Abstract
Objectives
Periodontal disease and diabetes have an extensively investigated bidirectional correlation. Non-surgical periodontal treatment (NSPT) was proven to contribute to glycemic control. Moreover, it may benefit from the association of adjunctive therapies. The aim of the present systematic review is to assess the clinical efficacy of NSPT in association with laser (LT) or photodynamic therapy (PDT) in controlled or uncontrolled diabetic patients, and to grade the level of evidence.
Materials and methods
Randomized controlled clinical trials with at least 3-month follow-up were searched in MEDLINE via OVID, EMBASE, and Cochrane Central, screened for inclusion, and grouped based on the performed treatments, follow-up time, type of diabetes, and level of glycemic control.
Results
Eleven RCTs with 504 total subjects were included. The adjunct of PDT showed a statistically significant 6-month difference in PD changes (with low certainty of evidence), but not in CAL changes, while a significant difference in 3-month PD and CAL changes was found with the adjunct of LT (low certainty of evidence). Patients treated with PDT registered a higher decrease in HbA1c levels at 3 months, but no significant difference was noted at 6 months; LT also led to better HbA1c changes at 3 months with a moderate certainty of evidence.
Conclusion
Despite the promising short-term HbA1c decrease, the results should be interpreted with caution due to the small effect sizes and the statistical heterogeneity, and further evidence from well-designed RCTs is needed to support the routine use of PDT or LT in adjunct to NSPT.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The bidirectional relationship between hyperglycemia (all types of diabetes) and periodontitis is well-known and widely documented in the scientific literature [1]. Several recent studies confirmed that diabetes represents a significant independent risk factor, it influences oral health in general, and it is a known cause of increased tooth loss rate [2,3,4]. Indeed, diabetes is considered one of the major risk factors for periodontal diseases, being the risk of having periodontitis in subjects with diabetes approximately three-fold higher than in healthy subjects [5].
Several mechanisms were pointed out to explain the linkage between diabetes mellitus and periodontitis. In general, diabetes can trigger an increase of the inflammatory response towards the oral microbiota (e.g., augmenting IL-1, IL-6, TNF-α) and can impair the immune host response, thus creating favorable conditions for the development and worsening of periodontal diseases in predisposed subjects [6, 7].
At the same time, periodontitis is responsible of increasing insulin resistance and may enhance the risk for diabetes or promote an impairment of glucose tolerance mechanisms. Based on the existing literature, there is evidence that periodontitis could be associated with an increased incidence of diabetes in specific cohorts of systemically compromised patients [8], as well as in the general population, since people with normal glycemic control and periodontitis are more prone to develop diabetes than periodontally healthy subjects [9]. Moreover, periodontitis represents an independent risk factor for microvascular complications in diabetic subjects, such as nephropathy, neuropathy, and retinopathy [10]. The biological plausibility of a correlation between periodontitis and diabetes finds a substantial support considering the low-grade inflammatory systemic status that is induced by periodontitis itself, which could be the basis of an increased susceptibility to diabetes in particularly predisposed subjects [11, 12]. Furthermore, periodontitis-induced systemic inflammation could also contribute to hematopoiesis by increasing the production of myeloid cells that are more responsive to inflammation, and this process might potentially be at the basis of different comorbidities [13].
Given the bidirectional correlation between diabetes and periodontitis, it was demonstrated that non-surgical periodontal treatment (NSPT) in subjects with periodontitis and diabetes could influence glycemic control [14,15,16]. A recent Cochrane systematic review, including 35 studies and accounting for a total of 3249 participants, found a reduction of HbA1c of 0.43% at 3–4 months after non-surgical treatment (any type of subgingival instrumentation), thus suggesting that periodontal therapy contributes to glycemic control [15].
Despite NSPT is considered to be generally effective in the treatment of periodontitis, we expect that a certain number of pockets (about 26% at 6/8 months) will not close because of local factors (e.g., depth of initial pocket, anatomy of the tooth and of the defect) and factors related to the patient (e.g., smoking, systemic diseases, compliance with oral hygiene) or operator (ability to successfully remove the deposits and to motivate the patient) [17]. Therefore, adjunctive measures that could enhance the outcomes of NSPT have been proposed [18,19,20,21,22]. Among these adjunctive therapies, the systematic review published by Salvi and coworkers, considered in the recently published S3-level treatment guideline of the European Federation of Periodontology, examined the efficacy of laser (LT) and photodynamic therapy (PDT) [20]. While the authors did not find differences when focusing on systemically healthy periodontitis patients, a specific analysis of the effects of laser or PDT in a particular susceptible group of subjects, such as diabetic patients, considering both periodontal and glycemic outcomes, is still missing. It might be hypothesized that LT and PDT, due to their anti-inflammatory effect and the ability of modulating the inflammatory response in other systemic clinical conditions [23], can be a valuable adjunctive therapy for the treatment of diabetic periodontitis patients. Moreover, the differences in the subgingival population that exist between diabetic and non-diabetic periodontal patients could be a further reason for the need of different/additional approaches for treating the periodontal disease in diabetic patients [24]. Despite some systematic reviews with heterogeneous methodology are available in this field [25, 26], no meta-analysis and critical appraisal of certainty of evidence have been published comparing PDT/LT as an adjunct to NSPT to NSPT alone. Moreover, the previously published studies reported inconclusive results.
There is therefore the need of systemically addressing the evidence about adjunctive periodontal treatments such as PDT and LT in subjects with diabetes, mainly because of the high prevalence of the disease and the need of considering the effect of this systemic disease on treatment outcome in studies designed for this specific purpose.
The present systematic review of the literature aimed to fill this knowledge gap and to assess the efficacy of NSPT performed with the adjunct of LT or PDT in patients with type II diabetes mellitus and to grade the level of available evidence.
Materials and methods
The protocol of the study was registered in PROSPERO database (number CRD42021237742) before study initiation. The protocol followed the instructions provided by the Cochrane Handbook for Systematic Review of Interventions – Second Edition [27].
The aim of this review was to answer the following focused question: in periodontitis patients affected by type II diabetes mellitus, what is the efficacy of PDT and LT as an adjunct to non-surgical periodontal therapy in terms of pocket closure, probing pocket depth (PPD) reduction, and clinical attachment level (CAL) gain?
Eligibility criteria
The criteria for considering studies for this review based on the PICOS are:
-
Population (P): ≥ 18 years old, previously untreated periodontitis patients (defined following the current and past classifications [28, 29] as stage II, stage III, or stage IV periodontitis (any grade) or moderate to severe periodontitis) affected by controlled or uncontrolled type II diabetes (T2DM) (code 5A11 following the International Classification of Diseases of the World Health Organization [30]), defined as presence of insulin resistance [31].
-
Intervention (I): (a) Physical treatment (e.g., LT, PDT) as an adjunct to non-surgical treatment (sub-gingival instrumentation) of periodontitis.
-
Control (C): The same non-surgical treatment of periodontitis associated with placebo or without adjunctive therapy, or performed according to a different protocol.
-
Outcomes (O):
Primary outcomes:
-
Proportion or number of pockets closed (defined as PPD < 5 mm and no bleeding on probing (BOP)); reduction in PPD, which is defined as the distance from the gingival margin to the base of the pocket as assessed with a standardized (UNC-15) periodontal probe with a force of 0.2/0.25N; changes in CAL, which is the measurement of the position of the soft tissue in relation to cemento-enamel junction (CEJ).
Secondary outcomes:
-
Site-specific response to subgingival instrumentation (in horizontal defects, intrabony defects and furcations)
-
Changes in HbA1c levels
-
Changes in BOP or gingival inflammation and in plaque levels
-
Number of teeth lost or extracted during the examination period
-
Patient-reported outcome measures (PROMs), including adverse events
-
-
Studies (S): Randomized controlled clinical trials with at least 3-month follow-up. Split-mouth studies were excluded due to the risk of carry-over effects
Search and study selection
The electronic search for pertinent articles was performed searching the following databases: MEDLINE via OVID, EMBASE, and Cochrane Central and by using the search strategy presented in Appendix 1. Grey literature was searched for pertinent articles interrogating Greylit and OpenGrey. Trials registers (ClinicalTrials.gov and EU Clinical Trials Register) were also searched through keywords. A manual search was performed for all the issues published since 1990 of the following journals: Journal of Clinical Periodontology, Journal of Periodontology, Journal of Periodontal Research, Journal of Dentistry, and Journal of Dental Research. Besides checking the reference list of all included papers, Scopus was consulted to check the articles citing the papers included. No language limitations were posed. Conference papers and abstracts were excluded.
The last electronic search was performed in all databases on 10 February 2022.
Two reviewers (SC, EC) independently screened titles and abstract for preliminary check of inclusion criteria (1st stage). The second stage of articles selection was performed by the same reviewers, by carefully screening the full texts of the papers retrieved after preliminary check. In case of disagreement, a third reviewer (ND) was interrogated to solve the dispute. Reasons for exclusion in the second step were recorded, and the level of concordance in each step of the selection process was assessed through Cohen’s kappa.
Data extraction
The process of data extraction was performed independently by two authors (AA, PE) who retrieved the following information from the included studies: authors’ names, year of publication, country, characteristics of the sample (age distribution, sex distribution, ethnicity, educational status, smoking status), characteristics of diabetes (definition and type, level of control of the disease, HbA1c levels, drugs), definition/assessment of periodontitis, characteristics of the periodontal treatment and of the adjunctive physical therapy, clinical data before and after the treatment (number of teeth lost, proportion of closed periodontal pockets, mean periodontal probing depth (PD), mean CAL, gingival bleeding indexes (gingival bleeding index, gingival index (GI), percentage of bleeding sites (BOP), plaque indexes (plaque index (PI), Turesky-modified plaque index, proportion of sites with visible plaque) or difference between baseline and follow-up values, occurrence of adverse events or complications, and patients’ reported outcomes (PROMs).
In case of missing/unclear information, an attempt was made to contact the authors by email.
Risk of bias evaluation and quality of evidence assessment
The risk of bias evaluation and the quality of evidence assessment were performed independently by two reviewers (SC, LF) and any disagreement resolved by discussion.
The criteria for evaluating the risk of bias in the included studies were the ones of the Cochrane risk-of-bias tool for randomized trials 2.0 [27]:
-
Bias arising from the randomization process
-
Bias due to deviations from intended interventions
-
Bias due to missing outcome data
-
Bias in measurement of the outcome
-
Bias in selection of the reported result
The overall risk-of-bias judgment was considered as high risk if the level of risk of bias was high for at least one domain or if the trial was judged to have some concerns for multiple domains (three). If the trial was judged to have some concerns for less than three domains, the overall risk of bias was “some concerns,” while the study had low risk of bias if all domains were judged to have low risk.
The funding bias was estimated by evaluating if authors disclosed their potential sources of competing conflict of interest and the source of funding for the studies they carried on (if any).
The quality of the available evidence was assessed for each comparison and for each outcome in the meta-analysis by using the Grading of Recommendations, Assessment, Development and Evaluations (GRADE) approach [32]. GRADE provides a system for rating quality of evidence and strength of recommendations that is explicit, comprehensive, transparent, and pragmatic.
Summary measures and synthesis of the results
In order to perform the meta-analysis, studies were grouped based on the treatments that were carried out, follow-up time, and, whenever possible, based on the type of diabetes and on level of control. In particular, we distinguished between photodynamic therapy (PDT) and direct laser application (LT). Meta-analysis was performed by using the software RevMan (Review Manager Version 5.3, 2014; The Nordic Cochrane Center, The Cochrane Collaboration, Copenhagen, Denmark) if at least three papers were available for each comparison.
For each continuous outcome, the difference between baseline and follow-up values was extracted with its specific error measure (standard deviation, standard error, or variance). When difference values were not reported, they were calculated as the difference between baseline and follow-up values and error (namely, standard deviation) was computed following the procedure described in Appendix 2. In the meta-analysis, the effect size was computed through the weighted mean method, and results were combined using the DerSimonian and Laird’s random-effect model [33], assuming heterogeneity among studies. Cochran’s test served to measure the consistency of the results, considering it significant if P < 0.1. I2 statistics was applied to measure heterogeneity (total variation across studies that was due to heterogeneity rather than to chance) [27].
Regression meta-analysis was performed to evaluate the effect of baseline HbA1c% on the primary outcome measures.
Small study effects, as proxy for publication bias, were assessed by testing for funnel plot asymmetry and by calculating Egger’s bias, as described in the Cochrane Handbook [27].
Results
The results of this systematic review are herein presented following the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) guidelines [34].
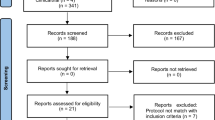
The summary of the article selection process is summarized in Fig. 1. Eleven RCTs were included in the analysis [35,36,37,38,39,40,41,42,43,44,45], which accounted for a total of 504 subjects, examined with a follow-up ranging from 1 to 6 months.
In particular, seven papers compared NSPT to NSPT and adjunctive PDT in subjects with diabetes [35,36,37, 40, 42, 43, 45]. In all the studies, in the test groups, non-thermal diode laser was used to irradiate a photosensitizer agent. In one study, NSPT was performed following a “Full-mouth disinfection” protocol in both groups [43].
Four studies compared NSPT to NSPT and adjunctive DL use (with settings varying between 0.8 and 1.8 W) in subjects with diabetes [38, 39, 41, 44]. In all studies, the control groups were treated according to a quadrant-based NSPT protocol. In four studies, the periodontal disease was classified following the 2017 classification [28], including stage II, stage III, and stage IV periodontitis and grade B or C [36, 40, 44, 45]. The other included studies used older classifications and diagnostic parameters [46].
Considering the characteristics of the population, three studies were performed in Saudi Arabia [35, 36, 40], three in Brazil [37, 42, 45], two in India [38, 44], two in Turkey [39, 41], and one in Pakistan [43]. In all studies, only T2DM was considered, with different level of controls defined on the basis of HbA1c: three studies included patients with HbA1c > 7% [39, 42, 45]; one included subjects with HbA1c > 6% [44]; one considered HbA1c ≥ 6.5% [43]; one < 7% [37]; and in one study, subjects with HbA1c between 5.7 and 8.5% were included [41], while other studies adopted different definitions [35, 36, 38, 40]. One study clearly stated that only subjects with decompensated T2DM were included [45], while in four studies, patients with major diabetic complications were excluded [35, 39, 42, 43]. Smokers were excluded in all studies.
Additional details about the characteristics of the studies are shown in Table 1.
Risk of bias evaluation
The results of risk of bias evaluation are reported in Table 2. Five studies raised some concerns about the risk of bias due to the methods of randomization and to the blinding of subjects [35, 38, 40, 44, 45], while six studies were at low risk [36, 37, 39, 41,42,43] (Fig. 2).
Synthesis of the results
Pocket closure, PD changes, CAL changes
-
NSPT versus NSPT and photodynamic therapy (PDT)
Meta-analysis based on 4 studies indicated a statistically significant difference in PD changes (favoring the test group) and CAL changes favoring control group 6 months after treatment with a low effect size (PD change: 0.26 mm, CI95%: 0.01, 0.50, I2: 57%, 137 subjects; CAL change: − 0.2 mm, CI95%: − 0.23, − 0.17, I2: 0%, 137 subjects) (Table 3).
Three studies reported data about the changes in pockets ≥ 5mm, but they could not be pooled in a meta-analysis because one study reported the mean number of pockets per patient [45] and the others presented the proportions [35, 40]. More specifically, at 3 months, Al-Zahrani and colleagues found a non-significant decrease in the proportion of sites with PD ≥ 5 mm from 11% ± 8% to 6% ± 7% in the test group and from 14% ± 14% to 8% ± 13% in the control group [35]. Likewise, Elsadek et al. [40] indicated a non-significant significant decrease after 3 months in the proportion of sites with PD ≥ 5 mm in both groups (from 12% ± 7% to 4% ± 6% in the test group and from 15% ± 15% to 9% ± 12% in control group). A more recent study reported a decrease in the number of pockets that was significant in both groups after 3 and 6 months from the treatment without a significant intergroup difference [45].
-
NSPT versus NSPT + diode laser (DL)
Meta-analysis was performed for PD and CAL change at 3 months post-treatment, and it involved 4 and 3 studies, respectively. As reported in Table 3, a significant difference in 3-month PD and CAL change is found when DL was applied as an adjunctive therapy (PD change: 0.59 mm, CI95%: 0.41 mm, 0.76 mm, I2: 80%, 170 subjects; CAL change: 0.84 mm, CI95%: 0.09 mm; 1.59 mm, I2: 86%, 112 subjects).
None of the studies reported data on pocket closure. Regression meta-analysis did not reveal any significant effect of baseline HbA1c% on the examined outcomes.
Secondary outcomes
-
NSPT versus NSPT and photodynamic therapy (PDT)
Based on 5 studies, meta-analysis indicated a statistically significant difference in 3-month BoP change between test and control groups (− 5.95% [− 9.92%, − 1.98%]), favoring the latter. However, at 6 months, this difference was no longer significant.
Remarkably, HbA1c decreased significantly more in the test groups than in control groups 3 months after the treatment (0.24, CI95%: 0.17, 0.32), but this outcome was not confirmed at 6 months.
No significant differences were suggested for PI changes (Table 3).
No patient-reported outcomes were reported.
-
NSPT versus NSPT + diode laser (DL)
The quantitative synthesis based on the data from 3 studies indicated a significant difference, in 3-month GI changes (0.34, CI95%: 0.21, 0.47) favoring the test groups (Table 3). Likewise, the adjunctive use of DL led to better HbA1c changes (0.18, CI95%: 0.07, 0.28) at 3 months (Table 3).
No patient-reported outcomes were reported.
Certainty of evidence
The results of the evaluation of the certainty of evidence are summarized in Table 3. Regarding the primary outcomes, in the comparison between NSPT and NSPT + PDT, the certainty was moderate for CAL reduction (6 months) and low for PD changes, while for the comparison between NSPT and NSPT + DL, the certainty of evidence was low. Regarding the secondary outcomes, the adjunctive use of PDT was associated with a moderate certainty of evidence in terms of BoP% reduction (3 months) and HbA1c reduction (3 months), while the adjunctive use of DL was associated with a moderate certainty of evidence both for GI reduction (3 months) and for HbA1c reduction (3 months).
Discussion
The results of the present systematic review and meta-analysis demonstrated a small but significant positive effect of the application of PDT as an adjunct to NSPT in type II diabetic patients regarding PD changes (6 months) and HbA1c changes (3 months) compared to control groups (NSPT only), the latter reporting more favorable CAL changes (6 months). Moreover, LT with DL as an adjunct to NSPT resulted in an enhanced effect for PD, CAL, GI, and HbA1c reductions at 3 months. However, these results need to be interpreted with caution due to the small effect sizes and the relatively high statistical heterogeneity.
Different from several other published systematic reviews that addressed mainly glycemic control [15, 16, 47, 48], the main focus of this systematic review was on post-treatment periodontal. As a matter of fact, our primary outcomes included the percentage of closed pockets, PD reduction, and CAL gain. Therefore, the results of the present research should be interpreted in the light of the existing literature that examined the same outcomes.
PDT was described as an effective antimicrobial strategy towards periodontal pathogens, and its activity depends on the creation of components that are noxious for the microorganisms (such as free radicals) following the activation, by the laser light, of the photosensitive component [49, 50]. Several laser types and applications were described as an adjunct for the treatment of periodontal diseases [51]. The rational of using laser for the treatment of periodontal pockets relates to the decontamination ability of the affected sites, particularly in situation of difficult access [52]. Moreover, laser application could result in accelerated healing and homeostasis, thus potentially improving the treatment outcomes [52].
Regarding the available evidence on the use of LT or PDT as an adjunct to NSPT, the systematic review published by Salvi and coworkers in 2020, using strict inclusion criteria, evaluated a total of 18 papers, of which only 2 could be included in the quantitative synthesis [20]. Their meta-analysis revealed a non-significant beneficial effect of PDT as an adjunct to NSPT in terms of PD changes [20]. Another systematic review about the application of LT for the management of untreated periodontitis and that performed meta-analysis on five papers did not suggest any significant effect on CAL or PD changes as well as PROMS over time [53]. Other recently published papers have provided further data on the topic without solving the controversy, as both favorable results [54, 55] and clinically insignificant benefits [56] were suggested. The results of our meta-analysis, although showing a significant effect in some comparisons of PDT/LT + NSPT, failed to clearly demonstrate a clinically relevant beneficial effect, being coherent with the previously cited studies.
While all the aforementioned systematic reviews focused on systemically healthy subjects, Abduljabbar and coworkers aimed at exploring the role of lasers as adjunct to NSPT in subjects with diabetes [25]. The authors adopted different inclusion criteria than those considered in the present study, and included six articles in the final qualitative synthesis, three about LT and three about PDT, without presenting conclusive results [25]. Another review of the same group on PDT included four RCTs and concluded that no difference between test and control group could be observed in terms of clinical parameters [26]. Compared to the works by Abduljabbar et al., our research included a higher number of recent papers by using different inclusion criteria, thus presenting updated data on the topic. Moreover, we performed a risk of bias evaluation with standardized methods, and we included in the meta-analysis more outcome variables. Additionally, the present research included the evaluation of the quality of evidence, which should be considered a crucial aspect for weighting the validity of the results.
Another important aspect to consider when dealing with diabetic patients is the effect that periodontal treatment might have on glycemic control. A recent Cochrane systematic review on the improvements in glycemic control (measured by the HbA1c changes) in subjects treated with NSPT compared to controls indicated a decrease of 0.43% (CI95%: 0.28–0.59) of HbA1c in test group at 3–4 months, with positive results also in longer follow-ups [15]. Although our main aim was not to assess changes in diabetes control, our meta-analysis suggested an adjunctive effect of PDT on HbA1c after 3 months of 0.24% (CI95%: 0.17–0.32), which was not confirmed after 6 months. Remarkably, studies on the efficacy of other adjunctive treatments to NSPT in subjects with diabetes, such as systemic antibiotics, found no significant additional effects in terms of glycaemic control [57, 58]. The regression meta-analysis performed in the present review failed to reveal a significant effect of baseline HbA1c% on PD changes and CAL changes. However, it should be noted that the relatively low number of papers available for each outcome and for each comparison may have limited the reliability of such analysis. Nevertheless, the risk of bias evaluation revealed a substantially moderate quality of the included studies, being six studies at low risk of bias. We can therefore reasonably assume that the results of the meta-analysis and the quality appraisal of the evidence were not affected by bias.
It is worth to acknowledge that the present systematic review had few shortcomings, as this might help to better consider the validity of the results and to interpret its findings. First, we should highlight that a substantial heterogeneity existed among the included study protocols regarding the characteristics of diabetes and the level of glycaemic control, the ethnicity of the population, the settings of the laser device, and the characteristic of periodontitis (namely severity), and this was probably the main cause of the statistical heterogeneity in the meta-analysis. Moreover, very limited data were available about the proportion of pocket closure, which is considered the most reliable outcome when evaluating the results of NSPT [59]. The lack of data about this outcome is a limiting factor, although PD and CAL changes are surrogate outcomes widely accepted and reported in the literature [60].
On the other hand, one strength of the present review is that to the best of our knowledge, this is the first systematic review on periodontitis and diabetes that also assessed the certainty of evidence for all the comparisons and outcomes included in the meta-analysis based on GRADE. The GRADE is a well-recognized tool for weighting the level of evidence of assumptions derived from a study, ideally a systematic review, in order to provide also clinical recommendations [32]. The GRADE is now fully integrated in Cochrane systematic reviews [27]; however, it is not frequently adopted in systematic reviews in the field of dentistry. In the authors’ opinion, considering the level of evidence and combining it with the statistical significance and the effect size can better inform on a clinically relevant topic such as the efficacy of PDT/LT. This comprehensive approach should be implemented whenever recommendations or clinically oriented guidelines are produced.
Finally, while it was not within the remit of this review to assess the cost-effectiveness of LT and PDT, the extra costs associated with the purchase and use of these physical therapies should be taken into account when considering whether or not to implement them in clinical practice and future studies are warranted to investigate the cost-effectiveness of these therapies.
In conclusion, taking all the aforementioned limitations into consideration, our review suggested that there is currently insufficient scientific evidence (and limited clinical relevance) to suggest the routine use of PDT or LT as an adjunct to NSPT in subjects with type II diabetes, although the promising results in terms of HbA1c decrease in the short term should be further explored in well-designed RCTs with > 6-month follow-up. It is recommended that future studies should consider the percentage of pocket closure as a primary outcome and explore the role of patient-reported outcome measures. It is also important that future studies will apply standard definitions of diabetes.
Data Availability
The data supporting the findings of this study are available on request from the authors.
References
Preshaw PM, Alba AL, Herrera D, Jepsen S, Konstantinidis A, Makrilakis K et al (2012) Periodontitis and diabetes: a two-way relationship. Diabetologia 55(1):21–31. https://doi.org/10.1007/s00125-011-2342-y
Ahmadinia AR, Rahebi D, Mohammadi M, Ghelichi-Ghojogh M, Jafari A, Esmaielzadeh F et al (2022) Association between type 2 diabetes (T2D) and tooth loss: a systematic review and meta-analysis. BMC Endocr Disord 22(1):100. https://doi.org/10.1186/s12902-022-01012-8
Aldosari M, Aldosari M, Aldosari MA, Agrawal P Diabetes mellitus and its association with dental caries, missing teeth and dental services utilization in the US adult population: results from the 2015-2018 National Health and Nutrition Examination Survey. Diabet Med 2022:e14826. https://doi.org/10.1111/dme.14826
Steigmann L, Miller R, Trapani VR, Giannobile WV, Braffett BH, Pop-Busui R et al (2022) Type 1 diabetes and oral health: findings from the epidemiology of diabetes interventions and complications (EDIC) study. J Diabetes Complications 36(4):108120. https://doi.org/10.1016/j.jdiacomp.2021.108120
Mealey BL, Ocampo GL (2007) Diabetes mellitus and periodontal disease. Periodontol 2000 44:127–153. https://doi.org/10.1111/j.1600-0757.2006.00193.x
Barutta F, Bellini S, Durazzo M, Gruden G (2022) Novel insight into the mechanisms of the bidirectional relationship between diabetes and periodontitis. Biomedicines 10(1). https://doi.org/10.3390/biomedicines10010178
Kocher T, Konig J, Borgnakke WS, Pink C, Meisel P (2018) Periodontal complications of hyperglycemia/diabetes mellitus: epidemiologic complexity and clinical challenge. Periodontol 2000 78(1):59–97. https://doi.org/10.1111/prd.12235
Zhang S, Philips KH, Moss K, Wu D, Adam HS, Selvin E et al (2021) Periodontitis and risk of diabetes in the atherosclerosis risk in communities (ARIC) study: a BMI-modified association. J Clin Endocrinol Metab 106(9):e3546–e3e58. https://doi.org/10.1210/clinem/dgab337
Saito T, Shimazaki Y, Kiyohara Y, Kato I, Kubo M, Iida M et al (2004) The severity of periodontal disease is associated with the development of glucose intolerance in non-diabetics: the Hisayama study. J Dent Res 83(6):485–490. https://doi.org/10.1177/154405910408300610
Park MS, Jeon J, Song TJ, Kim J (2022) Association of periodontitis with microvascular complications of diabetes mellitus: a nationwide cohort study. J Diabetes Complications 36(2):108107. https://doi.org/10.1016/j.jdiacomp.2021.108107
Hajishengallis G (2022) Interconnection of periodontal disease and comorbidities: evidence, mechanisms, and implications. Periodontol 2000 89(1):9–18. https://doi.org/10.1111/prd.12430
Pereira KKY, Jara CM, Antunes GL, Gomes MS, Rosing CK, Cavagni J et al (2022) Effects of periodontitis and periodontal treatment on systemic inflammatory markers and metabolic profile in obese and non-obese rats. J Periodontol. https://doi.org/10.1002/JPER.21-0575
Hajishengallis G, Chavakis T (2021) Local and systemic mechanisms linking periodontal disease and inflammatory comorbidities. Nat Rev Immunol 21(7):426–440. https://doi.org/10.1038/s41577-020-00488-6
D'Aiuto F, Gkranias N, Bhowruth D, Khan T, Orlandi M, Suvan J et al (2018) Systemic effects of periodontitis treatment in patients with type 2 diabetes: a 12 month, single-centre, investigator-masked, randomised trial. Lancet Diabetes Endocrinol 6(12):954–965. https://doi.org/10.1016/S2213-8587(18)30038-X
Simpson TC, Clarkson JE, Worthington HV, MacDonald L, Weldon JC, Needleman I et al (2022) Treatment of periodontitis for glycaemic control in people with diabetes mellitus. Cochrane Database Syst Rev. 4:CD004714. https://doi.org/10.1002/14651858.CD004714.pub4
Corbella S, Francetti L, Taschieri S, De Siena F, Fabbro MD (2013) Effect of periodontal treatment on glycemic control of patients with diabetes: a systematic review and meta-analysis. J Diabetes Investig 4(5):502–509. https://doi.org/10.1111/jdi.12088
Suvan J, Leira Y, Moreno Sancho FM, Graziani F, Derks J, Tomasi C (2020) Subgingival instrumentation for treatment of periodontitis. A systematic review. J Clin Periodontol 47(Suppl 22):155–175. https://doi.org/10.1111/jcpe.13245
Corbella S, Calciolari E, Alberti A, Donos N, Francetti L (2021) Systematic review and meta-analysis on the adjunctive use of host immune modulators in non-surgical periodontal treatment in healthy and systemically compromised patients. Sci Rep 11(1):12125. https://doi.org/10.1038/s41598-021-91506-7
Donos N, Calciolari E, Brusselaers N, Goldoni M, Bostanci N, Belibasakis GN (2020) The adjunctive use of host modulators in non-surgical periodontal therapy. A systematic review of randomized, placebo-controlled clinical studies. J Clin Periodontol 47(Suppl 22):199–238. https://doi.org/10.1111/jcpe.13232
Salvi GE, Stahli A, Schmidt JC, Ramseier CA, Sculean A, Walter C (2020) Adjunctive laser or antimicrobial photodynamic therapy to non-surgical mechanical instrumentation in patients with untreated periodontitis: a systematic review and meta-analysis. J Clin Periodontol 47(Suppl 22):176–198. https://doi.org/10.1111/jcpe.13236
Herrera D, Matesanz P, Martin C, Oud V, Feres M, Teughels W (2020) Adjunctive effect of locally delivered antimicrobials in periodontitis therapy: a systematic review and meta-analysis. J Clin Periodontol 47(Suppl 22):239–256. https://doi.org/10.1111/jcpe.13230
Teughels W, Feres M, Oud V, Martin C, Matesanz P, Herrera D (2020) Adjunctive effect of systemic antimicrobials in periodontitis therapy: a systematic review and meta-analysis. J Clin Periodontol 47(Suppl 22):257–281. https://doi.org/10.1111/jcpe.13264
Wickenheisser VA, Zywot EM, Rabjohns EM, Lee HH, Lawrence DS, Tarrant TK (2019) Laser light therapy in inflammatory, musculoskeletal, and autoimmune disease. Curr Allergy Asthma Rep 19(8):37. https://doi.org/10.1007/s11882-019-0869-z
Liu LS, Gkranias N, Farias B, Spratt D, Donos N (2018) Differences in the subgingival microbial population of chronic periodontitis in subjects with and without type 2 diabetes mellitus-a systematic review. Clin Oral Investig 22(8):2743–2762. https://doi.org/10.1007/s00784-018-2660-2
Abduljabbar T, Javed F, Shah A, Samer MS, Vohra F, Akram Z (2017) Role of lasers as an adjunct to scaling and root planing in patients with type 2 diabetes mellitus: a systematic review. Lasers Med Sci 32(2):449–459. https://doi.org/10.1007/s10103-016-2086-5
Abduljabbar T, Vohra F, Javed F, Akram Z (2017) Antimicrobial photodynamic therapy adjuvant to non-surgical periodontal therapy in patients with diabetes mellitus: a meta-analysis. Photodiagnosis Photodyn Ther 17:138–146. https://doi.org/10.1016/j.pdpdt.2016.11.008
Higgins JPT (2020) Cochrane Collaboration. Cochrane handbook for systematic reviews of interventions. In: Cochrane book series, 2nd edn. Wiley-Blackwell, Hoboken, NJ
Papapanou PN, Sanz M, Buduneli N, Dietrich T, Feres M, Fine DH et al (2018) Periodontitis: consensus report of workgroup 2 of the 2017 world workshop on the classification of periodontal and peri-implant diseases and conditions. J Periodontol 89(Suppl 1):S173–SS82. https://doi.org/10.1002/JPER.17-0721
Armitage GC (2004) Periodontal diagnoses and classification of periodontal diseases. Periodontol 2000 34:9–21. https://doi.org/10.1046/j.0906-6713.2002.003421.x
World Health Organization: International Classification of Diseases of the World Health Organization. https://icd.who.int/en (2022). Accessed 2021.
CDC Centers for Disease Control and Prevention: Diabetes. https://www.cdc.gov/diabetes/index.html (2022). Accessed 2022.
Guyatt GH, Oxman AD, Vist GE, Kunz R, Falck-Ytter Y, Alonso-Coello P et al (2008) GRADE: an emerging consensus on rating quality of evidence and strength of recommendations. BMJ 336(7650):924–926. https://doi.org/10.1136/bmj.39489.470347.AD
DerSimonian R, Laird N (1986) Meta-analysis in clinical trials. Controlled clinical trials 7(3):177–188
Moher D, Shamseer L, Clarke M, Ghersi D, Liberati A, Petticrew M et al (2015) Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015 statement. Syst Rev 4:1. https://doi.org/10.1186/2046-4053-4-1
Al-Zahrani MS, Bamshmous SO, Alhassani AA, Al-Sherbini MM (2009) Short-term effects of photodynamic therapy on periodontal status and glycemic control of patients with diabetes. J Periodontol 80(10):1568–1573
Al-Zawawi AS, Bukhari IA, Bello-Correa FO, Sheikh SA, Albaijan R, Vohra F (2020) Influence of root debridement with adjunct photodynamic therapy on periodontal parameters and gingival crevicular fluid cortisol levels among patients with and without type-2 diabetes mellitus. Photodiagnosis Photodyn Ther 32
Barbosa FI, Araújo PV, Machado LJC, Magalhães CS, Guimarães MMM, Moreira AN (2018) Effect of photodynamic therapy as an adjuvant to non-surgical periodontal therapy: periodontal and metabolic evaluation in patients with type 2 diabetes mellitus. Photodiagnosis Photodyn Ther 22:245–250
Chandra S, Shashikumar P (2019) Diode laser - a novel therapeutic approach in the treatment of chronic periodontitis in type 2 diabetes mellitus patients: a prospective randomized controlled clinical trial. J Lasers Med Sci 10(1):56–63
Dengizek Eltas S, Gursel M, Eltas A, Alptekin NO, Ataoglu T (2019) Evaluation of long-term effects of diode laser application in periodontal treatment of poorly controlled type 2 diabetic patients with chronic periodontitis. Int J Dent Hyg 17(4):292–299
Elsadek MF, Ahmed BM, Alkhawtani DM, Zia SA (2020) A comparative clinical, microbiological and glycemic analysis of photodynamic therapy and Lactobacillus reuteri in the treatment of chronic periodontitis in type-2 diabetes mellitus patients. Photodiagnosis Photodyn Ther 29
Koçak E, Sağlam M, Kayış SA, Dündar N, Kebapçılar L, Loos BG et al (2016) Nonsurgical periodontal therapy with/without diode laser modulates metabolic control of type 2 diabetics with periodontitis: a randomized clinical trial. Lasers Med Sci 31(2):343–353
Macedo Gde O, Novaes AB, Souza SL, Taba M, Palioto DB, Grisi MF (2014) Additional effects of aPDT on nonsurgical periodontal treatment with doxycycline in type II diabetes: a randomized, controlled clinical trial. Lasers Med Sci 29(3):881–886
Mirza S, Khan AA, Al-Kheraif AA, Khan SZ, Shafqat SS (2019) Efficacy of adjunctive photodynamic therapy on the clinical periodontal, HbA1c and advanced glycation end product levels among mild to moderate chronic periodontal disease patients with type 2 diabetes mellitus: a randomized controlled clinical trial. Photodiagnosis Photodyn Ther 28:177–182
Soi S, Bains VK, Srivastava R, Madan R (2021) Comparative evaluation of improvement in periodontal and glycemic health status of type 2 diabetes mellitus patients after scaling and root planing with or without adjunctive use of diode laser. Lasers Med Sci
Claudio MM, Nuernberg MAA, Rodrigues JVS, Belizario LCG, Batista JA, Duque C et al (2021) Effects of multiple sessions of antimicrobial photodynamic therapy (aPDT) in the treatment of periodontitis in patients with uncompensated type 2 diabetes: a randomized controlled clinical study. Photodiagnosis Photodyn Ther 35:102451. https://doi.org/10.1016/j.pdpdt.2021.102451
Armitage GC (1999) Development of a classification system for periodontal diseases and conditions. Ann Periodontol 4(1):1–6. https://doi.org/10.1902/annals.1999.4.1.1
Cao R, Li Q, Wu Q, Yao M, Chen Y, Zhou H (2019) Effect of non-surgical periodontal therapy on glycemic control of type 2 diabetes mellitus: a systematic review and Bayesian network meta-analysis. BMC Oral Health 19(1):176. https://doi.org/10.1186/s12903-019-0829-y
Hasuike A, Iguchi S, Suzuki D, Kawano E, Sato S (2017) Systematic review and assessment of systematic reviews examining the effect of periodontal treatment on glycemic control in patients with diabetes. Med Oral Patol Oral Cir Bucal 22(2):e167–ee76. https://doi.org/10.4317/medoral.21555
Sales LS, Miranda ML, de Oliveira AB, Ferrisse TM, Fontana CR, Milward M et al (2022) Effect of the technique of photodynamic therapy against the main microorganisms responsible for periodontitis: a systematic review of in-vitro studies. Arch Oral Biol 138:105425. https://doi.org/10.1016/j.archoralbio.2022.105425
Cieplik F, Deng D, Crielaard W, Buchalla W, Hellwig E, Al-Ahmad A et al (2018) Antimicrobial photodynamic therapy - what we know and what we don't. Crit Rev Microbiol 44(5):571–589. https://doi.org/10.1080/1040841X.2018.1467876
Cobb CM (2017) Lasers and the treatment of periodontitis: the essence and the noise. Periodontol 2000 75(1):205–295. https://doi.org/10.1111/prd.12137
Jiang Y, Feng J, Du J, Fu J, Liu Y, Guo L et al (2022) Clinical and biochemical effect of laser as an adjunct to non-surgical treatment of chronic periodontitis. Oral Dis 28(4):1042–1057. https://doi.org/10.1111/odi.13847
Lin Z, Strauss FJ, Lang NP, Sculean A, Salvi GE, Stahli A (2021) Efficacy of laser monotherapy or non-surgical mechanical instrumentation in the management of untreated periodontitis patients. A systematic review and meta-analysis. Clin Oral Investig 25(2):375–391. https://doi.org/10.1007/s00784-020-03584-y
Schar D, Ramseier CA, Eick S, Mettraux G, Salvi GE, Sculean A (2020) Transgingival photodynamic therapy (tg-aPDT) adjunctive to subgingival mechanical instrumentation in supportive periodontal therapy. A randomized controlled clinical study. Photodiagnosis Photodyn Ther 32:101971. https://doi.org/10.1016/j.pdpdt.2020.101971
Sukumar K, Tadepalli A, Parthasarathy H, Ponnaiyan D (2020) Evaluation of combined efficacy of photodynamic therapy using indocyanine green photosensitizer and non-surgical periodontal therapy on clinical and microbial parameters in the management of chronic periodontitis subjects: a randomized split-mouth design. Photodiagnosis Photodyn Ther 31:101949. https://doi.org/10.1016/j.pdpdt.2020.101949
Katsikanis F, Strakas D, Vouros I (2020) The application of antimicrobial photodynamic therapy (aPDT, 670 nm) and diode laser (940 nm) as adjunctive approach in the conventional cause-related treatment of chronic periodontal disease: a randomized controlled split-mouth clinical trial. Clin Oral Investig 24(5):1821–1827. https://doi.org/10.1007/s00784-019-03045-1
Yap KCH, Pulikkotil SJ (2019) Systemic doxycycline as an adjunct to scaling and root planing in diabetic patients with periodontitis: a systematic review and meta-analysis. BMC Oral Health 19(1):209. https://doi.org/10.1186/s12903-019-0873-7
Lira Junior R, Santos CMM, Oliveira BH, Fischer RG, Santos APP (2017) Effects on HbA1c in diabetic patients of adjunctive use of systemic antibiotics in nonsurgical periodontal treatment: a systematic review. J Dent 66:1–7. https://doi.org/10.1016/j.jdent.2017.08.001
Citterio F, Gualini G, Chang M, Piccoli GM, Giraudi M, Manavella V et al (2022) Pocket closure and residual pockets after non-surgical periodontal therapy: a systematic review and meta-analysis. J Clin Periodontol 49(1):2–14. https://doi.org/10.1111/jcpe.13547
Loos BG, Needleman I (2020) Endpoints of active periodontal therapy. J Clin Periodontol 47(Suppl 22):61–71. https://doi.org/10.1111/jcpe.13253
Follmann D, Elliott P, Suh I, Cutler J (1992) Variance imputation for overviews of clinical trials with continuous response. J Clin Epidemiol 45(7):769–773. https://doi.org/10.1016/0895-4356(92)90054-q
Abrams KR, Gillies CL, Lambert PC (2005) Meta-analysis of heterogeneously reported trials assessing change from baseline. Stat Med 24(24):3823–3844. https://doi.org/10.1002/sim.2423
Funding
Open access funding provided by Università degli Studi di Milano within the CRUI-CARE Agreement.
Author information
Authors and Affiliations
Contributions
SC: conceptualization, methodology, formal analysis, investigation, writing–original draft preparation; EC: conceptualization, methodology, investigation, writing–original draft preparation; ND: conceptualization, methodology, investigation, writing–review and editing, supervision; AA: conceptualization, investigation, data curation, writing–review and editing; PE: conceptualization, investigation, data curation, writing–review and editing; LF: conceptualization, methodology, investigation, writing–review and editing, supervision.
Corresponding author
Ethics declarations
Ethical approval
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendices
Appendix 1. Search strategy
Appendix 2. Procedure for calculating SD
For each presented outcome, the difference between baseline and follow-up values were extracted (with specific error measure such as standard deviation (SD) or standard error (SE) or variance). When such parameter was not presented, it was computed as the difference between baseline and follow-up values. In these cases, following the instructions of the Cochrane Handbook for Systematic Reviews when SDs of changes values were not presented and they were not provided by authors after contacting them by email, they were computed as follows: i) if similar studies were present (similar treatment, similar population, similar sample size), SD was imputed taking the value of the other study; ii) when P value is presented SD was computed by using T tables for retrieving SEs; iii) when P value is presented as a limit (e.g. < 0.05) a conservative value of P (e.g. 0.05 in case of < 0.05) was considered for computing SE as described before; iv) if P value was not present SDs of change values was imputed by using the following formula [27, 61, 62]:
being CORR the correlation coefficient, that could be imputed from similar studies if present, or it was assumed conservatively to be 0.2. For each measure, pooled estimate of 95% CI was calculated.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Corbella, S., Calciolari, E., Donos, N. et al. Laser treatments as an adjunct to non-surgical periodontal therapy in subjects with periodontitis and type 2 diabetes mellitus: a systematic review and meta-analysis. Clin Oral Invest 27, 1311–1327 (2023). https://doi.org/10.1007/s00784-023-04873-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00784-023-04873-y