Abstract
Introduction
Most myelomeningocele (MMC) cases present with ventriculomegaly or hydrocephalus, yet a comprehensive volumetric assessment of MMC intracranial structures is lacking. This study aimed to provide baseline data on intracranial structural volumes immediately after birth in MMC infants who underwent repair surgeries after birth (postnatal repair).
Methods
In this retrospective single-center study, we analyzed 52 MMC infants undergoing postnatal repair, utilizing head computed tomography scans at birth for volumetric assessment. Intracranial volume (ICV), lateral ventricles volume (LVV), choroid plexus volume (CPV), and posterior cranial fossa volume (PCFV) were measured. Hydrocephalus was classified into no hydrocephalus, progressive hydrocephalus, and hydrocephalus at birth. Comparative analysis employed the Wilcoxon rank-sum test. Receiver operating characteristic (ROC) analysis discriminated cases with and without ventriculoperitoneal shunt (VPS).
Results
The median values were 407.50 mL for ICV, 33.18 mL for LVV, 0.67 mL for CPV, and 21.35 mL for PCFV. Thirty-seven cases (71.15%) underwent VPS. ROC analysis revealed an LVV cut-off value of 6.74 mL for discriminating cases with and without VPS. Progressive hydrocephalus showed no significant difference in ICV but significantly larger LVV compared to no hydrocephalus. Hydrocephalus at birth demonstrated statistically larger ICV and LVV compared to the other two types.
Conclusion
Baseline volumetric data were provided, and volumetric analysis exhibited statistical differences among three hydrocephalus types. These findings enhance our understanding of intracranial volumetric changes in MMC, facilitating more objective assessments of MMC cases.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The most severe form of open spina bifida (dysraphism), myelomeningocele (MMC) [1], accounts for 98.8% of open spina bifida [2]. Hydrocephalus poses a significant concern [3], with comorbidity rates in MMC rising from 10–15% at birth to 65–85% within a few weeks [4,5,6]. A recent study has classified hydrocephalus treatment timing into simultaneous MMC repair with hydrocephalus treatment and delayed treatment [7]. These findings underscore the presence of progressive hydrocephalus in some MMC cases, which may remain undiagnosed at birth. As early as the 1970s, MMC cases were categorized into three hydrocephalus types: no hydrocephalus, progressive hydrocephalus, and hydrocephalus at birth [4]. However, quantitative volumetric assessment for these types is lacking, and concrete volumetric criteria for lateral ventricle volume to indicate shunt treatment in MMC infants are yet to be established.
Volumetric data on intracranial structures in MMC cases are lacking, and existing studies use ratios without establishing precise volume data [8,9,10]. This study aims to establish intracranial volumetric data calculated from head computed tomography (CT) scans of MMC cases who underwent repair procedures after birth (postnatal repair).
Methods
Patients and study setting
In this retrospective study, we included patients with MMC treated at our department from April 2006 to July 2023. All patients received head and spinal CT imaging after birth and underwent postnatal repair at our department. We specifically analyzed the initial head CT conducted immediately after birth. Before or after the repair surgeries, all cases underwent head and lumbar magnetic resonance imaging (MRI) to assess syringomyelia and Chiari malformation type II.
Data collection
We analyzed various medical variables, including sex, gestational week, birth weight, birthplace, fetal diagnosis (FD), cesarean section (CS), days of CT imaging, and information related to MMC and ventriculoperitoneal shunt (VPS). MMC lesions were categorized into four types based on the involved vertebrae: thoracic type (involving at least one thoracic vertebra), upper lumbar type (focused among 1st and 3rd lumbar vertebrae), lower lumbar type (mainly among 3rd and 5th lumbar vertebrae), and sacral type (focally presented in sacral vertebra). A lumbosacral type, where the lesion ranges from the lower lumbar to sacral vertebrae (e.g., from the fifth lumbar to the third sacral vertebrae), was categorized into the lower lumbar type. The vertebrae count affected by MMC lesions was measured using spinal CT and MRI imaging. The MMC lesions were defined as neural placode or impairment of epithelialization.
Hydrocephalus comorbidity in MMC was categorized into three types: no hydrocephalus (no VPS required), progressive hydrocephalus (developing ventricles enlargement after birth, requiring VPS), and hydrocephalus at birth (obvious hydrocephalus at birth, treated by VPS). The decision on the necessity of VPS was determined by tense fontanelle or increasing head circumstance [11].
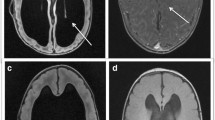
Quantitative assessment of intracranial volume (ICV), lateral ventricles volume (LVV), choroid plexus volume (CPV), and posterior cranial fossa volume (PCFV) in MMC patients used MATLAB R2023a (MathWorks, Natick, MA, USA) with digital imaging and communication in medicine (DICOM) CT data obtained immediately after birth. The target areas were manually segmented using the image segmenter app in MATLAB (https://www.mathworks.com/help/images/ref/imagesegmenter-app.html). Representative segmentation of ICV, LVV, CPV, and PCFV are shown in Fig. 1. Volume calculations (in milliliter, mL) were performed, and this method aligns with our previous studies [12,13,14,15]. LVV represented the volume of bilateral ventricles, and choroid plexus at the lateral ventricles and the foramen of Monro were assessed.
Representative segmentations. Segmentations for intracranial volume (ICV), lateral ventricles volume (LVV), choroid plexus volume (CPV), and posterior cranial fossa volume (PCFV) are colored as green, purple, yellow, and pink, respectively. Representative segmentations are presented from cases of no hydrocephalus (a), progressive hydrocephalus (b), and hydrocephalus at birth (c)
Statistical analyses
Categorical data were presented as frequencies (percentages), while continuous variables were expressed as mean ± SD for normally distributed variables and as median with 1st-3rd quartiles for non-normally distributed variables. Spearman correlation coefficients were used to assess parameter correlations, and the chi-squared test compared two categorical variables. The unpaired T-test was employed for parametric distributions. The Kruskal-Wallis and Wilcoxon rank-sum tests were utilized for non-parametric distributions. Bonferroni correction was applied for multiple comparisons. P-values < 0.05 and corrected p-values < 0.05 were considered statistically significant. The receiver operating characteristic (ROC) curve determined the area under the curve (AUC) and cut-off values using the maximal Youden index (sensitivity + specificity – 1). Statistical analyses were performed using MATLAB R2023a’s Statistical and Machine Learning Toolbox.
Results
Baseline characteristics
Fifty-two MMC cases (27 females, 51.92%) were followed up for 8.11 (3.95 – 11.66) years as of September 30, 2023 (Supplemental Fig. 1). Baseline characteristics are presented in Table 1. Among MMC-lower lumbar type, 90% were lumbosacral type. The delivery method was statistically relevant to MMC lesion types; 91.67% of thoracic types were delivered by CS, compared to 40.00% of sacral types (chi-squared test, p < 0.001). Progressive hydrocephalus and hydrocephalus at birth occurred in 14 (26.92%) and 23 (44.23%) patients, respectively, leading to VPS surgeries in 71.15% of cases.
Correlation analysis between continuous variables
Correlation analysis revealed positive correlations between gestational week and both weight at birth (r = 0.50, p < 0.001) and ICV (r = 0.32, p = 0.02). Weight at birth also positively correlated with ICV (r = 0.57, p < 0.001). In volumetric measurements, LVV demonstrated positive correlations with ICV (r = 0.67, p < 0.001) and CPV (r = 0.58, p < 0.001), and a negative correlation with PCFV (r = -0.69, p < 0.001) (Fig. 2a). MMC lesion vertebrae count exhibited positive correlations with ICV (r = 0.51, p < 0.001), LVV (r = 0.66, p < 0.001), and CPV (r = 0.29, p = 0.03), and a negative correlation with PCFV (r = -0.37, p = 0.006) (Fig. 2b). Significant differences in vertebrae count among MMC lesion types were noted (Kruskal-Wallis test, p < 0.001), with thoracic type having the highest counts compared to other types (Fig. 2c).
Continuous variable relationships. Scatter plots show correlations between lateral ventricles volume (LVV) and intracranial volume (ICV), posterior cranial fossa volume (PCFV), and choroid plexus volume (CPV) (a), and between vertebrae count and ICV, LVV, and PCFV (b). The correlation coefficients are denoted as “r”. Red lines indicate positive correlations, while blue lines indicate negative correlations. Myelomeningocele lesion vertebrae count distributions among lesion types are shown in a box-and-whisker plot overlaid with a bee-swarm plot (c). T, thoracic type; U-L, upper lumbar type; L-L, lower lumbar type; S, sacral type
Volumetric comparison
The median values were 407.50 mL for ICV, 33.18 mL for LVV, 0.67 mL for CPV, and 21.35 mL for PCFV (Table 2). MMC cases with VPS showed significantly larger volumes in ICV, LVV, and CPV, and a significantly lower volume in PCFV compared to those without VPS (using the Wilcoxon rank-sum test) (Table 2). Additionally, the association between VPS requirement and FD was evaluated. FD was statistically associated with VPS requirement; 83.78% of MMC cases with FD required VPS, compared to 40.00% of MMC cases without FD (chi-squared test, p = 0.002). MMC cases with FD showed significantly larger volumes in ICV, LVV, and CPV, and a significantly lower volume in PCFV compared to those without FD (using the Wilcoxon rank-sum test) (Table 2).
Hydrocephalus type impact
MMC cases with VPS included those with progressive hydrocephalus and hydrocephalus at birth, while those not requiring VPS corresponded to the no hydrocephalus type. Statistical relevance was observed in hydrocephalus types and MMC lesion types (Supplemental Fig. 2a, chi-squared test, p = 0.002) and Chiari malformation comorbidity (Supplemental Fig. 2b, chi-squared test, p < 0.001). In hydrocephalus at birth type, thoracic lesion type and Chiari malformation comorbidity were predominant.
While hydrocephalus at birth type showed significantly larger ICV than the others, there were no statistical differences between no hydrocephalus and progressive hydrocephalus (Fig. 3a). In LVV, even progressive hydrocephalus showed a significantly larger volume than no hydrocephalus (Fig. 3b). In CPV, both progressive hydrocephalus and hydrocephalus at birth demonstrated significantly larger volumes than no hydrocephalus; however, there was no statistical difference between these two types (Fig. 3c). Conversely, PCFV in no hydrocephalus exhibited significantly larger values than those in other two types (Fig. 3d). According to MMC lesion vertebrae count, hydrocephalus at birth demonstrated significantly larger values than no hydrocephalus (Fig. 3e). For exact values, refer to Supplemental Table 1.
Differences among three hydrocephalus types. The box-and-whisker plot overlaid with beeswarm plots is presented for intracranial volume (ICV) (a), lateral ventricles volume (LVV) (b), choroid plexus volume (CPV) (c), posterior cranial fossa volume (PCFV) (d), and Myelomeningocele (MMC) lesion vertebrae count (e). The Wilcoxon rank-sum test is used, and acquired p-values are corrected by Bonferroni correction, multiplied by three, for the solution of multiple comparisons. Statistically significant corrected-p values < 0.05 are denoted with an asterisk. Hydrocephalus types are presented as 0 for no hydrocephalus, 1 for progressive hydrocephalus, and 2 for hydrocephalus at birth
ROC analysis
To distinguish cases with VPS from those without VPS, ROC analysis revealed the highest AUC value in LVV (AUC = 0.99), with a cut-off value of 6.74 mL (sensitivity 1.00, specificity 0.93) (Fig. 4a). Similarly, for discriminating cases with progressive hydrocephalus and hydrocephalus at birth, LVV also exhibited the highest AUC value (AUC = 0.90), with a cut-off value of 35.97 mL (sensitivity 0.91, specificity 0.71) (Fig. 4b).
Receiver operating characteristic (ROC) curves. a ROC curves are calculated for discriminating cases requiring ventriculoperitoneal shunt (VPS) from those not requiring VPS using intracranial volume (ICV), lateral ventricles volume (LVV), choroid plexus volume (CPV), and posterior cranial fossa volume (PCFV). b ROC curves are calculated for discriminating cases with progressive hydrocephalus from those with hydrocephalus at birth using ICV, LVV, CPV, and PCFV
DICOM slice width
The width of DICOM slices varied from 1.00 mm to 6.98 mm, with a median value of 5.00 (5.00 – 5.33) mm. Slice width correlated positively with both ICV (r = 0.38, p = 0.006) and LVV (r = 0.40, p = 0.003) (Supplemental Fig. 3a and b). However, there was statistical significance between hydrocephalus types and slice width (chi-squared test, p = 0.02) (Supplemental Fig. 3c). Among patients with hydrocephalus at birth, who showed a statistically larger volume of ICV and LVV than the other two types, slice widths of 1 or 3 mm were not utilized at all. Thus, there was unevenness in the utilized slice width among hydrocephalus types. We mentioned this issue related to width-variation in the Discussion.
Discussion
Established baselines for intracranial structural volumes in MMC infants are lacking. In this study, we provide volumetric measurement values of ICV, LVV, CPV, and PCFV calculated from postnatally repaired MMC infants. Additionally, we demonstrated statistical differences in volumetric values among three hydrocephalus types: no hydrocephalus, progressive hydrocephalus, and hydrocephalus at birth. Moreover, the LVV cut-off value distinguishing between MMC cases with and without VPS was 6.74 mL.
The reported VPS rate for MMC with postnatal closure ranges from 60–80% [7, 11, 16,17,18,19,20,21,22,23], consistent with our results at 71.15%. We observed that 28.85% had no hydrocephalus, 26.92% had progressive hydrocephalus, and 44.23% had hydrocephalus at birth, with these three hydrocephalus types having been identified as early as the 1970s [4]. Progressive hydrocephalus following postnatal closure is believed to result from inadequate CSF resorption after repair surgery [17]. While the quantitative differences among these three types have remained unclear, we successfully demonstrated them using volumetric measurements. It is predictable that hydrocephalus at birth showed significantly larger ICV and LVV than the other two, and no hydrocephalus demonstrated significantly larger PCFV than the other two. However, it is interesting that progressive hydrocephalus showed no statistical differences in ICV compared to no hydrocephalus but significantly larger LVV than no hydrocephalus. This means that even in CT images taken immediately after birth, the characteristics of progressive hydrocephalus, where ICV is the same but LVV is higher than in no hydrocephalus, are apparent.
In a previous study on postnatal MMC closure, symptoms linked to hydrocephalus, such as apneic and bradycardic episodes, did not show statistical relevance to the need for CSF diversion. However, fontanelle characteristics, head circumference at birth, and the rate of head growth were significantly associated with CSF diversion [11]. Despite the absence of established volume criteria for VPS indication, our ROC analyses revealed a cut-off value of 6.74 mL for LVV in the necessity of VPS. This value, derived from our single-center experience, currently lacks scientific validation as a VPS criterion. Nevertheless, we anticipate that it could serve as a valuable reference for physicians managing MMC with hydrocephalus.
Our results on the correlations between MMC lesion vertebrae count and measured volumes reveal a clear trend: the more severe the MMC lesion (indicated by a higher vertebrae count), the smaller PCFV and the larger LVV. The thoracic type had the largest vertebrae count, signifying greater severity than the other three types. These findings align with previous studies, where larger spinal defects correlated with increased ventricular size [24]. The level of the lesion significantly affected the incidence of shunting, with more cephalad lesions correlating with higher rates: 97% in the thoracic, 87% in the lumbar, and 68% in the sacral spine [25]. In our study, all MMC cases with the thoracic type required treatment with VPS, while the rate of VPS in the sacral type was 51.85% (14/27 cases).
Small posterior fossa was observed in 84% of infants with MMC [19]. In a fetus with MMC, CSF leakage into the sac or amniotic fluid is thought to lead to hindbrain herniation, known as Chiari II malformation [26]. Hindbrain herniation is believed to play a key role in inducing hydrocephalus through increased resistance to cerebral venous outflow or abnormalities in CSF absorption or flow [5, 22, 27, 28]. Therefore, it is reasonable to infer that MMC induces small PCFV, leading to the enlargement of LVV and subsequent hydrocephalus. Thus, the negative correlation between PCFV and LVV in our study is acceptable and aligns with previous findings on the negative correlation between posterior cranial fossa hypoplasia and ventriculomegaly [10]. Our results also demonstrated a positive correlation between ICV and LVV, and we infer that the enlargement of LVV caused the enlargement of ICV.
Additionally, a positive correlation between LVV and CPV was observed, consistent with prior studies on this relationship [14]. Considering that the choroid plexus is a soft tissue, we infer that small ventricles compress the choroid plexus, while large ventricles induce a larger volume of the choroid plexus by releasing compressive pressure. Therefore, there is a possibility of underestimation or overestimation of CPV influenced by the size of the ventricles. In various neurological diseases such as Alzheimer’s disease [29] and stroke [30], some papers have reported the importance of the choroid plexus in neuropathology. However, it remains unknown how MMC pathology affects the growth of choroid plexus volume. Further research is needed to elucidate whether the larger size of CPV observed in larger LVV is a neuropathological result or merely an overestimation.
Our correlation analysis revealed positive correlations between ICV and slice width and between LVV and slice width, supporting concerns about overestimation. However, a statistical unevenness emerged between hydrocephalus types and the slice width; all slice widths for hydrocephalus at birth were over 5 mm, with none at 1 mm to 3 mm. This means that slice widths of 1 mm to 3 mm were only used in cases of no hydrocephalus or progressive hydrocephalus. Considering that ICV and LVV of hydrocephalus at birth were significantly larger than those of no hydrocephalus or progressive hydrocephalus, it is reasonable to conclude that the statistical distributional unevenness among hydrocephalus types and slice widths affected the positive correlation among them. Additionally, as there were no correlations between slice width and PCFV, or CPV, we judged that the influence of the non-consistency of slice width was minimal. However, it is crucial to note that this non-consistency of the utilized CT scans’ slice width is a significant limitation of this study.
Limitations
Firstly, conducting a single-center and retrospective posed challenges in ensuring consistency in slice width. Secondly, we lacked concrete criteria for VPS operation, and VPS procedures were performed based on empirical judgment by neurosurgeons in our department. Further studies are needed to establish a reliable and clinically feasible LVV criterion for VPS. Finally, there may be overestimation or underestimation when calculating CPV.
Conclusions
We presented intracranial volumetric data obtained immediately after birth from infants with MMC who underwent postnatal repair and highlighted volumetric differences among three hydrocephalus types. Our data could serve as a valuable reference for a more objective assessment of hydrocephalus in MMC cases.
Data availability
No datasets were generated or analysed during the current study.
Abbreviations
- AUC:
-
Area under the curve
- CPV:
-
Choroid plexus volume
- CS:
-
Cesarean section
- CSF:
-
Cerebrospinal fluid
- CT:
-
Computed tomography
- DICOM:
-
Digital imaging and communication in medicine
- FD:
-
Fetal diagnosis
- ICV:
-
Intracranial volume
- LVV:
-
Lateral ventricles volume
- mL:
-
Milliliter
- MMC:
-
Myelomeningocele
- MRI:
-
Magnetic resonance imaging
- PCFV:
-
Posterior cranial fossa volume
- ROC:
-
Receiver operating characteristic
- SD:
-
Standard deviation
- VPS:
-
Ventriculoperitoneal shunt
References
Copp AJ, Adzick NS, Chitty LS, Fletcher JM, Holmbeck GN, Shaw GM (2015) Spina bifida. Nat Rev Dis Primers 1:15007. https://doi.org/10.1038/nrdp.2015.7
Tortori-Donati P, Rossi A, Cama A (2000) Spinal dysraphism: a review of neuroradiological features with embryological correlations and proposal for a new classification. Neuroradiology 42:471–491. https://doi.org/10.1007/s002340000325
Blount JP, Hopson BD, Johnston JM, Rocque BG, Rozzelle CJ, Oakes JW (2023) What has changed in pediatric neurosurgical care in spina bifida? A 30-year UAB/Children’s of Alabama observational overview. Childs Nerv Syst 39:1791–1804. https://doi.org/10.1007/s00381-023-05938-9
Stein SC, Schut L (1979) Hydrocephalus in myelomeningocele. Pediatr Neurosurg 5:413–419. https://doi.org/10.1159/000119836
Tamburrini G, Frassanito P, Iakovaki K, Pignotti F, Rendeli C, Murolo D, Di Rocco C (2013) Myelomeningocele: the management of the associated hydrocephalus. Childs Nerv Syst 29:1569–1579. https://doi.org/10.1007/s00381-013-2179-4
Ntimbani J, Kelly A, Lekgwara P (2020) Myelomeningocele - A literature review. Interdiscip Neurosurg 19:100502. https://doi.org/10.1016/j.inat.2019.100502
McCarthy DJ, Sheinberg DL, Luther E, McCrea HJ (2019) Myelomeningocele-associated hydrocephalus: nationwide analysis and systematic review. Neurosurg Focus 47:E5. https://doi.org/10.3171/2019.7.FOCUS19469
Van Roost D, Solymosi L, Funke K (1995) A characteristic ventricular shape in myelomeningocele-associated hydrocephalus? A CT stereology study. Neuroradiology 37:412–417. https://doi.org/10.1007/BF00588025
Krogness K, Nyland H (1978) Posterior fossa measurements II. Size of the posterior fossa in myelomeningocele. Pediatr Radiol 6:198–202. https://doi.org/10.1007/BF00975536
Calandrelli R, Pilato F, Massimi L, Panfili M, Di Rocco C, Colosimo C (2020) Posterior cranial fossa maldevelopment in infants with repaired open myelomeningoceles: double trouble or a dynamic process of posterior cranial fossa abnormalities? World Neurosurg 141:e989–e997. https://doi.org/10.1016/j.wneu.2020.06.106
Phillips BC, Gelsomino M, Pownall AL, Ocal E, Spencer HJ, O’Brien MS, Albert GW (2014) Predictors of the need for cerebrospinal fluid diversion in patients with myelomeningocele: Clinical article. J Neurosurg Pediatr 14:167–172. https://doi.org/10.3171/2014.4.PEDS13470
Hashimoto H, Maruo T, Kimoto Y, Nakamura M, Fujinaga T, Nakamura H, Ushio Y (2023) The association between diffusion-weighted imaging-Alberta Stroke Program Early Computed Tomography Score and the outcome following mechanical thrombectomy of anterior circulation occlusion. Interdiscip Neurosurg 33:101758. https://doi.org/10.1016/j.inat.2023.101758
Hashimoto H, Maruo T, Kimoto Y, Nakamura M, Fujinaga T, Ushio Y (2023) Burr hole locations are associated with recurrence in single burr hole drainage surgery for chronic subdural hematoma. World Neurosurg: X 19:100204. https://doi.org/10.1016/j.wnsx.2023.100204
Hashimoto H, Takemoto O, Nishimoto K, Moriguchi G, Nakamura M, Chiba Y (2023) Normal growth curve of choroid plexus in children: implications for assessing hydrocephalus due to choroid plexus hyperplasia. J Neurosurg Pediatr 32:627–637. https://doi.org/10.3171/2023.7.PEDS23218
Hashimoto H, Takemoto O, Chiba Y (2023) Growth patterns and ratios of posterior cranial fossa structures in the Japanese pediatric population: a study utilizing CT scans. Neuroradiology 65:1835–1844. https://doi.org/10.1007/s00234-023-03229-3
Adzick NS, Thom EA, Spong CY, Sutton LN et al (2011) A randomized trial of prenatal versus postnatal repair of myelomeningocele. N Engl J Med 364:993–1004. https://doi.org/10.1056/NEJMoa1014379
Clemmensen D, Rasmussen MM, Mosdal C (2010) A retrospective study of infections after primary VP shunt placement in the newborn with myelomeningocele without prophylactic antibiotics. Childs Nerv Syst 26:1517–1521. https://doi.org/10.1007/s00381-010-1113-2
Tulipan N, Wellons JC, Thom EA, Rand L et al (2015) Prenatal surgery for myelomeningocele and the need for cerebrospinal fluid shunt placement. J Neurosurg Pediatr 16:613–620. https://doi.org/10.3171/2015.7.PEDS15336
Alexiou GA, Zarifi MK, Georgoulis G, Prodromou N et al (2011) Cerebral abnormalities in infants with myelomeningocele. Neurol Neurochir Pol 45:18–23. https://doi.org/10.1016/s0028-3843(14)60055-4
Talamonti G, D’Aliberti G, Collice M (2007) Myelomeningocele: long-term neurosurgical treatment and follow-up in 202 patients. J Neurosurg Pediatr 107:368–386. https://doi.org/10.3171/PED-07/11/368
Alatas I, Canaz G, Kayran NA, Kara N, Canaz H (2018) Shunt revision rates in myelomeningocele patients in the first year of life: a retrospective study of 52 patients. Childs Nerv Syst 34:919–923. https://doi.org/10.1007/s00381-017-3663-z
Gualberto IJN, Medeiros GA, Santos MV, da Silva LL, Machado HR, Sbragia L (2022) Is there a role in the central nervous system development for using corticosteroids to treat meningomyelocele and hydrocephalus? Childs Nerv Syst 38:1849–1854. https://doi.org/10.1007/s00381-022-05615-3
Flanders TM, Heuer GG, Madsen PJ, Adzick NS et al (2020) Detailed analysis of hydrocephalus and hindbrain herniation after prenatal and postnatal myelomeningocele closure: report from a single institution. Neurosurgery 86:637–645. https://doi.org/10.1093/neuros/nyz302
Nagaraj UD, Bierbrauer KS, Stevenson CB, Peiro JL, Lim F-Y, Zhang B, Kline-Fath BM (2018) Myelomeningocele versus myelocele on fetal mr images: are there differences in brain findings? Am J Roentgenol 211:1376–1380. https://doi.org/10.2214/ajr.18.20088
Rintoul NE, Sutton LN, Hubbard AM, Cohen B, Melchionni J, Pasquariello PS, Adzick NS (2002) A new look at myelomeningoceles: functional level, vertebral level, shunting, and the implications for fetal intervention. Pediatrics 109:409–413. https://doi.org/10.1542/peds.109.3.409
McLone DG, Knepper PA (1989) The cause of Chiari II malformation: a unified theory. Pediatr Neurosurg 15:1–12
George E, MacPherson C, Pruthi S, Glenn OA et al (2023) Long-Term Imaging Follow-up from the Management of Myelomeningocele Study. Am J Neuroradiol 44:861–866. https://doi.org/10.3174/ajnr.A7926
Williams H (2008) A unifying hypothesis for hydrocephalus, Chiari malformation, syringomyelia, anencephaly and spina bifida. Cerebrospinal Fluid Res 5:7. https://doi.org/10.1186/1743-8454-5-7
Choi JD, Moon Y, Kim H-J, Yim Y, Lee S, Moon W-J (2022) Choroid plexus volume and permeability at brain mri within the alzheimer disease clinical spectrum. Radiology 304:635–645. https://doi.org/10.1148/radiol.212400
Egorova N, Gottlieb E, Khlif MS, Spratt NJ, Brodtmann A (2019) Choroid plexus volume after stroke. Int J Stroke 14:923–930. https://doi.org/10.1177/1747493019851277
Acknowledgements
Since not all authors were native English speakers, we utilized ChatGPT 3.5 to review the manuscript grammatically.
Funding
Open Access funding provided by Osaka University. The Japan Society for the Promotion of Science (JSPS) KAKENHI [JP21K16629 (Hiroaki Hashimoto)] supported this work.
Author information
Authors and Affiliations
Contributions
H.H. conceived the study, collected and analyzed the data, developed the MATLAB program, created all figures and tables, and was primarily responsible for writing the manuscript. All authors provided clinical care and evaluated the patients. O.T. and Y.C. supervised the study. All authors have reviewed the manuscript.
Corresponding author
Ethics declarations
Ethical approval and consent to participate
Ethical approval was obtained from the Ethics Committee of Osaka Women’s and Children’s Hospital (Izumi, Japan, approval no. 1659), adhering to the Declaration of Helsinki. Informed consent was obtained through the opt-out method on our center’s website, considering the retrospective and noninvasive nature of the study.
Consent for publication
All authors reviewed the complete manuscript and consented for publication.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hashimoto, H., Irizato, N., Takemoto, O. et al. Intracranial volumetric evaluation in postnatally repaired myelomeningocele infants. Childs Nerv Syst (2024). https://doi.org/10.1007/s00381-024-06444-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00381-024-06444-2