Abstract
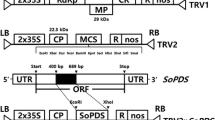
Virus-induced gene silencing (VIGS) is widely used for functional analysis of genes in plants. Because of the variation in its effectiveness among plant species, the VIGS system is normally optimized using the phytoene desaturase (PDS) gene as a visible indicator. In this study, we optimized an efficient VIGS system for petunias using the PDS gene isolated from pepper (Capsicum annuum) plants as a visible indicator. Application of the CaPDS gene effectively induced gene silencing in the petunia cultivars Mirage Rose, Mirage Pink, and Picobella Blue. However, silencing effectiveness was observed to be limited by genotypes and inoculation methods; apical meristem application was the appropriate method, whereas the highest silencing effectiveness among the cultivars was found in Picobella Blue. In addition, it was found that higher silencing effectiveness was associated with higher degradation of the endogenous PhPDS mRNA. Moreover, in terms of plant age and temperatures, 3-week-old plants grown at 20 °C day/18 °C night showed high silencing effectiveness for all cultivars. Taken together, infection of apical meristem of 3-week-old plants grown at 20 °C day/18 °C night with pTRV2-CaPDS exhibited the highest silencing effectiveness of all cultivars. Therefore, this study indicated the effectiveness of the CaPDS in the VIGS experiment with petunias, as well as the involvement of different factors in the mechanism influencing silencing effectiveness in petunias. These results will enable high-throughput functional analysis of genes associated with commercially important traits in petunias.
Key Message
CaPDS gene effectively induced gene silencing in the petunia cultivars, however, higher silencing effectiveness was observed to be limited by genotypes, inoculation methods, plant age, inoculation temperature, and presence of endogenous PhPDS mRNA.








Similar content being viewed by others
Abbreviations
- CaMV:
-
Cauliflower mosaic virus
- GOI:
-
Gene of interest
- PTGS:
-
Post transcriptional gene silencing
- PDS:
-
Phytoene desaturase
- siRNA:
-
Short interference RNA
- TRV:
-
Tobacco rattle virus
- VIGS:
-
Virus-induced gene silencing
References
Ai TN, Naing AH, Arun M, Lim SH, Kim CK (2016) Sucrose-induced anthocyanin accumulation in vegetative tissue of Petunia plants requires anthocyanin regulatory transcription factors. Plant Sci 252:144–150
Anand A, Vaghchhipawala Z, Ryu CM, Kang L, Wang K, del-Pozo O, Martin GB, Mysore KS (2007) Identification and characterization of plant genes involved in Agrobacterium-mediated plant transformation by virus-induced gene silencing. MPMI 187:41–52
Baek SA, Han T, Ahn SK, Kang H, Cho MR, Lee SC, Im KH (2012) Effects of heavy metals on plant growths and pigment contents in Arabidopsis thaliana. Plant Pathol J 28(4):446–452
Benedito VA, Visser PB, Angenent GC, Krens FA (2004) The potential of virus-induced gene silencing for speeding up functional characterization of plant genes. Genet Mol Res 3:323–341
Brigneti G, Martín-Hernández AM, Jin H (2004) Virus-induced gene silencing in Solanum species. Plant J 39:264–272
Broderick SR, Jones ML (2014) An optimized protocol to increase virus-induced gene silencing efficiency and minimize viral symptoms in Petunia. Plant Mol Biol Rep 32:219–233
Burch-Smith TM, Anderson JC, Martin GB, Dinesh-Kumar SP (2004) Applications and advantages of virus-induced gene silencing for gene function studies in plants. Plant J 39:734–746
Burch-Smith TM, Schiff M, Liu Y, Dinesh-Kumar SP (2006) Efficient virus-induced gene silencing in Arabidopsis. Plant Physiol 142:21–27
Caplan J, Dinesh-Kumar SP (2006) Using viral vectors to silence endogenous genes. Curr Protoc Microbiol 1(1):161.6.1–161.6.13
Chen J, Jiang C, Gookin T, Hunter D, Clark D, Reid M (2004) Chalcone synthase as a reporter in virus-induced gene silencing studies of flower senescence. Plant Mol Biol 55:521–530
Chung E, Seong E, Kim YC, Chung EJ, Oh SK, Lee S, Park JM, Joung YH, Choi D (2004) A method of high frequency virus-induced gene silencing in chili pepper (Capsicum annuum L. cv. Bukang). Mol Cells 17:377–380
Donaire L, Barajas D, Martínez-García B, Martínez-Priego L, Pagán I, Llave C (2008) Structural and genetic requirements for the biogenesis of tobacco rattle virus-derived small interfering RNAs. J Virol 82:5167–5177
Fu DQ, Zhu BZ, Zhu HL (2006) Enhancement of virus-induced gene silencing in tomato by low temperature and low humidity. Mol Cells 21:153–160
Jin Z, Yan T, Chang C, Liu Z, Wang Y, Tang Z, Yu F (2016) Application of virus-induced gene silencing approach in Camptotheca acuminata. Plant Cell Tissue Organ Cult 126:533–540
Krishnan A, Mahadevan C, Mani T, Sakuntala M (2015) Virus-induced gene silencing (VIGS) for elucidation of pathogen defense role of serine/threonine protein kinase in the non-model plant Piper colubrinum link. Plant Cell Tissue Organ Cult 122:269–283
Kumagai M, Donson J, Della-Cioppa G, Harvey D, Hanley K, Grill L (1995) Cytoplasmic inhibition of carotenoid biosynthesis with virus derived RNA. Proc Natl Acad Sci USA 92:1679–1683
Liu YL, Schiff M, Dinesh-Kumar SP (2002) Virus-induced gene silencing in tomato. Plant J 31:777–786
Lu R, Martin-Hernandez AM, Peart JR, Malcuit I, Baulcombe DC (2003) Virus-induced gene silencing in plants. Methods 30:296–303
Muruganantham M, Moskovitz Y, Haviv S (2009) Grapevine virus A-mediated gene silencing in Nicotiana benthamiana and Vitis vinifera. J Virol Methods 155:167–174
Qu F, Ye X, Hou G, Sato S, Clemente TE, Morris TJ (2005) RDR6 has a broad-spectrum but temperature-dependent antiviral defense role in Nicotiana benthamiana. J Virol 79:15209–15217
Rhee SY, Dickerson J, Xu D (2006) Bioinformatics and its applications in plant biology. Annu Rev Plant Biol 57:335–360
Robertson D (2004) VIGS vectors for gene silencing: many targets, many tools. Annu Rev Plant Biol 55:495–519
Ryu C, Anand A, Kang L, Mysore K (2004) Agrodrench: a novel and effective agroinoculation method for virus-induced gene silencing in roots and diverse solanaceous species. Plant J 40:322–331
Senthil-Kumar M, Mysore KS (2011) Virus-induced gene silencing can persist for more than 2 years and also be transmitted to progeny seedlings in Nicotiana benthamiana and tomato. Plant Biotechnol J 9:797–806
Senthil-Kumar M, Hema R, Anand A, Kang L, Udayakumar M, Mysore KS (2007) A systematic study to determine the extent of gene silencing in Nicotiana benthamiana and other Solanaceae species when heterologous gene sequences are used for virus-induced gene silencing. New Phytol 176:782–791
Spitzer B, Zvi MMB, Ovadis M (2007) Reverse genetics of floral scent: application of tobacco rattle virus-based gene silencing in petunia. Plant Physiol 145:1241–1250
Sung YC, Lin CP, Chen JC (2014) Optimization of virus-induced gene silencing in Catharanthus roseus. Plant Pathol 63:1159–1167
Wang C, Cai X, Wang X, Zheng Z (2006) Optimisation of tobacco rattle virus-induced gene silencing in Arabidopsis. Funct Plant Biol 33:347–355
Watson JM, Fusaro AF, Wang M, Waterhouse PM (2005) RNA silencing platforms in plants. FEBS Lett 579:5982–5987
Wellburn AR (1994) The spectral determination of chlorophylls a and b, as well as total carotenoids, using various solvents with spectrophotometers of different resolution. J Plant Physiol 144:307–313
Xu P, Zhang Y, Kang L, Roossinck MJ, Mysore KS (2006) Computational estimation and experimental verification of off-target silencing during post-transcriptional gene silencing in plants. Plant Physiol 142:429–440
Yan H, Fu D, Zhu B, Liu H, Shen X, Luo Y (2012) Sprout vacuum infiltration: a simple and efficient agroinoculation method for virus induced gene silencing in diverse solanaceous species. Plant Cell Rep 31:1713–1722
Zhang J, Tian J, Tai DQ, Li KT, Zhu YJ, Yao YC (2016) An optimized TRV-based virus-induced gene silencing protocol for Malus crabapple. Plant Cell Tissue Organ Cult 126:499–509
Funding
This work was supported by a grant from the Next-Generation BioGreen 21 Program (Project no. PJ01368505), Rural Development Administration, Republic of Korea.
Author information
Authors and Affiliations
Contributions
AHN and CKK designed the experiment. JML generated the construct. HYS performed all experimental steps such as planting of the petunia plants, preparation of inoculum, Agro-infiltration, analysis of qRT-PCR, and detection of total chlorophyll contents. KBL assisted in data analysis and molecular analysis AHN wrote and revised the manuscript. CKK supervised the project. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by Sergio Rosales-Mendoza.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Naing, A.H., Song, H.Y., Lee, J.M. et al. Development of an efficient virus-induced gene silencing method in petunia using the pepper phytoene desaturase (PDS) gene. Plant Cell Tiss Organ Cult 138, 507–515 (2019). https://doi.org/10.1007/s11240-019-01646-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-019-01646-y