukraine [230].
Łukasiewicz research network — Krakow institute of technology
Krakow Institute of Technology is a part of the Łukasiewicz Research Network, the third largest research network in Europe [231]. At the Institute of Advanced Manufacturing Technology in Krakow, Doctor Maria Zybura-Skrabalak was working on electrochemical machining (ECM). The experiments were performed within the INNOTECH project. The aim of the project was the implementation of the proposed process. The investigation of the influence, inter alia, of the features of ECM processes on the surface roughness of the workpiece material was performed [232]. The modeling of the process considering the application of the flat rectangular universal electrode in the ECM [233] or current in the ECDM (electrochemical discharge machining) process [234] was made among others by Doctor Zybura-Skrabalak. In her work, she also investigated the possible application of the electrode, made by the milling process, for electrochemical smoothing [235].
Conclusions
Krakow is the headquarter of many research groups working in the field of electrochemistry. There are a few common topics for all, e.g., electrodeposition of metal, its alloys, and composites; the phenomenon of the corrosion and enhancement of anti-corrosion properties of coatings; and catalytic activity of the samples. The new directions, such as photovoltaics, biomaterials, and green chemistry, are in accordance with the interests of scientists all over the world. The teams collaborate with research facilities from various countries but also with each other. It results in high-quality results, new projects, and implementations.
References
Historia nauki polskiej. https://pl.wikipedia.org/wiki/Historia_nauki_polskiej
Higher Education Institutions. https://study.gov.pl/higher-education-institutions. Accessed 12 Oct 2022
NIK o instytutach badawczych. https://www.nik.gov.pl/aktualnosci/nik-o-instytutach-badawczych.html
Burzyńska L, Zabiński P (2000) Analysis of the composition and morphology of products of the anodic dissolution of Cu2S. Arch Metall 45:286–302
Burzyńska L (2001) Comparison of the spontaneous and anodic processes during dissolution of brass. Corros Sci 43:1053–1069. https://doi.org/10.1016/S0010-938X(00)00130-X
Burzyńska L, Gumowska W, Rudnik E (2004) Influence of the composition of Cu–Co–Fe alloys on their dissolution in ammoniacal solutions. Hydrometallurgy 71:447–455. https://doi.org/10.1016/j.hydromet.2003.08.007
Burzyńska L, Gumowska W, Rudnik E, Partyka J (2008) Mechanism of the anodic dissolution of Cu70–Co4–Fe14–Pb7 alloy originated from reduced copper converter slag in an ammoniacal solution. Hydrometallurgy 92:34–41. https://doi.org/10.1016/j.hydromet.2008.01.009
Burzyńska L, Rudnik E, Gumowska W (2004) The influence of phase structure on the dissolution of Cu–Co–Fe alloys in sulphuric acid solution and the metals recovery. Hydrometallurgy 71:457–463. https://doi.org/10.1016/S0304-386X(03)00084-7
Burzyńska L, Rudnik E, Barteczko P (2006) Recovery of copper and cobalt from low copper Cu-Co-Fe alloy. Arch Metall Mater 51:299–308
Burzyńska L, Rudnik E, Błaz L et al (2003) The influence of current density and bath composition on the electrodeposition of nickel and nickel/silicon carbide composite. Trans IMF 81:193–198. https://doi.org/10.1080/00202967.2003.11871539
Burzyńska L, Rudnik E, Koza J et al (2008) Electrodeposition and heat treatment of nickel/silicon carbide composites. Surf Coatings Technol 202:2545–2556. https://doi.org/10.1016/j.surfcoat.2007.09.020
Rudnik E, Burzyńska L, Jędruch J, Błaż L (2009) Codeposition of SiC particles with electrolytic cobalt in the presence of Cs+ ions. Appl Surf Sci 255:7164–7171. https://doi.org/10.1016/j.apsusc.2009.03.053
Burzyńska L, Rudnik E (2000) The influence of electrolysis parameters on the composition and morphology of Co–Ni alloys. Hydrometallurgy 54:133–149. https://doi.org/10.1016/S0304-386X(99)00060-2
Burzyńska L, Rudnik E (2000) The influence of saccharin and sodium lauryl sulfate on the electrodeposition process of Co-Ni alloys. Arch Metall 45:276–285
Zabinski PR, Jarek A, Kowalik R (2009) Effect of applied external magnetic field on electrodeposition of cobalt alloys for hydrogen evolution in 8MNaOH. Magnetohydrodynamics 45:275–280. https://doi.org/10.22364/mhd.45.2.18
Zabinski P, Górski M, Kowalik R (2009) Influence of superimposed external magnetic fHiLD onto electrodeposition of co-P alloys for hydrogen evolution. Arch Metall Mater 54:1157–1166
Kowalik R, Żabiński P, Fitzner K (2008) Electrodeposition of ZnSe. Electrochim Acta 53:6184–6190. https://doi.org/10.1016/j.electacta.2007.12.009
Zabinski P, Franczak A, Kowalik R (2012) Electrodeposition of functional Ni-Re alloys for hydrogen evolution. ECS Trans 41:39–48. https://doi.org/10.1149/1.3702411
Zabiński PR, Kowalik R, Piwowarczyk M (2007) Cobalt-tungsten alloys for hydrogen evolution in hot 8 M NaOH. Arch Metall Mater 52:627–634
Kowalik R, Kutyła D, Mech K, Żabiński P (2016) Analysis of tellurium thin films electrodeposition from acidic citric bath. Appl Surf Sci 388:817–824. https://doi.org/10.1016/j.apsusc.2016.03.127
Kowalik R, Kutyła D, Mech K et al (2015) Electrowinning of tellurium from acidic solutions. Arch Metall Mater 60:591–596. https://doi.org/10.1515/amm-2015-0178
Dobosz I, Gumowska W, Czapkiewicz M (2013) Magnetic properties of Co-Fe nanowires electrodeposited in pores of alumina membrane. Arch Metall Mater 58:663–671. https://doi.org/10.2478/amm-2013-0052
Gumowska W, Dobosz I, Wrzoszczyk B (2014) The morphology of the alumina films formed tn the anodization process of aluminium in the orthophosphoric acid solution. The Co-Fe alloys electrodeposition into obtained alumina pores. Arch Metall Mater 59:137–143. https://doi.org/10.2478/amm-2014-0022
Dobosz I, Kutyła D, Kac M et al (2020) The influence of homogenous external magnetic field on morphology and magnetic properties of CoRu nanowire arrays. Mater Sci Eng B Solid-State Mater Adv Technol 262:. https://doi.org/10.1016/j.mseb.2020.114795
Electrocatalytic properties of alloys deposited with superimposed magnetic field (2012) Magnetohydrodynamics 48:243–250. https://doi.org/10.22364/mhd.48.2.1
Zabiński PR, Franczak A, Kowalik R (2012) Electrocatalytically active Ni-Re binary alloys electrodeposited with superimposed magnetic field. Arch Metall Mater 57:495–501. https://doi.org/10.2478/v10172-012-0051-2
Magnetic field effect on properties of galvanostatically deposited Co-Pd alloys (2014) Magnetohydrodynamics 50:75–82. https://doi.org/10.22364/mhd.50.1.8
Wojtysiak M, Jędraczka A, Stępień M et al (2021) Electrodeposition of Pd–Se thin films. Electrochem commun 127:107053. https://doi.org/10.1016/j.elecom.2021.107053
Kwiecińska AM, Kutyła D, Kołczyk-Siedlecka K et al (2019) Electrochemical analysis of co-deposition cobalt and selenium. J Electroanal Chem 848:113278. https://doi.org/10.1016/j.jelechem.2019.113278
Kowalik R, Szaciłowski K, Żabiński P (2012) Photoelectrochemical study of ZnSe electrodeposition on Cu electrode. J Electroanal Chem 674:108–112. https://doi.org/10.1016/j.jelechem.2012.03.002
Kutyła D, Kołczyk-Siedlecka K, Kwiecińska A et al (2019) Preparation and characterization of electrodeposited Ni-Ru alloys: morphological and catalytic study. J Solid State Electrochem 23:3089–3097. https://doi.org/10.1007/s10008-019-04374-7
Kutyła D, Salcı A, Kwiecińska A et al (2020) Catalytic activity of electrodeposited ternary Co–Ni–Rh thin films for water splitting process. Int J Hydrogen Energy 45:34805–34817. https://doi.org/10.1016/j.ijhydene.2020.05.196
Chat-Wilk K, Rudnik E, Włoch G, Osuch P (2022) Codeposition of zinc with nickel from gluconate solutions. J Solid State Electrochem 26:1715–1731. https://doi.org/10.1007/s10008-022-05205-y
Chat-Wilk K, Rudnik E, Włoch G, Osuch P (2022) Codeposition of zinc with manganese from different gluconate baths. Russ J Electrochem 58:168–183. https://doi.org/10.1134/S1023193522030053
Chat-Wilk K, Rudnik E, Włoch G (2022) Effect of chloride and sulfate ions on electrodeposition and surface properties of alloys produced from zinc-nickel-manganese gluconate baths. J Electrochem Soc 169:092515. https://doi.org/10.1149/1945-7111/ac8eec
Żabiński R, Skibińsk K-SK, Kutył K, Kwiecińsk D, Kowali A (2019) Influence of magnetic field on electroless metallization of 3D prints by copper and nickel. Arch Metall Mater 64:17–22. https://doi.org/10.24425/amm.2019.126212
Żabiński R, Zborowsk KK, Kutył W, Kwiecińsk D, Kowali A (2018) Investigation of two-step metallization process of plastic 3D prints fabricated by SLA method. Arch Metall Mater 63:1031–1036. https://doi.org/10.24425/122438
Kołczyk-Siedlecka K, Kutyła D, Skibińska K et al (2021) Well-ordered 3D printed Cu/Pd- decorated catalysts for the methanol electrooxidation in alkaline solutions. Technologies 9:6. https://doi.org/10.3390/technologies9010006
Skibińska K, Wojtaszek K, Krause L et al (2023) Tuning up catalytical properties of electrochemically prepared nanoconical Co-Ni deposit for HER and OER. Appl Surf Sci 607:155004. https://doi.org/10.1016/j.apsusc.2022.155004
Skibińska K, Kornaus K, Yang X et al (2022) One-step synthesis of the hydrophobic conical Co-Fe structures – the comparison of their active areas and electrocatalytic properties. Electrochim Acta 140127. https://doi.org/10.1016/j.electacta.2022.140127
Skibinska K, Kolczyk-Siedlecka K, Kutyla D et al (2021) Electrocatalytic properties of Co nanoconical structured electrodes produced by a one-step or two-step method. Catalysts 11:544. https://doi.org/10.3390/catal11050544
Skibińska K, Semeniuk S, Kutyła D et al (2021) Study on synthesis and modification of conical Ni structures by one-step method. 66:861–869. https://doi.org/10.24425/amm.2021.136391
Rudnik E, Chat K (2018) A brief review on bio-inspired superhydrophobic electrodeposited nickel coatings. Trans IMF 96:185–192. https://doi.org/10.1080/00202967.2018.1475931
Skibińska K, Huang M, Mutschke G et al (2022) On the electrodeposition of conically nano-structured nickel layers assisted by a capping agent. J Electroanal Chem 904:115935. https://doi.org/10.1016/j.jelechem.2021.115935
Huang M, Eckert K, Mutschke G (2021) Magnetic-field-assisted electrodeposition of metal to obtain conically structured ferromagnetic layers. Electrochim Acta 365:137374. https://doi.org/10.1016/j.electacta.2020.137374
Skibińska K, Kutyła D, Yang X et al (2022) Rhodium-decorated nanoconical nickel electrode synthesis and characterization as an electrochemical active cathodic material for hydrogen production. Appl Surf Sci 592:153326. https://doi.org/10.1016/j.apsusc.2022.153326
Koza JA, Mühlenhoff S, Żabiński P et al (2011) Hydrogen evolution under the influence of a magnetic field. Electrochim Acta 56:2665–2675. https://doi.org/10.1016/j.electacta.2010.12.031
Zabinski PR, Meguro S, Asami K, Hashimoto K (2006) Electrodeposited Co-Ni-Fe-C alloys for hydrogen evolution in a hot 8 kmol·m-3 NaOH. Mater Trans 47:2860–2866. https://doi.org/10.2320/matertrans.47.2860
Zabinski PR, Meguro S, Asami K, Hashimoto K (2003) Electrodeposited Co-Fe and Co-Fe-C alloys for hydrogen evolution in a Hot 8 kmol m-3 NaOH solution. Mater Trans 44:2350–2355. https://doi.org/10.2320/matertrans.44.2350
Zabinski PR, Nemoto H, Meguro S et al (2003) Electrodeposited Co-Mo-C cathodes for hydrogen evolution in a hot concentrated NaOH solution. J Electrochem Soc 150:C717. https://doi.org/10.1149/1.1604788
Zabinski PR, Meguro S, Asami K, Hashimoto K (2004) Electrodeposited Co-Fe and Co-Fe-C alloys for hydrogen evolution in a Hot 8 kmol m-3 NaOH solution. J Japan Inst Met 68:456–461. https://doi.org/10.2320/jinstmet.68.456
Hashimoto K, Kumagai N, Izumiya K et al (2013) The use of renewable energy in the form of methane via electrolytic hydrogen generation / Zastosowanie Odnawialnej Energii W Formie Metanu Na Drodze Elektrolitycznej Produkcji Wodoru. Arch Metall Mater 58:231–239. https://doi.org/10.2478/v10172-012-0179-0
Aaboubi O, Douglade J, Zabinski P, Chopart J (2012) Magnetic field effect on molybdenum-based alloys 48:271–278
Chouchane S, Levesque A, Zabinski P et al (2010) Electrochemical corrosion behavior in NaCl medium of zinc–nickel alloys electrodeposited under applied magnetic field. J Alloys Compd 506:575–580. https://doi.org/10.1016/j.jallcom.2010.07.099
Dobrovolska T, Krastev I, Żabiński P et al (2011) Oscillations and self-organization phenomena during electrodeposition of silver-indium alloys. Experimental Study. Arch Metall Mater 56:. https://doi.org/10.2478/v10172-011-0070-4
Dobrovolska T, Kowalik R, Zabinski P, Krastev I (2008) Investigations of the surface morphology of electrodeposited Ag-In coatings by means of optical, scanning-electron and atomic-force microscopy. Bulg Chem Commun 40:254–260
Kołczyk-Siedlecka K, Bernasconi R, Lucotti A et al (2020) Self-assembled monolayers assisted all wet metallization of SU-8 negative tone photoresist. J Electrochem Soc 167:142506. https://doi.org/10.1149/1945-7111/abc844
Elezovic N, Zabinski P, Krstajic-Pajic M et al (2018) Electrochemical deposition and characterization of AgPd alloy layers. J Serbian Chem Soc 83:593–609. https://doi.org/10.2298/JSC171103011E
Lačnjevac U, Vasilić R, Tokarski T et al (2018) Deposition of Pd nanoparticles on the walls of cathodically hydrogenated TiO2 nanotube arrays via galvanic displacement: a novel route to produce exceptionally active and durable composite electrocatalysts for cost-effective hydrogen evolution. Nano Energy 47:527–538. https://doi.org/10.1016/j.nanoen.2018.03.040
Mech K (2017) Influence of organic ligands on electrodeposition and surface properties of nickel films. Surf Coatings Technol 315:232–239. https://doi.org/10.1016/j.surfcoat.2017.02.042
Mech K (2019) Electrodeposition of composite Ni-TiO2 coatings from aqueous acetate baths. Metall Mater Trans A 50:4275–4287. https://doi.org/10.1007/s11661-019-05325-7
Mech K (2019) A novel magnetoelectrochemical method of synthesis of photoactive Ni-TiO2 coatings from glycinate electrolytes. Mater Des 182:108055. https://doi.org/10.1016/j.matdes.2019.108055
Mech K, Gajewska M, Marzec M, Szaciłowski K (2020) On the influence of magnetic field on electrodeposition of Ni–TiO2 composites from a citrate baths. Mater Chem Phys 255:123550. https://doi.org/10.1016/j.matchemphys.2020.123550
Mech K (2020) MHD supported electroreduction of formate nickel complexes with simultaneous incorporation of TiO2 particles. Arch Metall Mater 65:219–227. https://doi.org/10.24425/amm.2019.131118
Moskalewicz T, Warcaba M, Łukaszczyk A et al (2022) Electrophoretic deposition, microstructure and properties of multicomponent sodium alginate-based coatings incorporated with graphite oxide and hydroxyapatite on titanium biomaterial substrates. Appl Surf Sci 575:151688. https://doi.org/10.1016/j.apsusc.2021.151688
Iordache I, Bouzek K, Paidar M et al (2019) The hydrogen context and vulnerabilities in the central and Eastern European countries. Int J Hydrogen Energy 44:19036–19054. https://doi.org/10.1016/j.ijhydene.2018.08.128
Stygar M, Brylewski T (2017) Hydrogen-based energy market in Poland. In: Hydrogen in an International Context: Vulnerabilities of Hydrogen Energy in Emerging Markets. River Publishers, pp 105–126
Brylewski T, Przybylski K (2008) Perovskite and spinel functional coatings for SOFC metallic interconnects. Mater Sci Forum 595–598:813–822. https://doi.org/10.4028/www.scientific.net/MSF.595-598.813
Kruk A, Stygar M, Brylewski T (2013) Mn–Co spinel protective–conductive coating on AL453 ferritic stainless steel for IT-SOFC interconnect applications. J Solid State Electrochem 17:993–1003. https://doi.org/10.1007/s10008-012-1952-8
Bednarz M, Molin S, Bobruk M, Stygar M, Długoń E, Sitarz M, Brylewski T (2019) High-temperature oxidation of the Crofer 22 H ferritic steel with Mn1.45Co1.45Fe0.1O4 and Mn1.5Co1.5O4 spinel coatings under thermal cycling conditions and its properties. Mater Chem Phys 225:227–238. https://doi.org/10.1016/j.matchemphys.2018.12.090
Lubszczyk M, Wyrwa J, Wojteczko K et al (2021) Electrical and mechanical properties of ZrO2-Y2O3-Al2O3 composite solid electrolytes. J Electron Mater 50:5933–5945. https://doi.org/10.1007/s11664-021-09125-x
Thorel AS, Abreu J, Ansar SA et al (2013) Proof of concept for the dual membrane cell. J Electrochem Soc 160:F360–F366. https://doi.org/10.1149/2.051304jes
Filipek R, Szyszkiewicz-Warzecha K, Szczudło J (2020) Corrosion of steel in concrete - modeling of electrochemical potential measurement in 3D geometry. Arch Metall Mater 65:117–124. https://doi.org/10.24425/amm.2019.131104
Szyszkiewicz K, Jasielec JJ, Danielewski M et al (2017) Modeling of electrodiffusion processes from nano to macro scale. J Electrochem Soc 164:E3559–E3568. https://doi.org/10.1149/2.0571711jes
Jasielec J, Stec J, Szyszkiewicz-Warzecha K et al (2020) Effective and apparent diffusion coefficients of chloride ions and chloride binding kinetics parameters in mortars: non-stationary diffusion–reaction model and the inverse problem. Materials (Basel) 13:5522. https://doi.org/10.3390/ma13235522
Filipek R, Szyszkiewicz K, Dziembaj P et al (2012) Modeling of reactive diffusion: mechanism and kinetics of the intermetallics growth in Ag/Ag interconnections. J Mater Eng Perform 21:638–647. https://doi.org/10.1007/s11665-012-0131-5
Danielewski M, Filipek R (2000) Interdiffusion in oxide solid solutions, simulation of the process and calculation of intrinsic diffusivities. Mol Cryst Liq Cryst Sci Technol Sect A Mol Cryst Liq Cryst 341:277–282. https://doi.org/10.1080/10587250008026153
Kupis J, Mattinen U, Kisiel A et al (2012) Biomimetic membranes made of conducting polymers doped with adenosine diphosphate (ADP). Electrochim Acta 77:23–28. https://doi.org/10.1016/j.electacta.2012.04.058
Madej M, Fendrych K, Porada R et al (2021) Application of Fe(III)-exchanged clinoptilolite/graphite nanocomposite for electrochemical sensing of amitriptyline. Microchem J 160:105648. https://doi.org/10.1016/j.microc.2020.105648
Porada R, Fendrych K, Baś B (2021) Development of novel Mn-zeolite/graphite modified screen-printed carbon electrode for ultrasensitive and selective determination of folic acid. Measurement 179:109450. https://doi.org/10.1016/j.measurement.2021.109450
Blaz T, Migdalski J, Lewenstam A (2005) Junction-less reference electrode for potentiometric measurements obtained by buffering pH in a conducting polymer matrix. Analyst 130:637. https://doi.org/10.1039/b418384c
Kupis-Rozmysłowicz J, Wagner M, Bobacka J et al (2016) Biomimetic membranes based on molecularly imprinted conducting polymers as a sensing element for determination of taurine. Electrochim Acta 188:537–544. https://doi.org/10.1016/j.electacta.2015.12.007
Migdalski J, Błaż T, Lewenstam A (2014) Conducting polymers - mechanisms of cationic sensitivity and the methods of inducing thereof. Electrochim Acta 133:316–324. https://doi.org/10.1016/j.electacta.2014.03.169
Migdalski J, Lewenstam A (2022) Electrically enhanced sensitivity (EES) of ion-selective membrane electrodes and membrane-based ion sensors. Membranes (Basel) 12:763. https://doi.org/10.3390/membranes12080763
Węgiel K, Grabarczyk M, Kubiak WW, Baś B (2017) A reliable and sensitive voltammetric determination of Mo(VI) at the in situ renovated bismuth bulk annular band electrode. J Electrochem Soc 164:H352–H357. https://doi.org/10.1149/2.1161706jes
Piech R, Kubiak WW (2007) Determination of trace arsenic with DDTC-Na by cathodic stripping voltammetry in presence of copper ions. J Electroanal Chem 599:59–64. https://doi.org/10.1016/j.jelechem.2006.09.004
Jakubowska M, Baś B, Ciepiela F, Kubiak WW (2010) A calibration strategy for stripping voltammetry of lead on silver electrodes. Electroanalysis 22:1757–1764. https://doi.org/10.1002/elan.200900510
Górski Ł, Sordoń W, Ciepiela F et al (2016) Voltammetric classification of ciders with PLS-DA. Talanta 146:231–236. https://doi.org/10.1016/j.talanta.2015.08.027
Górski Ł, Jakubowska M, Baś B, Kubiak WW (2012) Application of genetic algorithm for baseline optimization in standard addition voltammetry. J Electroanal Chem 684:38–46. https://doi.org/10.1016/j.jelechem.2012.08.014
Górski Ł, Kubiak WW, Jakubowska M (2016) Independent components analysis of the overlapping voltammetric signals. Electroanalysis 28:1470–1477. https://doi.org/10.1002/elan.201501089
Jakubowska M, Piech R, Dzierwa T et al (2003) The evaluation method of smoothing algorithms in voltammetry. Electroanalysis 15:1729–1736. https://doi.org/10.1002/elan.200302751
Rękas M (2015) Electrolytes for intermediate temperature solid oxide fuel cells. Arch Metall Mater 60:891–896. https://doi.org/10.1515/amm-2015-0225
Godlewska E, Mars K, Mania R, Zimowski S (2011) Combustion synthesis of Mg2Si. Intermetallics 19:1983–1988. https://doi.org/10.1016/j.intermet.2011.06.013
Godlewska E, Mars K, Zawadzka K (2012) Alternative route for the preparation of CoSb3 and Mg2Si derivatives. 227–230
Mitoraj M, Godlewska EM (2013) Oxidation of Ti–46Al–8Ta in air at 700 °C and 800 °C under thermal cycling conditions. Intermetallics 34:112–121. https://doi.org/10.1016/j.intermet.2012.10.014
Mitoraj M, Mars K, Matula M, Godlewska E (2015) Hot corrosion behaviour of Cr-Si coated titanium alloys. Ann Chim Sci des Matériaux 39:141–148. https://doi.org/10.3166/acsm.39.141-148
Godlewska EM, Mitoraj-Królikowska M, Czerski J et al (2020) Corrosion of Al(Co)CrFeNi high-entropy alloys. Front Mater 7:. https://doi.org/10.3389/fmats.2020.566336
Godlewska EM, Mars K, Drozdz P et al (2016) Reaction and diffusion phenomena in Ag-doped Mg 2 Si. J Alloys Compd 657:755–764. https://doi.org/10.1016/j.jallcom.2015.10.174
Godlewska E, Mitoraj M, Morgiel J (2009) Reaction and diffusion phenomena upon oxidation of a (γ+α 2) TiAlNb alloy in air. Mater High Temp 26:99–103. https://doi.org/10.3184/096034009X440263
Łańcucki Ł, Mizera A, Łącz A et al (2017) Development and chemical stability evaluation of enhanced surface LaFe1−xTixO3 (LFT) perovskites using polystyrene nanospheres as templating agent. J Alloys Compd 727:863–870. https://doi.org/10.1016/j.jallcom.2017.08.205
Łańcucki Ł, Lach R, Nieroda P et al (2019) Impact of calcium doping on structure, catalytic and conductive properties of lanthanum strontium iron oxide. Process Appl Ceram 13:411–417. https://doi.org/10.2298/PAC1904411L
Popardowski A, Pasierb P (2022) Influence of electrolyte and redox active electrode materials properties on working mechanism and performance of manganese oxide-based supercapacitors. Electrochim Acta 416:140257. https://doi.org/10.1016/j.electacta.2022.140257
Nieroda P, Kolezynski A, Leszczynski J et al (2019) The structural, microstructural and thermoelectric properties of Mg2Si synthesized by SPS method under excess Mg content conditions. J Alloys Compd 775:138–149. https://doi.org/10.1016/j.jallcom.2018.10.064
Trenczek-Zajac A, Kusior A, Radecka M (2016) CdS for TiO2-based heterostructures as photoactive anodes in the photoelectrochemical cells. Int J Hydrogen Energy 41:7548–7562. https://doi.org/10.1016/j.ijhydene.2015.12.219
Trenczek-Zajac A, Banas J, Radecka M (2016) TiO 2 -based photoanodes modified with GO and MoS 2 layered materials. RSC Adv 6:102886–102898. https://doi.org/10.1039/C6RA22979D
Radecka M, Rekas M, Trenczek-Zajac A, Zakrzewska K (2008) Importance of the band gap energy and flat band potential for application of modified TiO2 photoanodes in water photolysis. J Power Sources 181:46–55. https://doi.org/10.1016/j.jpowsour.2007.10.082
Kusior A, Klich-Kafel J, Trenczek-Zajac A et al (2013) TiO2–SnO2 nanomaterials for gas sensing and photocatalysis. J Eur Ceram Soc 33:2285–2290. https://doi.org/10.1016/j.jeurceramsoc.2013.01.022
Zakrzewska K, Radecka M (2007) TiO2–SnO2 system for gas sensing—photodegradation of organic contaminants. Thin Solid Films 515:8332–8338. https://doi.org/10.1016/j.tsf.2007.03.019
Redel K, Kulka A, Plewa A, Molenda J (2019) High-performance Li-rich layered transition metal oxide cathode materials for Li-ion batteries. J Electrochem Soc 166:A5333–A5342. https://doi.org/10.1149/2.0511903jes
Molenda J (2011) Material problems and prospects of Li -ion batteries for vehicles applications. Funct Mater Lett 04:107–112. https://doi.org/10.1142/S1793604711001816
Baster D, Maziarz W, Świerczek K et al (2015) Structural and electrochemical properties of Na0.72CoO2 as cathode material for sodium-ion batteries. J Solid State Electrochem 19:3605–3612. https://doi.org/10.1007/s10008-015-2977-6
Molenda J (2017) Electronic structure ‘engineering’ in the development of materials for Li-ion and Na-ion batteries. Adv Nat Sci Nanosci Nanotechnol 8:015007. https://doi.org/10.1088/2043-6254/aa5955
Gędziorowski B, Świerczek K, Molenda J (2012) La1−xBaxCo0.2Fe0.8O3−δ perovskites for application in intermediate temperature SOFCs. Solid State Ionics 225:437–442. https://doi.org/10.1016/j.ssi.2012.05.025
Molenda J, Świerczek K, Zając W (2007) Functional materials for the IT-SOFC. J Power Sources 173:657–670. https://doi.org/10.1016/j.jpowsour.2007.05.085
Świerczek K, Klimkowicz A, Niemczyk A et al (2014) Oxygen storage-related properties of substituted BaLnMn 2 O 5+δ A-site ordered manganites. Funct Mater Lett 07:1440004. https://doi.org/10.1142/S1793604714400049
Klimkowicz A, Świerczek K, Takasaki A et al (2015) Crystal structure and oxygen storage properties of BaLnMn2O5+δ (Ln: Pr, Nd, Sm, Gd, Dy, Er and Y) oxides. Mater Res Bull 65:116–122. https://doi.org/10.1016/j.materresbull.2015.01.041
Zając W, Rusinek D, Zheng K, Molenda J (2013) Applicability of Gd-doped BaZrO3, SrZrO3, BaCeO3 and SrCeO3 proton conducting perovskites as electrolytes for solid oxide fuel cells. Open Chem 11:471–484. https://doi.org/10.2478/s11532-012-0144-9
Kulka A, Hu Y, Dezanneau G, Molenda J (2011) Investigation of GdBaCo 2–x Fe x O 5.5-δ as a cathode material for intermediate temperature solid oxide fuel cells. Funct Mater Lett 04:157–160. https://doi.org/10.1142/S1793604711001737
Zając W, Suescun L, Świerczek K, Molenda J (2009) Structural and electrical properties of grain boundaries in Ce0.85Gd0.15O1.925 solid electrolyte modified by addition of transition metal ions. J Power Sources 194:2–9. https://doi.org/10.1016/j.jpowsour.2008.12.020
Zając W, Molenda J (2011) Properties of doped ceria solid electrolytes in reducing atmospheres. Solid State Ionics 192:163–167. https://doi.org/10.1016/j.ssi.2010.05.025
Zheng K, Świerczek K (2016) Evaluation of W-containing Sr1−Ba Fe0.75W0.25O3– (x= 0, 0.5, 1) anode materials for solid oxide fuel cells. Solid State Ionics 288:124–129. https://doi.org/10.1016/j.ssi.2015.11.022
Zheng K, Klimkowicz A, Świerczek K et al (2015) Chemical diffusion and surface exchange in selected Ln–Ba–Sr–Co–Fe perovskite-type oxides. J Alloys Compd 645:S357–S360. https://doi.org/10.1016/j.jallcom.2014.12.110
Niemczyk A, Olszewska A, Du Z et al (2018) Assessment of layered La2-x(Sr, Ba)xCuO4-δ oxides as potential cathode materials for SOFCs. Int J Hydrogen Energy 43:15492–15504. https://doi.org/10.1016/j.ijhydene.2018.06.119
Moździerz M, Dąbrowa J, Stępień A et al (2021) Mixed ionic-electronic transport in the high-entropy (Co,Cu,Mg,Ni,Zn)1-Li O oxides. Acta Mater 208:116735. https://doi.org/10.1016/j.actamat.2021.116735
Zhang Y, Zhang B, Zhao H et al (2021) Electrochemical performance and structural durability of Mg-doped SmBaMn2O5+δ layered perovskite electrode for symmetrical solid oxide fuel cell. Catal Today 364:80–88. https://doi.org/10.1016/j.cattod.2020.05.057
Olszewska A, Zhang Y, Du Z et al (2019) Mn-rich SmBaCo0.5Mn1.5O5+δ double perovskite cathode material for SOFCs. Int J Hydrogen Energy 44:27587–27599. https://doi.org/10.1016/j.ijhydene.2019.08.254
Skubida W, Zheng K, Stępień A et al (2021) SrCe0.9In0.1O3-δ-based reversible symmetrical protonic ceramic cell. Mater Res Bull 135:111154. https://doi.org/10.1016/j.materresbull.2020.111154
Klimkowicz A, Świerczek K, Kobayashi S et al (2018) Improvement of oxygen storage properties of hexagonal YMnO3+δ by microstructural modifications. J Solid State Chem 258:471–476. https://doi.org/10.1016/j.jssc.2017.10.037
Kawalec M, Krawiec H (2015) Corrosion resistance of high-alloyed white cast iron. Arch Metall Mater 60:301–303. https://doi.org/10.1515/amm-2015-0048
Kozina I, Krawiec H, Starowicz M, Kawalec M (2021) Corrosion resistance of MgZn alloy covered by chitosan-based coatings. Int J Mol Sci 22:8301. https://doi.org/10.3390/ijms22158301
Krawiec H, Lelito J, Tyrała E, Banaś J (2009) Relationships between microstructure and pitting corrosion of ADI in sodium chloride solution. J Solid State Electrochem 13:935–942. https://doi.org/10.1007/s10008-008-0636-x
Krawiec H, Vignal V, Latkiewicz M, Herbst F (2018) Structure and corrosion behaviour of electrodeposited Co-Mo/TiO2 nano-composite coatings. Appl Surf Sci 427:1124–1134. https://doi.org/10.1016/j.apsusc.2017.08.111
Lelek-Borkowska U, Gruszka M, Banas J (2021) Effect of cathodic protection on corrosion of water-pipe network in kraków-case study. Arch Foundry Eng 21:59–64. https://doi.org/10.24425/afe.2021.138666
Bisztyga M, Lelek-Borkowska U, Proniewicz E, Banaś J (2016) Cathodic behaviour of nickel in alcohol solutions of electrolytes. Electrochim Acta 207:1–8. https://doi.org/10.1016/j.electacta.2016.04.156
Starowicz M, Stypuła B (2008) Electrochemical synthesis of ZnO nanoparticles. Eur J Inorg Chem 2008:869–872. https://doi.org/10.1002/ejic.200700989
Stypuła B, Banaś J, Starowicz M et al (2006) Production of nanoparticles of copper compounds by anodic dissolution of copper in organic solvents. J Appl Electrochem 36:1407–1414. https://doi.org/10.1007/s10800-006-9233-9
Starowicz M, Stypuła B, Banaś J (2006) Electrochemical synthesis of silver nanoparticles. Electrochem commun 8:227–230. https://doi.org/10.1016/j.elecom.2005.11.018
Światowska-Mrowiecka J, Banaś J (2005) Anodic behaviour of zinc in methanol solutions of lithium perchlorate. Electrochim Acta 50:1829–1840. https://doi.org/10.1016/j.electacta.2004.08.035
Krawiec H, Vignal V, Banas J (2006) Macroscopic and local electrochemical studies of austempered ductile iron in perchlorate solutions. J Electrochem Soc 153:B231. https://doi.org/10.1149/1.2197635
Banas J, Lelek-Borkowska U, Starowicz M (2004) Electrochemical behaviour of p-Si in methanol solutions of chlorides. J Solid State Electrochem 8:422–429. https://doi.org/10.1007/s10008-003-0475-8
Lelek-Borkowska U, Banaś J (2006) Passivation and local corrosion of p-silicon in anhydrous organic solutions of chlorides. In: Passivation of Metals and Semiconductors, and Properties of Thin Oxide Layers. Elsevier 245–250
Proniewicza E, Tąta A, Starowicz M et al (2021) Is the electrochemical or the “green chemistry” method the optimal method for the synthesis of ZnO nanoparticles for applications to biological material? Characterization and SERS on ZnO. Colloids Surfaces A Physicochem Eng Asp 609:125771. https://doi.org/10.1016/j.colsurfa.2020.125771
Proniewicz E, Tąta A, Wójcik A et al (2020) SERS activity and spectroscopic properties of Zn and ZnO nanostructures obtained by electrochemical and green chemistry methods for applications in biology and medicine. Phys Chem Chem Phys 22:28100–28114. https://doi.org/10.1039/D0CP03517C
Krawiec H, Vignal V, Lelito J et al (2021) In-situ monitoring of the corrosion behaviour of austempered ductile iron (ADI) under cyclic salt spray exposure. Corros Sci 185:109437. https://doi.org/10.1016/j.corsci.2021.109437
Grevey A-L, Vignal V, Krawiec H et al (2020) Microstructure and long-term corrosion of archaeological iron alloy artefacts. Herit Sci 8:57. https://doi.org/10.1186/s40494-020-00398-9
Heintz O, Vignal V, Krawiec H, Loch J (2017) Passivity and corrosion behaviour of Ti-10Mo-4Zr and Ti-6Al-4V alloys after long-term ageing in Ringer’s solution at 37 °C. J Solid State Electrochem 21:1445–1455. https://doi.org/10.1007/s10008-017-3506-6
Krawiec H, Vignal V, Schwarzenboeck E, Banas J (2013) Role of plastic deformation and microstructure in the micro-electrochemical behaviour of Ti–6Al–4V in sodium chloride solution. Electrochim Acta 104:400–406. https://doi.org/10.1016/j.electacta.2012.12.029
Krawiec H, Vignal V, Akid R (2008) Numerical modelling of the electrochemical behaviour of 316L stainless steel based upon static and dynamic experimental microcapillary-based techniques. Electrochim Acta 53:5252–5259. https://doi.org/10.1016/j.electacta.2008.02.063
Major L, Krawiec H, Lackner JM et al (2020) Nanoscale characterization of corrosion mechanisms in advanced Zr/ZrxN and Zr/ZrxN+a-C:H nano-multilayer coatings for medical tools. Mater Charact 168:110565. https://doi.org/10.1016/j.matchar.2020.110565
Eiler K, Krawiec H, Kozina I et al (2020) Electrochemical characterisation of multifunctional electrocatalytic mesoporous Ni-Pt thin films in alkaline and acidic media. Electrochim Acta 359:136952. https://doi.org/10.1016/j.electacta.2020.136952
Overview. https://en.uj.edu.pl/en_GB/about-university/overview
Zaraska L, Kurowska E, Sulka GD, Jaskuła M (2012) Porous alumina membranes with branched nanopores as templates for fabrication of Y-shaped nanowire arrays. J Solid State Electrochem 16:3611–3619. https://doi.org/10.1007/s10008-012-1795-3
Zaraska L, Sulka GD, Jaskuła M (2011) Anodic alumina membranes with defined pore diameters and thicknesses obtained by adjusting the anodizing duration and pore opening/widening time. J Solid State Electrochem 15:2427–2436. https://doi.org/10.1007/s10008-011-1471-z
Zaraska L, Sulka GD, Jaskuła M (2010) Porous anodic alumina membranes formed by anodization of AA1050 alloy as templates for fabrication of metallic nanowire arrays. Surf Coatings Technol 205:2432–2437. https://doi.org/10.1016/j.surfcoat.2010.09.038
Sulka GD, Brzózka A, Zaraska L, Jaskuła M (2010) Through-hole membranes of nanoporous alumina formed by anodizing in oxalic acid and their applications in fabrication of nanowire arrays. Electrochim Acta 55:4368–4376. https://doi.org/10.1016/j.electacta.2010.01.048
Zaraska L, Bobruk M, Sulka GD (2015) Formation of nanoporous tin oxide layers on different substrates during anodic oxidation in oxalic acid electrolyte. Adv Condens Matter Phys 2015:1–11. https://doi.org/10.1155/2015/302560
Gawlak K, Knapik A, Sulka GD, Zaraska L (2022) Improving the photoelectrochemical performance of porous anodic SnOx films by adjusting electrosynthesis conditions. Int J Energy Res 46:17465–17477. https://doi.org/10.1002/er.8414
Zaraska L, Mika K, Hnida KE et al (2017) High aspect-ratio semiconducting ZnO nanowires formed by anodic oxidation of Zn foil and thermal treatment. Mater Sci Eng B 226:94–98. https://doi.org/10.1016/j.mseb.2017.09.003
Zaraska L, Mika K, Syrek K, Sulka GD (2017) Formation of ZnO nanowires during anodic oxidation of zinc in bicarbonate electrolytes. J Electroanal Chem 801:511–520. https://doi.org/10.1016/j.jelechem.2017.08.035
Sulka GD, Kapusta-Kołodziej J, Brzózka A, Jaskuła M (2010) Fabrication of nanoporous TiO2 by electrochemical anodization. Electrochim Acta 55:4359–4367. https://doi.org/10.1016/j.electacta.2009.12.053
Kapusta-Kołodziej J, Tynkevych O, Pawlik A et al (2014) Electrochemical growth of porous titanium dioxide in a glycerol-based electrolyte at different temperatures. Electrochim Acta 144:127–135. https://doi.org/10.1016/j.electacta.2014.08.055
Jarosz M, Pawlik A, Kapusta-Kołodziej J et al (2014) Effect of the previous usage of electrolyte on growth of anodic titanium dioxide (ATO) in a glycerol-based electrolyte. Electrochim Acta 136:412–421. https://doi.org/10.1016/j.electacta.2014.05.077
Zaraska L, Sulka GD, Jaskuła M (2012) Fabrication of free-standing copper foils covered with highly-ordered copper nanowire arrays. Appl Surf Sci 258:7781–7786. https://doi.org/10.1016/j.apsusc.2012.04.148
Zaraska L, Kurowska E, Sulka GD, Jaskuła M (2012) Template-assisted fabrication of tin and antimony based nanowire arrays. Appl Surf Sci 258:9718–9722. https://doi.org/10.1016/j.apsusc.2012.06.018
Hnida K, Mech J, Sulka GD (2013) Template-assisted electrodeposition of indium–antimony nanowires – comparison of electrochemical methods. Appl Surf Sci 287:252–256. https://doi.org/10.1016/j.apsusc.2013.09.135
Sulka GD, Hnida K, Brzózka A (2013) pH sensors based on polypyrrole nanowire arrays. Electrochim Acta 104:536–541. https://doi.org/10.1016/j.electacta.2012.12.064
Szuwarzyński M, Zaraska L, Sulka GD, Zapotoczny S (2013) Pulsatile releasing platform of nanocontainers equipped with thermally responsive polymeric nanovalves. Chem Mater 25:514–520. https://doi.org/10.1021/cm303930y
Brudzisz A, Rajska D, Gajewska M et al (2020) Controlled synthesis and characterization of AgPd nanowire arrays for electrocatalytic applications. J Electroanal Chem 873:114373. https://doi.org/10.1016/j.jelechem.2020.114373
Zaraska L, Syrek K, Hnida KE et al (2016) Nanoporous tin oxides synthesized via electrochemical anodization in oxalic acid and their photoelectrochemical activity. Electrochim Acta 205:273–280. https://doi.org/10.1016/j.electacta.2016.02.023
Zych M, Syrek K, Pisarek M, Sulka GD (2022) Synthesis and characterization of anodic WO3 layers in situ doped with C, N during anodization. Electrochim Acta 411:140061. https://doi.org/10.1016/j.electacta.2022.140061
Sołtys-Mróz M, Syrek K, Pięta Ł et al (2022) Photoelectrochemical performance of nanotubular Fe2O3–TiO2 electrodes under solar radiation. Nanomaterials 12:1546. https://doi.org/10.3390/nano12091546
Mika K, Syrek K, Uchacz T et al (2022) Dark nanostructured ZnO films formed by anodic oxidation as photoanodes in photoelectrochemical water splitting. Electrochim Acta 414:140176. https://doi.org/10.1016/j.electacta.2022.140176
Gawlak K, Popiołek D, Pisarek M et al (2022) CdS-decorated porous anodic SnOx photoanodes with enhanced performance under visible light. Materials (Basel) 15:3848. https://doi.org/10.3390/ma15113848
Syrek K, Grudzień J, Sennik-Kubiec A et al (2019) Anodic titanium oxide layers modified with gold, silver, and copper nanoparticles. J Nanomater 2019:1–10. https://doi.org/10.1155/2019/9208734
Kurowska E, Brzózka A, Jarosz M et al (2013) Silver nanowire array sensor for sensitive and rapid detection of H2O2. Electrochim Acta 104:439–447. https://doi.org/10.1016/j.electacta.2013.01.077
Kurowska-Tabor E, Gawlak K, Hnida K et al (2016) Synthesis of porous thin silver films and their application for hydrogen peroxide sensing. Electrochim Acta 213:811–821. https://doi.org/10.1016/j.electacta.2016.08.007
Zaraska L, Gawlak K, Kurowska-Tabor E et al (2017) Template-assisted synthesis of rough Ag nanorods and their application for amperometric sensing of H 2 O 2. Comptes Rendus Chim 20:693–696. https://doi.org/10.1016/j.crci.2017.03.001
Hnida KE, Socha RP, Sulka GD (2013) Polypyrrole–Silver composite nanowire arrays by cathodic Co-deposition and their electrochemical properties. J Phys Chem C 130916100825004. https://doi.org/10.1021/jp4038304
Ryczek K, Kozieł M, Wiercigroch E et al (2020) Fast fabrication of nanostructured semiconducting oxides by anodic oxidation of brass. Mater Sci Semicond Process 113:105035. https://doi.org/10.1016/j.mssp.2020.105035
Jarosz M, Pawlik A, Szuwarzyński M et al (2016) Nanoporous anodic titanium dioxide layers as potential drug delivery systems: drug release kinetics and mechanism. Colloids Surfaces B Biointerfaces 143:447–454. https://doi.org/10.1016/j.colsurfb.2016.03.073
Pawlik A, Rehman MAU, Nawaz Q et al (2019) Fabrication and characterization of electrophoretically deposited chitosan-hydroxyapatite composite coatings on anodic titanium dioxide layers. Electrochim Acta 307:465–473. https://doi.org/10.1016/j.electacta.2019.03.195
Golda-Cepa M, Syrek K, Brzychczy-Wloch M et al (2016) Primary role of electron work function for evaluation of nanostructured titania implant surface against bacterial infection. Mater Sci Eng C 66:100–105. https://doi.org/10.1016/j.msec.2016.04.079
Pawlik A, Jarosz M, Syrek K, Sulka GD (2017) Co-delivery of ibuprofen and gentamicin from nanoporous anodic titanium dioxide layers. Colloids Surfaces B Biointerfaces 152:95–102. https://doi.org/10.1016/j.colsurfb.2017.01.011
Pawlik A, Jarosz M, Socha RP, Sulka GD (2021) The impacts of crystalline structure and different surface functional groups on drug release and the osseointegration process of nanostructured TiO2. Molecules 26:1723. https://doi.org/10.3390/molecules26061723
Wierzbicka E, Szultka-Młyńska M, Buszewski B, Sulka GD (2016) Epinephrine sensing at nanostructured Au electrode and determination its oxidative metabolism. Sensors Actuators B Chem 237:206–215. https://doi.org/10.1016/j.snb.2016.06.073
Wierzbicka E, Sulka GD (2016) Fabrication of highly ordered nanoporous thin Au films and their application for electrochemical determination of epinephrine. Sensors Actuators B Chem 222:270–279. https://doi.org/10.1016/j.snb.2015.08.066
Wierzbicka E, Sulka GD (2016) Nanoporous spongelike Au–Ag films for electrochemical epinephrine sensing. J Electroanal Chem 762:43–50. https://doi.org/10.1016/j.jelechem.2015.12.013
Jarosz M, Grudzień J, Kamiński K et al (2019) Novel bioelectrodes based on polysaccharide modified gold surfaces and electrochemically active Lactobacillus rhamnosus GG biofilms. Electrochim Acta 296:999–1008. https://doi.org/10.1016/j.electacta.2018.11.154
Brzózka A, Fic K, Bogusz J et al (2019) Polypyrrole–nickel hydroxide hybrid nanowires as future materials for energy storage. Nanomaterials 9:307. https://doi.org/10.3390/nano9020307
Pawlik A, Socha RP, Hubalek Kalbacova M, Sulka GD (2018) Surface modification of nanoporous anodic titanium dioxide layers for drug delivery systems and enhanced SAOS-2 cell response. Colloids Surfaces B Biointerfaces 171:58–66. https://doi.org/10.1016/j.colsurfb.2018.07.012
Mika K, Socha RP, Nyga P et al (2019) Electrochemical synthesis and characterization of dark nanoporous zinc oxide films. Electrochim Acta 305:349–359. https://doi.org/10.1016/j.electacta.2019.03.052
Thomas R, Gurgul M, Xavier B et al (2022) Lithium and sodium storage performance of tin oxyphosphate anode materials. Appl Surf Sci 579:152126. https://doi.org/10.1016/j.apsusc.2021.152126
Gurgul M, Lytvynenko AS, Jarosz M et al (2020) Hierarchical nanoporous Sn/SnOx systems obtained by anodic oxidation of electrochemically deposited Sn nanofoams. Nanomaterials 10:410. https://doi.org/10.3390/nano10030410
Góral-Kurbiel M, Drelinkiewicz A, Kosydar R et al (2014) The effect of Nafion ionomer on electroactivity of palladium–polypyrrole catalysts for oxygen reduction reaction. J Solid State Electrochem 18:639–653. https://doi.org/10.1007/s10008-013-2299-5
Góral-Kurbiel M, Kosydar R, Gurgul J et al (2016) Carbon supported PdxPty nanoparticles for oxygen reduction. The effect of Pd: Pt ratio. Electrochim Acta 222:1220–1233. https://doi.org/10.1016/j.electacta.2016.11.096
Góral-Kurbiel M, Drelinkiewicz A, Kosydar R et al (2014) Palladium content effect on the electrocatalytic activity of palladium–polypyrrole nanocomposite for cathodic reduction of oxygen. Electrocatalysis 5:23–40. https://doi.org/10.1007/s12678-013-0155-0
Socha RP, Nowak P, Laajalehto K, Väyrynen J (2004) Particle-electrode surface interaction during nickel electrodeposition from suspensions containing SiC and SiO2 particles. Colloids Surfaces A Physicochem Eng Asp 235:45–55. https://doi.org/10.1016/j.colsurfa.2004.01.011
Socha R, Laajalehto K, Nowak P (2002) Influence of the surface properties of silicon carbide on the process of SiC particles codeposition with nickel. Colloids Surfaces A Physicochem Eng Asp 208:267–275. https://doi.org/10.1016/S0927-7757(02)00153-X
Socha RP, Laajalehto K, Nowak P (2002) Oxidation of the silicon carbide surface in Watts’ plating bath. Surf Interface Anal 34:413–417. https://doi.org/10.1002/sia.1329
Pacuła A, Nowak P, Socha RP et al (2013) Preparation and characterization of the electroactive composites containing nickel nanoparticles and carbon nanotubes. Electrochim Acta 90:563–572. https://doi.org/10.1016/j.electacta.2012.12.062
Kopeć M, Szczepanowicz K, Warszyński P, Nowak P (2016) Liquid-core polyelectrolyte nanocapsules produced by membrane emulsification as carriers for corrosion inhibitors. Colloids Surfaces A Physicochem Eng Asp 510:2–10. https://doi.org/10.1016/j.colsurfa.2016.08.056
Haber J, Nowak P, Żurek P (2008) Charge transfer in photocatalytic systems: V and Mo doped TiO2/Ti electrodes. Catal Letters 126:43–48. https://doi.org/10.1007/s10562-008-9652-9
Haber J, Nowak P, Socha RP, Żurek P (2008) Preparation of TiO2 for photocatalytic applications - doping of TiO2/Ti with transition metal ions. Pol J Chem 82:1753–1766
Haber J, Nowak P, Żurek P (2003) Electrodeposition of hedgehog-shaped gold crystallites on TiO 2 surface and their behavior in anodic oxidation of oxalic acid. Langmuir 19:196–199. https://doi.org/10.1021/la020793y
Dziedzic J, Wodka D, Nowak P, Warszyński P, Simon C, Kumakiri I (2010) Photocatalytic degradation of the humic species as a method of their removal from water - comparison of UV and artificial sunlight irradiation. Physicochem Probl Miner Process 45:15–28
Mosiałek M, Nowak P, Dudek M, Mordarski G (2014) Oxygen reduction at the Ag|Gd0.2Ce0.8O1.9 interface studied by electrochemical impedance spectroscopy and cyclic voltammetry at the silver point electrode. Electrochim Acta 120:248–257. https://doi.org/10.1016/j.electacta.2013.12.071
Mosiałek M, Dudek M, Nowak P et al (2013) Changes in the morphology and the composition of the Ag|Gd0.2Ce0.8O1.9 interface caused by polarization. Electrochim Acta 104:474–480. https://doi.org/10.1016/j.electacta.2013.01.117
Mosiałek M, Bielańska E, Socha RP et al (2012) Changes in the morphology and the composition of the Ag|YSZ and Ag|LSM interfaces caused by polarization. Solid State Ionics 225:755–759. https://doi.org/10.1016/j.ssi.2012.03.011
Szaleniec M, Hagel C, Menke M et al (2007) Kinetics and mechanism of oxygen-independent hydrocarbon hydroxylation by ethylbenzene dehydrogenase. Biochemistry 46:7637–7646. https://doi.org/10.1021/bi700633c
Tataruch M, Heider J, Bryjak J et al (2014) Suitability of the hydrocarbon-hydroxylating molybdenum-enzyme ethylbenzene dehydrogenase for industrial chiral alcohol production. J Biotechnol 192:400–409. https://doi.org/10.1016/j.jbiotec.2014.06.021
Choi H, Demeke D, Kang F-A et al (2003) Synthetic studies on the marine natural product halichondrins. Pure Appl Chem 75:1–17. https://doi.org/10.1351/pac200375010001
Cole DC, Olland AM, Jacob J et al (2010) Identification and characterization of acidic mammalian chitinase inhibitors. J Med Chem 53:6122–6128. https://doi.org/10.1021/jm100533p
Chen JJ, Cole DC, Ciszewski G et al (2010) Identification of a new class of small molecule C5a receptor antagonists. Bioorg Med Chem Lett 20:662–664. https://doi.org/10.1016/j.bmcl.2009.11.058
Nowak P, Laajalehto K (2000) Oxidation of galena surface – an XPS study of the formation of sulfoxy species. Appl Surf Sci 157:101–111. https://doi.org/10.1016/S0169-4332(99)00575-9
Wodka D, Socha RP, Bielańska E et al (2014) Photocatalytic activity of titanium dioxide modified by Fe2O3 nanoparticles. Appl Surf Sci 319:173–180. https://doi.org/10.1016/j.apsusc.2014.08.010
Bigos A, Wolowicz M, Janusz-Skuza M et al (2021) Citrate-based baths for electrodeposition of nanocrystalline nickel coatings with enhanced hardness. J Alloys Compd 850:156857. https://doi.org/10.1016/j.jallcom.2020.156857
Beltowska-Lehman E, Ozga P, Swiatek Z, Lupi C (2002) Influence of structural factor on corrosion rate of functional Zn–Ni coatings. Cryst Eng 5:335–345. https://doi.org/10.1016/S1463-0184(02)00045-X
Beltowska-Lehman E, Indyka P, Bigos A et al (2015) Ni–W/ZrO2 nanocomposites obtained by ultrasonic DC electrodeposition. Mater Des 80:1–11. https://doi.org/10.1016/j.matdes.2015.04.049
Beltowska-Lehman E, Indyka P, Bigos A et al (2016) Effect of current density on properties of Ni–W nanocomposite coatings reinforced with zirconia particles. Mater Chem Phys 173:524–533. https://doi.org/10.1016/j.matchemphys.2016.02.050
Beltowska-Lehman E, Indyka P, Bigos A et al (2016) Effect of hydrodynamic conditions of electrodeposition process on microstructure and functional properties of Ni-W/ZrO2 nanocomposites. J Electroanal Chem 775:27–36. https://doi.org/10.1016/j.jelechem.2016.05.003
Beltowska-Lehman E, Swiatek Z, Lipinski M et al (2000) The influence of surface modification on optoelectronic properties of monocrystalline silicon solar cells. In: Conference Record of the Twenty-Eighth IEEE Photovoltaic Specialists Conference - (Cat. No.00CH37036). IEEE 331–334
Lipinski M, Panek P, Bełtowska E, Czternastek H (2003) Reduction of surface reflectivity by using double porous silicon layers. Mater Sci Eng B 101:297–299. https://doi.org/10.1016/S0921-5107(02)00720-1
Ozga P (2006) Electrodeposition of Sn-Ag and Sn-Ag-Cu alloys from thiourea aqueous solutions. Arch Metall Mater 51:413–422
Ozga P, Świątek Z, Michalec M, Onderka B, Bonarski J (2008) Phase structure and texture of electrodeposited InSn alloys on copper substrate. Arch Metall Mater 53:307–315
Slupska M, Ozga P (2014) Electrodeposition of Sn-Zn-Cu alloys from citrate solutions. Electrochim Acta 141:149–160. https://doi.org/10.1016/j.electacta.2014.07.039
Beltowska-Lehman E, Ozga P, Swiatek Z, Lupi C (2002) Electrodeposition of Zn–Ni protective coatings from sulfate–acetate baths. Surf Coatings Technol 151–152:444–448. https://doi.org/10.1016/S0257-8972(01)01614-0
Ozga P, Bielańska E (2003) Determination of the corrosion rate of Zn and Zn–Ni layers by the EDS technique. Mater Chem Phys 81:562–565. https://doi.org/10.1016/S0254-0584(03)00075-0
Kazimierczak H, Świątek Z, Ozga P (2020) Electrodeposition of tin-zinc-bismuth alloys from aqueous citrate-EDTA baths. Electrochim Acta 338:135889. https://doi.org/10.1016/j.electacta.2020.135889
Pohrelyuk IM, Tkachuk OV, Proskurnyak RV et al (2020) Cytocompatibility evaluation of Ti-6Al-4V alloy after gas oxynitriding. J Mater Eng Perform 29:7785–7792. https://doi.org/10.1007/s11665-020-05265-z
Łukasiewicz Research Network – Krakow Institute of Technology. https://kit.lukasiewicz.gov.pl/en/company-presentation/. Accessed 13 Oct 2022
Kozak J, Zybura-Skrabalak M (2016) Some problems of surface roughness in electrochemical machining (ECM). Procedia CIRP 42:101–106. https://doi.org/10.1016/j.procir.2016.02.198
Ruszaj A, Zybura-Skrabalak M (2001) The mathematical modelling of electrochemical machining with flat ended universal electrodes. J Mater Process Technol 109:333–338. https://doi.org/10.1016/S0924-0136(00)00816-5
Skrabalak G, Zybura-Skrabalak M, Ruszaj A (2004) Building of rules base for fuzzy-logic control of the ECDM process. J Mater Process Technol 149:530–535. https://doi.org/10.1016/j.jmatprotec.2003.11.058
Zybura-Skrabalak M, Ruszaj A (2000) The influence of electrode surface geometrical structure on electrochemical smoothing process. J Mater Process Technol 107:288–292. https://doi.org/10.1016/S0924-0136(00)00696-8
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Skibińska, K., Żabiński, P. Electrochemistry at Krakowian research institutions. J Solid State Electrochem 27, 1675–1685 (2023). https://doi.org/10.1007/s10008-023-05391-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10008-023-05391-3