Abstract
Main conclusion
CaLCuV-based VIGS effectively works in cabbage and contributes to efficient functional genomics research in Brassica crop species.
Abstract
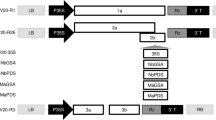
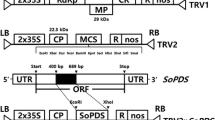
Virus-induced gene silencing (VIGS), a posttranscriptional gene silencing method, is an effective technique for analysing the functions of genes in plants. However, no VIGS vectors have been available for Brassica oleracea until now. Here, tobacco rattle virus (TRV), pTYs and cabbage leaf curl virus (CaLCuV) gene-silencing vectors (PCVA/PCVB) were chosen to improve the VIGS system in cabbage using the phytoene desaturase (PDS) gene as an efficient visual indicator of VIGS. We successfully silenced the expression of PDS and observed photobleaching phenomena in cabbage in response to pTYs and CaLCuV, with the latter being more easy to operate and less expensive. The parameters potentially affecting the silencing efficiency of VIGS by CaLCuV in cabbage, including the targeting fragment strategy, inoculation method and incubation temperature, were then compared. The optimized CaLCuV-based VIGS system involves the following: an approximately 500 bp insert sequence, an Agrobacterium OD600 of 1.0, use of the vacuum osmosis method applied at the bud stage, and an incubation temperature of 22 °C. Using these parameters, we achieved a stable silencing efficiency of 65%. To further test the effectiveness of the system, we selected the Mg-chelatase H subunit (ChlH) gene in cabbage and knocked down its expression, and we observed yellow leaves, as expected. We successfully applied the CaLCuV-based VIGS system to two other representative Brassica crop species, B. rapa and B. nigra, and thus expanded the application scope of this system. Our VIGS system described here will contribute to efficient functional genomics research in Brassica crop species.







Similar content being viewed by others
Availability of data and materials
The datasets used and/or analysed during the current study are available from the corresponding author upon reasonable request.
Abbreviations
- CaLCuV:
-
Cabbage leaf curl virus
- ChlH:
-
Chelatase H subunit
- PDS:
-
Phytoene desaturase
- TRV:
-
Tobacco rattle virus
- VIGS:
-
Virus-induced gene silencing
References
Ayako O, Takumi O, Chie K, Kanako K, Mizue I, Jun I, Nobuya K (2018) CRISPR/Cas9-mediated genome editing of the fatty acid desaturase 2 gene in Brassica napus. Plant Physiol Biochem 131:63–69. https://doi.org/10.1016/j.plaphy.2018.04.025
Beyene G, Chauhan RD, Taylor NJ (2017) A rapid virus-induced gene silencing (VIGS) method for assessing resistance and susceptibility to cassava mosaic disease. Virol J 14:47. https://doi.org/10.1186/s12985-017-0716-6
Burch-Smith TM, Anderson JC, Martin GB, Dinesh-Kumar SP (2004) Applications and advantages of virus-induced gene silencing for gene function studies in plants. Plant J 39:734–746. https://doi.org/10.1111/j.1365-313x.2004.02158.x
Burch-Smith TM, Schiff M, Liu Y, Dinesh-Kumar SP (2006) Efficient virus-induced gene silencing in Arabidopsis. Plant Physiol 142:21–27. https://doi.org/10.1104/pp.106.084624
Cai XZ, Xu QF, Wang CC, Zheng Z (2006) Development of a virus-induced gene silencing system for functional analysis of the RPS2-dependent resistance signaling pathway in Arabidopsis. Plant Mol Biol 62:223–232. https://doi.org/10.1007/s11103-006-9016-z
Chen G, Zeng F, Wang J, Wang J, Ye X, Zhu S, Yuan L, Hou J, Wang C (2019) Transgenic Wucai (Brassica campestris L.) produced via Agrobacterium-mediated anther transformation in planta. Plant Cell Rep 38:577–586. https://doi.org/10.1007/s00299-019-02387-0
Chen I, Chiu MH, Cheng SF, Hsu YH, Tsai CH (2013) The glutathione transferase of Nicotiana benthamiana NbGSTU4 plays a role in regulating the early replication of bamboo mosaic virus. New Phytol 199:749–757. https://doi.org/10.1111/nph.12304
Chen JC, Jiang CZ, Gookin T, Hunter D, Clark D, Reid M (2004) Chalcone synthase as a reporter in virus induced gene silencing studies of flower senescence. Plant Mol Biol 55:521–530. https://doi.org/10.1007/s11103-004-0590-7
Chen WW, Zhang Q, Kong JH, Hu F, Li B, Wu CQ, Qin C, Zhang PC, Shi NN, Hong YG (2015) Mr VIGS: microRNA-based virus-induced gene silencing in plants. Methods Mol Biol 1287:147–157. https://doi.org/10.1007/978-1-4939-2453-0_11
Dinesh-Kumar SP, Anandalakshmi R, Marathe R, Schiff M, Liu Y (2011) Virus-induced gene silencing. Methods Mol Biol 236:287–294. https://doi.org/10.1385/1-59259-413-1:287
Faivre-Rampant O, Gilroy EM, Hrubikova K, Hein I, Millam S, Loake GJ, Birch P, Taylor M, Lacomme C (2004) Potato virus X-induced gene silencing in leaves and tubers of potato. Plant Physiol 134:1308–1316. https://doi.org/10.1104/pp.103.037507
Fang ZY, Sun PT, Liu YM, Wang XW, Hou AF, Bian CS (1997) A male sterile line with dominant gene (Ms) in cabbage (Brassica oleracea var. capitata) and its utilization for hybrid seed production. Euphytica 97:265–268. https://doi.org/10.1023/A:1003026523150
Fauquet C, Fargette D, Thouvenel JC (1988) Some aspects of the epidemiology of African cassava mosaic virus in ivory coast. Pans Pest Artic News Summ 34:92–96. https://doi.org/10.1080/09670878809371217
Fernández-Calvino L, Guzmán-Benito I, Del TF, Donaire L, Castro-Sanz AB, Ruíz-Ferrer V, Llave C (2016) Activation of senescence-associated dark-inducible (DIN) genes during infection contributes to enhanced susceptibility to plant viruses. Mol Plant Pathol 17:3–15. https://doi.org/10.1111/mpp.12257
Fernandez-Moreno JP, Orzaez D, Granell A (2013) VIGS: a tool to study fruit development in Solanum lycopersicum. Methods Mol Biol 975:183. https://doi.org/10.1007/978-1-62703-278-0_14
Fofana IBF, Sangaré A, Collier R, Taylor C, Fauquet CM (2014) A geminivirus-induced gene silencing system for gene function validation in cassava. Plant Mol Biol 56:613–624. https://doi.org/10.1007/s11103-004-0161-y
Fu DQ, Zhu BZ, Zhu HL, Jiang WB, Luo YB (2005) Virus-induced gene silencing in tomato fruit. Plant J 43:299–308. https://doi.org/10.1111/j.1365-313x.2005.02441.x
George GM, Bauer R, Blennow A, Kossmann J, Lloyd JR (2012) Virus-induced multiple gene silencing to study redundant metabolic pathways in plants: silencing the starch degradation pathway in Nicotiana benthamiana. Biotechnol J 7:884–890. https://doi.org/10.1002/biot.201100469
Godge MR, Purkayastha A, Dasgupta I, Kumar PP (2008) Virus-induced gene silencing for functional analysis of selected genes. Plant Cell Rep 27:209–219. https://doi.org/10.1007/s00299-007-0460-2
Gosselé V, Faché I, Meulewaeter F, Cornelissen M, Metzlaff M (2003) SVISS—a novel transient gene silencing system for gene function discovery and validation in tobacco plants. Plant J 32:859–866. https://doi.org/10.1046/j.1365-313X.2002.01471.x
Hamilton AJ, Baulcombe DC (1999) A species of small antisense RNA in posttranscriptional gene silencing in plants. Science 286:950–952. https://doi.org/10.1126/science.286.5441.950
Han F, Yuan K, Kong C, Zhang X, Yang YM, Zhuang M, Zhang YY, Li ZS, Fang ZY, Lv HH (2018) Fine mapping and candidate gene identification of the genic male-sterile gene ms3 in cabbage 51S. Theor Appl Genet 131:2651–2661. https://doi.org/10.1007/s00122-018-3180-9
Han FQ, Yang C, Fang ZY, Yang YM, Zhuang M, Lv HH, Liu YM, Li ZS, Liu B, Yu HL, Liu XP, Zhang YY (2015) Inheritance and InDel markers closely linked to petal color gene (CPC-1) in Brassica oleracea. Mol Breed 35:160. https://doi.org/10.1007/s11032-015-0354-x
Hands P, Vosnakis N, Betts D, Irish VF, Drea S (2011) Alternate transcripts of a floral developmental regulator have both distinct and redundant functions in Opium poppy. Ann Bot 107:1557–1566. https://doi.org/10.1093/aob/mcr045
Hericourt F, Jupin I (1993) Infection of Arabidopsis thaliana plants by Turnip yellow mosaic virus (TYMV). Virologie 3:133–134
Hidalgo OM, Bartholmes CM, Gleissberg S (2012) Virus-induced gene silencing (VIGS) in Cysticapnos vesicaria, a zygomorphic-flowered Papaveraceae (Ranunculales basal eudicots). Ann Bot 109:911–920. https://doi.org/10.1093/aob/mcs008
Hiriart JB, Aro EM, Lehto K (2003) Dynamics of the VIGS-mediated chimeric silencing of the Nicotiana benthamiana CHLH gene and of the tobacco mosaic virus vector. Mol Plant Microbe In 16:99–106. https://doi.org/10.1094/mpmi.2003.16.2.99
Holzberg S, Brosio P, Gross C, Pogue GP (2010) Barley stripe mosaic virus-induced gene silencing in a monocot plant. Plant J 3:315–327. https://doi.org/10.1046/j.1365-313X.2002.01291.x
Hsieh MH, Pan ZJ, Lai PH, Lu HC, Yeh HH, Hsu CC, Wu WL, Chung MC, Wang SS, Chen WH, Chen HH (2013) Virus-induced gene silencing unravels multiple transcription factors involved in floral growth and development in Phalaenopsis orchids. J Exp Bot 64:3869–3884. https://doi.org/10.1093/jxb/ert218
Igarashi A, Yamagata K, Sugai T, Takahashi Y, Sugawara E, Tamura A, Yaegashi H, Yamagishi N, Takahashi T, Isogai M, Takahashi H, Yoshikawa N (2009) Apple latent spherical virus vectors for reliable and effective virus-induced gene silencing among a broad range of plants including tobacco, tomato, Arabidopsis thaliana, cucurbits, and legumes. Virology 386:407–416. https://doi.org/10.1016/j.virol.2009.01.039
Jeuken MJW, Zhang NW, McHale LK, Pelgrom K, den Boer E, Lindhout P, Michelmore RW, Visser RGF, Niks RE (2009) Rin4 causes hybrid necrosis and race-specific resistance in an interspecific lettuce hybrid. Plant Cell 21:3368–3378. https://doi.org/10.1105/tpc.109.070334
Jeyabharathy C, Shakila H, Usha R (2015) Development of a VIGS vector based on the β-satellite DNA associated with bhendi yellow vein mosaic virus. Virus Res 195:73–78. https://doi.org/10.1016/j.virusres.2014.08.009
Jun L, Yan Y, Xiao-Hui Z, Xuan-Yun J, Sheng-You B, Yong Z (2018) Establishment and optimization of virus-induced gene silencing in eggplants. Jiangsu J Agric Sci
Kang L, Udayakumar M, Mysore KS (2010) A systematic study to determine the extent of gene silencing in Nicotiana benthamiana and other Solanaceae species when heterologous gene sequences are used for virus-induced gene silencing. New Phytol 176:782–791. https://doi.org/10.1111/j.1469-8137.2007.02225.x
Kim J, Park M, Jeong ES, Lee JM, Choi D (2017) Harnessing anthocyanin-rich fruit: a visible reporter for tracing virus-induced gene silencing in pepper fruit. Plant Methods 13:3. https://doi.org/10.1016/j.virusres.2014.08.009
Lacomme C, Hrubikova K, Hein I (2003) Enhancement of virus-induced gene silencing through viral-based production of inverted-repeats. Plant J 34:543–553. https://doi.org/10.1046/j.1365-313X.2003.01733.x
Lawrenson T, Shorinola O, Stacey N (2015) Induction of targeted, heritable mutations in barley and Brassica oleracea using RNA-guided Cas9 nuclease. Genome Biol 16:258. https://doi.org/10.1186/s13059-015-0826-7
Lee MK, Kim HS, Kim JS, Kim SH, Park YD (2004) Agrobacterium-mediated transformation system for large-scale producion of transgenic chinese cabbage (Brassica rapa L. ssp. pekinensis) plants for insertional mutagenesis. J Plant Biol 47:300–306. https://doi.org/10.1007/bf03030544
Liu E, Page JE (2008) Optimized cDNA libraries for virus-induced gene silencing (VIGS) using tobacco rattle virus. Plant Methods 4:5. https://doi.org/10.1186/1746-4811-4-5
Liu X, Han FQ, Kong CC, Fang ZY, Yang LM, Zhang YY, Zhuang M, Liu YM, Li ZS, Lv HH (2017) Rapid introgression of the Fusarium wilt resistance gene into an elite cabbage line through the combined application of a microspore culture, genome background analysis, and disease resistance-specific marker assisted foreground selection. Front Plant Sci 8:354. https://doi.org/10.3389/fpls.2017.00354
Liu XP, Yang C, Han FQ, Fang ZY, Yang LM, Zhuang M, Lv HH, Liu YM, Li ZS, Zhang YY (2016) Genetics and fine mapping of a yellow-green leaf gene (ygl-1) in cabbage (Brassica oleracea var. capitata L.). Mol Breed 36:82. https://doi.org/10.1007/s11032-016-0509-4
Liu Y, Nakayama N, Schiff M, Litt A, Irish VF, Dinesh-Kumar SP (2004) Virus induced gene silencing of a deficiens ortholog in Nicotiana benthamiana. Plant Mol Biol 54:701–711. https://doi.org/10.1023/B:PLAN.0000040899.53378.83
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔ c t method. Methods 25:402–408. https://doi.org/10.1006/meth.2001.1262
Manmathan H, Shaner D, Snelling J, Tisserat N, Lapitan N (2013) Virus-induced gene silencing of Arabidopsis thaliana gene homologues in wheat identifies genes conferring improved drought tolerance. J Exp Bot 64:1381–1392. https://doi.org/10.1093/jxb/ert003
Navarro Gallón SM, Elejalde-Palmett C, Daudu D (2017) Virus-induced gene silencing of the two squalene synthase isoforms of apple tree (Malus × domestica L.) negatively impacts phytosterol biosynthesis, plastid pigmentation and leaf growth. Planta 246:1–16. https://doi.org/10.1007/s00425-017-2681-0
Naylor M, Reeves J, Cooper JI, Edwards ML, Wang H (2005) Construction and properties of a based on Poplar mosaic virus (genus Carlavirus). J Virol Methods 124:27–36. https://doi.org/10.1016/j.jviromet.2004.10.007
Nooduan M, Christophe B, Herve V, Dominique R (2004) Geminivirus VIGS of endogenous genes requires SGS2/SDE1 and SGS3 and defines a new branch in the genetic pathway for silencing in plants. Plant J 38:1004–1014. https://doi.org/10.1111/j.1365-313X.2004.02103.x
Pandey P, Choudhury NR, Mukherjee SK (2009) A geminiviral amplicon (VA) derived from tomato leaf curl virus (ToLCV) can replicate in a wide variety of plant species and also acts as a VIGS vector. Virol J 6:152. https://doi.org/10.1186/1743-422X-6-152
Peng Y, Cui XF, Zhou XP (2002) Plant viruses-novel vectors for expressing foreign genes. J Zhejiang Uni-SC A 28:465–472. https://doi.org/10.1007/s11769-002-0028-6
Qu J, Guo XQ, Ci XY, Qi D, Wen FJ (2003) Recent advances on molecular biology of potato virus X and its application to gene expression vector. Virol Sin 18:87–92
Ratcliff F, Harrison BD, Baulcombe DC (1997) A similarity between viral defense and gene silencing in plants. Science 276:1558–1560. https://doi.org/10.1126/science.276.5318.1558
Richard TJ, Haigler CH, Dominique R (2012) Method: low-cost delivery of the cotton leaf crumple virus-induced gene silencing system. Plant Methods 8:27. https://doi.org/10.1186/1746-4811-8-27
Sasaki S, Yamagishi N, Yoshikawa N (2011) Efficient virus-induced gene silencing in apple, pear and Japanese pear using apple latent spherical virus vectors. Plant Methods 7:15. https://doi.org/10.1186/1746-4811-7-15
Shao Y, Zhu HL, Tian HQ, Wang XG, Lin XJ, Zhu BZ, Xie YH, Luo YB (2003) Virus-induced gene silencing in plant species. Russ J Plant Physiol 30:296–303. https://doi.org/10.1134/s1021443708020027
Spitzer-Rimon B, Cna'Ani A, Vainstein A (2013) Virus-aided gene expression and silencing using TRV for functional analysis of floral scent-related genes. Methods Mol Biol 975:139–148. https://doi.org/10.1007/978-1-62703-278-0_10
Szittya G, Dániel S, Attila M, Zoltán H, Agnes L, Lóránt L, Bánfalvi Z, Burgyán J (2003) Low temperature inhibits RNA silencing-mediated defence by the control of siRNA generation. EMBO J 22:633–640. https://doi.org/10.1093/emboj/cdg74
Tang Y, Lai Y, Liu Y (2013) Virus-induced gene silencing using artificial miRNAs in Nicotiana benthamiana. Methods Mol Biol 975:99–107. https://doi.org/10.1007/978-1-62703-278-0_8
Thomas CL, Jones L, Baulcombe DC, Maule AJ (2010) Size constraints for targeting post-transcriptional gene silencing and for RNA-directed methylation in Nicotiana benthamiana using a potato virus X vector. Plant J 25:417–425. https://doi.org/10.1046/j.1365-313x.2001.00976.x
Thomas W, Markus N, Thomas D, Manfred K, Marco H (2017) Efficient generation of mutations mediated by CRISPR/Cas9 in the hairy root transformation system of Brassica carinata. PLoS ONE 12:e0185429. https://doi.org/10.1371/journal.pone.0185429
Tuttle JR, Idris A, Brown JK, Haigler CH, Robertson D (2008) Geminivirus-mediated gene silencing from Cotton leaf crumple virus is enhanced by low temperature in cotton. Plant Physiol 148:41–50. https://doi.org/10.1104/pp.108.123869
Xiao Z, Hu Y, Zhang XL, Xue YQ, Fang ZY, Yang LM, Zhang YY, Liu YM, Li ZS, Liu X, Liu ZZ, Lv HH, Zhuang M (2017) Fine mapping and transcriptome analysis reveal candidate genes associated with hybrid lethality in cabbage (Brassica oleracea). Genes 8:2–17. https://doi.org/10.3390/genes8060147
Xiong XP, Liu WM, Jiang JX, Xu LA, Cao JS (2019) Efficient genome editing of Brassica campestris based on the CRISPR/Cas9 system. Mol Genet Genomics 5:1251–1261. https://doi.org/10.1007/s00438-019-01564-w
Xu H, Xu LF, Yang PP, Cao YW, Tang YC, He GR, Yuan SX, Ming J (2018) Tobacco rattle virus-induced PHYOENEN DESATURASE (PDS) and Mg-chelatase H subunit (ChlH) gene silencing in Solanum pseudocapsicum L. Peer J 6:e4424. https://doi.org/10.7717/peerj.4424
Yu J, Yang XD, Wang Q, Gao LW, Yang Y, Xiao D, Liu TK, Li Y, Hou XL, Zhang CW (2018) Efficient virus-induced gene silencing in Brassica rapa using a turnip yellow mosaic virus vector. Biol Plant 62:826–834. https://doi.org/10.1007/s10535-018-0803-6
Zhang J, Yu DH, Zhang Y, Liu K, Xu KD, Zhang FL, Wang J, Tan GX, Nie XH, Ji QH, Zhao L, Li CW (2017) Vacuum and co-cultivation agroinfiltration of (germinated) seeds results in tobacco rattle virus (TRV) mediated whole-plant virus-induced gene silencing (VIGS) in wheat and maize. Front Plant Sci 8:393. https://doi.org/10.3389/fpls.2017.00393
Zhao Y, Mao WW, Chen YT, Wang W, Dai ZR, Dou ZC, Zhang K, Wei LZ, Li TY, Zeng BZ, Liu T, Fan YJ, Yan YJ, Li BB, Jia WS (2019) Optimization and standardization of transient expression assays for gene functional analyses in strawberry fruits. Hortic Res 6:53. https://doi.org/10.1038/s41438-019-0135-5
Zhao Z, Liu FZ, Zhang Y, Qi DX, Chen YH, Lian Y (2015) VIGS expression vector construction and expression analyses of SmMsrA gene in eggplant. Acta Hortic Sin 42(8):1495–1504
Zheng SJ, Snoeren TA, Hogewoning SW, van Loon JJ, Dicke M (2010) Disruption of plant carotenoid biosynthesis through virus-induced gene silencing affects oviposition behaviour of the butterfly Pieris rapae. New Phytol 186:733–745. https://doi.org/10.1111/j.1469-8137.2010.03213.x
Acknowledgements
The work was performed at the Key Laboratory of Biology and Genetic Improvement of Horticultural Crops, Ministry of Agriculture, Beijing 100081, China. We thank Su Xiaoming (Shandong Academy of Agricultural Sciences, Jinan, Shandong Province, China) for providing the TRV vectors, Prof Zhang Changwei (Nanjing Agricultural University, Nanjing, Jiangsu Province, China) for providing the pTYs vectors and Liu Yule (Tsinghua University, Beijing, China) for providing the PCVA/PCVB vectors.
Funding
This study was supported by grants from the Key Projects of the National Key Research and Development Program of China (2016YFD0100307), the National Natural Science Foundation of China (31572139), Central Public-interest Scientific Institution Basal Research Fund (Y2020PT01), the Science and Technology Innovation Program of the Chinese Academy of Agricultural Sciences (CAAS-ASTIP-IVFCAAS) and the earmarked fund for the Modern Agro-Industry Technology Research System, China (CARS-23).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethics approval and consent to participate
No human subjects were involved in this study.
Additional information
Communicated by Dorothea Bartels.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Xiao, Z., Xing, M., Liu, X. et al. An efficient virus-induced gene silencing (VIGS) system for functional genomics in Brassicas using a cabbage leaf curl virus (CaLCuV)-based vector. Planta 252, 42 (2020). https://doi.org/10.1007/s00425-020-03454-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00425-020-03454-7