Abstract
Purpose
To document long-term morphological changes of Chiari type II malformation (CM-II) following closure of spina bifida manifesta (SBM).
Methods
We retrospectively evaluated postnatal magnetic resonance images of the CM-II and posterior fossa (PF) in 28 consecutive cases. We measured changes in vertebral level and length of the cerebellar peg (CP), cerebrospinal fluid (CSF) spaces anterior and posterior to the cerebrospinal junction, PF area, and the anteroposterior diameters of the foramen magnum (FM) and C1 vertebra. We examined the morphological differences between the cases with and without ventriculoperitoneal (VP) shunting and derived predicted means by nonlinear mixed-effect modeling.
Results
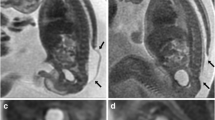
At birth, there were significant differences in CP length, PF area, and FM and C1 diameters between those who underwent VP shunting and those who did not. In cases with a CP below C1, VP shunting was required in every case but one. In those with visible CSF space at birth, VP shunts were not required. In 17 of 18 cases with a CP below C1, the vertebral level ascended by mean two vertebral levels (range 0–5 levels) within 4–6 months of delivery. In the remaining case, slowly progressive hydrocephalus and delayed CP descent required VP shunting at 8 months. Predicted mean CP length and FM and C1 diameters were greater in those who underwent VP shunting, but there was no difference in predicted mean PF area.
Conclusion
The morphology of CM-II and the presence of hydrocephalus influence each other in children who have undergone postnatal SBM repair.






Similar content being viewed by others
References
Adzick NS, Thom EA, Spong CY, Brock JW 3rd, Burrows PK, Johnson MP, Howell LJ, Farrell JA, Dabrowiak ME, Sutton LN, Gupta N, Tulipan NB, D’Alton ME, Farmer DL, Investigators MOMS (2011) A randomized trial of prenatal versus postnatal repair of myelomeningocele. N Engl J Med 364:993–1004. doi:10.1056/NEJMoa1014379
Bell WO, Charney EB, Bruce DA, Sutton LN, Schut L (1987) Symptomatic Arnold-Chiari malformation: review of experience with 22 cases. J Neurosurg 66:812–816
Boltshauser E, Schneider J, Kollias S, Waibel P, Weissert M (2002) Vanishing cerebellum in myelomeningocoele. Eur J Paediatr Neurol 6:109–113
Bruner JP, Tulipan N, Paschall RL, Boehm FH, Walsh WF, Silva SR, Hernanz-Schulman M, Lowe LH, Reed GW (1999) Fetal surgery for myelomeningocele and the incidence of shunt-dependent hydrocephalus. JAMA 282:1819–1825
Caldarelli M, DiRocco C, LaMarca F (1996) Shunt complications in the first postoperative year in children with meningomyelocele. Childs Nerv Syst 12:748–754
Casselman JW, Kuhweide R, Deimling M, Ampe W, Dehaene I, Meeus L (1993) Constructive interference in steady state 3DFT MR imaging of the inner ear and cerebellopontine angle. AJNR Am J Neuroradiol 14:47–57
Charney EB, Rorke LB, Sutton LN, Schut L (1987) Management of Chiari II complications in infants with myelomeningocele. J Pediatr 111:364–371
Danzer E, Johnson MP, Bebbington M, Simon EM, Wilson RD, Bilaniuk LT, Sutton LN, Adzick NS (2007) Fetal head biometry assessed by fetal magnetic resonance imaging following in utero myelomeningocele repair. Fetal Diagn Ther 22:1–6
Danzer E, Johnson MP (2014) Fetal surgery for neural tube defects. Semin Fetal Neonatal Med 19:2–8. doi:10.1016/j.siny.2013.09.004
Fichter MA, Dornseifer U, Henke J, Schneider KT, Kovacs L, Biemer E, Bruner J, Adzick NS, Harrison MR, Papadopulos NA (2008) Fetal spina bifida repair—current trends and prospects of intrauterine neurosurgery. Fetal Diagn Ther 23:271–286. doi:10.1159/000,123,614
Fütterer JJ, Scheenen TW, Huisman HJ, Klomp DW, van Dorsten FA, Hulsbergen-van de Kaa CA, Witjes JA, Heerschap A, Barentsz JO (2004) Initial experience of 3 T endorectal coil magnetic resonance imaging and 1H-spectroscopic imaging of the prostate. Invest Radiol 39:671–680
Gaskill SJ (2004) Primary closure of open myelomeningocele. Neurosurg Focus 16:E3
Grant RA, Heuer GG, Carrión GM, Adzick NS, Schwartz ES, Stein SC, Storm PB, Sutton LN (2011) Morphometric analysis of posterior fossa after in utero myelomeningocele repair. J Neurosurg Pediatr 7:362–368. doi:10.3171/2011.1.PEDS10234
Hashiguchi K, Morioka T, Fukui K, Miyagi Y, Mihara F, Yoshiura T, Nagata S, Sasaki T (2005) Usefulness of constructive interference in steady-state magnetic resonance imaging in the presurgical examination for lumbosacral lipoma. J Neurosurg 103(6 Suppl):537–543
Hashiguchi K, Morioka T, Yoshida F, Miyagi Y, Mihara F, Yoshiura T, Nagata S, Sasaki T (2007) Feasibility and limitation of constructive interference in steady-state (CISS) MR imaging in neonates with lumbosacral myeloschisis. Neuroradiology 49:579–585
Hashiguchi K, Morioka T, Murakami N, Suzuki SO, Hiwatashi A, Yoshiura T, Sasaki T (2010) Utility of 3-T FLAIR and 3D short tau inversion recovery MR imaging in the preoperative diagnosis of hippocampal sclerosis: direct comparison with 1.5-T FLAIR MR imaging. Epilepsia 51:1820–1828. doi:10.1111/j.1528-1167.2010.02685.x.
Hirose S, Farmer DL (2009) Fetal surgery for myelomeningocele. Clin Perinatol 36:431–438. doi:10.1016/j.clp.2009.03.008
Kronenburg A, Han KS, Gooskens R, Esposito G, Cochrane D, Woerdeman P (2013) Imaging the course of a hypoplastic cerebellum in a spina bifida newborn. Childs Nerv Syst 29:2131–2135. doi:10.1007/s00381-013-2156-y.
Lu H, Nagae-Poetscher LM, Golay X, Lin D, Pomper M, van Zijl PC (2005) Routine clinical brain MRI sequences for use at 3.0 Tesla. J Magn Reson Imaging 22:13–22
Luccichenti G, Giugni E, Péran P, Cherubini A, Barba C, Bivona U, Formisano R, Sabatini U (2010) 3 Tesla is twice as sensitive as 1.5 tesla magnetic resonance imaging in the assessment of diffuse axonal injury in traumatic brain injury patients. Funct Neurol 25:109–114
McLone DG (1983) Results of treatment of children born with a myelomeningocele. Clin Neurosurg 30:407–412
McLone DG, Knepper PA (1989) The cause of Chiari II malformation: a unified theory. Pediatr Neurosci 15:1–12
McLone DG (1992) Continuing concepts in the management of spina bifida. Pediatr Neurosurg 18:254–256
McLone DG, Dias MS (2003) The Chiari II malformation: cause and impact. Childs Nerv Syst 19:540–550
Morota N, Ihara S (2008) Postnatal ascent of the cerebellar tonsils in Chiari malformation type II following surgical repair of myelomeningocele. J Neurosurg Pediatr 2:188–193
Murakami N, Morioka T, Hashiguchi K, Yoshiura T, Hiwatashi A, Suzuki SO, Nakamizo A, Amano T, Hata N, Sasaki T (2013) Usefulness of three-dimensional T1-weighted spoiled gradient-recalled echo and three-dimensional heavily T2-weighted images in preoperative evaluation of spinal dysraphism. Childs Nerv Syst 29:1905–1914. doi:10.1007/s00381-013-2140-6
Nöbauer-Huhmann IM, Ba-Ssalamah A, Mlynarik V, Barth M, Schöggl A, Heimberger K, Matula C, Fog A, Kaider A, Trattnig S (2002) Magnetic resonance imaging contrast enhancement of brain tumors at 3 T versus 1.5 T. Invest Radiol 37:114–119
Park TS, Hoffman HJ, Hendrick EB, Humphreys RP (1983) Experience with surgical decompression of the Arnold–Chiari malformation in young infants with myelomeningocele. Neurosurgery 13:147–152
Pollack IF, Pang D, Albright AL, Krieger D (1992) Outcome following hindbrain decompression of symptomatic Chiari malformations in children previously treated with myelomeningocele closure and shunts. J Neurosurg 77:881–888
Pollack IF, Kinnunen D, Albright AL (1996) The effect of early craniocervical decompression on functional outcome in neonates and young infants with myelodysplasia and symptomatic Chiari II malformations: results from a prospective series. Neurosurgery 38:703–710
Rauzzino M, WJ O (1985) Chiari II malformation and syringomyelia. Neurosurg Clin N Am 1995 6:293–309
Rintoul NE, Sutton LN, Hubbard AM, et al. (2002) A new look at myelomeningoceles: functional level, vertebral level, shunting, and the implication for fetal intervention. Pediatrics 109:409–413
Stevenson KL (2004) Chiari Type II malformation: past, present, and future. Neurosug Focus 16(2):E5
Sutton LN, Adzick NS, Bilaniuk LT, Johnson MP, Crombleholme TM, Flake AW (1999) Improvement in hindbrain herniation demonstrated by serial fetal magnetic resonance imaging following fetal surgery for myelomeningocele. JAMA 282(19):1826–1831
Talamonti G, D’Alberti G, Collice M (2007) Myelomeningocele: long-term neurosurgical treatment and follow-up in 202 patients. J Neurosurg 107(5 Suppl):368–386
Thompson DN (2009) Postnatal management and outcome for neural tube defects including spina bifida and encephalocoeles. Prenat Diagn 29:412–419. doi:10.1002/pd.2199
Tubbs RS, Oakes WJ (2004) Treatment and management of the Chiari II malformation: an evidence-based review of the literature. Childs Nerv Syst 20:375–381
Tsai T, Bookstein FL, Levey E, Kinsman SL (2002) Chiari-II malformation: a biometric analysis. Eur J Pediatr Surg 12(Suppl 1):S12–S18
Tulipan N, Hernanz-Schulman M, Bruner JP (1998) Reduced hindbrain herniation after intrauterine myelomeningocele repair: a report of four cases. Pediatr Neurosurg 29:274–278
Tulipan N, Bruner JP, Hernanz-Schulman M, Lowe LH, WalshWF ND, Oakes WJ (1999) A) Effect of intrauterine myelomeningocele repair on central nervous system structure and function. Pediatr Neurosurg 31:183–188
Tulipan N, Hernanz-Schulman M, Lowe LH, Bruner JP (1999) Intrauterine myelomeningocele repair reverses preexisting hindbrain herniation. Pediatr Neurosurg 31:137–142
Tulipan N, Sutton LN, Bruner JP, Cohen BM, Johnson M, Adzick NS (2003) The effect of intrauterine myelomeningocele repair on the incidence of shunt-dependent hydrocephalus. Pediatr Neurosurg 38:27–33
Yousry I, Camelio S, Schmid UD, Horsfield MA, Wiesmann M, Bruckmann H, Yousry TA (2000) Visualization of cranial nerves I–XII: value of 3D CISS and T2-weighted FSE sequences. Eur Radiol 10:1061–1067
Vandertop WP, Asai A, Hoffman HJ, Drake JM, Humphreys RP, Rutka JT, Becker LE (1992) Surgical decompression for symptomatic Chiari II malformation in neonates with myelomeningocele. J Neurosurg 77:541–544
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Hashiguchi, K., Morioka, T., Murakami, N. et al. Sequential morphological change of Chiari malformation type II following surgical repair of myelomeningocele. Childs Nerv Syst 32, 1069–1078 (2016). https://doi.org/10.1007/s00381-016-3041-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00381-016-3041-2