Abstract
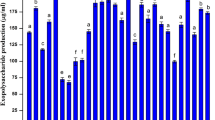
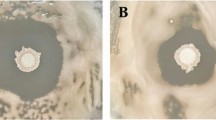
Soil pH conditions have important consequences for microbial community structure, their dynamics, ecosystem processes, and interactions with plants. Low soil pH affects the growth and functional activity of bacterial biocontrol agents which may experience a paradigm shift in their ability to act antagonistically against fungal phytopathogens. In this study, the antifungal activity of an acid-tolerant soil bacterium Bacillus amyloliquefaciens MBNC was evaluated under low pH and compared to its activity in neutral pH conditions. Bacterial supernatant from 3-day-old culture (approximately 11.2 × 108 cells/mL) grown in low pH conditions was found more effective against fungal pathogens. B. amyloliquefaciens MBNC harboured genes involved in the synthesis of secondary metabolites of which surfactin homologues, with varying chain length (C11–C15), were identified through High-Resolution Mass Spectroscopy. The pH of the medium influenced the production of these metabolites. Surfactin C15 was exclusive to the extract of pH 4.5; production of iturinA and surfactin C11 was detected only in pH 7.0, while surfactin C12, C13 and C14 were detected in extracts of both the pH conditions. The secretion of phytohormones viz. indole acetic acid and gibberellic acid by B. amyloliquefaciens MBNC was detected in higher amounts in neutral condition compared to acidic condition. Although, secretion of metabolites and phytohormones in B. amyloliquefaciens MBNC was influenced by the pH condition of the medium, the isolate retained its antagonistic efficiency against several fungal phyto-pathogens under acidic condition.





Similar content being viewed by others
Availability of data and material
The ITS and 16S rRNA gene information is publicly available in the NCBI database.
References
Aleti G, Sessitsch A, Brader G (2015) Genome mining: prediction of lipopeptides and polyketides from Bacillus and related Firmicutes. Comput Struct Biotechnol J 13:192–203. https://doi.org/10.1016/j.csbj.2015.03.003
Arrebola E, Jacobs R, Korsten L (2010) Iturin A is the principal inhibitor in the biocontrol activity of Bacillus amyloliquefaciens PPCB004 against postharvest fungal pathogens. J Appl Microbiol 108:386–395. https://doi.org/10.1111/j.1365-2672.2009.04438.x
Athukorala SNP, Fernando WGD, Rashid KY (2009) Identification of antifungal antibiotics of Bacillus species isolated from different microhabitats using polymerase chain reaction and MALDI-TOF mass spectrometry. Can J Microbiol 55:1021–1032. https://doi.org/10.1139/W09-067
Bartal A, Vigneshwari A, Bóka B et al (2018) Effects of different cultivation parameters on the production of surfactin variants by a Bacillus subtilis strain. Molecules 23:2675. https://doi.org/10.3390/molecules23102675
Binagwa PH, Bonsi CK, Msolla SN (2016) Evaluation of common bean (Phaseolus vulgaris) genotypes for resistance to root rot disease caused by Pythium aphanidermatum and Pythium splendens under screen house conditions. J Nat Sci Res 6:36–43
Burianek LL, Yousef AE (2000) Solvent extraction of bacteriocins from liquid cultures. Lett Appl Microbiol 31:193–197. https://doi.org/10.1046/j.1365-2672.2000.00802.x
Carrillo C, Teruel JA, Aranda FJ, Ortiz A (2003) Molecular mechanism of membrane permeabilization by the peptide antibiotic surfactin. Biochem Biophys Acta 1611(1–2):91–97. https://doi.org/10.1016/s0005-2736(03)00029-4
Chen K, Tian Z, Luo Y et al (2018) Antagonistic activity and the mechanism of Bacillus amyloliquefaciens DH-4 against citrus green mold. Phytopathology 108:1253–1262. https://doi.org/10.1094/PHYTO-01-17-0032-R
Chowdhury N, Goswami G, Boro RC, Barooah M (2021) A pH-dependent gene expression enables Bacillus amyloliquefaciens MBNC to adapt to acid stress. Curr Microbiol 78:3104–3114. https://doi.org/10.1007/s00284-021-02573-y
Deka P, Goswami G, Das P et al (2019) Bacterial exopolysaccharide promotes acid tolerance in Bacillus amyloliquefaciens and improves soil aggregation. Mol Biol Rep 46:1079–1091. https://doi.org/10.1007/s11033-018-4566-0
Essghaier B, Fardeau ML, Cayol JL et al (2009) Biological control of grey mould in strawberry fruits by halophilic bacteria. J Appl Microbiol 106:833–846. https://doi.org/10.1111/j.1365-2672.2008.04053.x
Goswami G, Deka P, Das P et al (2017) Diversity and functional properties of acid-tolerant bacteria isolated from tea plantation soil of Assam. 3 Biotech. https://doi.org/10.1007/s13205-017-0864-9
Goswami G, Panda D, Samanta R et al (2018) Bacillus megaterium adapts to acid stress condition through a network of genes: insight from a genome-wide transcriptome analysis. Sci Rep 8:16105. https://doi.org/10.1038/s41598-018-34221-0
Hazarika DJ, Goswami G, Gautom T et al (2019) Lipopeptide mediated biocontrol activity of endophytic Bacillus subtilis against fungal phytopathogens. BMC Microbiol 19:71. https://doi.org/10.1186/s12866-019-1440-8
Hendrix FF, Campbell WA (1973) Pythiums as plant pathogens. Annu Rev Phytopathol 11:77–98. https://doi.org/10.1146/annurev.py.11.090173.000453
Hmidet N, Ben Ayed H, Jacques P, Nasri M (2017) Enhancement of surfactin and fengycin production by Bacillus mojavensis A21: application for diesel biodegradation. Biomed Res Int 2017:1–8. https://doi.org/10.1155/2017/5893123
Horak I, Engelbrecht G, van Rensburg PJ, Claassens S (2019) Microbial metabolomics: essential definitions and the importance of cultivation conditions for utilizing Bacillus species as bionematicides. J Appl Microbiol 127(2):326–343. https://doi.org/10.1111/jam.14218
Horikoshi K (2008) Past, present and future of extremophiles. Extremophiles 12:1–2. https://doi.org/10.1007/s00792-007-0127-5
Jaivel N, Ramasamy R, Sivakumar U, Marimuthu P (2019) Antimicrobial activity and spectroscopic characterization of surfactin class of lipopeptides from Bacillus amyloliquefaciens SR1. Microb Pathog. https://doi.org/10.1016/j.micpath.2019.01.037
Ji SH, Paul NC, Deng JX et al (2013) Biocontrol activity of Bacillus amyloliquefaciens CNU114001 against fungal plant diseases. Mycobiology 41:234–242. https://doi.org/10.5941/MYCO.2013.41.4.234
Kadaikunnan S, Rejiniemon T, Khaled JM et al (2015) In-vitro antibacterial, antifungal, antioxidant and functional properties of Bacillus amyloliquefaciens. Ann Clin Microbiol Antimicrob 14:9. https://doi.org/10.1186/s12941-015-0069-1
Kumar P, Dubey RC, Maheshwari DK (2012) Bacillus strains isolated from rhizosphere showed plant growth promoting and antagonistic activity against phytopathogens. Microbiol Res 167:493–499. https://doi.org/10.1016/J.MICRES.2012.05.002
Lee T, Park D, Kim K et al (2017) Characterization of Bacillus amyloliquefaciens DA12 showing potent antifungal activity against mycotoxigenic Fusarium species. Plant Pathol J 33:499–507. https://doi.org/10.5423/PPJ.FT.06.2017.0126
Leyva-Madrigal KY, Sandoval-Castro E, Calderón-Vázquez CL et al (2017) Pathogenic and genetic variability of Fusarium verticillioides from maize in northern Mexico. Can J Plant Pathol 39:486–496. https://doi.org/10.1080/07060661.2017.1378726
Li S, Liu Y, Wang J et al (2017) Soil Acidification aggravates the occurrence of bacterial wilt in South China. Front Microbiol. https://doi.org/10.3389/fmicb.2017.00703
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 25:402–408. https://doi.org/10.1006/meth.2001.1262
Mahadevakumar S, Tejaswini GS, Janardhana GR, Yadav V (2015) First report of Sclerotium rolfsii causing southern blight and leaf spot on common bean (Phaseolus vulgaris) in India. Plant Dis 99:1280–1280. https://doi.org/10.1094/PDIS-01-15-0125-PDN
Marajan C, Alias S, Ramasamy K, Abdul-Talib S (2018) The effect of incubation time, temperature and pH variations on the surface tension of biosurfactant produced by Bacillus spp. AIP conference. AIP, p 020047
Marcenaro D, Valkonen JPT (2016) Seedborne pathogenic fungi in common bean (Phaseolus vulgaris cv. INTA Rojo) in Nicaragua. PLoS ONE 11:e0168662. https://doi.org/10.1371/journal.pone.0168662
Mariam SH, Zegeye N, Tariku T et al (2014) Potential of cell-free supernatants from cultures of selected lactic acid bacteria and yeast obtained from local fermented foods as inhibitors of Listeria monocytogenes, Salmonella spp. and Staphylococcus aureus. BMC Res Notes 7:606. https://doi.org/10.1186/1756-0500-7-606
Mizumoto S, Shoda M (2007) Medium optimization of antifungal lipopeptide, iturin A, production by Bacillus subtilis in solid-state fermentation by response surface methodology. Appl Microbiol Biotechnol 76:101–108. https://doi.org/10.1007/s00253-007-0994-9
Monteiro FP, de Medeiros F, Henrique Vasconcelos Ongena M, Franzil L, de Souza P, Estevão de Souza JT (2016) Effect of temperature, pH and substrate composition on production of lipopeptides by Bacillus amyloliquefaciens 629. African J Microbiol Res 10:506–1512
Niehaus F, Bertoldo C, Kähler M, Antranikian G (1999) Extremophiles as a source of novel enzymes for industrial application. Appl Microbiol Biotechnol 51:711–729. https://doi.org/10.1007/s002530051456
Nunes CA (2012) Biological control of postharvest diseases of fruit. Eur J Plant Pathol 133:181–196. https://doi.org/10.1007/s10658-011-9919-7
Patel PS, Huang S, Fisher S et al (1995) Bacillaene, a novel inhibitor of procaryotic protein synthesis produced by Bacillus subtilis: production, taxonomy, isolation, physico-chemical characterization and biological activity. J Antibiot (tokyo) 48:997–1003
Patten CL, Glick BR (2002) Role of Pseudomonas putida indoleacetic acid in development of the host plant root system. Appl Environ Microbiol 68:3795–3801. https://doi.org/10.1128/AEM.68.8.3795-3801.2002
Rousk J, Brookes PC, Bååth E (2009) Contrasting soil pH effects on fungal and bacterial growth suggest functional redundancy in carbon mineralization. Appl Environ Microbiol 75:1589–1596. https://doi.org/10.1128/AEM.02775-08
Saraf M, Pandya U, Thakkar A (2014) Role of allelochemicals in plant growth promoting rhizobacteria for biocontrol of phytopathogens. Microbiol Res 169:18–29. https://doi.org/10.1016/j.micres.2013.08.009
Sarwar A, Nadeem M, Imran M et al (2018) Biocontrol activity of surfactin A purified from Bacillus NH-100 and NH-217 against rice bakanae disease. Microbiol Res 209:1–13. https://doi.org/10.1016/j.micres.2018.01.006
Schneider K, Chen X-H, Vater J et al (2007) Macrolactin is the polyketide biosynthesis product of the pks2 cluster of Bacillus amyloliquefaciens FZB42. J Nat Prod 70:1417–1423. https://doi.org/10.1021/np070070k
Velho RV, Caldas DGG, Medina LFC et al (2011) Real-time PCR investigation on the expression of sboA and ituD genes in Bacillus spp. Lett Appl Microbiol 52:660–666. https://doi.org/10.1111/j.1472-765X.2011.03060.x
Vikram A, Hamzehzarghani H, Alagawadi AR et al (2007) Production of plant growth promoting substances by phosphate solubilizing bacteria isolated from vertisols. J Plant Sci 2:326–333. https://doi.org/10.3923/jps.2007.326.333
Wang H, Fewer DP, Holm L et al (2014) Atlas of nonribosomal peptide and polyketide biosynthetic pathways reveals common occurrence of nonmodular enzymes. Proc Natl Acad Sci 111:9259–9264. https://doi.org/10.1073/pnas.1401734111
Xiong H, Li Y, Cai Y, Cao Yu, Wang Y (2015) Isolation of Bacillus amyloliquefaciens JK6 and identification of its lipopeptides surfactin for suppressing tomato bacterial wilt. RSC Adv 5(100):82042–82049. https://doi.org/10.1039/C5RA13142A
Yu S (2009) Biological control of postharvest green and blue mold rots of citrus fruits by Bacillus amyloliquefaciens BCL251. Chungnam National University, Daejeon
Yu GY, Sinclair JB (1996) Evaluation of Bacillus amyloliquefaciens B94 for control of Rhizoctonia seedling disease on soybeans. Phytopathology 86:S54
Yu G, Sinclair J, Hartman G, Bertagnolli B (2002) Production of iturin A by Bacillus amyloliquefaciens suppressing Rhizoctonia solani. Soil Biol Biochem 34:955–963. https://doi.org/10.1016/S0038-0717(02)00027-5
Zhang W, Jiang F, Ou J (2011) Global pesticide consumption and pollution: with China as a focus. Proc Int Acad Ecol Environ Sci 1:125–144
Zhang S, Jiang W, Li J et al (2016) Whole genome shotgun sequence of Bacillus amyloliquefaciens TF28, a biocontrol entophytic bacterium. Stand Genom Sci. https://doi.org/10.1186/s40793-016-0182-6
Acknowledgements
The authors wish to acknowledge the Department of Biotechnology (DBT), Government of India, for financial support for the Project, “Screening of soil microbes for acid tolerance gene” under DBT-North East Centre for Agricultural Biotechnology (DBT-NECAB), Assam Agricultural University, Jorhat, India. The authors are thankful to Dr. M. K. Modi, Head, Department of Agricultural Biotechnology and Dr. B. K. Sarmah, Director DBT-AAU Centre, AAU, Jorhat for providing the requisite facilities and recommendations. The authors gratefully acknowledge the Department of Plant Pathology, Assam Agricultural University, Jorhat, India for providing the fungal isolates.
Funding
This study was supported by funds received from the Department of Biotechnology (DBT), Government of India for the project “Screening of soil microbes for acid tolerance gene” under DBT-NECAB, Assam Agricultural University (AAU), Jorhat, India.
Author information
Authors and Affiliations
Contributions
Conceptualization and Methodology: MB, GG, NC; Formal analysis and investigation: NC, GG, DJH, US, SB; Data Curation: NC, GG, DJH; Writing—original draft preparation: NC; Writing—review and editing: NC, GG, DJH, RCB, MB; Resources: MB, RCB; Supervision: MB.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
All the authors have given consent for publication.
Additional information
Communicated by Erko Stackebrandt.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Chowdhury, N., Hazarika, D.J., Goswami, G. et al. Acid tolerant bacterium Bacillus amyloliquefaciens MBNC retains biocontrol efficiency against fungal phytopathogens in low pH. Arch Microbiol 204, 124 (2022). https://doi.org/10.1007/s00203-021-02741-5
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00203-021-02741-5