Abstract
Purpose
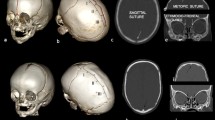
The changes in the proportion of posterior cranial fossa structures during pediatric development remain unclear. This retrospective study aimed to investigate the growth patterns and ratios of these structures using CT scans.
Methods
Head CT scans of pediatric patients with minor head trauma from Osaka Women’s and Children’s Hospital between March 2006 and May 2023 were analyzed. The study segmented the intracranial volume (ICV), posterior cranial fossa volume (PCFV), cerebellum volume (CBMV), and brainstem volume (BSV). Correlation coefficients were calculated among the parameters. Patients aged 0 to 10 years were divided into 15 age-related clusters, and mean and standard deviation values were measured. Growth curves were created by plotting mean values sequentially. Ratios such as PCFV/ICV and (CBMV + BSV)/PCFV were examined. Statistical analyses, including unpaired t tests and logarithmic curve fitting, were performed.
Results
A total of 234 CT scans (97 from females, 115 from infants under 1 year of age) were analyzed. Positive correlations were observed among the parameters, with the strongest between PCFV and CBMV. The growth curves for ICV, PCFV, CBMV, and BSV exhibited a two-phase process, with rapid growth until approximately 4 years of age, followed by stabilization. The ratios PCFV/ICV and (CBMV + BSV)/PCFV showed increasing trends from birth onwards, stabilizing by 4 and 1 years of age, respectively.
Conclusion
This study provides insights into the growth patterns and ratios of posterior cranial fossa structures in the pediatric population. The findings demonstrate a two-phase growth process and increasing trends in the examined ratios.






Similar content being viewed by others
Abbreviations
- BSV:
-
Brainstem volume
- CBMV:
-
Cerebellum volume
- CGCS:
-
Modified child’s Glasgow coma scales
- CI:
-
Confidence interval
- CT:
-
Computed tomography
- DICOM:
-
Digital Imaging and Communications in Medicine
- ICV:
-
Intracranial volume
- m:
-
Month
- MRI:
-
Magnetic resonance imaging
- PCFV:
-
Posterior cranial fossa volume
- SD:
-
Standard deviation
- y:
-
Year
References
Khalsa SSS, Siu A, DeFreitas TA, Cappuzzo JM, Myseros JS, Magge SN, Oluigbo CO, Keating RF (2017) Comparison of posterior fossa volumes and clinical outcomes after decompression of Chiari malformation Type I. J Neurosurg Pediatr 19:511–517. https://doi.org/10.3171/2016.11.PEDS16263
Sweeney KJ, Caird J, Sattar MT, Allcutt D, Crimmins D (2013) Spinal level of myelomeningocele lesion as a contributing factor in posterior fossa volume, intracranial cerebellar volume, and cerebellar ectopia. J Neurosurg Pediatr 11:154–159. https://doi.org/10.3171/2012.10.PEDS12177
Prassopoulos P, Cavouras D, Golfinopoulos S (1996) Developmental changes in the posterior cranial fossa of children studied by CT. Neuroradiology 38:80–83. https://doi.org/10.1007/BF00593229
Coll G, Lemaire J-J, Di Rocco F, Barthélémy I, Garcier J-M, De Schlichting E, Sakka L (2016) Human foramen magnum area and posterior cranial fossa volume growth in relation to cranial base synchondrosis closure in the course of child development. Neurosurgery 79:722–735. https://doi.org/10.1227/neu.0000000000001309
Kamdar MR, Gomez RA, Ascherman JA (2009) Intracranial volumes in a large series of healthy children. Plast Reconstr Surg 124:2072–2075. https://doi.org/10.1097/PRS.0b013e3181bcefc4
Hauptmann M, Byrnes G, Cardis E, Bernier M-O, Blettner M, Dabin J, Engels H, Istad TS, Johansen C, Kaijser M, Kjaerheim K, Journy N, Meulepas JM, Moissonnier M, Ronckers C, Thierry-Chef I, Le Cornet L, Jahnen A, Pokora R et al (2023) Brain cancer after radiation exposure from CT examinations of children and young adults: results from the EPI-CT cohort study. Lancet Oncol 24:45–53. https://doi.org/10.1016/S1470-2045(22)00655-6
Muhm M, Danko T, Henzler T, Luiz T, Winkler H, Ruffing T (2015) Pediatric trauma care with computed tomography—criteria for CT scanning. Emerg Radiol 22:613–621. https://doi.org/10.1007/s10140-015-1332-7
Astrand R, Rosenlund C, Undén J, for the Scandinavian Neurotrauma C (2016) Scandinavian guidelines for initial management of minor and moderate head trauma in children. BMC Med 14:33. https://doi.org/10.1186/s12916-016-0574-x
Kirkham FJ, Newton CR, Whitehouse W (2008) Paediatric coma scales. Dev Med Child Neurol 50:267–274. https://doi.org/10.1111/j.1469-8749.2008.02042.x
Hashimoto H, Maruo T, Kimoto Y, Nakamura M, Fujinaga T, Nakamura H, Ushio Y (2023) The association between diffusion-weighted imaging-Alberta Stroke Program Early Computed Tomography Score and the outcome following mechanical thrombectomy of anterior circulation occlusion. Interdiscip Neurosurg 33:101758. https://doi.org/10.1016/j.inat.2023.101758
Hashimoto H, Maruo T, Kimoto Y, Nakamura M, Fujinaga T, Ushio Y (2023) Burr hole locations are associated with recurrence in single burr hole drainage surgery for chronic subdural hematoma. World Neurosurg: X 19:100204. https://doi.org/10.1016/j.wnsx.2023.100204
Gao K, Kemp DE, Ganocy SJ, Gajwani P, Xia G, Calabrese JR (2008) Antipsychotic-induced extrapyramidal side effects in bipolar disorder and schizophrenia: a systematic review. J Clin Psychopharmacol 28:203–209. https://doi.org/10.1097/JCP.0b013e318166c4d5
Hashimoto H, Takemoto O, Nishimoto K, Moriguchi G, Nakamura M, Chiba Y (2023) Normal growth curve of choroid plexus in children: implications for assessing hydrocephalus due to choroid plexus hyperplasia. J Neurosurg Pediatr:1–11. https://doi.org/10.3171/2023.7.PEDS23218
Bianchi F, Benato A, Frassanito P, Tamburrini G, Massimi L (2021) Functional and morphological changes in hypoplasic posterior fossa. Childs Nerv Syst 37:3093–3104. https://doi.org/10.1007/s00381-021-05193-w
Rijken BFM, Lequin MH, van der Lijn F, van Veelen-Vincent M-LC, de Rooi J, Hoogendam YY, Niessen WJ, Mathijssen IMJ (2015) The role of the posterior fossa in developing Chiari I malformation in children with craniosynostosis syndromes. J Craniomaxillofac Surg 43:813–819. https://doi.org/10.1016/j.jcms.2015.04.001
Rijken BF, Lequin MH, Van Veelen M-LC, de Rooi J, Mathijssen IM (2015) The formation of the foramen magnum and its role in developing ventriculomegaly and Chiari I malformation in children with craniosynostosis syndromes. J Craniomaxillofac Surg 43:1042–1048. https://doi.org/10.1016/j.jcms.2015.04.025
Zhang CH, D'Arco F, Borghi A, Picariello S, Cheung M, Irving M, Thompson D (2023) Unravelling the pathogenesis of foramen magnum stenosis in patients with severe achondroplasia: a CT-based comparison with age-matched controls and FGFR3 craniosynostosis syndromes. Childs Nerv Syst. https://doi.org/10.1007/s00381-023-06005-z
Botelho RV, Heringer LC, Botelho PB, Lopes RA, Waisberg J (2020) Posterior fossa dimensions of Chiari malformation patients compared with normal subjects: systematic review and meta-analysis. World Neurosurg 138:521-529. e522. https://doi.org/10.1016/j.wneu.2020.02.182
Badie B, Mendoza D, Batzdorf U (1995) Posterior fossa volume and response to suboccipital decompression in patients with Chiari I malformation. Neurosurgery 37:214–218. https://doi.org/10.1227/00006123-199508000-00004
Trigylidas T, Baronia B, Vassilyadi M, Ventureyra E (2008) Posterior fossa dimension and volume estimates in pediatric patients with Chiari I malformations. Childs Nerv Syst 24:329–336. https://doi.org/10.1007/s00381-007-0432-4
Tubbs RS, Hill M, Loukas M, Shoja MM, Oakes WJ (2008) Volumetric analysis of the posterior cranial fossa in a family with four generations of the Chiari malformation Type I. J Neurosurg Pediatr 1:21–24. https://doi.org/10.3171/PED-08/01/021
Tubbs RS, Wellons JC III, Smyth MD, Bartolucci AA, Blount JP, Oakes WJ, Grabb PA (2003) Children with growth hormone deficiency and Chiari I malformation: a morphometric analysis of the posterior cranial fossa. Pediatr Neurosurg 38:324–328. https://doi.org/10.1159/000070416
Sgouros S, Kountouri M, Natarajan K (2006) Posterior fossa volume in children with Chiari malformation Type I. J Neurosurg Pediatr 105:101–106. https://doi.org/10.3171/ped.2006.105.2.101
McLone DG, Dias MS (2003) The Chiari II malformation: cause and impact. Childs Nerv Syst 19:540–550. https://doi.org/10.1007/s00381-003-0792-3
Messing-Jünger M, Röhrig A (2013) Primary and secondary management of the Chiari II malformation in children with myelomeningocele. Childs Nerv Syst 29:1553–1562. https://doi.org/10.1007/s00381-013-2134-4
Talamonti G, Marcati E, Mastino L, Meccariello G, Picano M, D’Aliberti G (2020) Surgical management of Chiari malformation type II. Childs Nerv Syst 36:1621–1634. https://doi.org/10.1007/s00381-020-04675-7
Kim I, Hopson B, Aban I, Rizk EB, Dias MS, Bowman R, Ackerman LL, Partington MD, Castillo H, Castillo J (2018) Decompression for Chiari malformation type II in individuals with myelomeningocele in the National Spina Bifida Patient Registry. J Neurosurg Pediatr 22:652–658. https://doi.org/10.3171/2018.5.PEDS18160
Vurdem ÜE, Acer N, Ertekin T, Savranlar A (2012) Inci MF (2012) Analysis of the volumes of the posterior cranial fossa, cerebellum, and herniated tonsils using the stereological methods in patients with Chiari type I malformation. Sci World J. https://doi.org/10.1100/2012/616934
Farin A, Deutsch R, Biegon A, Marshall LF (2003) Sex-related differences in patients with severe head injury: greater susceptibility to brain swelling in female patients 50 years of age and younger. J Neurosurg 98:32–36. https://doi.org/10.3171/jns.2003.98.1.0032
Coll G, Arnaud E, Collet C, Brunelle F, Sainte-Rose C, Di Rocco F (2015) Skull base morphology in fibroblast growth factor receptor type 2-related faciocraniosynostosis: a descriptive analysis. Neurosurgery 76:571–583. https://doi.org/10.1227/NEU.0000000000000676
Calandrelli R, Panfili M, D’Apolito G, Zampino G, Pedicelli A, Pilato F, Colosimo C (2017) Quantitative approach to the posterior cranial fossa and craniocervical junction in asymptomatic children with achondroplasia. Neuroradiology 59:1031–1041. https://doi.org/10.1007/s00234-017-1887-y
Tomita Y, Kameda M, Senoo T, Tokuyama E, Sugahara C, Yabuno S, Okazaki Y, Kawauchi S, Hosomoto K, Sasaki T (2022) Growth curves for intracranial volume and two-dimensional parameters for Japanese children without cranial abnormality: toward treatment of craniosynostosis. Neurol Med Chir 62:89. https://doi.org/10.2176/nmc.oa.2021-0208
De Bonis P, Anile C (2020) Post-traumatic hydrocephalus: the Cinderella of neurotrauma. Expert Rev Neurother 20:643–646. https://doi.org/10.1080/14737175.2020.1779059
Khasawneh AH, Garling RJ, Harris CA (2018) Cerebrospinal fluid circulation: what do we know and how do we know it? Brain Circ 4:14. https://doi.org/10.4103/bc.bc_3_18
Hashimoto H, Khoo HM, Yanagisawa T, Tani N, Oshino S, Hirata M, Kishima H (2022) Frequency band coupling with high-frequency activities in tonic-clonic seizures shifts from θ to δ band. Clin Neurophysiol 137:122–131. https://doi.org/10.1016/j.clinph.2022.02.015
Funding
The Japan Society for the Promotion of Science (JSPS) KAKENHI [JP21K16629 (Hiroaki Hashimoto)] supported this work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors report no relevant financial or non-financial interests to disclose.
Ethical approval
The Ethics Committee of Osaka Women’s and Children’s Hospital (Izumi, Japan, approval no. 1568) provided ethical approval for this study, which was conducted in accordance with the Declaration of Helsinki guidelines for experiments involving humans.
Informed consent
Informed consent was obtained using the opt-out method from our center’s website because of the retrospective and noninvasive nature of the study.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Previous Presentations: This paper has not been published and is not under consideration for publication elsewhere. This paper has already been submitted to the preprint server medRxiv: https://doi.org/10.1101/2023.07.07.23292362.
Supplementary information
ESM 1
(PDF 248 kb)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hashimoto, H., Takemoto, O. & Chiba, Y. Growth patterns and ratios of posterior cranial fossa structures in the Japanese pediatric population: a study utilizing CT scans. Neuroradiology 65, 1835–1844 (2023). https://doi.org/10.1007/s00234-023-03229-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00234-023-03229-3