Abstract
Seabirds often engage in coordinated, cooperative foraging to improve detection and capture of prey. An extreme example of such coordinated behavior is synchronicity, whereby the movements of individuals are aligned temporally and spatially. Synchronous diving among penguins has been reported, but simultaneous observations of predation by synchronously diving individuals have not. We instrumented chinstrap penguins (Pygoscelis antarcticus) during their incubation period in December 2019 from Cape Shirreff, Livingston Island, Antarctica (60.79°W, 62.46°S) with video and depth recorders to monitor predator foraging behavior and prey consumption rates. Serendipitously, two instrumented individuals, accompanied by a third, banded individual, engaged in synchronous foraging activities on Antarctic krill (Euphausia superba) for 9.25 h. This group formed ashore, transited together to their foraging area, remained in close visual contact after dives, synchronously timed diving behavior, and foraged at similar depths. Prey capture rates were positively correlated across dives and total consumption estimates were equivalent for the two instrumented birds during the video observation period. Video loggers confirmed that synchronous diving and foraging behavior are among the behavioral repertoire of chinstrap penguins and demonstrated equivalent prey capture rates by synchronously foraging predators. The results further suggest that group formation while ashore and group cohesion during a foraging trip may facilitate shared foraging success among group members.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Seabirds often engage in group hunting to improve detection and capture of dense, but patchy, prey resources (Götmark et al. 1986; Ballance and Pitman 1999; Sutton et al. 2015; McInnes et al. 2017). An extreme example of such coordinated hunting behavior is synchronicity, whereby the movements of individual predators in a group are aligned temporally and spatially. Among penguins, synchronicity of diving behavior has been reported based on visual observations from the surface (Ainley 1972; Siegfried et al. 1975; Wilson et al. 1986). More recently, data from animal-borne sensors demonstrated that synchrony extended beyond the timing of dives to include similar dive profiles among individuals in several species (Tremblay and Cherel 1999; Takahashi et al. 2004b; Pütz and Cherel 2005; Berlincourt and Arnould 2014; Wantanabe and Takahashi 2013).
Synchronous diving behavior of conspecifics may indicate complex cooperative foraging strategies that benefit all group members (Tremblay and Cherel 1999). The degree of coordination necessary for prey capture, however, can vary among predator species and be affected by the distribution and anti-predator behaviors exhibited by the target prey (Takahashi et al. 2004b; Thiebault et al. 2014, 2015; Handley et al. 2018). In particular, predator coordination may differ when targeting fish schools versus swarming crustaceans, or when targeting dense versus loose aggregations of prey. Such coordination, however, may not represent optimal foraging behavior for each participant in synchronous foraging given variation in physical or morphological constraints of individuals (Takahashi et al. 2004b). Simultaneous observations of predator behavior and prey consumption rates necessary to assess foraging performance of predators engaged in synchronous foraging behaviors within the same prey field have not been reported previously.
The advent of video-logging equipment suitable for use on diving seabirds has facilitated direct observations of foraging behavior, including cryptic social interactions and prey encounters that occur underwater (Ponganis et al. 2000; Takahashi et al. 2004a; Mattern et al. 2018). Furthermore, the identification of stereotypical behaviors readily observed in video data, like the rapid movement of the head during predation events (Kokubun et al. 2011; del Caño et al. 2021), helps to quantify observable penguin–prey interactions. Analyses of predation events recorded by animal-borne video loggers have revealed foraging strategies that vary by prey type and location (Handley et al. 2018; Mattern et al. 2018; Takahashi et al. 2008), unique predation events on large or rare prey (Pistorius et al. 2020), kleptoparasitism (Handley and Pistorius 2015), and identified the wide-spread, but under-appreciated, role of gelatinous animals in the diets of several penguin species (Thiebot et al. 2017). The stream of novel insights on predator–prey interactions derived from video loggers underscore their utility for studying foraging ecology and behavior.
Here, we report direct observations of diving and foraging behavior from synchronously foraging chinstrap penguins (Pygoscelis antarcticus) outfitted with video loggers and depth recorders. Specifically, we ask if predation rates of simultaneously foraging penguins are similar during their period of synchrony. We compare dive characteristics of the two instrumented companions during and after the period of synchrony, quantify predation rates from video data to compare individual foraging success, and explore the role of visual contact in maintaining group cohesion and coordination while foraging.
Materials and methods
Study site
We studied the diving and foraging behavior of breeding chinstrap penguins from Cape Shirreff, Livingston Island (60.79°W, 62.46°S) in the northern Antarctic Peninsula region (Supplementary Fig. S1). Chinstrap penguins at Cape Shirreff have been monitored by the United States Antarctic Marine Living Resources Program (U.S. AMLR) since 1996/97, during which time breeding populations have declined from 7744 breeding pairs in 1997/98 to 2170 breeding pairs in 2019/20. Data presented here were collected in late December 2019, near the end of the incubation period, when foraging trips typically last < 3 days (Lishman 1985).
Instrumentation
We used a video logger (Little Leonardo DVL400M065, 61 × 21x15 mm, 29 g) and a depth recorder (Lotek LAT1800FP, 36 × 13x11 mm, 9 g) to record foraging behaviors of adult chinstrap penguins. The video loggers initiate recording based on pre-set timers that were configured to delay recording for several hours after release and then to record continuously until battery exhaustion. The cameras have an expected recording life of ≈5 h at 30 frames per second with a pixel resolution of 1280 × 960. The underwater field of view of the camera was 31° horizontally and 24° vertically. Depth and wet/dry status of the tag were recorded every second during deployment. The depth loggers have an accuracy of ± 2 m and a resolution of ± 0.1 m.
Birds selected for tagging were chosen from nests distributed across the colony. Each bird was captured at their nest in the presence of their mate to ensure incubation of eggs was uninterrupted during the tagging procedure. The tags were attached to dorsal contour feathers over the spine with cyanoacrylate glue. Small plastic cable ties (2.5 × 150 mm) were threaded through underlying feathers and closed over the tags as an additional fastener. Video loggers were placed anterior to the depth logger and between the scapulae to improve forward-looking views for capturing predation events. Birds were recaptured for tag recovery when first encountered ashore at least 24 h after deployment. In total, we tagged three chinstrap penguins with video and depth loggers and report data from the two birds that were observed behaving synchronously. For reference, we present the dive data from the third chinstrap video logger deployment, along with the location and dive data from four other chinstrap penguins that were tracked during the incubation period of the same year with GPS and depth loggers in Supplementary Fig. S1.
We note that tagging effects on behavior and foraging performance are unavoidable when using external devices to monitor that activity of free-ranging penguins (Culik and Wilson 1991). Tag weight, shape, and placement are factors that can affect the performance of free-ranging penguins. As a rule of thumb, total instrument weight should not exceed 5% of bird weight (Kenward 2001) and our total instrument weight (39 g in air) represented ≈1% of bird weight in this study. With respect to shape, the video logger presents a blunt face that is not an ideal hydrodynamic shape (Culik et al. 1994). However, no instrument contained an external antenna, which can also increase drag while swimming (Wilson et al. 2004). To improve video capture, we placed loggers more-anteriorly than recommended (Bannasch et al. 1994), which induces additional drag. On balance, negative effects on foraging performance of the penguins due to instrumentation are expected. However, given short deployments for single trips, our analysis of similarly tagged individuals should provide valid comparisons of the synchronous foraging behaviors observed here. In particular, both penguins successfully fed during the trip and returned to their nests weighing an estimated 500 g heavier than when tagged (Table 1). Such weight gain is similar to the long-term (1997–2014) mean diet mass of chinstrap penguins (608 ± 46 g, U.S. AMLR, unpublished data) collected via stomach lavage (Wilson 1984) at the study site during the chick-rearing period.
Analysis of dive data
We estimated the start time of each foraging trip as the time when each bird initiated transit away from the beach, as observed in the video. The estimate of the time when the foraging trip ended was based on a visual analysis of the dive data, including the wet/dry status of each reading, to verify cessation of diving activity and return to land. We then used the R (R Core Team 2020) package ‘diveMove’ (Luque 2007) to identify individual dives during the foraging trip. First, depth readings were adjusted to account for potential sensor bias, drift, and variation in atmospheric pressure at the ocean surface to ensure that surface readings averaged 0 m depth. Visual analysis of the raw data indicated that a constant bias, rather than drift or temporal variation, was the major factor affecting surface depth readings for these relatively short deployments. Therefore, we used a fixed correction factor for each tag, estimated as the median recorded depth from each tag, respectively. We note that > 50% of recorded depths occurred at the surface in these deployments. We then specified a depth threshold of 5 m for dive identification, consistent with prior analyses of diving behaviors for chinstrap penguins (Miller and Trivelpiece 2008) and conforming with the manual video annotations (see below) that consistently identified underwater movements to depths > 5 m as dives. Dive and dive phases, including the descent, bottom, and ascent phases of the dive, were identified by examining the rate of change in depths in the dive data (Luque 2007). Once each dive was identified, we paired the dives that were initiated within 10 s of one another during the synchronous and asynchronous periods. Pairing dives across the entire foraging trip enabled a quantification of the degree of similarity in dive-level behavior during the synchronous and asynchronous periods. We used Pearson correlations and paired t tests to assess differences in the maximum depths achieved during the dive, the mean depths used during the bottom phase of the dive, the durations the each dive, the duration of time spent in the bottom phase of the dive, and the duration of the surface intervals between dives.
Analysis of video data
Manual annotations of video recordings were conducted by each of the authors, yielding five independent annotations for each video. For each annotation, time stamps in the depth records were aligned with the video time stamps. Once aligned, each annotator viewed all videos using the freely available VLC media player and annotated the corresponding depth records, to the nearest second, when video observations indicated that a dive began and ended and when a predation attempt occurred. We also noted the associated prey type. A description of annotator training and examples of the raw annotations are provided in Supplementary Fig. S2.
Predation attempts, hereafter strikes, on krill (Euphausia superba), fish, or unidentifiable prey were identified based on clear observation of prey in the beak or, more commonly, based on the rapid movement of the head into, and out of, the video frame in the presence of prey items. Prior studies using accelerometers suggest that such rapid head movement is a reliable indicator of prey strikes (Kokubun et al. 2011; Wantanabe and Takahashi 2013). Note that, given placement of the camera, the head was either stationary or not visible in the imagery while the penguin was swimming normally.
Krill generally represent the majority of prey eaten by chinstrap penguins at the study site (Miller et al. 2010), so we estimated the total mass of krill consumed during the video period based on the number of strikes observed and an estimate of the mean mass of an individual krill. We based our estimate of krill mass on the mean length of krill measured in a diet study conducted at the same site during the 2019–20 season. Stomach samples were obtained from 18 chick-provisioning penguins and krill lengths were measured to the nearest mm (unpublished data). We then estimated the wet weight of the average-length krill with a length–weight relationship previously reported for the Antarctic Peninsula region (Siegel et al. 2013).
Variability in strike rates (total strikes per dive) across birds were assessed with linear mixed-effects models implemented in R (R Core Team 2020) package ‘lme4’ (Bates et al. 2015). We considered a set of four nested models and selected, via AIC, the best-fitting model for reporting results. The base model included a fixed effect for bird identity as an explanatory factor to assess differences between birds, and random effects for the observers to account for differences in krill strike rate arising from observer bias. The three alternative models were constructed to assess variation in strike rates that might arise from changes in the accessibility of prey during the foraging trip. Temporal trends in accessibility could occur, inter alia, from depletion of prey by predation, changes in prey behavior or distribution that limits their vulnerability to predation (e.g., diel vertical migration), or satiation of the predator that slows consumption over time. Note we cannot differentiate these hypotheses with the data. Nonetheless, we can assess general trends in consumption rate and therefore included time of day, represented as minutes elapsed since the start of the first dive, in the model. Similarly, longer dive durations could reflect changes in the distribution of accessible prey that affect consumption rates at the dive level. We, therefore, included dive duration as a predictor of strike rates. The alternative models, therefore, included one or both of the fixed effects for time of day and dive duration. Time of day and dive duration were not highly correlated (r = 0.10), supporting their inclusion as potential predictors of strike rate.
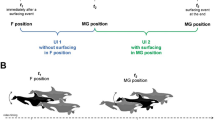
During manual video annotation, one annotator recorded the presence of other individually identifiable companions to the nearest second. Identifiable marks of other penguins encountered during this study included the back-mounted instrumentation from this study or a stainless steel flipper band used in long-term demographic monitoring (Hinke et al. 2007). These three companions were observed together throughout the video record (Supplementary Fig. S5). We assumed that variation in the proximity of companions, quantified in the frequency of sightings, reflects behavioral processes that affect group cohesion. For example, an imbalance in sightings of group members may suggest a social hierarchy, whereby a following individual may view a leading individual more frequently. We used a chi-square test to ask whether the individually identifiable companions were observed more frequently by one camera than the other. Second, if visual cues help maintain group cohesion and enhance synchrony, then we would expect sighting frequency to increase prior to dive bouts as birds prepare for coordinated diving. We tested this hypothesis with linear regression and modeled trends in the sighting frequency of companions during the six, relatively long surface intervals (LSI) when the birds were at rest before diving bouts (Fig. 1b). Average sighting frequency during the LSI was estimated by binning all sightings of companions into 1-min intervals and averaging the total sightings within each bin across all LSIs in which that bin was present.
Depth records of two chinstrap penguins from Cape Shirreff, Livingston Island that include synchronous periods. a Depths recorded from release until recovery. Grey polygons indicate the time of local night. b Depths recorded during the period of video observations. Black bars indicate inter-bout surface intervals. c Representative 30 min of synchronous diving behavior on 20 December 2019 with black bars as in panel (b). d Synchronous and asynchronous diving behavior before and after, respectively, 03:45 on 21 December 2019. The dotted red line indicates the start of the video-logging period. The dashed red line indicates the end of the video-logging period. The solid red line indicates the end of synchronous diving behavior
Results
We recorded synchronous diving and foraging behaviors with video and depth loggers for a female and a male chinstrap penguin during their incubation periods. The foraging trip made by the female lasted 20.7 h, while the male spent 43.5 h at sea (Table 1). Note that we use the male and female nomenclature to identify individual-level results, not generic sex-based differences. Depth and video data demonstrated that the two birds exhibited 9.25 h of synchronous movements, including departure from the colony, transit to foraging areas, and foraging during dives to depths up to 30 m (Fig. 1b, c). During the recorded foraging trip (18:45–23:00; Table 1), each bird exhibited the same pattern of repeated diving bouts interrupted by resting surface intervals that averaged 12 min in duration (Fig. 1b). The pattern of synchronous behaviors diverged at night (03:45), around the time of local solar nadir (03:59; Fig. 1a, d).
Dive analysis
Dive durations, maximum dive depths, mean depth of the bottom phase of the dive, and inter-dive surface intervals were highly correlated during the period of synchronous behavior, but not during the asynchronous period (Table 2). Mean values for these paired dive parameters were more similar across periods (Supplementary Table S1). The male tended to dive deeper than the female (Table 2), but the mean difference was within the accuracy limit of the pressure sensor on the tag.
Video analysis—predation
The first 4.5 h of the foraging trip for each bird were recorded with video loggers (Table 1). Annotators identified 85 synchronous dives in the video, excluding 3 short, shallow dives that were identified in the analysis of the depth records (Table 2). During most dives, both birds encountered and consumed prey, primarily krill (Table 3). Krill strikes were recorded throughout the observed depth range, including predation just below the surface, with a mean depth of predation occurring at roughly 15 m (Table 3). Strikes occurred regularly on lone krill encountered in the water column, within swarms, and on the periphery of swarms (Fig. 2). The female exhibited more prey strikes early in the foraging trip relative to the end of the video record (Fig. 3a), but krill strikes per dive were positively correlated (r = 0.56, p < 0.01) between the birds (Fig. 3b). The best-fitting mixed-effect model of krill strikes, based on AIC, included significant terms for time of day and dive duration. Strikes decreased at a rate of − 0.043 ± 0.004 (95% CI) per minute over the observation period, while strike rates increased by 0.14 ± 0.02 (95% CI) per second of dive duration. There was no significant effect of bird identity on strike rate (− 0.4 ± 0.53; 95% CI). Further, the random effect of observer identity (SD = 0.47) was small relative to residual error (SD = 3.9), suggesting no strong bias associated with inter-individual variation in annotations. On average, variation in total krill strikes among observers exhibited a coefficient of variation of 8.9% (Supplementary Fig. S2). Additionally, cumulative observed prey strikes were similar at the end of the video record, with each bird having consumed roughly 258 g of krill (Fig. 3c). On average, each bird successfully captured krill in ≥ 77% of the dives, with roughly 5.5 krill consumed per dive (Table 3).
Krill strikes observed during paired dives for each bird. a Mean and 95% CI for krill strikes per dive by each bird. b Relationship of krill strikes per dive for the male and female chinstrap penguins. A reference line and correlation coefficient are provided for reference. c Accumulation of mean ± 95% CI of estimated krill biomass consumed by the male and female chinstrap penguins during the video observation period
Video analysis—companions
Three identifiable individuals initiated the foraging trip together. Two birds carried video loggers and one bird was marked with a flipper band. At no time during the video were more than 5 birds observed swimming together (Supplementary Fig. S5). This group formed ashore and remained in close proximity for the duration of the video recording (Fig. 4a, Supplementary Fig. S5). On average, sightings of identifiable companions occurred regularly (2.6 sightings per minute), with 95% of sightings occurring within 2 m of the surface (Fig. 4b). Sightings were most common while birds were at rest on the surface (63%) and while transiting (25%). Sightings during foraging dives were less common (12%), and typically occurred close to the surface during ascent or descent phases of the dive. We did not observe companions during active foraging within krill swarms. There was no difference in the number of times a particular tagged or flipper-banded bird was observed in the videos (χ2 = 2.50, p = 0.11), indicating no bias for one camera to view other companions more frequently. During the six long surface intervals (Fig. 2b), the frequency of sightings increased as the time of dive bout initiation neared (R2 = 0.36, F1,14 = 7.77, p = 0.015; Fig. 4c).
Timing, depth distribution, and trend in sightings of identifiable individuals during the video-logging period. a Sightings over time, with the six surface intervals that precede diving bouts highlighted with gray polygons. b Depth distribution of sightings prior to departure (ashore), during shallow, directed swimming (transit), while resting at the surface (surface), and during underwater movements to depths > 5 m (dive). c Increasing trend in average sightings per minute during the six long surface intervals prior to diving bouts
Discussion
Synchronous foraging behaviors in penguins have been reported in several studies (Ainley 1972; Siegfried et al. 1975; Tremblay and Cherel 1999; Takahashi et al. 2004a, b; Pütz and Cherel 2005; Berlincourt and Arnould 2014) but were limited to analysis of diving behavior. The miniaturization of video-logging equipment suitable for deployment on small seabirds enabled us to contribute to this small, but growing, body of observations with a serendipitous recording of sustained, synchronous diving and foraging behaviors of chinstrap penguins. Synchronous diving behavior may indicate cooperative foraging (Tremblay and Cherel 1999), whereby multiple predators coordinate their movements to locate and disrupt prey swarms to improve capture efficiency (Wilson and Wilson 1990; McInnes et al. 2017). The video and diving data presented here demonstrate a clear synchronicity in diving behavior and foraging performance by the two birds. Each bird was observed to repeatedly strike at prey irrespective of location or apparent density of krill in the vicinity, yielding positively correlated numbers of krill strikes per dive and equivalent total consumption after 4.5 h of video monitoring (Fig. 3). On balance, the synchronicity observed among foraging chinstrap penguins likely enabled similar capture efficiencies within the krill swarms they encountered.
While the video did not capture clear examples of coordinated underwater behaviors within or near krill aggregations, it seems likely that the birds were aware of, and reacted to, the other bird while foraging. In particular, we note that depth use by both birds during the synchronous period was correlated (Table 2) and both birds consistently initiated ascent at the same time (Supplementary Fig. S3). Such awareness of companion activity, even if not observed in the video, could be explained by the primacy of prey pursuit and capture during active foraging, the visual field of birds, and the limited field of view in the forward-looking camera. For many birds, including penguins, the forward-looking, binocular visual field is narrow relative to the lateral, monocular visual field (Martin 2007). In particular, the functional binocular field for penguins is roughly 20° (Martin 2007) relative to the field of view of the camera (31°). Based on our observations of predation occurring only within the field of view of the camera, it seems reasonable to assume that prey capture mainly occurs in a forward-looking, binocular field of view. This suggest that coordination with companions while foraging may depend on their relatively larger, lateral field of view. A side-by-side configuration and peripheral awareness of coordinating birds would naturally limit observations of companions in the forward-looking imagery obtained with a video logger during active foraging.
An alternative explanation for the similar timing of dive duration and depth use may owe to reaching physiological limits of breath-hold diving. However, this appears to be unlikely for several reasons. First, allometric estimates of maximum dive duration suggest dive thresholds near 4 min for chinstrap penguins (Watanuki and Burger 1999). Recorded maximum dive durations for chinstraps exceed 3 min, while typical dives are reported to be roughly one minute (Bengston et al. 1993; Miller et al. 2010), similar to the mean dive duration reported here (Table 3). Second, the timing of ascent initiation was similar across the range of observed dive durations (43–94 s). Taken together, the dive durations observed in the video are within physiological limits that would constrain behavior across individuals and we infer that the shared synchronous diving and foraging behavior arise from behavioral coordination, rather than physiological limitation.
Synchronous foraging behaviors in penguins are typically associated with relatively small groups where visual and vocal cues are key factors that promote cohesion of the group (Ainley 1972; Choi et al. 2017; Thiebault et al. 2019). For example, synchronous swimming was largely absent among African penguins in groups larger than 17 birds (Wilson et al. 1986). The maximum number of birds (5) observed at one time in the video conforms to this small-group constraint. We do not have recordings of vocalizations to assess their effect on group formation and cohesion, but several lines of evidence suggest that visual cues were important for maintaining group cohesion in this group. First, sightings of each group member were frequent and consistent throughout the video. This suggests that all group members maintained visual contact, particularly at the surface. Second, the synchronous diving behaviors ended near the time of solar nadir. Reduced solar incidence and underwater light attenuation could limit the ability of chinstrap penguins to remain in visual contact while foraging at night. Alternatively, dissolution of the foraging group during night could arise from changes in prey distributions that make group foraging less effective. Krill swarms generally shoal at night, thus being more accessible to diving predators like chinstrap penguins. Less clear, however, is whether krill swarm density increases or decreases at night (e.g., Everson 1982; Zhou and Dorland 2004; Bestley et al. 2018) and how chinstrap penguin foraging behavior responds to such variation in krill swarm density. Such uncertainty provides opportunity for further research on factors that affect group foraging dynamics. Finally, the trend for increasing sightings of other identifiable individuals during the inter-bout surface intervals strongly suggests that visual contact is a key mechanism that maintains group cohesion.
The video data also shed light on the role of group formation and cohesion over the course of transit and foraging bouts that span several hours. The group of penguins we tracked coalesced before transit from the breeding colony began. The two birds with video loggers made several short excursions into the surf zone, but returned to land each time before acquiring the banded bird and finally initiating a transit out to sea. Group formation, or flocking, on beaches prior to departure is a common feature among Pygoscelis penguins and thought to be primarily an anti-predator tactic (Ainley 1972; Krause and Ruxton 2002). Indeed, leopard seal (Hydrurga leptonyx) predation on penguins at the study site is common (Krause et al. 2020). The risk of predation while at sea, due primarily to ecotype-B killer whales (Orcinus orca) (Pitman and Durban 2010), is also a factor that may favor group transit and group cohesion while foraging. However, the relatively high rates of successful foraging dives and the similar rates of prey capture within dives also suggests that pre-trip flocking may help form stable foraging groups that promote coordination of prey capture. For example, little penguins (Eudyptula minor) can form small groups with non-random membership prior to departure from breeding beaches (Daniel et al. 2007). Such selective group membership suggests that groups form for reasons other than simply increasing the size of the group to dilute predation risk. Among chinstrap penguins, our results indicate that group formation while ashore may confer benefits that include improved foraging success.
Conclusion
Video loggers confirmed that synchronous diving and foraging behavior are among the behavioral repertoire of chinstrap penguins. The birds transited together from shore to their foraging area, remained in visual contact after dives, and matched their timing of dive phases and depth use. Predation rates on krill were correlated across dives and total consumption estimates were equivalent for the two birds during their period of synchrony. While generalization from this small sample size is challenging, the results prompt questions for further research. For example, is there evidence for satiation in the flattening of consumption curves estimated from the observed prey strikes? If coordinated foraging benefits group members, how does dissociation of the group affect subsequent foraging performance? Is the similarity of foraging performance of group members sensitive to variation in prey size structure or availability? Future research using video loggers and other sensors (Watanabe and Takahashi 2013) to continue monitoring in situ predator foraging behavior may help answer such questions and expand our scope of knowledge of predator–prey interactions in marine ecosystems.
Data availability
The datasets generated and/or analysed during the current study are available from the NOAA National Centers for Environmental Information repository under accession number 0239297 and available at https://doi.org/10.25921/jzzg-wk59.
References
Ainley DG (1972) Flocking in Adélie penguins. Ibis 114:388–390. https://doi.org/10.1111/j.1474-919X.1972.tb00836.x
Ballance LT, Pitman RL (1999) Foraging ecology of tropical seabirds. In: Adams NJ, Slotow RH (eds) Foraging ecology of tropical seabirds. BirdLife South Africa, Johannesburg, pp 2057–2071
Bannasch R, Wilson RP, Culik B (1994) Hydrodynamic aspects of design and attachment of a back-mounted device in penguins. J Exp Biol 194:83–96. https://doi.org/10.1242/jeb.194.1.83
Bates D, Maechler M, Bolker B, Walker S (2015) Fitting linear mixed-effects models using lme4. J Stat Software 67:1–48. https://doi.org/10.18637/jss.v067.i01
Bengston JL, Cross DA, Goebel ME (1993) Diving behavior of chinstrap penguins at Seal Island. Ant Sci 5:9–15. https://doi.org/10.1017/S0954102093000033
Berlincourt M, Arnould JPY (2014) At-sea associations in foraging Little Penguins. PLoS ONE 9(8):e105065. https://doi.org/10.1371/journal.pone.0105065
Bestley S, Raymond B, Gales NJ, Harcourt RG, Hindell MA, Jonsen ID, Nicol S, Péron C, Sumner MD, Weimerskirch H, Wotherspoon SJ, Cox MJ (2018) Predicting krill swarm characteristics important for marine predators foraging off East Antarctica. Ecography 41:996–1012. https://doi.org/10.1111/ecog.03080
Choi N, Kim J-H, Kokobun N, Park S, Chung H, Lee W (2017) Group association and vocal behaviour during foraging trips in gentoo penguins. Sci Rep 7:7570. https://doi.org/10.1038/s41598-017-07900-7
Culik BM, Wilson RP (1991) Swimming energetics and performance of instrumented Adélie penguins (Pygoscelis adeliae). J Exp Biol 158:355–368. https://doi.org/10.1242/jeb.158.1.355
Culik BM, Bannasch R, Wilson RP (1994) External devices on penguins: how important is shape? Mar Biol 118:353–357. https://doi.org/10.1007/BF00350291
Daniel TA, Chiaradia A, Logan M, Quinn GP, Reina RD (2007) Synchronized group association in the little penguin (Eudyptula minor). Anim Behav 74:1241–1248. https://doi.org/10.1016/j.anbehav.2007.01.029
del Caño M, Quintana F, Yoda K, Dell’Omo G, Blanco GS, Gómez-Laich A (2021) Fine-scale body and head movements allow to determine prey capture events in the Magellanic Penguin (Spheniscus magellanicus). Mar Biol 168:84. https://doi.org/10.1007/s00227-021-03892-1
Everson I (1982) Diurnal variations in mean volume backscattering strength of an Antarctic krill (Euphausia superba) patch. J Plankton Res 4:155–162. https://doi.org/10.1093/plankt/4.1.155
Götmark F, Winkler DW, Andersson M (1986) Flock-feeding on fish schools increases individual success in gulls. Nature 319:589–591. https://doi.org/10.1038/319589a0
Handley JM, Pistorius P (2015) Kleptoparasitism in foraging gentoo penguins Pygoscelis papua. Polar Biol 39:391–395. https://doi.org/10.1007/s00300-015-1772-2
Handley JM, Thiebault A, Stanworth A, Schutt D, Pistorius P (2018) Behaviourally mediated predation avoidance in penguin prey: in situ evidence from animal-borne camera loggers. Roy Soc Open Sci 5:171449. https://doi.org/10.1098/rsos.171449
Hinke JT, Salwicka K, Trivelpiece SG, Watters GM, Trivelpiece WZ (2007) Divergent responses of Pygoscelis penguins reveal a common environmental driver. Oecologia 153:845–855. https://doi.org/10.1007/s00442-007-0781-4
Kenward RE (2001) A manual for wildlife radio tagging. Academic Press, London
Kokubun N, Kim J-H, Shin H-C, Naito Y, Takahashi A (2011) Penguin head movement detected using small accelerometers: a proxy of prey encounter rate. J Exp Biol 214:3760–3767. https://doi.org/10.1242/jeb.058263
Krause J, Ruxton GD (2002) Living in groups. Oxford University Press, New York
Krause DJ, Goebel ME, Kurle CM (2020) Leopard seal diets in a rapidly warming polar region vary by year, season, sex, and body size. BMC Ecol 20:32. https://doi.org/10.1186/s12898-020-00300-y
Lishman GS (1985) The comparative breeding biology of Adelie and chinstrap penguins Pygoscelis adeliae and P. antarctica at Signy Island, South Orkney Islands. Ibis 127:84–89. https://doi.org/10.1111/j.1474-919X.1985.tb05039.x
Luque SP (2007) Diving behaviour analysis in R. R News 7(3):8–14
Martin GR (2007) Visual fields and their functions in birds. J Ornithol 148:S547–S562. https://doi.org/10.1007/s10336-007-0213-6
Mattern T, McPherson MD, Ellenberg U, van Heezik Y, Seddon PJ (2018) High definition video loggers provide new insights into behaviour, physiology, and the oceanic habitat of a marine predator, the yellow-eyed penguin. PeerJ 6:e5459. https://doi.org/10.7717/peerj.5459
McInnes AM, McGeorge C, Ginsberg S, Pichegru L, Pistorius PA (2017) Group foraging increases foraging efficiency in a piscivorous diver, the African penguin. R Soc Open Sci 4:170918. https://doi.org/10.1098/rsos.170918
Miller AK, Trivelpiece WZ (2008) Chinstrap penguins alter foraging and diving behavior in response to the size of their principle prey, Antarctic krill. Mar Biol 154:201–208. https://doi.org/10.1007/s00227-008-0909-z
Miller AK, Kappes MA, Trivelpiece SG, Trivelpiece WZ (2010) Foraging-niche separation of breeding gentoo and chinstrap penguins, South Shetland Islands, Antarctica. Condor 112:683–695. https://doi.org/10.1525/cond.2010.090221
Pistorius PA, Green DB, Seddon PJ, Thiebault A (2020) In situ observation of a record-sized squid prey consumed by a gentoo penguin. Polar Biol 43:279–283. https://doi.org/10.1007/s00300-020-02630-5
Pitman RL, Durban JW (2010) Killer whale predation on penguins in Antarctica. Polar Biol 33:1589–1594. https://doi.org/10.1007/s00300-010-0853-5
Ponganis PJ, VanDam RP, Marshall G, Knower T, Levenson DH (2000) Sub-ice foraging behavior of emperor penguins. J Exp Biol 203:3275–3278. https://doi.org/10.1242/jeb.203.21.3275
Pütz C, Cherel Y (2005) The diving behaviour of brooding king penguins (Aptenodytespatagonicus) from the Falkland Islands: variation in dive profiles and synchronous underwater swimming provide new insights into their foraging strategies. Mar Biol 147:281–290. https://doi.org/10.1007/s00227-005-1577-x
R Core Team (2020) R: A language and environment for statistical computing. Vienna, Austria: R Foundation for Statistical Computing. https://cran.r-project.org/
Siegel V, Reiss CS, Dietrich KS, Haraldsson M, Rohardt G (2013) Distribution and abundance of Antarctic krill (Euphausia superba) along the Antarctic Peninsula. Deep Sea Res I 77:63–74. https://doi.org/10.1016/j.dsr.2013.02.005
Siegfried WR, Frost PGH, Kinahan JB, Cooper J (1975) Social behaviour of Jackass Penguins at sea. Zool Africana 10:87–100. https://doi.org/10.1080/00445096.1975.11447495
Sutton GJ, Hoskins AJ, Arnould JPY (2015) Benefits of group foraging depend on prey type in a small marine predator, the Little Penguin. PLoS ONE 10(12):e0144297. https://doi.org/10.1371/journal.pone.0144297
Takahashi A, Sato K, Naito Y, Dunn MJ, Trathan PN, Croxall JP (2004a) Penguin-mounted cameras glimpse underwater group behavior. Proc Roy Soc Lond B 271:S281–S282. https://doi.org/10.1098/rsbl.2004.0182
Takahashi A, Sato K, Nishikawa J, Watanuki Y, Naito Y (2004b) Synchronous diving behavior of Adélie penguins. J Ethol 22:5–11. https://doi.org/10.1007/s10164-003-0111-1
Takahashi A, Kokobun N, Mori Y, Shin H-C (2008) Krill-feeding behavior of gentoo penguins as shown by animal-borne camera loggers. Polar Biol 31:1291–1294. https://doi.org/10.1007/s00300-008-0502-4
Thiebault A, Mullers RHE, Pistorius PA, Tremblay Y (2014) Local enhancement in a seabird: reaction distance and foraging consequence of predator aggregations. Behav Ecol 25:1302–1310. https://doi.org/10.1093/beheco/aru132
Thiebault A, Semeria M, Lett C, Tremblay Y (2015) How to capture fish in a school? Effect of successive predator attacks on seabird feeding success. J Anim Ecol 85:157–167. https://doi.org/10.1111/1365-2656.12455
Thiebault A, Charrier I, Aubin T, Green DB, Pistorius PA (2019) First evidence of underwater vocalisations in hunting penguins. PeerJ 7:e8240. https://doi.org/10.7717/peerj.8240
Thiebot J-B, Arnould JPY, Gómez-Laich A, Ito K, Kato A, Mattern T, Mitamura H, Noda T, Poupart T, Quintana F, Raclot T, Ropert-Coudert Y, Sala JE, Seddon PJ, Sutton GJ, Yoda K, Takahashi A (2017) Jellyfish and other gelata as food for four penguin species—insights from predator-borne videos. Front Ecol Eviron 15:437–441. https://doi.org/10.1002/fee.1529
Tremblay Y, Cherel Y (1999) Synchronous underwater foraging behavior in penguins. Condor 101:179–185. https://doi.org/10.2307/1370462
Wantanabe YY, Takahashi A (2013) Linking animal-borne video to accelerometers reveals prey capture variability. Proc Natl Acad Sci USA 110:2199–2204. https://doi.org/10.1073/pnas.1216244110
Watanuki Y, Burger AE (1999) Body mass and dive duration in alcids and penguins. Can J Zool 77:1838–1842. https://doi.org/10.1139/z99-157
Wilson RP (1984) An improved stomach pump for penguins and other seabirds. J Field Ornithol 55:109–112
Wilson RP, Wilson MPT (1990) Foraging ecology of breeding Spheniscus penguins. In: Davies LS, Darby JT (eds) Penguin Biology. Academic Press, San Diego, pp 181–206
Wilson RP, Wilson MPT, McQuaid L (1986) Group size in foraging African penguins (Spheniscus dermesus). Ethology 72:338–341. https://doi.org/10.1111/j.1439-0310.1986.tb00634.x
Wilson RP, Kreye JM, Lucke K, Urquhart H (2004) Antennae on transmitters on penguins: balancing energy budgets on the high wire. J Exp Biol 207:2649–2662. https://doi.org/10.1242/jeb.01067
Zhou M, Dorland RD (2004) Aggregation and vertical migration behavior of Euphausia superba. Deep-Sea Res II Top Stud Oceanogr 51:221–2137. https://doi.org/10.1016/j.dsr2.2004.07
Acknowledgements
We thank G. Kooyman, D. Krause, G. Watters, N. Kokubun, and T. Mattern for insightful and constructive comments that improved this manuscript. Reference to any specific commercial products, process, or service by trade name, trademark, manufacturer, or otherwise, does not constitute or imply its recommendation, or favoring by the United States Government or NOAA/National Marine Fisheries Service.
Funding
No special funding contributed to this research.
Author information
Authors and Affiliations
Contributions
JTH conceived the study; SLW and LB collected data in the field; JTH and TMR conducted statistical analyses; JTH drafted the manuscript; all authors analyzed data and critically revised the manuscript. All authors gave final approval for publication and agree to be held accountable for the work performed therein.
Corresponding author
Ethics declarations
Conflicts of interest
We declare we have no competing interests.
Ethical approval
All animal handling procedures and research protocols are approved by the Southwest Fisheries Science Center/Pacific Islands Fisheries Science Institutional Animal Care and Use Committee (# SWPI 2020-01). All field research activities are permitted under the U.S. Antarctic Conservation Act (Permit #2017-012).
Additional information
Responsible Editor: V. Paiva.
Reviewers: N. Kokubun and T. Mattern.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Ocean Associates, Inc. Arlington, VA, USA. Under contract to: Antarctic Ecosystem Research Division, Southwest Fisheries Science Center, National Marine Fisheries Service, National Oceanic and Atmospheric Administration. La Jolla, USA.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hinke, J.T., Russell, T.M., Hermanson, V.R. et al. Serendipitous observations from animal-borne video loggers reveal synchronous diving and equivalent simultaneous prey capture rates in chinstrap penguins. Mar Biol 168, 135 (2021). https://doi.org/10.1007/s00227-021-03937-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00227-021-03937-5