Abstract
Rationale
The 5-methoxy-N-methyl-N-isopropyltryptamine (5-MeO-MiPT, known online as “Moxy”) is a new psychedelic tryptamine first identified on Italian national territory in 2014. Its hallucinogen effects are broadly well-known; however, only few information is available regarding its pharmaco-toxicological effects.
Objectives
Following the seizure of this new psychoactive substances by the Arm of Carabinieri and the occurrence of a human intoxication case, in the current study we had the aim to characterize the in vivo acute effects of systemic administration of 5-MeO-MiPT (0.01–30 mg/kg i.p.) on sensorimotor (visual, acoustic, and overall tactile) responses, thermoregulation, and stimulated motor activity (drag and accelerod test) in CD-1 male mice. We also evaluated variation on sensory gating (PPI, prepulse inhibition; 0.01–10 mg/kg i.p.) and on cardiorespiratory parameters (MouseOx and BP-2000; 30 mg/kg i.p.). Lastly, we investigated the in silico ADMET (absorption, distribution, metabolism, excretion, toxicity) profile of 5-MeO-MiPT compared to 5-methoxy-N,N-diisopropyltryptamine (5-MeO-DIPT) and N,N-dimethyltryptamine (DMT).
Results
This study demonstrates that 5-MeO-MiPT dose-dependently inhibits sensorimotor and PPI responses and, at high doses, induces impairment of the stimulated motor activity and cardiorespiratory changes in mice. In silico prediction shows that the 5-MeO-MiPT toxicokinetic profile shares similarities with 5-MeO-DIPT and DMT and highlights a cytochrome risk associated with this compound.
Conclusions
Consumption of 5-MeO-MiPT can affect the ability to perform activities and pose a risk to human health status, as the correspondence between the effects induced in mice and the symptoms occurred in the intoxication case suggests. However, our findings suggest that 5-MeO-MiPT should not be excluded from research in the psychiatric therapy field.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The synthesis and the spread of new psychoactive substances (NPS) are a growing phenomenon that constantly pose challenges for law enforcements, clinicians, and global public health. Indeed, in 2020 more than 350 laboratories were dismantled with almost 7 tons of NPS seized and 880 NPS monitored (EMCDDA 2021, 2022). Despite hallucinogens represent a class of drugs with a prevalence of use in Europe generally low compared to others, the European Monitoring Centre for Drugs and Drug Addiction (EMCDDA) reported that 163 new psychedelics were monitored in 2021 (57 tryptamines and 106 phenethylamines; EMCDDA 2022). Moreover, between 2022 and 2023, 82 countries reported a total of 167 classic hallucinogens, representing 14% of the NPS registered (UNODC 2023). Nowadays psychedelics are divided into three groups based on their chemical structure (Baumeister et al. 2015): lysergamides (including LSD and its analogues), tryptamines (including psilocin and its analogues), and phenethylamines (including mescaline and its analogues). These substances alter perception, thought, and state of consciousness acting on serotonin (5HT) receptor 2A (5HT2A; Halberstadt 2015), with a possible interaction with other serotonin receptor subtypes such as 1A (5HT1A) and 2C (Halberstadt and Geyer 2011; Zamberlan et al. 2018). Regarding tryptamines, the core structure contains an indole ring linked by an ethyl side chain to an amino group, which can be mono- or di-substituted by alkyl groups, and the indole ring can support substitution on position 4 or 5 (Fantegrossi et al. 2008). These compounds present a non-selective action on 5HT receptors with quite different potency and affinity profiles. It has been shown that the 4-hydroxylation or the 5-methoxylation is associated with increased potency in comparison to the analogues with different substitutions (Araújo et al. 2015). Some of them, like N,N-dimethyltryptamine (DMT; Fig. 1c) and derivates, bind adrenergic-α1 receptors and are substrate for SERT (serotonin transporter; Halberstadt 2015). They bind various targets in vitro, including dopaminergic, adrenergic, and histaminergic receptors, as well as monoaminergic transporters (Luethi and Liechti 2020). Tryptamines are generally consumed with the aim to alter perception but can induce psychological disturbance, including psychosis. Adverse effects of these compounds are restlessness, disorientation, confusion, hallucinate state, amnesia, mydriasis, tachypnoea, hypertension, and tachycardia. Cases of acute renal failure and rhabdomyolysis associated with synthetic tryptamine intoxications have been reported, as well as deaths (Luethi and Liechti 2020). From 2009 to 2013, eight tryptamines were intercepted on Italian territory (Serpelloni et al. 2013) and during 2013 the Italian National Early Warning System (SNAP) reported the seizure of tryptamines, such as 5-MeO-DALT, 4-AcO-DMT, and DET (Allerta Dronetplus 2016).
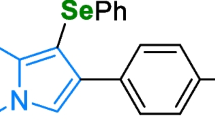
The 5-methoxy-N-methyl-N-isopropyltryptamine (5-MeO-MiPT; Fig. 1a), also known as “Moxy” in the illicit drug market, is an analogue of 5-methoxy-N,N-diisopropyltryptamine (5-MeO-DIPT, “Foxy”; Fig. 1b; Altuncı et al. 2021). Its synthesis was first reported by Repke et al. (1985) and its psychoactive effects were first documented by Shulgin and Shulgin (1997). This analogue has been involved in cases of intoxications in Japan (Itowaka et al. 2007; Shimizu et al. 2007) and in a case of homicide as a result of a sever psychotic state induced by the simultaneous intake of 5-MeO-MiPT and 5-MeO-DIPT (Matsumoto and Okada 2006). In 2017, a suicide associated to 3-MeO-PCP has been reported, in which post-mortem analysis revealed the presence of 5-MeO-MiPT (concentration of 0.13 µg/g in femoral blood; Johansson et al. 2017). The compound was first reported to the National Early Warning System (SNAP) in November 2013 and has been identified for the first time on Italian territory in 2014 (Dipartimento Politiche Antidroga 2015). In fact, on 18 May 2018 5-MeO-MiPT has been included in Table I of narcotic and psychotropic substances (DPR 309/90). The compound is available online in the form of powder or liquid and it can be orally consumed (4–6 mg) or inhaled (12–20 mg; Erowid 2015a, b, c; Tittarelli et al. 2015). According to users’ report, the psychoactive effect following the oral ingestion appears after 15–20 min with a peak approximately between 45 and 60 min and seems to decrease in 10 h. On the other hand, if inhaled the onset is immediate and the offset is about 2–5 h later. The effects reported by users vary from sleepy to speedy and from sense enhancing to mind-numbing (Erowid 2015a, b, c). In particular, it induces a full range of high-level hallucinatory states, visual distortions, colour and visual acuity enhancement, enhanced sense of touch, relaxation, and sleep disturbances (Tittarelli et al. 2015; Psychonautwiki 2023a, b). Adverse effects described are stomach discomfort, gas and vomiting, anxious stimulation, and muscle tension (Erowid 2015a, b, c). This is in line with symptoms associated with its analogue 5-MeO-DIPT and DMT, which induce stomach discomfort, nausea and vomiting, disorientation, tachycardia, hypertension, confusion, headache, and speech difficulties, basing on information reported by consumers (Erowid 2015a, b, c; Psychonautwiki 2022, 2023a, b) and case reports (Smolinske et al. 2005; Muller 2004; Heise and Brooks 2017; Bouso et al. 2022). In vitro studies by Rickli and colleagues showed that 5-MeO-MIPT is a full agonist of 5HT2A receptor (activation efficacy up to 80%) with a value of EC50 of 0.023 ± 0.04 µM, but it presents a higher affinity with 5HT1A receptor than with 5HT2A (values of Ki respectively of 0.058 ± 0.01 µM and 0.163 ± 0.03 µM; Rickli et al. 2016). Concerning interaction with other targets, it has been found that 5-MeO-MiPT inhibited serotonin and norepinephrine uptake in rats (Nagai et al. 2007), but in contrast with these results another study showed that the substance does not interact with rat monoamine transporters (Blough et al. 2014).
In 2021, the Arm of Carabinieri seized 5-MeO-MIPT in a form of powder and, in 2022, the Pavia Poison Control Centre (PCC)—National Toxicology Information Centre alerted our laboratory about a non-fatal human intoxication involving 5-MeO-MiPT. Both these events underlined the urgent need to expand the knowledge on the pharmaco-toxicology of this tryptamine analogue. Indeed, as a collaborative centre for the Italian National Early Warning System, committed to the monitoring and prevention of NPS abuse, we intended to characterize the toxicological effects of this above reported NPS through our preclinical investigations.
With this purpose, the current study aimed to evaluate the in vivo acute effects of 5-MeO-MIPT on neurological, sensorimotor (visual, tactile, and acoustic), motor activity, and thermoregulation, together with the possible alteration of the sensorimotor gating system through PPI (prepulse inhibition) in murine model. Also, we investigated the effects on cardiovascular and respiratory parameters in mice. Finally, we evaluated in silico ADMET (absorption, distribution, metabolism, excretion, toxicity) profile of 5-MeO-MiPT in comparison to 5-MeO-DIPT and DMT to provide a rapid screening of the toxic potential of these compounds.
Materials and methods
Animals
A total of 110 adult male ICR (CD-1®) mice, 3–4 months old, weighing 25–30 g (ENVIGO Harlan Italy, Italy; bred inside the Laboratory for Preclinical Research, LARP, of University of Ferrara, Italy), were group-housed (5 mice per cage; a floor area per animal of 80 cm2; a minimum enclosure height of 12 cm on a 12:12-h light–dark cycle (light on at 6:30 AM), the temperature of 20–22 °C, the humidity of 45–55%) and were provided ad libitum access to food (Diet 4RF25 GLP; Mucedola, Settimo Milanese, Milan, Italy) and water. Experiments were performed during the light phase of the light–dark cycle (lights on between 6:30 AM and 6:30 PM). The experimental protocols followed in the present study were in accordance with the UK Animals (Scientific Procedures) Act of 1986 and associated guidelines and the new European Communities Council Directive of September 2010 (2010/63/EU). The Italian Ministry of Health (licence n. 223/2021-PR and extension CBCC2.46.EXT.21) and the Animal Welfare Body of the University of Ferrara approved experimental protocols. According to the ARRIVE guidelines, all possible efforts were made to minimize the number of animals used, to minimize the animals’ pain and discomfort. For the present study, mice were divided into different groups, as follows. In safety pharmacology studies (visual, acoustic, and tactile sensorimotor responses; body core and surface temperature; accelerod and drag tests), eight mice (total of 48 mice) were employed for each treatment (vehicle or five doses: 0.01, 0.1, 1, 10, and 30 mg/kg). In the PPI test, eight mice (total of 40 mice) were used for each treatment (vehicle or four doses: 0.01, 0.1, 1, and 10 mg/kg). In cardiorespiratory studies, six mice (total of 12 mice) were utilized for each treatment (vehicle or 30 mg/kg). In blood pressure studies, five mice (total of 10 mice) were employed for each treatment (vehicle or 30 mg/kg).
As indicated by the latest European School Survey Project on Alcohol and Other Drugs report (ESPAD 2020), the lifetime prevalence of use of hallucinogens is higher in male (2.4%) than female (1.7%) population. Moreover, the majority of intoxications with designer tryptamines reported in literature concerns male subjects (Muller 2004; Smolinske et al. 2005; Matsumoto and Okada 2006; Shimizu et al. 2007; Johansson et al. 2017; Bilhimer et al. 2018). Furthermore, the intoxication case reported by the Pavia Poison Control Centre (PCC)—National Toxicology Information Centre involved a 23-year-old male. All these observations led us to investigate the effects of 5-MeO-MiPT at first in male subjects. However, we are aware that abuse of those compounds can as well involve female population, as the case reported by Itowaka and colleagues suggests (Itokawa et al. 2007). Moreover, it is known that gender and sex can importantly affect the responses to drugs (Fattore et al. 2020). In this regard, such studies have been conducted on murine model to evaluate any sex-related differences after hallucinogens administration. For example, female rats were reported to be more sensitive than males to the effects of 5-MeO-DMT (Shephard and Broadhurst 1983), while other studies stated that females are lesser affected than males by behavioural changes induced by LSD (Pálenícek et al. 2010) and psilocin (Tylš et al. 2016). Basing on this evidence, it can be assumed that male mice could be more sensitive to the effects of 5-MeO-MiPT on locomotor activity and sensory gating alterations than females. However, due to the higher sensitivity of females to the effect on hyponeophagia induced by 5-MeO-DMT showed by Shephard and Broadhurst, further research must be conducted to clarify this conflicting aspect.
Drug preparation and dose selection
The 5-MeO-MiPT was provided by the Forensic Science Department of Arm of Carabinieri (Roma’s R.I.S., referent Colonel Adolfo Gregori). The drug was dissolved in Tween 80 (2%) and ethanol (5%), brought to the final volume with saline (0.9% NaCl), and administered by intraperitoneal (i.p.) injection at a volume of 4 µL/g. Tween 80 (2%), ethanol (5%), and saline were also used as the vehicle. Doses of 5-MeO-MiPT were chosen based on the behavioural and neurological effects reported on internet experiences with the compound (Erowid 2015a, b, c) and other tryptamine hallucinogens (5-MeO-DIPT, 5-MeO-DMT, DMT). The human doses were converted to animal doses using the body surface area (BSA) normalization method (Reagan-Shaw et al. 2008). In particular, the lower doses (0.01 and 0.1 mg/kg; human equivalent doses of 0.00081 mg/kg and 0.0081 mg/kg in a 60 kg subject) tested in this study are equivalent to a human subthreshold dose, the intermediate dosage (1 mg/kg; human equivalent dose of 0.081 mg/kg) tested amounts to a common human oral dose, while the higher dosages (10 and 30 mg/kg; human equivalent doses of 0.81 and 2.43 mg/kg) tested exceed the highest values consumed by users (Psychonautwiki 2023a, b). Regarding PPI test, basing on our previous preclinical studies on hallucinogens (Tirri et al. 2022) and preliminary data, we decided to use a different range of doses (0.01–10 mg/kg). Instead, in cardiorespiratory and blood pressure tests we injected the highest dose of 5-MeO-MiPT (30 mg/kg) to simulate the heaviest intoxication.
Behavioural tests
The effect of 5-MeO-MiPT were investigated using a battery of behavioural tests widely employed in our laboratory for safety pharmacology studies for the preclinical characterization of NPS in rodents (Bilel et al. 2020, 2023; Corli et al. 2023). Mice were evaluated using different tests carried out in a consecutive manner, as follows. Observation of visual object (frontal and lateral) and placing responses, acoustic and tactile (pinna, vibrissae, and corneal reflexes) responses, were measured 10, 30, 60, 120, 180, 240, and 300 min after injection of the compound. The body core and surface temperatures were measured 15, 35, 65, 125, 185, 245, and 305 min after treatment. Accelerod test was measured at 15, 40, 70, 130, 190, 250, and 310 min, while drag test was measured at 45, 70, 105, 160, 220, 280, and 340 min. For the visual object, acoustic, and tactile sensorimotor tests, each mouse was housed in an experimental chamber (350 × 350 × 350 mm) made with black methacrylate walls and a transparent front door. A camera (B/W USB Camera day and night with varifocal lens; Ugo Basile, Italy) was placed at the top and/or side of the box. For the evaluation of effects of 5-MeO-MiPT on acoustic sensorimotor gating, the startle/PPI technique (Ugo Basile apparatus, Milan, Italy) was used. Behavioural tests were conducted in the Centralized Preclinical Research Laboratory of University of Ferrara at a thermostated temperature of 20–22 °C, humidity of approximately 45–55%, and controlled light (approximately 150 lx) with a background noise of approximately 40 ± 4 dB. All experiments were performed between 8:30 AM and 2:00 PM and conducted blindly by trained observers working in pairs.
Evaluation of visual response (object and placing response tests)
Visual response was measured by two behavioural tests, which evaluated the ability of the mouse to capture visual information when it is stationary (visual object response) or moving (visual placing response). The visual object response test consists of approaching an object (respectively a white horizontal bar and a small dentist’s mirror) from the front (frontal view) or the side (lateral view) to the mouse and evaluate the animal ability to see it, if when the mouse typically shifts or turns the head, or retreat (Ossato et al. 2015; Marti et al. 2019; Bilel et al. 2020, 2023). Both frontal and lateral visual response tests were repeated three times. The assigned score was 1 if there was a reflection in the mouse movement, or 0 if it was not present. The total value was calculated by adding the single scores of frontal and lateral visual response tests (overall score: 9).
Visual placing response test was performed using a tail suspension modified apparatus able to bring down the mouse towards the floor at a constant speed of 10 cm/s (Ossato et al. 2015). Mice were suspended 20 cm above the floor by an adhesive tape that was placed approximately 1 cm from the tip of the tail. The downward movement of the mouse was videotaped by a camera (B/W USB Camera day and night with varifocal lens; Ugo Basile, Italy) placed at the base of the tail suspension apparatus. Movies were analysed off-line by a trained operator who did not know the drug treatments performed. The analysis frame by frame allowed to evaluate the beginning of the reaction of the mouse while it was close to the floor. The first movement of the mouse when it perceives the floor is the extension of the front legs. When the mouse started the reaction, an electronic ruler evaluated the perpendicular distance in millimetres between the first-mice movement reaction and the floor.
Evaluation of overall tactile and acoustic responses
Tactile response of the mice was verified through vibrissae, corneal, and pinnae reflexes (Ossato et al. 2015). Vibrissae reflex was evaluated by touching vibrissae (right and left) with a gavage needle once for side, a score of 1 was given if there was a response (the mouse turns the head to the side of touch or vibrissae movement), and a score of 0 was given if there was no response (overall score: 2). Corneal reflex was assessed gently touching the cornea of the mouse with a gavage needle and evaluating the response, assigning a value of 1 if the mouse responded closing the eyelid and a value of 0 if there was no response. The test was repeated 3 times and was conducted bilaterally (overall score: 6). Pinna reflex was assessed by touching pavilions (left and right) with a gavage needle, first the interior and then the external pavilions. The test is repeated twice for side giving a value of 1 if there was a reflex and 0 if not present (overall score: 4). Data are expressed as the sum of the three parameters (overall score: 12).
Evaluation of acoustic response measures the reflexes of the mouse in response to an acoustic stimulus produced behind the animal (Ossato et al. 2015). Four acoustic stimuli of different intensities and frequencies were tested: a snap of the fingers, a sharp click (produced by a metal instrument), an acute (frequency of about 5.0–5.1 kHz, produced by an audiometer), and a sever (frequency of about 125–150 kHz, produced by an audiometer). Each sound was repeated three times, a score of 1 was given if there was a response, and a score of 0 was given if there was no response (total score of 3 for each sound). The acoustic total score was calculated by adding scores obtained in the four tests (overall score: 12).
Evaluation of core temperature and body surface temperature
The core temperature was evaluated by a probe (1 mm diameter) that was gently inserted, after lubrication with liquid Vaseline, into the rectum of the mouse (to about 2 cm) and left in position until the stabilization of the temperature (about 10 s; Vigolo et al. 2015). The probe was connected to a digital thermometer (Cole Parmer, model 8402). A cut-off for core body temperature was set at 22 °C (room temperature) as the lowest value reached by animals. The body surface temperature was measured by a Microlife FR 1DZ1 digital infrared digital thermometer (Microlife AG Swiss Corporation, Widnau, Switzerland) placed at 1 cm from the surface of the abdomen of the mouse and left in position until the indication of the temperature on the display of the instrument.
Stimulated motor activity assessment
Alteration of stimulated motor activity induced by 5-MeO-MiPT in mice was evaluated through two tests in which the animal is forced to move (accelerod and drag tests; Canazza et al. 2016; Bilel et al. 2020; De-Giorgio et al. 2020). For the accelerod test, mice were placed on a rotating cylinder (Ugo Basile, Milan, Italy) at 5-min intervals. The speed automatically and constantly increased (0–60 rotation/min). Time spent on the moving cylinder was measured with a cut-off of 300 s. The drag test consisted of lifting the mouse by the tail, leaving the front paws on the table, and dragging the animal backward at a constant speed of about 20 cm/s for a distance of 100 cm. The number of steps of each paw was recorded by two different observers. For each animal, five measurement were collected.
Startle/prepulse inhibition analysis
As previously reported (Bilel et al. 2020; Corli et al. 2023), mice were tested for acoustic startle reactivity in startle chamber (Ugo Basile apparatus, Milan, Italy) consisting of a sound-attenuated, lighted, and ventilated enclosure holding a transparent non-restrictive Perspex® cage (90 × 45 × 50 mm). Acoustic stimuli were produced by a loudspeaker mounted laterally the holder. Peak and amplitudes of the startle response were detected by a loadcell. At the onset of the startling stimulus, 300-ms readings were recorded and the wave amplitude evoked by the movement of the animal startle response was measured. Acoustic startle test session consisted of startle trials (pulse-alone) and prepulse trials (prepulse + pulse). The pulse-alone trials consisted of a 40-ms 120-dB pulse. Prepulse + pulse trials sequence consisted of a 20-ms acoustic prepulse, a 80-ms delay, followed by a 40-ms 120-dB startle pulse (100-ms onset-onset). Between the trials, there was an average of 15 s (range: from 9 to 21 s). Each startle session began with a 10-min acclimatation period with a 65-dB broadband white noise, continuously present throughout the session. The test session contained 40 trials composed by pulse-alone and prepulse + pulse trials, with three different prepulses of 68-dB, 75-dB, and 85-dB, presented in a pseudorandomized order. Animals were placed in the startle chambers 5 min after treatment and the entire PPI test lasted 20 min. As previously reported, 5-MeO-MiPT (0.01–10 mg/kg) was administered intraperitoneally, and startle/PPI responses were recorded 15 and 120 min after drug injection. The prepulse inhibition (% PPI) was expressed as the percentage decrease in the amplitude of the startle reactivity caused by the presentation of the prepulse.
Evaluation of cardiorespiratory and blood pressure changes
Cardiorespiratory parameters were monitored in awake and freely moving animals, without using invasive instruments and handling, a collar equipped with a sensor monitored heart rate, breath rate, oxygen saturation (SpO2), and pulse distension with an acquisition frequency of 15 Hz (Bilel et al. 2019; Marchetti et al. 2023). During the procedure, mice freely moved around their cages (30 × 30 × 20 cm), while having no access to food or water and while being monitored by the sensor collar through the software MouseOx Plus (STARR Life Sciences® Corp, Oakmont, PA, USA). To minimize the potential stress of mice during the experiment, a fake collar, similar in design to the one used in the test, was used in the first hour of acclimatation. After that, the collar with the sensor was applied, while the baseline parameters were monitored for 60 min. Then, 5-MeO-MiPT (30 mg/kg) or vehicle was administered, and data were recorded for 5 h.
Systolic and diastolic blood pressure were measured by tail-cuff plethysmography using a BP-2000 blood pressure analysis system (Visitech Systems, Apex, NC, USA; Ossato et al. 2018). Mice were placed in a metal box restraint with the tail passing through the optical sensor and compression cuff before finally being tapped to the platform. A traditional tail-cuff occlude was placed proximally on the animal’s tail, which was then immobilized with tape in a V-shaped block between a light source above and a photoresistor below. Upon inflation, the occlude stopped blood flow through the tail, while upon deflation, the sensor detected the blood flow return. The restraint platform was maintained at a temperature of 37 °C. Before experiments, mice were acclimated to restraint and tail-cuff inflation for 5–7 days. On the test day, 10 measurements were made to collect basal blood pressure. Upon the tenth analysis, the software was paused, and mice were injected with 5-MeO-MiPT (30 mg/kg) or vehicle; animals were then repositioned in the restraints, and 60 measurements were acquired.
Data and statistical analysis
In sensorimotor response experiments, data are expressed in arbitrary units (visual object response, vibrissae, corneal and pinnae reflex, acoustic response) and percentage of baseline (visual placing response, accelerod, drag tests). Core temperature and body surface temperature values were expressed as the difference between control temperature (before injection) and temperature following drug administration (Δ°C). The amount of PPI was calculated as a percentage score for each prepulse + pulse trial type: % PPI = 100 − {[(startle response for prepulse + pulse trial)/(startle response for pulse-alone trial)] × 100}. Startle magnitude was calculated as the average response to all the pulse-alone trials. All data are shown as mean ± SEM of 8 independent experimental replications. Statistical analysis of the effects of 5-MeO-MiPT at different concentrations over time was performed by two-way ANOVA followed by Bonferroni’s test for multiple comparisons, while statistical analysis of PPI results was carried out with one-way ANOVA followed by the Bonferroni’s test for multiple comparisons and expressed in histograms. Data related to heart rate (heart beats per min; bpm), pulse distention (vessel diameter changes; µm), breath rate (respiratory rate per minute; rrpm), and SpO2 saturation (oxygen blood saturation) were expressed as % changes of basal values. The statistical analysis of the effects of 5-MeO-MiPT (30 mg/kg) was performed by two-way ANOVA followed by Bonferroni’s test for multiple comparisons. Changes in systolic and diastolic blood pressure were expressed as absolute values (mmHg). The effects of 5-MeO-MiPT (30 mg/kg) over time were analysed by a two-way ANOVA followed by Bonferroni’s test for multiple comparisons. All statistical analyses were performed using GraphPad Prism software (GraphPad Prism, San Diego, CA, USA).
In silico ADMET prediction of 5-MeO-MiPT, 5-MeO-DIPT, and DMT
The in silico ADMET prediction of 5-MeO-MiPT, 5-MeO-DIPT, and DMT (chemical structures, Fig. 1) profiles was conducted through Simulations Plus ADMET Predictor® version 10.4 (× 64) on a Windows 11 operating system (ADMET Predictor®, Simulations-Plus Inc). The program predicts the physicochemical, pharmacokinetics, and toxicity properties based on the molecular structures of compounds. It uses artificial neural network ensemble (ANNE) models, which were trained to ensemble with data sets that share the same “architecture” (i.e. same inputs and number of neurons) from well-defined drugs, using the 2D structure and the atomic descriptors for data selection. The criteria used to set the ADMET scores are illustrated in Table 1.
Results
Case report
In July 2022, a 23-year-old male, transported via 112 emergency ambulance, was admitted to the Emergency Department (ED) of a Northern Italy Hospital manifesting tachycardia, catatonia, slowdown, drowsiness, and muteness. The anamnestic evaluation revealed a medical history characterized by a pre-existing depressive disorder, medicated with sertraline and alprazolam. The mother reported that, together with the ingestion of alprazolam, the son had smoked and sniffed an unknown substance, purchased via an internet-order. Therefore, the patient was temporarily diagnosed as suffering from a toxidrome with an unknown substance. His temperature was 36.0 °C, pulse rate 107 bpm, breath rate 19 breaths/min, SpO2 99%, and blood pressure 150/90 mmHg. Standard laboratory investigations assessed borderline haematochemical parameters. The patient’s treatment consisted of a supportive care and symptom management, conducted until the complete remission of symptomatology. The patient was discharged home after 24 h, without any known sequel. During hospitalization, blood and urine samples were collected and then processed by high-performance liquid chromatography-tandem mass spectrometry (LC–MS/MS) and gas chromatography/mass spectrometry (GC/MS) for the toxicological confirmation analyses, also with the support of electronic libraries. The analytical results indicated the presence of 5-MeO-MiPT, 4F-MPH, and DCK. The same samples tested negative for other substances (Table 2).
Behavioural tests
Evaluation of visual response (object and placing response tests)
Visual object (both frontal and lateral; Fig. 2a) and placing (Fig. 2b) responses did not change in vehicle-treated mice during 5 h of observation. Systemic administration of low doses (0.01–0.1 mg/kg i.p.) of 5-MeO-MiPT did not affect the visual object response (Fig. 1a). The dose of 1 mg/kg reduced the visual object response of mice ~ 33% compared to basal values during the first 30 min of the experiment [significant effect of treatment (F5,336 = 20.65, p < 0.0001), time (F7,336 = 17.35, p < 0.0001), and time × treatment interaction (F35,336 = 3.700, p < 0.0001)]. Ten and 30 mg/kg (i.p.; Fig. 2a) more deeply decreased the visual reflexes of animals in a transient manner (~ 55% at 10 min). In particular, after 120 min of treatment with 10 mg/kg 5-MeO-MiPT the response was equal to basal values, while for the 30 mg/kg was persistent. Systemic administration of 5-MeO-MiPT dose-dependently (Fig. 2b) reduced the visual placing response in mice [significant effect of treatment (F5,336 = 97.11, p < 0.0001), time (F7,336 = 59.28, p < 0.0001), and time × treatment interaction (F35,336 = 9.836, p < 0.0001)]. All the doses transiently induced an inhibition of the response, except the lowest one (0.01 mg/kg) that did not affect the visual placing response. The 0.1 mg/kg of 5-MeO-MiPT had a mild effect only during the first 5 min, while the highest dose (30 mg/kg) impaired the visual reflexes of mice to ~ 80% compared to the basal values during the first 5 min (p < 0.001) and the effect persisted up to 180 min (~ 30%) of observation.
Effects of 5-MeO-MiPT (0.01, 0.1, 1, 10, and 30 mg/kg; i.p.) on visual object (a) and placing (b) responses in mice. Data are expressed as mean ± SEM (n = 8/group). Statistical analysis was performed by two-way ANOVA followed by Bonferroni’s test for multiple comparisons for the dose–response curve of each compound at different time points (*p < 0.05, **p < 0.01, ***p < 0.001 versus vehicle)
Evaluation of overall tactile and acoustic responses
Overall tactile and acoustic responses did not change in vehicle-treated mice during 5 h of observation (Fig. 3a, b). Systemic administration of 5-MeO-MiPT (0.01–1 mg/kg) did not affect the tactile responses of mice. The injection of 10–30 mg/kg induced a significant enhancement of the reflex to tactile stimulation during the first 60 min of the experiment (Fig. 3a) and the effect disappeared after 120 min of treatment [significant effect of treatment (F5,336 = 58.99, p < 0.0001), time (F7,336 = 33.12, p < 0.0001), and time × treatment interaction (F35,336 = 14.04, p < 0.0001)].
Effects of 5-MeO-MiPT (0.01, 0.1, 1, 10, and 30 mg/kg; i.p.) on overall tactile (a) and acoustic (b) responses in mice. Data are expressed as mean ± SEM (n = 8/group). Statistical analysis was performed by two-way ANOVA followed by Bonferroni’s test for multiple comparisons for the dose–response curve of each compound at different time points (***p < 0.001 versus vehicle)
Conversely, the systemic administration of 5-MeO-MiPT (0.01–30 mg/kg) did not significantly affect the acoustic response of mice (Fig. 3b). Two-way ANOVA detected a significant effect of treatment [F5,336 = 3.160, p = 0.0084], time [F7,336 = 1.410, p = 0.2004], and time × treatment interaction [F35,336 = 0.5742, p = 0.9760].
Evaluation of core temperature and body surface temperature
Core temperature and body surface temperature of vehicle-treated animals remained constant during 5 h of observation (Fig. 4a, b). Only the highest dose of 5-MeO-MiPT (30 mg/kg i.p.) induced a hypothermic effect (Fig. 4a) at 35 min from the administration [significant effect of treatment (F5,294 = 4.486, p = 0.0006), time (F6,294 = 14.38, p < 0.0001), and time × treatment interaction (F30,294 = 1.625, p = 0.0238)]. On body surface temperature (Fig. 4b), two-way ANOVA revealed a significant decrease at the dose of 10 mg/kg at 35 min after treatment [significant effect of treatment (F5,294 = 11.85, p < 0.0001), time (F6,294 = 8.400, p < 0.0001), and time × treatment interaction (F30,294 = 1.262, p = 1.2692)].
Effects of 5-MeO-MiPT (0.01, 0.1, 1, 10, and 30 mg/kg; i.p.) on core body temperature (a) and body surface temperature (b) in mice. Data are expressed as mean ± SEM (n = 8/group). Statistical analysis was performed by two-way ANOVA followed by Bonferroni’s test for multiple comparisons for the dose–response curve of each compound at different time points (***p < 0.001 versus vehicle)
Stimulated motor activity assessment
Vehicle administration did not change motor activity in both accelerod and drag tests over the 5 h observation (Fig. 5a, b) in mice. The motor performance was not significantly affected in the groups of mice treated with the doses of 0.01, 0.1, 1, and 10 mg/kg of 5-MeO-MIPT (Fig. 4a). The highest dose induced a strong inhibition of the motor performance in the accelerod test (~ 70% of the basal), the effect appeared at 40 min after the administration, at 130 min there was still a mild effect, and in the last 2 h of the experiment the performance returned to basal values. Two-way ANOVA detected a significant effect of treatment [F5,336 = 20.16, p < 0.0001], time [F7,336 = 1.745, p = 0.0978], and time × treatment interaction [F35,336 = 1.163, p = 0.2480]. Similarly, the systemic administration of 5-MeO-MiPT (0.01–30 mg/kg) did not induce any effect on drag test, except with the highest one (Fig. 5b). In fact, the injection of 30 mg/kg induced ~ 60% of reduction of the number of steps performed with the front paws of mice after 45 min from the injection and the effect persisted up to 135 min of observation [significant effect of treatment (F5,336 = 7.673, p < 0.0001), time (F7,336 = 4.116, p = 0.0002), and time × treatment interaction (F35,336 = 1.828, p = 0.0038)].
Effects of 5-MeO-MiPT (0.01, 0.1, 1, 10, and 30 mg/kg; i.p.) on time on rod (a) and number of steps (b) in mice. Data are expressed as mean ± SEM (n = 8/group). Statistical analysis was performed by two-way ANOVA followed by Bonferroni’s test for multiple comparisons for the dose–response curve of each compound at different time points (*p < 0.05, **p < 0.01, ***p < 0.001 versus vehicle)
Startle/prepulse inhibition (PPI) analysis
Startle and PPI response did not change after the vehicle injection in mice (Fig. 6a–c). The systemic administration of 5-MeO-MiPT reduced the acoustic startle amplitude only at the dose of 10 mg/kg (Fig. 6a) at 120 min of analysis [significant effect of treatment (F4,35 = 3.862, p = 0.0106)]. In addition, PPI was also impaired after 1 mg/kg of 5-MeO-MiPT treatment and one-way ANOVA detected a significant effect at 15 min (Fig. 6b) at 68 dB [significant effect of treatment F4,35 = 7.340, p = 0.0002] and at 120 min (Fig. 6c) at 85 dB [significant effect of treatment F4,35 = 6.431, p = 0.0005].
Effects of 5-MeO-MiPT (0.01, 0.1, 1, 10, and 30 mg/kg; i.p.) on startle amplitude (a) and prepulse inhibition (PPI; b, c) in mice. Effects on PPI are shown for the three prepulse intensities (68, 75, 85 dB) 15 min (b) and 120 min (c) after treatment. Startle amplitude was expressed in absolute values (in dB) and the values represent the mean ± SEM (n = 8/group). PPI was expressed as the percentage decrease in the amplitude of the startle reactivity caused by presentation of the prepulse (% PPI; see “Materials and methods” section), values represent mean ± SEM (n = 8/group). Statistical analysis was performed by one-way ANOVA followed by Bonferroni’s test for multiple comparisons (*p < 0.05, **p < 0.01, and ***p < 0.001 vs. vehicle)
The injection of 10 mg/kg of 5-MeO-MiPT caused an impairment of the prepulse inhibition at 15 min (Fig. 6b) at 68 dB [significant effect of treatment (F4,35 = 7.340, p = 0.0002)], 75 dB [significant effect of treatment (F4,35 = 4.362, p = 0.0058)], and 85 dB [significant effect of treatment (F4,35 = 4.631, p = 0.0042)]. The effect persisted after 120 min of analysis (Fig. 6c); one-way ANOVA detected a reduction of the PPI at 68 dB [significant effect of treatment (F4,35 = 2.953, p = 0.0334)], 75 dB [significant effect of treatment (F4,35 = 5.258, p = 0.0020)], and 85 dB [significant effect of treatment (F4,35 = 6.431, p = 0.0005)].
Cardiorespiratory analysis
Since the most pronounced effects were observed after the systemic administration of the highest dose tested of 5-MeO-MiPT (30 mg/kg), we decided to investigate its possible adverse effects on cardiorespiratory system in mice, simulating a heavy intoxication. Basal heart rate (679 ± 14 bpm), breath rate (202 ± 2.42 rrpm), pulse distension (vessel diameter, 344 ± 16.83 µm), and SpO2 (92.22 ± 0.37 SpO2) did not change in animals treated with the vehicle (Fig. 7a–d). The systemic administration of 5-MeO-MiPT evoked a triphasic effect in heart rate in mice (Fig. 7a). After an initial enhancement right after the injection, a significant reduction in heart rate (between 40 and 20% comparing to basal values) was observed and the bradycardic effect lasted about 2 and a half hours of analysis. Then, from 255 min after the treatment the heart rate increased again, the maximal increase (~ 20%) was recorded at 330 min of the experiment. Two-way ANOVA revealed a significant effect of treatment [F1,760 = 412.8, p < 0.0001], time [F75,760 = 34.66, p < 0.0001], and time × treatment interaction [F75,760 = 43.93, p < 0.0001]. Simultaneously, the breath rate raised in the first hour of injection (Fig. 7b). The effect disappeared at 120 min after injection and manifested again at 140 min of analysis. The maximal effect (~ 150%) was registered at 155 min and the tachypnoea persisted up to the end of the experiment [significant effect of treatment (F1,760 = 366.2, p < 0.0001), time (F75,760 = 3.180, p < 0.0001), and time × treatment interaction (F75,760 = 5.380, p < 0.0001)]. The pulse distention (Fig. 7c) also immediately increased (~ 150% of basal values) after the 5-MeO-MiPT injection and then significantly decreased during the last 3 h of the experiment [significant effect of treatment (F1,760 = 374.9, p < 0.0001), time (F75,760 = 5.744, p < 0.0001), and time × treatment interaction (F75,760 = 4.768, p < 0.0001)]. At the same time, administration of 5-MeO-MIPT did not cause pronounced variation in SpO2 (Fig. 7d), two-way ANOVA detected a mild but statistically significant increase of Sp02 in comparison to basal values from 90 min after the treatment, and it remained constant during the following hours of the experiment [significant effect of treatment (F1,760 = 386.6, p < 0.0001), time (F75,760 = 2.259, p < 0.0001), and time × treatment interaction (F75,760 = 5.371, p < 0.0001)].
Effects of 5-MeO-MiPT (30 mg/kg; i.p.) on heart rate (a), breath rate (b), pulse distension (c), and SpO2 saturation (d) in mice. Data are expressed as percentage of basal values in the form mean ± SEM (n = 6/group). Statistical analysis was performed by two-way ANOVA, followed by Bonferroni’s test for multiple comparisons (*p < 0.05, **p < 0.01, *** p < 0.001 versus vehicle)
Basal systolic (103 ± 2 mmHg) and diastolic (73 ± 2 mmHg) pressure did not change in the group of mice treated with the vehicle (Fig. 8a, b). The systemic administration of 5-MeO-MIPT induced an immediate decrease of the systolic pressure (~ 80 mmHg; Fig. 8a) during the first 10 min after the injection followed by an increase that lasted up to the end of registration. Two-way ANOVA detected significant effect of treatment [F1,630 = 376.9, p < 0.0001], time [F69,360 = 5.641, p < 0.0001], and time × treatment interaction [F69,630 = 4.870, p < 0.0001]. Diastolic pressure also decreased during the first minutes after the injection (~ 40 mmHg), then it raised after 10 min (Fig. 8b). Then, ANOVA analysis detected a statistically mild significant increase of diastolic pressure only at 49 min of the experiment [significant effect of treatment (F1,560 = 3.239, p = 0.0724), time (F69, 560 = 5.342, p < 0.0001), and time × treatment interaction (F69,560 = 3.094, p < 0.0001)].
Effects of 5-MeO-MiPT (30 mg/kg; i.p.) on systolic (a) and diastolic (b) blood pressure. Data are expressed as absolute values (mm/Hg) in the form mean ± SEM (n = 5/group). Statistical analysis was performed by two-way ANOVA, followed by Bonferroni’s test for multiple comparisons (*p < 0.05, **p < 0.01, ***p < 0.001 versus vehicle)
ADMET prediction of 5-MeO-MiPT, 5-MeO-DIPT, and DMT
ADMET Predictor® calculated the risk scores for 5-MeO-MiPT, 5-MeO-DIPT, and DMT (Table 3). The ADMET risk of 5-MeO-MiPT, its analogue 5-MeO-DIPT, and the indole-alkaloid DMT were below the threshold of 7; however, ADMET predicted potential risk codes related to the volume of distribution (Vd) for 5-MeO-DIPT and DMT. The predictor calculated no absorption risk for the three compounds, and no toxicity risk for 5-MeO-MiPT and 5-MeO-DIPT. The predicted toxicity risk for DMT was under the threshold of 2, with a risk code related to acute rat toxicity (Table 3). ADMET predicted no cytochrome risk for DMT, whereas the scores of 5-MeO-MiPT and 5-MeO-DIPT were below the cytochrome risk threshold (< 2). Cytochrome risk code of high clearance by cytochrome P450 1A2 (CYP1A2) was predicted for both compounds, while risk code of high clearance by cytochrome P450 2C9 (CYP2C9) was predicted only for 5-MeO-MiPT. For DMT, a potential mutation risk was also predicted (yellow flag plot, Table 3); however, this risk is not considered in our study. As shown in Table 4, the three compounds were not predicted to induce elevated levels of hepatic enzymes in human serum, except for DMT which is predicted to induce elevated levels of serum aspartic acid transaminase (Ser_AST).
Table 5 describes the absorption profile of 5-MeO-MiPT, 5-MeO-DIPT, and DMT. The predictor calculated a high jejunal permeability (S + Peff) and a very high kidney permeability predicted in Madin-Darby canine kidney cells (MDCK) for the three compounds. They were all predicted to have a high blood brain barrier (BBB) penetration (99%). None of the compounds was predicted to be substrate and/or inhibitor of P-glycoprotein (P-gp) and substrate of the organic anion transporter 1 (OAT1) and organic anion transporter 3 (OAT3).
Some pharmacokinetic properties were also predicted (Table 6) for the three compounds. The predicted percent unbound to blood plasma proteins in human (hum_fup%) of the three drugs were similar to the values predicted for mice (mou_fup%). The calculated hum_fup% of the compounds were all over 30%. DMT was predicted to have a blood to plasma ratio in human (hum_RBP) slightly higher than 5-MeO-MiPT and 5-MeO-DIPT, which showed values lower than 1. Concerning the predicted mouse blood to plasma ratio (mou_RBP), the values of the three drugs were above 1.
The predicted volumes of distribution (Vd) of the three compounds were high, indicating that they are widely distributed in tissues after administration. The predicted values were higher for 5-MeO-DIPT and DMT than 5-MeO-MiPT, with a resulting potential ADME risk described in Table 3 for the former two compounds.
Regarding the inhibition of the bile salt export pump (BSEP), values of IC50 below the threshold of 60 μM indicate an inhibition of the transporter. The three drugs are not predicted to inhibit BSEP, since the IC50 values exceeded the threshold. The primary clearance mechanism predicted for 5-MeO-MiPT, 5-MeO-DIPT, and DMT was metabolism; in fact, the three compounds are classified as class_2 according to Extended Clearance Classification System (ECCS).
The metabolic profile of 5-MeO-MiPT was also predicted (Table 7). 5-MeO-MiPT was predicted to be a substrate but not inhibitor for CYP1A2 and CYP2C9, a substrate for cytochromes P450 2A6 (CYP2A6) and 2B6 (CYP2B6). The compound is not expected to be a substrate for cytochrome P450 2E1 (CYP2E1). 5-MeO-DIPT is predicted to be a substrate but not inhibitor for CYP1A2 and CYP2C9, a substrate for CYP2B6 and CYP2E1, while it is not predicted to be a substrate for CYP2A6. DMT is expected to be a substrate but not inhibitor for CYP1A2 and CYP2C9 and a substrate for CYP2A6 and CYP2E1. The three compounds were predicted to be both substrates and inhibitors for cytochrome P450 2D6 (CYP2D6). None of the compounds was predicted to be inhibitor or substrate for cytochrome P450 3A4 (CYP3A4). ADMET predicted the metabolic profile of 5-MeO-MiPT in human liver microsomes (HLM). ADMET predictor showed that 5-MeO-MiPT could produce four main metabolites (Fig. 9): N-deisopropyl metabolite (M1); N-demethyl metabolite (M2); O-demethyl metabolite (M3); and 1-OH-isopropyl metabolite (M4). On the other hand, two metabolites from 5-MeO-DIPT are estimated to be produced (Fig. 10): N-demethyl metabolite (M1) and O-demethyl metabolite (M2). Lastly, DMT is expected to be metabolized to (Fig. 11): N-oxide metabolite (M1), 5-OH metabolite (M2), and N-demethyl metabolite (M3).
Discussion
This study shows that the 5-Meo-MIPT identified in the seizures of law enforcement and responsible for the intoxication of a human subject causes relevant pharmaco-toxicological effects in the preclinical model.
Behavioural tests
Evaluation of visual response (object and placing response tests)
The manifestation of pharmacological effects after the administration of 5-MeO-MiPT was dose-dependent. Our results first showed that 5-MeO-MiPT induces a decrease of response to visual stimulation that has been seen through both object and placing response tests. In the visual object response test, 5-MeO-MiPT starts to have an effect at the 1 mg/kg dose, while it immediately reduces the visual placing starting from the 0.1 mg/kg. This trend of the visual response has also been described by our work group after the administration of LSD in mice (Tirri et al. 2022). The effect on visual functionality can be explained with the presence of 5HT2A receptors in primary visual cortex (V1) of mammals. In this regard, a study conducted on macaque demonstrated a modulatory effect of 5HT2A and serotonin receptor 1B (5HT1B) that depends on level of neuronal response. Activation of 5HT1B caused a facilitation of visual response from V1 high-firing neurons and suppressed low-firing ones. Administration of DOI, a 5-HT2A agonist, induced an opposite modulatory effect, suppressing the response from the high-firing neurons and facilitating responses from low-firing ones. Therefore, it is thought that 5HT2A receptor has a gain control in response to visual stimulation (Watakabe et al. 2009). Moreover, 5HT2A receptors’ role in reduction of visual response has been demonstrated in murine model. In fact, it has been showed that the administration of DOI induces a decrease in visual response and the surround suppression in primary visual cortex (Michaiel et al. 2019).
Evaluation of overall tactile and acoustic responses
The effect of hyperstimulation of the overall tactile response observed after the injection of the high doses (10–30 mg/kg) of 5-MeO-MiPT is in accordance with our previous preclinical studies conducted with LSD (Tirri et al. 2022), while the effect of 5-MeO-MiPT seems to be opposite to the one observed after the injection of DOB, that induces an inhibition of tactile response that may be caused by the stimulation of 5HT2A receptors in the cortical areas of the brain (Tirri et al. 2020). Regarding acoustic response, our results suggest that 5-MeO-MiPT at the doses tested do not induce significant effects.
Evaluation of core temperature and body surface temperature
Concerning thermoregulation, 5-MeO-MiPT causes a significant hypothermic effect only at the highest dose (30 mg/kg) and in a transient manner. Its effect deviates from LSD, which induces a dose-dependent increase of core temperature (Holze et al. 2022a). A study conducted on thermoregulation induced by 36 different tryptamines in rat and rabbits showed that just few of them caused variation in core temperature. Among them, 5-MeO-DIPT induced hypothermia during the period of multiple administration in rats, but 24 h after the last injection hyperthermia occurred (Araújo et al. 2015). Thus, it cannot be excluded a similar profile of effect on body temperature for 5-MeO-MiPT and further studies are required to clarify this aspect. The fact that hallucinogens can alter thermoregulation is in accordance with the roles of serotonin in maintenance of body homeostasis: activation of 25HT1, 5HT2, 5HT3, and 5HT7 receptors participates to thermoregulation through different mechanisms, as O2 consumption, CO2 excretion, or heat loss (Ishiwata 2014; Barnes et al. 2021; Voronova 2021). Specifically, previous studies showed that central 5HT1A receptor’s activation induces hypothermia in rats, affecting both heat production and heat loss processes. Contrary, central 5HT2 receptors have an opposite effect on rat body temperature, leading to hyperthermia. Also, it has been observed that peripheral 5HT2 receptors mediate the hypothermic effect described after the injection of 5HT in ICR mice (Voronova 2021). Moreover, it is been widely described the serotonin syndrome, a condition associated with drugs that alter the regulation of serotonin (such as monoamine oxidase inhibitors, MAOI, serotonin reuptake inhibitors, SSRI, or serotonin agonist), that, among all the symptoms, causes hyperthermia (Wang et al. 2016).
Stimulated motor activity assessment
Administration of 5-MeO-MiPT at the dose of 30 mg/kg significantly reduced the time spent on the rotarod and the number of steps performed by mice in a transient manner, suggesting that high doses of 5-MeO-MiPT produce an impairment of motor coordination. At date, no study on the effect on stimulated motor activity of tryptamines has been conducted but it is worth noting that designer tryptamines cause a decrease in spontaneous locomotor activity in rats (Krebs-Thomson et al. 2006; Williams et al. 2007; Pic-Taylor et al. 2015). Moreover, a similar effect has been observed in mice after the administration of psilocin and 5-MeO-DMT and it was demonstrated that the mechanism responsible for it is the activation of 5HT1A receptors (Halberstadt et al. 2011). Also, it is important to highlight that one of the symptoms observed in the patient of the case we reported was catatonia, accordingly to previous investigations that have shown that hallucinogens, especially at high doses, can produce withdrawal and catatonia-like state (Halberstadt and Geyer 2013).
Startle/prepulse inhibition (PPI) analysis
Prepulse inhibition (PPI) describes a phenomenon where the response to a sensorial stimulus (startle) is attenuated when preceded by a weaker stimulus (prepulse; Halberstadt 2015). PPI is often used as a measure of sensorial gating and it is one of the most typical tests conducted to characterize hallucinogen compounds, which have been postulated to disrupt sensory filtering mechanism and to cause sensory overload and cognitive dysfunction (Halberstadt and Geyer 2018).
From our analysis, we found that 5-MeO-MIPT causes a dose-dependent impairment of sensorial gating, according to previous studies that demonstrated the inhibition of PPI in murine model as a result of administration of hallucinogens, such as LSD, DOB, 2C-B, and “-NBOMe” serie phenethylamines (Halberstadt 2015; Halberstadt and Geyer 2018; Tirri et al. 2022). To date, the mechanism by which 5-MeO-MiPT can alter PPI is still unknown and more studies are needed and different mechanisms might be involved. The impairment of sensorial gating induced in rats by LSD and DOI has been attributed to the activation of 5HT2A receptors, because this effect is blocked by the 5HT2 antagonist M100907, but not by selective antagonists to subtypes 2B, 2C, and 1A (Halberstadt 2016). 5-MeO-DMT has also been found to disrupt PPI in rats, but the effect is primarily mediated by 5HT1A and not by 5HT2A receptors (Krebs-Thomson et al. 2006; Halberstadt 2016). Interestingly, it has been found that tryptamine hallucinogens have an opposite effect on PPI in mice. It has been shown that 5-MeO-DMT and psilocin dose-dependently increase PPI in mice and the effect of 5-MeO-DMT was partially attenuated by 5HT1A selective antagonist WAY-100635 (Halberstadt and Geyer 2011).
The effect on sensory functionality we found in mice fits in well with the action of hallucinogen compounds on human; in fact, it is known that these substances alter the mechanisms implicated in the processing of sensorial and sensorimotor information of the cortex (Preller et al. 2019). Specifically, 5HT2A receptors are expressed in the prefrontal cortex (PFC), which is involved in the sense of awareness of the external environment and in higher cognitive functions, and in the thalamus, where reticular nucleus regulates the flow of information between thalamus and cortex (Nichols 2016).
Cardiorespiratory analysis
In addition to behavioural effects, we have demonstrated through our study that systemic administration of 30 mg/kg of 5-MeO-MiPT can alter cardiorespiratory parameters. In fact, it affects heart rate, inducing an initial tachycardia followed by a significant bradycardia and reappearance of tachycardia. It also causes a decrease in pulse distention and an alteration of the breath rate. Tachycardia was one of the symptoms of 5-MeO-MiPT of the intoxication case reported above and was also described by some users on online forum (Psychonaut 2011). BP-2000 analysis showed that the compound elicits an increase of both systolic and diastolic blood pressure after an initial fall. All these effects fall within the profile of tryptamine designer drugs which as previously mentioned, cause in human tachypnoea, hypertension, and tachycardia, and are in accordance with the ones evoked by severe classic psychedelics (Schlag et al. 2022), including psilocybin (Carbonaro et al. 2018; Holze et al. 2022b) and DMT (Strassman and Qualls 1994). Conversely, administration of 5-MeO-DMT in rats has been observed to induce bradycardia and a biphasic effect in blood pressure, with a hypotension preceded by a primary hypertension (Dabire et al. 1987). It is well known that serotonin has a variety of actions on cardiovascular (Kaumann and Levy 2006) and respiratory systems (Barnes et al. 2021) and mediates the neuronal regulation of blood pressure (Watts et al. 2012). Specifically, previous findings suggest that the stimulation of 5HT1A induces central sympathoinhibition and decreases blood pressure, whereas 5HT2A mediate sympathomimetic effects such as increased heart rate, vasomotor tone, and blood pressure (Villalón and Centurión 2007; Reckweg et al. 2022). As previous studies (Chaouche-Teyara et al. 1994; Buchborn et al. 2020) conducted on psychedelics suggest, the mechanism behind the effects of 5-MeO-MiPT on blood pressure may also involve the stimulation of peripheral serotonin receptors located on the vascular smooth muscle and the endothelium, by which 5HT acts as a direct vasopressor regulator (Frishman and Grewall 2000; Villalón and Centurión 2007).
In silico ADMET prediction
Considering the lack of knowledge of the toxicokinetic effects of 5-MeO-MiPT, we decided to apply in silico ADMET prediction for 5-MeO-MiPT in comparison to 5-MeO-DIPT and DMT. The ADMET risk scores of the three compounds and their predicted pharmacokinetic properties were similar. They are shown to have a high permeability and to almost cross the BBB completely. In line with these findings, all the three compounds are very lipophilic; in fact, the drug permeability across membranes by passive diffusion is strongly related to its lipophilicity (Fan and de Lannoy 2014). Moreover, none of the drugs is predicted to be a substrate of P-gp, OAT1, and OAT3, which are efflux transporters expressed in the BBB and can limit the bioavailability of the drugs in the brain (Löscher and Potschka 2005; Nigam et al. 2015). In addition, they present high values of predicted Vd and their percentage unbound to blood plasma protein are over 30%. These data taken together reveal that the three compounds have a good ability to distribute and reach the target tissues (Zhang et al. 2012). Notably, only 5-MeO-DIPT and DMT are shown to have a potential risk code related to Vd, whereas 5-MeO-MiPT presents a lower predicted Vd and fits the threshold risk of ADMET (< 3.7 L/Kg).
The three compounds are not predicted to induce elevated levels of serum alanine transaminase (Ser_ALT), serum lactate dehydrogenase (Ser_LDH), serum alkaline phosphatase (Ser_AlkPhos), and serum gamma-glutamyl transferase (Ser_GGT) in human. An alteration of these hepatic enzymes can be associated to liver damages or alteration of the bile flow (Giannini et al. 2005). Moreover, 5-MeO-MiPT, 5-MeO-DIPT, and DMT are not predicted to inhibit the bile salt export pump, as the BSEP_IC50 predicted are beyond the threshold. Liver damage or alteration of the bile flow can be related to an alteration of hepatic enzyme levels (Giannini et al. 2005) and the inhibition of BSEP, which leads to an increase of intracellular bile salt concentration to toxic level, is one of the causes of drug induced liver injury (DILI; Kis et al. 2012). These evidences show that 5-MeO-MiPT, 5-MeO-DIPT, and DMT are not expected to induce hepatotoxicity. It is worth to note that Altuncı and colleagues previously conducted an in vivo experiment on female mice and found that high dosages of 5-MeO-MiPT may induce histopathological effects on liver, kidneys, and brain. Also, high doses of 5-MeO-MiPT caused an increase of levels of caspase-3 and caspase-8 in the tissues, suggesting that the compound could trigger apoptotic cell death (Altuncı et al. 2021). However, their findings are not in contradiction with our prediction; in fact, we estimated the hepatotoxic risk of the compound only in human and not in murine model. Finally, the primary clearance mechanism predicted for 5-MeO-MiPT, 5-MeO-DIPT, and DMT was metabolism and ADMET Predictor® highlights a cytochrome risk related to 5-MeO-MiPT and 5-MeO-DIPT. Specifically, 5-MeO-MiPT is exposed to high clearance by CYP1A2 and CYP2C9, while 5-MeO-DIPT only by CYP1A2. Moreover, all the three drugs are predicted to be both substrate and inhibitor to CYP2D6, an enzyme that takes part in metabolizing many commonly used drugs (Taylor et al. 2020) and psychoactive substances (Kirchheiner et al. 2011), and the inhibition of which could lead to drug-drug interaction.
The described metabolisms of 5-MeO-MiPT, 5-MeO-DIPT, and DMT involve reactions of N-dealkylation, O-demethylation, and hydroxylation of the parent drug. According to in silico ADMET prediction, 5-MeO-MiPT and DMT are both expected to be metabolized to 5-MeO-NIPT. These results are partially in line with a previous study conducted by Grafinger and colleagues, who analysed three samples from an intoxication case associated to 5-MeO-MiPT and proposed a possible metabolic pathway for the compound, using both in vivo and in vitro samples. Their study showed the production of 5-MeO-NIPT and 5-OH-MiPT in all the sample types (Grafinger et al. 2018). The proposed pathway is similar to the one described for 5-MeO-DIPT, and it involves CYP1A2, CYP2C19, CYP3A4, and CYP2D6 (Kamata et al. 2010).
The in silico ADMET prediction has already been used by our workgroup (Arfè et al. 2023) and it once again proved to be a useful tool in preliminary characterization of the pharmaco-toxicological effect on NPS.
Conclusion
The present study is the result of a collaboration among the Police Forces (Arma of Carabinieri), the Pavia Poison Control Centre (PCC)—National Toxicology Information Centre, and the preclinical drug-toxicology laboratory (Collaborative Centre of DPA) and represents an integrated competence model (Fig. 12) for the pharmaco-toxicological evaluation (in vivo preclinical studies and in silico ADMET prediction) of the effects of NPS seized in the national territory, which can cause intoxications in humans.
Schematic representation of the competency model for the pharmaco-toxicological evaluation (in vivo preclinical studies and in silico ADMET prediction) of the effects of NPS seized in the national territory and involved in human intoxications, as a result of the collaboration among the Police Forces (Arma of Carabinieri), the Pavia Poison Control Centre (PCC)—National Toxicology Information Centre, and the preclinical drug-toxicology laboratory (Collaborative Centre of DPA)
The preclinical in vivo study demonstrates that 5-MeO-MiPT inhibits, in a dose-dependent manner, the visual response and the sensorial gating. Also, we demonstrated that high doses of the compound impair the motor coordination and induce cardiorespiratory alterations in mice. Moreover, we emphasize that the human intoxication case includes symptoms that match the effects evoked by 5-Meo-MiPT in mice, suggesting that 5-MeO-MiPT, among the other compounds found in blood and urine samples, played a role in the induction of the patient health status. In silico ADMET prediction revealed a cytochrome risk of 5-MeO-MiPT and the possible interaction with other substances by inhibiting CYP2D6. Worthily noting, the compound is not predicted to have a risk of hepatotoxicity. The exposure to high doses of this compound may lead to a risk due to its effects on cardiorespiratory system. Moreover, the psychomotor and sensory impairment together with the hallucinatory state that it induces can profoundly affect the daily activities of the consumer and, for example, have implications for driving safety. In this regard, driving under the influence of hallucinogens has been associated with risk-taking and impulsive behaviours such as speeding or disregarding traffic signals (Salas-Wright et al. 2021). On the other hand, we highlight that the heaviest effects we found in mice were induced by doses of 5-MeO-MiPT that strongly exceed the human dose commonly consumed in order to experience the hallucinogen effects and that so far, the compound has never been associated with fatal intoxication. In those circumstances, we tend to consider that the risk of 5-MeO-MiPT is limited to the assumption of very high dosages, the combination with other substances, or the intake of the substance under conditions requiring the full ability of the subject to perform his activities. Given that and the in silico ADMET profile obtained, we consider 5-MeO-MiPT a compound of which the therapeutic potential in the treatment of psychiatric disorders, such as depression, anxiety, post-traumatic stress disorder, addiction, and eating disorders, can be explored. In fact, in recent years there has been a revival of interest in psychedelics in this area and several studies and clinical trials have suggested their effectiveness in the treatment of alcohol dependence, anxiety, and major depression disorder (Bogenschutz et al. 2015; Dos Santos et al. 2016; Davis et al. 2021; De Gregorio et al. 2021).
Abbreviations
- 4F-MPH:
-
4-Fluoromethylphenidate
- 5HT:
-
Serotonin
- 5HT1A :
-
Serotonin receptor 1A
- 5HT1B :
-
Serotonin receptor 1B
- 5HT2A :
-
Serotonin receptor 2A
- 5-MeO-DIPT:
-
5-Methoxy-N,N-diisopropyltryptamine
- 5-MeO-MiPT:
-
5-Methoxy-N-methyl-N-isopropyltryptamine
- Absn_Risk:
-
Absorption risk
- ADMET:
-
Absorption, distribution, metabolism, excretion, toxicity
- ADMET_Risk:
-
Absorption, distribution, metabolism, excretion, toxicity risk
- BBB:
-
Blood brain barrier
- BSEP:
-
Bile salt export pump
- CYP_Risk:
-
Cytochrome risk
- CYP1A2:
-
Cytochrome P450 1A2
- CYP2A6:
-
Cytochrome P450 2A6
- CYP2B6:
-
Cytochrome P450 2B6
- CYP2C9:
-
Cytochrome P450 2C9
- CYP2E1:
-
Cytochrome P450 2E1
- CYP3A4:
-
Cytochrome P450 3A4
- CYPs:
-
Cytochromes P450
- DCK:
-
Deschloroketamine
- DET:
-
N,N-diethyltryptamine
- DMT:
-
N,N-dimethyltryptamine
- ECCS:
-
Extended Clearance Classification System
- Fup:
-
Percent unbound to blood plasma proteins
- hERG:
-
Human voltage-dependent K + channel inhibition
- HLM:
-
Human liver microsomes
- Hum_fup:
-
Percent unbound to blood plasma proteins in human
- Hum_RBP:
-
Blood to plasma ratio in human
- MDCK:
-
Madin-Darby canine kidney
- Mou_fup:
-
Percent unbound to blood plasma proteins in mouse
- Mou_RBP:
-
Blood to plasma ratio in mouse
- NET:
-
Norepinephrine transporter
- NPS:
-
Novel psychoactive substances
- OAT1:
-
Organic anion transporter 1
- OAT3:
-
Organic anion transporter 3
- Peff:
-
Jejunal permeability
- Pgp:
-
P-glycoprotein
- PPI:
-
Prepulse inhibition
- RBP:
-
Blood to plasma ratio
- S + CL_Mech:
-
Clearance mechanism
- Ser_AlkPhos:
-
Serum alkaline phosphatase
- Ser_ALT:
-
Serum alanine transaminase
- Ser_AST:
-
Serum aspartic acid transaminase
- Ser_GGT:
-
Serum gamma-glutamyl transferase
- Ser_LDH:
-
Serum lactate dehydrogenase
- SERT:
-
Serotonin transporter
- SNAP:
-
National Early Warning System
- SpO2:
-
Oxygen blood saturation
- TOX_Risk:
-
Toxicity risk
- Vd:
-
Volume of distribution
References
Allerta Dronetplus (2016) Triptamine. [Online] Available at: http://allerta.dronetplus.eu/triptamine.html. Accessed 28 July 2023. http://allerta.dronetplus.eu/triptamine.html
Altuncı YA, Aydoğdu M, Açıkgöz E, Güven Ü, Düzağaç F, Atasoy A, Dağlıoğlu N, Annette Akgür S (2021) New psychoactive substance 5-MeO-MiPT in vivo acute toxicity and hystotoxicological study. Balkan Med J 38(1):34–42. https://doi.org/10.4274/balkanmedj.galenos.2020.2019.11.68
Araújo AM, Carvalho F, MdeL B, Guedes de Pinho P, Carvalho M (2015) The hallucinogenic world of tryptamines: an updated review. Arch Toxicol 89(8):1151–1173. https://doi.org/10.1007/s00204-015-1513-x
Arfè R, Bilel S, Tirri M, Corli G, Bergamin E, Serpelloni G, Bassi M, Borsari M, Boccuto F, Bernardi T, Caruso L, Alkilany AM, Rachid O, Botrè F, De-Giorgio F, Marti M (2023) Comprehensive evaluation of the pharmacological and toxicological effects of γ-valerolactone as compared to γ-hydroxybutyric acid: insights from in vivo and in silico models. Drug Alcohol Depend 252:110951. https://doi.org/10.1016/j.drugalcdep.2023.110951
Barnes NM, Ahern GP, Becamel C, Bockaert J, Camilleri M, Chaumont-Dubel S, Claeysen S, Cunningham KA, Fone KC, Gershon M, Di Giovanni G, Goodfellow NM, Halberstadt AL, Hartley RM, Hassaine G, Herrick-Davis K, Hovius R, Lacivita E, Lambe EK, Leopoldo M, Levy FO, Lummis SCR, Marin P, Maroteaux L, McCreary AC, Nelson DL, Neumaier JF, Newman-Tancredi A, Nury H, Roberts A, Roth BL, Roumier A, Sanger GJ, Teitler M, Sharp T, Villalón CM, Vogel H, Watts SW, Hoyer D (2021) International Union of Basic and Clinical Pharmacology. CX. Classification of receptors for 5-hydroxytryptamine; pharmacology and function. Pharmacol Rev 73(1):310–520. https://doi.org/10.1124/pr.118.015552
Baumeister D, Tojo LM, Tracy DK (2015) Legal highs: staying on top of the flood of novel psychoactive substances. Ther Adv Psychopharmacol 5(2):97–132. https://doi.org/10.1177/2045125314559539
Bilel S, Tirri M, Arfè R, Stopponi S, Soverchia L, Ciccocioppo R, Frisoni P, Strano-Rossi S, Miliano C, De-Giorgio F, Serpelloni G, Fantinati A, De Luca MA, Neri M, Marti M (2019) Pharmacological and behavioral effects of the synthetic cannabinoid AKB48 in rats. Front Neurosci 13:1163. https://doi.org/10.3389/fnins.2019.01163
Bilel S, Tirri M, Arfè R, Ossato A, Trapella C, Serpelloni G, Neri M, Fattore L, Marti M (2020) Novel halogenated synthetic cannabinoids impair sensorimotor functions in mice. Neurotoxicology 76:17–32. https://doi.org/10.1016/j.neuro.2019.10.002
Bilel S, Giorgetti A, Tirri M, Arfè R, Cristofori V, Marchetti B, Corli G, Caruso L, Zauli G, Giorgetti R, Marti M (2023) Sensorimotor alterations induced by novel fentanyl analogs in mice: possible impact on human driving performances. Curr Neuropharmacol 21(1):87–104. https://doi.org/10.2174/1570159X21666221116160032
Bilhimer MH, Schult RF, Higgs KV, Wiegand TJ, Gorodetsky RM, Acquisto NM (2018) Acute intoxication following dimethyltryptamine ingestion. Case Reports in Emergency Medicine 3452691. https://doi.org/10.1155/2018/3452691
Blough BE, Landavazo A, Decker AM, Partilla JS, Baumann MH, Rothman RB (2014) Interaction of psychoactive tryptamines with biogenic amine transporters and serotonin receptor subtypes. Psychopharmacology 231(21):4135–4144. https://doi.org/10.1007/s00213-014-3557-7
Bogenschutz MP, Forcehimes AA, Pommy JA, Wilcox CE, Barbosa PC, Strassman RJ (2015) Psilocybin-assisted treatment for alcohol dependence: a proof-of-concept study. J Psychopharmacol 29(3):289–299. https://doi.org/10.1177/0269881114565144
Bouso JC, Andión Ó, Sarris JJ, Scheidegger M, Tófoli LF, Opaleye ES, Schubert V, Perkins D (2022) Adverse effects of ayahuasca: results from the Global Ayahuasca Survey. PLOS Global Public Health 2(11):e0000438. https://doi.org/10.1371/journal.pgph.0000438
Buchborn T, Lyons T, Song C, Feilding A, Knöpfel T (2020) The serotonin 2A receptor agonist 25CN-NBOH increases murine heart rate and neck-arterial blood flow in a temperature-dependent manner. J Psychopharmacol 34(7):786–794. https://doi.org/10.1177/0269881120903465
Canazza I, Ossato A, Trapella C, Fantinati A, De Luca MA, Margiani G, Vincenzi F, Rimondo C, Di Rosa F, Gregori A, Varani K, Borea PA, Serpelloni G, Marti M (2016) Effect of the novel synthetic cannabinoids AKB48 and 5F-AKB48 on “tetrad”, sensorimotor, neurological and neurochemical responses in mice. In vitro and in vivo pharmacological studies. Psychopharmacology 233(21–22):3685–3709. https://doi.org/10.1007/s00213-016-4402-y
Carbonaro TM, Johnson MW, Hurwitz E, Griffiths RR (2018) Double-blind comparison of the two hallucinogens psilocybin and dextromethorphan: similarities and differences in subjective experiences. Psychopharmacology 235(2):521–534. https://doi.org/10.1007/s00213-017-4769-4
Chaouche-Teyara K, Fournier B, Safar M, Dabiré H (1994) Systemic and regional haemodynamic effects of 1-(2,5-dimethoxy-4-iodo-phenyl)-2-aminopropane (DOI) and alpha-methyl-5-HT, in the anaesthetised rat. Clin Exp Hypertens 16(6):779–798. https://doi.org/10.3109/10641969409078025
Corli G, Tirri M, Bilel S, Arfè R, Coccini T, Roda E, Marchetti B, Vincenzi F, Zauli G, Borea PA, Locatelli CA, Varani K, Marti M (2023) MAM-2201 acute administration impairs motor, sensorimotor, prepulse inhibition, and memory functions in mice: a comparison with its analogue AM-2201. Psychopharmacology 240(7):1435–1452. https://doi.org/10.1007/s00213-023-06378-8
Dabire H, Cherqui C, Fournier B, Schmitt H (1987) Comparison of effects of some 5-HT1 agonists on blood pressure and heart rate of normotensive anaesthetized rats. Eur J Pharmacol 140(3):259–266. https://doi.org/10.1016/0014-2999(87)90282-2
Davis AK, Barrett FS, May DG, Cosimano MP, Sepeda ND, Johnson MW, Finan PH, Griffiths RR (2021) Effects of psilocybin-assisted therapy on major depressive disorder: a randomized clinical trial. JAMA Psychiat 78(5):481–489. https://doi.org/10.1001/jamapsychiatry.2020.3285
De Gregorio D, Aguilar-Valles A, Preller KH, Heifets BD, Hibicke M, Mitchell J, Gobbi G (2021) Hallucinogens in mental health: preclinical and clinical studies on LSD, psilocybin, MDMA, and ketamine. J Neurosci 41(5):891–900. https://doi.org/10.1523/JNEUROSCI.1659-20.2020
De-Giorgio F, Bilel S, Tirri M, Arfè R, Trapella C, Camuto C, Foti F, Frisoni P, Neri M, Botrè F, Marti M (2020) Methiopropamine and its acute behavioral effects in mice: is there a gray zone in new psychoactive substances users? Int J Legal Med 134(5):1695–1711. https://doi.org/10.1007/s00414-020-02302-3
Dipartimento Politiche Antidroga (2015) Relazione annuale al Parlamento 2015 sullo stato delle tossicodipendenze in Italia [Online] Available at: https://www.politicheantidroga.gov.it/it/attivita/relazioni-annuali-al-parlamento/relazione-annuale-al-parlamento-sul-fenomeno-delle-tossicodipendenze-in-italia-anno-2015-dati-2014/. Accessed 28 July 2023.
Dos Santos RG, Osório FL, Crippa JA, Riba J, Zuardi AW, Hallak JE (2016) Antidepressive, anxiolytic, and antiaddictive effects of ayahuasca, psilocybin and lysergic acid diethylamide (LSD): a systematic review of clinical trials published in the last 25 years. Ther Adv Psychopharmacol 6(3):193–213. https://doi.org/10.1177/2045125316638008
EMCDDA (2021) European drug report 2021: trends and developments. Publications Office of the European Union, Luxembourg. https://doi.org/10.2810/18539
EMCDDA (2022) European drug report 2022: trends and developments. Publications Office of the European Union, Luxembourg. https://doi.org/10.2810/75644
Erowid (2015a) 5-MeO-DIPT, effects [Online] Available at: https://erowid.org/chemicals/5meo_dipt/5meo_dipt_effects.shtml Accessed on 14/12/2023.
Erowid (2015b) 5-MeO-MiPT, effects [Online] Available at: https://erowid.org/chemicals/5meo_mipt/5meo_mipt_effects.shtml. Accessed 28 July 2023.
Erowid (2015c) DMT, effects [Online] Available at: https://erowid.org/chemicals/dmt/dmt_effects.shtml Accessed on 14/12/2023.
ESPAD (2020) ESPAD Report 2019: results from the european school survey project on alcohol and other drugs, EMCDDA Joint Publications, Publications Office of the European Union, Luxembourg
Fan J, de Lannoy IA (2014) Pharmacokinetics. Biochem Pharmacol 87(1):93–120. https://doi.org/10.1016/j.bcp.2013.09.007
Fantegrossi WE, Murnane KS, Reissig CJ (2008) The behavioral pharmacology of hallucinogens. Biochem Pharmacol 75(1):17–33. https://doi.org/10.1016/j.bcp.2007.07.018
Fattore L, Marti M, Mostallino R, Castelli MP (2020) Sex and gender differences in the effects of novel psychoactive substances. Brain Sci 10(9):606. https://doi.org/10.3390/brainsci10090606
Frishman WH, Grewall P (2000) Serotonin and the heart. Ann Med 32(3):195–209. https://doi.org/10.3109/07853890008998827
Giannini EG, Testa R, Savarino V (2005) Liver enzyme alteration: a guide for clinicians. Can Med Assoc J 172(3):367–379. https://doi.org/10.1503/cmaj.1040752
Grafinger KE, Hädener M, König S, Weinmann W (2018) Study of the in vitro and in vivo metabolism of the tryptamine 5-MeO-MiPT using human liver microsomes and real case samples. Drug Test Anal 10(3):562–574. https://doi.org/10.1002/dta.2245
Halberstadt AL (2015) Recent advances in the neuropsychopharmacology of serotonergic hallucinogens. Behav Brain Res 277:99–120. https://doi.org/10.1016/j.bbr.2014.07.016
Halberstadt AL (2016) Behavioral and pharmacokinetic interactions between monoamine oxidase inhibitors and the hallucinogen 5-methoxy-N, N-dimethyltryptamine. Pharmacol Biochem Behav 143:1–10. https://doi.org/10.1016/j.pbb.2016.01.005
Halberstadt AL, Geyer MA (2011) Multiple receptors contribute to the behavioral effects of indoleamine hallucinogens. Neuropharmacology 61(3):364–381. https://doi.org/10.1016/j.neuropharm.2011.01.017
Halberstadt AL, Geyer MA (2013) Serotonergic hallucinogens as translational models relevant to schizophrenia. Int J Neuropsychopharmacol 16(10):2165–2180. https://doi.org/10.1017/S1461145713000722
Halberstadt AL, Geyer MA (2018) Effect of hallucinogens on unconditioned behavior. Curr Top Behav Neurosci 36:159–199. https://doi.org/10.1007/7854_2016_466
Halberstadt AL, Koedood L, Powell SB, Geyer MA (2011) Differential contributions of serotonin receptors to the behavioral effects of indoleamine hallucinogens in mice. J Psychopharmacol 25(11):1548–1561. https://doi.org/10.1177/0269881110388326
Heise CW, Brooks DE (2017) Ayahuasca exposure: descriptive analysis of calls to US poison control centers from 2005 to 2015. J Med Toxicol 13(3):245–248. https://doi.org/10.1007/s13181-016-0593-1
Holze F, Caluori TV, Vizeli P, Liechti ME (2022a) Safety pharmacology of acute LSD administration in healthy subjects. Psychopharmacology 239(6):1893–1905. https://doi.org/10.1007/s00213-021-05978-6
Holze F, Ley L, Müller F, Becker AM, Straumann I, Vizeli P, Kuehne SS, Order MA, Duthaler U, Kolaczynska KE, Varghese N, Eckert A, Liechti ME (2022b) Direct comparison of the acute effects of lysergic acid diethylamide and psilocybin in a double-blind placebo-controlled study in healthy subjects. Neuropsychopharmacology 47(6):1180–1187. https://doi.org/10.1038/s41386-022-01297-2
Ishiwata T (2014) Role of serotonergic system in thermoregulation in rats. J Phys Fit Sports Med 3(4):445–450. https://doi.org/10.7600/jpfsm.3.445
Itokawa M, Iwata K, Takahashi M, Sugihara G, Sasaki T, Abe Y, Uno M, Hobo M, Jitoku D, Inoue K, Arai M, Yasuda I, Shintani M (2007) Acute confusional state after designer tryptamine abuse. Psychiatry Clin Neurosci 61(2):196–199. https://doi.org/10.1111/j.1440-1819.2007.01638.x
Johansson A, Lindstedt D, Roman M, Thelander G, Nielsen EI, Lennborn U, Sandler H, Rubertsson S, Ahlner J, Kronstrand R, Kugelberg FC (2017) A non-fatal intoxication and seven deaths involving the dissociative drug 3-MeO-PCP. Forensic Sci Int 275:76–82. https://doi.org/10.1016/j.forsciint.2017.02.034
Kamata T, Katagi M, Tsuchihashi H (2010) Metabolism and toxicological analyses of hallucinogenic tryptamine analogues being abused in Japan. Forensic Toxicol 28:1–8. https://doi.org/10.1007/s11419-009-0087-9
Kaumann AJ, Levy FO (2006) 5-Hydroxytryptamine receptors in the human cardiovascular system. Pharmacol Ther 111(3):674–706. https://doi.org/10.1016/j.pharmthera.2005.12.004
Kirchheiner J, Seeringer A, Godoy AL, Ohmle B, Maier C, Beschoner P, Sim EJ, Viviani R (2011) CYP2D6 in the brain: genotype effects on resting brain perfusion. Mol Psychiatry 16:333–341. https://doi.org/10.1038/mp.2010.42
Kis E, Ioja E, Rajnai Z, Jani M, Méhn D, Herédi-Szabó K, Krajcsi P (2012) BSEP inhibition: in vitro screens to assess cholestatic potential of drugs. Toxicol Vitro 26(8):1294–1299. https://doi.org/10.1016/j.tiv.2011.11.002
Krebs-Thomson K, Ruiz EM, Masten V, Buell M, Geyer MA (2006) The roles of 5-HT1A and 5-HT2 receptors in the effects of 5-MeO-DMT on locomotor activity and prepulse inhibition in rats. Psychopharmacology 189(3):319–329. https://doi.org/10.1007/s00213-006-0566-1
Löscher W, Potschka H (2005) Blood-brain barrier active efflux transporters: ATP-binding cassette gene family. NeuroRx 2(1):86–98. https://doi.org/10.1602/neurorx.2.1.86
Luethi D, Liechti ME (2020) Designer drugs: mechanism of action and adverse effects. Arch Toxicol 94(4):1085–1133. https://doi.org/10.1007/s00204-020-02693-7
Marchetti B, Bilel S, Tirri M, Corli G, Roda E, Locatelli CA, Cavarretta E, De-Giorgio F, Marti M (2023) Acute cardiovascular and cardiorespiratory effects of JWH-018 in awake and freely moving mice: mechanism of action and possible antidotal interventions? Int J Mol Sci 24(8):7515. https://doi.org/10.3390/ijms24087515
Marti M, Neri M, Bilel S, Di Paolo M, La Russa R, Ossato A, Turillazzi E (2019) MDMA alone affects sensorimotor and prepulse inhibition responses in mice and rats: tips in the debate on potential MDMA unsafety in human activity. Forensic Toxicol 37:132–144. https://doi.org/10.1007/s11419-018-0444-7
Matsumoto T, Okada T (2006) Designer drugs as a cause of homicide. Addiction 101(11):1666–1667. https://doi.org/10.1111/j.1360-0443.2006.01573.x
Michaiel AM, Parker PRL, Niell CM (2019) A hallucinogenic serotonin-2A receptor agonist reduces visual response gain and alters temporal dynamics in mouse V1. Cell Rep 26(13):3475-3483.e4. https://doi.org/10.1016/j.celrep.2019.02.104
Muller AA (2004) New drugs of abuse update: foxy methoxy. J Emerg Nurs 30(5):507–508. https://doi.org/10.1016/j.jen.2004.07.037
Nagai F, Nonaka R, Satoh Hisashi Kamimura K (2007) The effects of non-medically used psychoactive drugs on monoamine neurotransmission in rat brain. Eur J Pharmacol 559(2–3):132–137. https://doi.org/10.1016/j.ejphar.2006.11.075
Nichols DE (2016) Psychedelics. Pharmacol Rev 68(2):264–355. https://doi.org/10.1124/pr.115.011478
Nigam SK, Bush KT, Martovetsky G, Ahn SY, Liu HC, Richard E, Bhatnagar V, Wu W (2015) The organic anion transporter (OAT) family: a systems biology perspective. Physiol Rev 95(1):83–123. https://doi.org/10.1152/physrev.00025.2013
Ossato A, Vigolo A, Trapella C, Seri C, Rimondo C, Serpelloni G, Marti M (2015) JWH-018 impairs sensorimotor functions in mice. Neuroscience 300:174–188. https://doi.org/10.1016/j.neuroscience.2015.05.021
Ossato A, Bilel S, Gregori A, Talarico A, Trapella C, Gaudio RM, De-Giorgio F, Tagliaro F, Neri M, Fattore L, Marti M (2018) Neurological, sensorimotor and cardiorespiratory alterations induced by methoxetamine, ketamine and phencyclidine in mice. Neuropharmacology 141:167–180. https://doi.org/10.1016/j.neuropharm.2018.08.017
Pálenícek T, Hlinák Z, Bubeníková-Valesová V, Novák T, Horácek J (2010) Sex differences in the effects of N, N-diethyllysergamide (LSD) on behavioural activity and prepulse inhibition. Prog Neuropsychopharmacol Biol Psychiatry 34(4):588–596. https://doi.org/10.1016/j.pnpbp.2010.02.008
Pic-Taylor A, da Motta LG, de Morais JA, Junior WM, Santos Ade F, Campos LA, Mortari MR, von Zuben MV, Caldas ED (2015) Behavioural and neurotoxic effects of ayahuasca infusion (Banisteriopsis caapi and Psychotria viridis) in female Wistar rat. Behav Processes 118:102–110. https://doi.org/10.1016/j.beproc.2015.05.004
Preller KH, Razi A, Zeidman P, Stämpfli P, Friston KJ, Vollenweider FX (2019) Effective connectivity changes in LSD-induced altered states of consciousness in humans. Proc Natl Acad Sci USA 116(7):2743–2748. https://doi.org/10.1073/pnas.1815129116
Psychonaut (2011) 5-MEO-MIPT e 5MM + AMT, experiences [Online] Available at: https://www.psychonaut.fr/Thread-5-MEO-MIPT-e-5MM-AMT?pid=410964#pid410964. Accessed 28 July 2023.
Psychonautwiki (2022) 5-MeO-DIPT [Online] Available at: https://psychonautwiki.org/wiki/5-MeO-DiPT Accessed on 14/12/23.
Psychonautwiki (2023a) 5-MeO-MiPT [Online] Available at: https://psychonautwiki.org/wiki/5-MeO-MiPT. Accessed 28 July 2023.
Psychonautwiki (2023b) DMT [Online] Available at: https://psychonautwiki.org/wiki/DMT Accessed on 14/12/2023.
Reagan-Shaw S, Nihal M, Ahmad N (2008) Dose translation from animal to human studies revisited. FASEB J 22(3):659–661. https://doi.org/10.1096/fj.07-9574LSF
Reckweg JT, Uthaug MV, Szabo A, Davis AK, Lancelotta R, Mason NL, Ramaekers JG (2022) The clinical pharmacology and potential therapeutic applications of 5-methoxy-N, N-dimethyltryptamine (5-MeO-DMT). J Neurochem 162(1):128–146. https://doi.org/10.1111/jnc.15587
Repke DB, Grotjahn DB, Shulgin AT (1985) Psychotomimetic N-methyl-N-isopropyltryptamines. Effects of variation of aromatic oxygen substituents. J Med Chem 28(7):892–896. https://doi.org/10.1021/jm00145a007
Rickli A, Moning OD, Hoener MC, Liechti ME (2016) Receptor interaction profiles of novel psychoactive tryptamines compared with classic hallucinogens. Eur Neuropsychopharmacol 26(8):1327–1337. https://doi.org/10.1016/j.euroneuro.2016.05.001
Salas-Wright CP, Cano M, Hodges J, Oh S, Hai AH, Vaughn MG (2021) Driving while under the influence of hallucinogens: prevalence, correlates, and risk profiles. Drug Alcohol Depend 228:109055. https://doi.org/10.1016/j.drugalcdep.2021.109055
Schlag AK, Aday J, Salam I, Neill JC, Nutt DJ (2022) Adverse effects of psychedelics: from anecdotes and misinformation to systematic science. J Psychopharmacol 36(3):258–272. https://doi.org/10.1177/02698811211069100
Serpelloni G, Macchia T, Locatelli C, Rimondo C, Seri C (2013) New drugs. Nuove Sostanze Psicoattive (NSP). Schede tecniche relative alle molecole registrate dal Sistema Nazionale di Allerta Precoce. [Online] Available at: http://allerta.dronetplus.eu/modules/pubbdetails/690/Nuove+Sostanze+Psicoattive+(NSP):+schede+tec.html. Accessed 28 July 2023.
Shephard RA, Broadhurst PL (1983) Hyponeophagia in the Roman rat strains: effects of 5-methoxy-N, N-dimethyltryptamine, diazepam, methysergide and the stereoisomers of propranolol. Eur J Pharmacol 95(3–4):177–184. https://doi.org/10.1016/0014-2999(83)90632-5
Shimizu E, Watanabe H, Kojima T, Hagiwara H, Fujisaki M, Miyatake R, Hashimoto K, Iyo M (2007) Combined intoxication with methylone and 5-MeO-MIPT. Prog Neuropsychopharmacol Biol Psychiatry 31(1):288–291. https://doi.org/10.1016/j.pnpbp.2006.06.012
Shulgin A, Shulgin A (1997) TiHKAL: the continuation. Transform Press, United States
Smolinske SC, Rastogi R, Schenkel S (2005) Foxy methoxy: a new drug of abuse. J Med Toxicol 1(1):22–25
Strassman RJ, Qualls CR (1994) Dose-response study of N, N-dimethyltryptamine in humans. I. Neuroendocrine, autonomic, and cardiovascular effects. Arch Gen Psychiatry 51(2):85–97. https://doi.org/10.1001/archpsyc.1994.03950020009001
Taylor C, Crosby I, Yip V, Maguire P, Pirmohamed M, Turner RM (2020) A review of the important role of CYP2D6 in pharmacogenomics. Genes 11(11):1295. https://doi.org/10.3390/genes11111295
Tirri M, Ponzoni L, Bilel S, Arfè R, Braida D, Sala M (2020) Acute DOB and PMA administration impairs motor and sensorimotor responses in mice and causes hallucinogenic effects in adult zebrafish. Brain Sci 10:E586. https://doi.org/10.3390/brainsci10090586
Tirri M, Bilel S, Arfè R, Corli G, Marchetti B, Bernardi T, Boccuto F, Serpelloni G, Botrè F, De-Giorgio F, Golembiowska K, Marti M (2022) Effect of -NBOMe compounds on sensorimotor, motor, and prepulse inhibition responses in mice in comparison with the 2C analogs and lysergic acid diethylamide: from preclinical evidence to forensic implication in driving under the influence of drugs. Front Psychiatry 13:875722. https://doi.org/10.3389/fpsyt.2022.875722
Tittarelli R, Mannocchi G, Pantano F, Romolo FS (2015) Recreational use, analysis and toxicity of tryptamines. Curr Neuropharmacol 13(1):26–46. https://doi.org/10.2174/1570159X13666141210222409
Tylš F, Páleníček T, Kadeřábek L, Lipski M, Kubešová A, Horáček J (2016) Sex differences and serotonergic mechanisms in the behavioural effects of psilocin. Behav Pharmacol 27(4):309–320. https://doi.org/10.1097/FBP.0000000000000198
UNODC (2023) Early warning advisory on NPS - summary dashboard. [ONLINE] Available at: https://www.unodc.org/LSS/Page/NPS/DataVisualisations. Accessed 28 July 2023
Varma MV, Steyn SJ, Allerton C, El-Kattan AF (2015) Predicting clearance mechanism in drug discovery: extended clearance classification system (ECCS). Pharmaceut Res 32(12):3785–3802. https://doi.org/10.1007/s11095-015-1749-4
Vigolo A, Ossato A, Trapella C, Vincenzi F, Rimondo C, Seri C, Varani K, Serpelloni G, Marti M (2015) Novel halogenated derivates of JWH-018: behavioral and binding studies in mice. Neuropharmacology 95:68–82. https://doi.org/10.1016/j.neuropharm.2015.02.008
Villalón CM, Centurión D (2007) Cardiovascular responses produced by 5-hydroxytriptamine: a pharmacological update on the receptors/mechanisms involved and therapeutic implications. Naunyn Schmiedebergs Arch Pharmacol 376(1–2):45–63. https://doi.org/10.1007/s00210-007-0179-1
Voronova IP (2021) 5-HT receptors and temperature homeostasis. Biomolecules 11(12):1914. https://doi.org/10.3390/biom11121914
Wang RZ, Vashistha V, Kaur S, Houchens NW (2016) Serotonin syndrome: preventing, recognizing, and treating it. Cleve Clin J Med 83(11):810–817. https://doi.org/10.3949/ccjm.83a.15129
Watakabe A, Komatsu Y, Sadakane O, Shimegi S, Takahata T, Higo N, Tochitani S, Hashikawa T, Naito T, Osaki H, Sakamoto H, Okamoto M, Ishikawa A, Hara S, Akasaki T, Sato H, Yamamori T (2009) Enriched expression of serotonin 1B and 2A receptor genes in macaque visual cortex and their bidirectional modulatory effects on neuronal responses. Cereb Cortex 19(8):1915–1928. https://doi.org/10.1093/cercor/bhn219
Watts SW, Morrison SF, Davis RP, Barman SM (2012) Serotonin and blood pressure regulation. Pharmacol Rev 64(2):359–388. https://doi.org/10.1124/pr.111.004697
Williams MT, Herring NR, Schaefer TL, Skelton MR, Campbell NG, Lipton JW, McCrea AE, Vorhees CV (2007) Alterations in body temperature, corticosterone, and behavior following the administration of 5-methoxy-diisopropyltryptamine (‘foxy’) to adult rats: a new drug of abuse. Neuropsychopharmacology 32(6):1404–1420. https://doi.org/10.1038/sj.npp.1301232
Zamberlan F, Sanz C, Martínez Vivot R, Pallavicini C, Erowid F, Erowid E, Tagliazucchi E (2018) The varieties of the psychedelic experience: a preliminary study of the association between the reported subjective effects and the binding affinity profiles of substituted phenethylamines and tryptamines. Front Integr Neurosci 12:54. https://doi.org/10.3389/fnint.2018.00054
Zhang F, Xue J, Shao J, Jia L (2012) Compilation of 222 drugs’ plasma protein binding data and guidance for study designs. Drug Discov Today 17(9–10):475–485. https://doi.org/10.1016/j.drudis.2011.12.018
Funding
Open access funding provided by Università degli Studi di Ferrara within the CRUI-CARE Agreement. This research has been funded by the Anti-Drug Policies Department, Presidency of the Council of Ministers, Italy (project: “Effects of NPS: development of a multicentre research for the information enhancement of the Early Warning System” to M. Marti), and local funds from the University of Ferrara (FAR 2021, FAR 2022 to M. Marti; project: “Implementation of the identification and study of the effects of NPS: Development of a multicentric research to strengthen the database of the National Monitoring Centre for Drug Addiction and the Early Warning System” to M. Marti).
Author information
Authors and Affiliations
Contributions
Authors declare that MB, SB, and MM designed the studies, with refinements contributed by MT, GC, AMA, and OR. MT, MB, GC, and SB performed the research, conducted initial data analysis, created figures, and conducted statistical analysis of data. MM, SB, MB, MT, and ER wrote the major drafts of the paper. AG, FDR, AMA, OR, CAL, ER, and GZ edited sections on the manuscript. All authors have approved the submitted version of the manuscript.
Corresponding author
Ethics declarations
Ethical statements
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. All procedures performed in the studies involving animals were in accordance with the ethical standards of the institution or practice at which the studies were conducted. Project activated in collaboration with the Presidency of the Council of Ministers-DPA Anti-Drug Policies (Italy).
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Bassi, M., Bilel, S., Tirri, M. et al. Pharmaco-toxicological effects of the novel tryptamine hallucinogen 5-MeO-MiPT on motor, sensorimotor, physiological, and cardiorespiratory parameters in mice—from a human poisoning case to the preclinical evidence. Psychopharmacology 241, 489–511 (2024). https://doi.org/10.1007/s00213-024-06526-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-024-06526-8