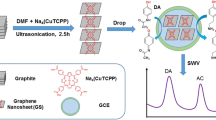
Abstract
A strategy is presented for doping graphene into layered double hydroxide films (LDHs) as a means of improving charge transport of the LDH film in a modified glassy carbon electrode. This result in an enhanced electrocatalytic current for dopamine (DA) and a good separation of the potentials of DA, uric acid and ascorbic acid. Under selected conditions, the square wave voltammetric response of the electrode to DA is linear in the concentration range from 1.0 to 199 μM even in the presence of 0.1 mM ascorbic acid, and the detection limit is 0.3 μM at a signal-to-noise ratio of 3. The method was applied to the determination of DA in pharmaceutical injections with satisfactory results.

A strategy is presented for doping graphene into layered double hydroxide films (LDHs) as a means of improving charge transport of the LDH film in a modified glassy carbon electrode. This result in an enhanced electrocatalytic current for dopamine (DA) and the square wave voltammetric response of the electrode to DA is linear in the concentration range from 1.0 to 199 μM with the detection limit of 0.3 μM. The method was applied to the determination of DA in pharmaceutical injections with satisfactory results.





Similar content being viewed by others
References
Braterman PS, Xu Z, Yarberry F (2004) In: Auerbach SM, Carrado KA, Dutta PK (eds) Handbook of layered materials, Part II. Marcel Dekker, New York, p 373
Wang X, Lu J, Shi W, Li F, Wei M, Evans DG, Duan X (2010) A thermochromic thin film based on host − guest interactions in a layered double hydroxide. Langmuir 26:1247
Li L, Feng Y, Li Y, Zhao W, Shi J (2009) Fe3O4 core/layered double hydroxide shell nanocomposite:versatile magnetic matrix for anionic functional materials. Angew Chem Int Ed 48:5888
Mousty C (2010) Biosensing applications of clay-modified electrodes: a review. Anal Bioanal Chem 396:315
Li M, Chen S, Ni F, Wang Y, Wang L (2008) Layered double hydroxides functionalized with anionic surfactant: direct electrochemistry and electrocatalysis of hemoglobin. Electrochim Acta 53:7255
Ni F, Wang Y, Zhang D, Gao F, Li M (2010) Electrochemical oxidation of epinephrine and uric acid at a layered double hydroxide film modified glassy carbon electrode and its application. Electroanalysis 22:1130
Li M, Xu S, Ni F, Wang Y, Chen S, Wang L (2009) Fast and sensitive non-enzymatic glucose concentration determination using electroactive anionic clay modified electrode. Microchim Acta 166:203
Li M, Ni F, Wang Y, Xu S, Zhang D, Chen S, Wang L (2009) Sensitive and facile determination of catechol and hydroquinone simultaneously under coexistence of resorcinol with a Zn/Al layered double hydroxide film modified glassy carbon electrode. Electroanalysis 21:1521
Wang Y, Liu L, Zhang D, Xu S, Li M (2010) A new strategy for immobilization of electroactive species on the surface of solid electrode. Electrocatalysis 12:30
Mondal D, Villemure G (2009) Effect of the presence of [Co(bpy)3]2+ on the electrochromic responses of films of a redox active Ni–Al-layered double hydroxide. J Electroanal Chem 628:67
Wang Y, Zhang D, Tang M, Xu S, Li M (2010) Electrocatalysis of gold nanoparticles/layered double hydroxides nanocomposites toward methanol electro-oxidation in alkaline medium. Electrochim Acta 55:4045
Novoselov KS, Geim AK, Morozov SV, Jiang D, Zhang Y, Dubonos SV, Grigorieva IV, Firsov AA (2004) Electric field effect in atomically thin carbon films. Science 306:666
Wang X, Zhi L, Mullen K (2008) Transparent, conductive graphene electrodes for dye-sensitized solar cells. Nano Lett 8:323
Pumera M (2010) Graphene-based nanomaterials and their electrochemistry. Chem Soc Rev 39:4146
Brownson DAC, Banks CE (2010) Graphene electrochemistry: an overview of potential applications. Analyst 135:2768
Gilje S, Han S, Wang K, Kaner RB (2007) A chemical route to graphene for device applications. Nano Lett 7:3394
Wang C, Zhang L, Guo Z, Xu J, Wang H, Zhai K, Zhuo X (2010) A novel hydrazine electrochemical sensor based on the high specific surface area grapheme. Microchim Acta 169:1
Li Y, Tang L, Li J (2009) Preparation and electrochemical performance for methanol oxidation of pt/graphene nanocomposites. Electrochem Commun 11:846
Bunch JS, van der Zande AM, Verbridge SS, Frank IW, Tanenbaum DM, Parpia JM, Craighead HG, McEuen PL (2007) Electromechanical resonators from graphene sheets. Science 315:490
Zhang Y, Li H, Pan L, Lu T, Sun Z (2009) Capacitive behavior of graphene–ZnO composite film for supercapacitors. J Electroanal Chem 634:68
Li F, Wang Y, Yang Q, Evans DG, Forano C, Duan X (2005) Study on adsorption of glyphosate (N-phosphonomethyl glycine) pesticide on MgAl-layered double hydroxides in aqueous solution. J Hazard Mater 125:89
He F, Zhang L (2007) Organo-modified ZnAl layered double hydroxide as new catalyst support for the ethylene polymerization. J Colloid Interface Sci 315:439
Li M, Xu S, Tang M, Liu L, Gao F, Wang Y (2011) Direct electrochemistry of horseradish peroxidase on graphene-modified electrode for electrocatalytic reduction towards H2O2. Electrochim Acta 56:1144
Xu Z, Stevenson G, Lu C, Lu G (2006) Dispersion and size control of layered double hydroxide nanoparticles in aqueous solutions. J Phys Chem B 110:16923
Orazem ME, Tribollet B (2008) Electrochemical impedance spectroscopy. Wiley, New York
Kharitonov AB, Alfonta L, Katz E, Willner I (2000) Probing of bioaffinity interactions at interfaces using impedance spectroscopy and chronopotentiometry. J Electroanal Chem 487:133
Tan Y, Deng W, Li Y, Huang Z, Meng Y, Xie Q, Ma M, Yao S (2010) Polymeric bionanocomposite cast thin films with in situ laccase-catalyzed polymerization of dopamine for biosensing and biofuel cell applications. J Phys Chem B 114:5016
Zhang Y, Li B, Chen X (2010) Simple and sensitive detection of dopamine in the presence of high concentration of ascorbic acid using gold nanoparticles as colorimetric probes. Microchim Acta 168:107
Sun Y, Fei J, Hou J, Zhang Q, Liu Y, Hu B (2009) Simultaneous determination of dopamine and serotonin using a carbon nanotubes-ionic liquid gel modified glassy carbon electrode. Microchim Acta 165:373
Wang G, Sun J, Zhang W, Jiao S, Fang B (2009) Simultaneous determination of dopamine, uric acid and ascorbic acid with LaFeO3 nanoparticles modified electrode. Microchim Acta 164:357
Li Y, Huang X, Chen Y, Wang L, Lin X (2009) Simultaneous determination of dopamine and serotonin by use of covalent modification of 5-hydroxytryptophan on glassy carbon electrode. Microchim Acta 164:107
Li J, Wei W, Luo S (2010) A novel one-step electrochemical codeposition of carbon nanotubes-DNA hybrids and tiron doped polypyrrole for selective and sensitive determination of dopamine. Microchim Acta 171:109
Alwarappan S, Erdem A, Liu C, Li C (2009) Probing the electrochemical properties of graphene nanosheets for biosensing applications. J Phys Chem C 113:8853
Wang Y, Li Y, Tang L, Lu J, Li J (2009) Application of graphene-modified electrode for selective detection of dopamine. Electrochem Commun 11:889
Zhou M, Zhai Y, Dong S (2009) Electrochemical sensing and biosensing platform based on chemically reduced graphene oxide. Anal Chem 81:5603
Wang AJ, Feng JJ, Li YF, Xi JL, Dong WJ (2010) In-situ decorated gold nanoparticles on polyaniline with enhanced electrocatalysis toward dopamine. Microchim Acta 171:431
Joshi P, Joshi HC, Sanghi SK, Kundu S (2010) Immobilization of monoamine oxidase on eggshell membrane and its application in designing an amperometric biosensor for dopamine. Microchim Acta 169:338
Zhang L (2008) Covalent modification of glassy carbon electrode with cysteine for the determination of dopamine in the presence of ascorbic acid. Microchim Acta 161:191
Lai GS, Zhang HL, Han DY (2008) Electrocatalytic oxidation and voltammetric determination of dopamine at a Nafion/carbon-coated iron nanoparticles-chitosan composite film modified electrode. Microchim Acta 160:233
Acknowledgements
This work is supported by National Natural Science Foundation of China (Grant Nos. 20801001, 21055001, and 21075001) and Anhui Provincial Natural Science Foundation (Grant No. 11040606 M46). We also deeply appreciate the support of the foundation for doctor science research of Anhui Normal University.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Wang, Y., Peng, W., Liu, L. et al. Enhanced conductivity of a glassy carbon electrode modified with a graphene-doped film of layered double hydroxides for selectively sensing of dopamine. Microchim Acta 174, 41–46 (2011). https://doi.org/10.1007/s00604-011-0593-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00604-011-0593-4