Abstract
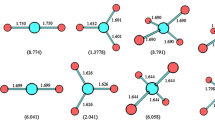
Ab initio calculations have been performed on complexes of CFCl3 with SO2. Ten complexes were found stable, the interaction energies that reflect their stability were corrected by the basis set superposition error and the correction of zero-point energy. The natures of these interactions were investigated by the analysis of natural bond orbital and the atoms in molecules. The results from theoretical calculation indicated that there were the interactions of Cl···O, F···O, Cl···S and F···S between CFCl3 and SO2, furthermore, the non-covalent bonds of Cl···O and Cl···S were the major interaction forces, which provided some data and information for studying the environment problem such as greenhouse effect relevant to CFCl3 and SO2.

Similar content being viewed by others
References
Bader RFW (1990) Atom in molecules: a quantum theory, internationalseries of monographs in chemistry. Oxford University Press, Oxford
Bader RFW (1998) A bond path: a universal indicator of bonded interactions. J Phys Chem A 102:7314–7323
Biegler-Konig F, Bader RF (2002) AIM 2000, version 2. University of Applied Science, Bielefeld
Bondi A (1964) Van der Waals volumes and radii. J Phys Chem 68:441–451
Boys SF, Bernardi F (1970) The calculations of small molecular interaction by the difference of separate total energies, some procedures with reduced error. Mol Phys 19:553–566
Davies MJ (2005) The oxidative environment and protein damage. Biochim Biophys Acta 1703:93–109
Esrafili MD, Behzadi H, Hadipour NL (2008) Theoretical study of N-HO hydrogen bonding properties and cooperativity effects in linear acetamide clusters. Theor Chem Account 121:135–146
Foster JP, Weinhold F (1980) Natural hybrid orbitals. J Am Chem Soc 102:7211–7218
Frisch MJ (2004) Gaussian 03, revision E.01. Gaussian, Inc, Wallingford
Frisch MJ, Trucks GW, Schlegel HB, Pople JA (2003) Gaussian03.Gaussian. Gaussian, Inc, Pittsburgh
Meng ZQ, Liu YX (2007) Cell morphological ultrastructural changes in various organs from mice exposed by inhalation to sulfur dioxide. Inhal Toxicol 19:543–551
Mohajeri A, Karimi E (2006) AIM and NBO analyses of cation-.π. interaction. Theochem 774:71–76
Reed AE, Curtiss LA, Weinhold F (1988) Intermolecular interactions from a natural bond orbital, donor-acceptor viewpoint. Chem Rev 88:899–926
Ren XH, Wang HJ, Shan YY, Dong WB (2008) A theoretical study of the interactions between N, N-dimethylformamide and xylenes. Struct Chem 19:233–238
Riley KE, Hobza P (2008) Investigations into the nature of halogen bonding including symmetry adapted perturbation theory analyses. J Chem Theory Comput 4:232–242
Rowland FS, Molina M (1974) Stratospheric sink for chlorofluoromethanes. Chlorine atom-catalyzed destruction of ozone. Nature 249:810–814
Rowland FS, Molina M (1975) Chlorofluoromethanes in the environment. Rev Geophys Space Phys 13:1–5
Saltzman ES, Cooper DJ (1989) Dimethyl sulfide and hydrogen sulfide in marine air. Am Chem Soc 393:31–43
Shan YY, Ren XH, Wang HJ, Dong WB (2007) A theoretical study of the interactions between N, N-dimethylformamide and aromatic hydrocarbons. Struct Chem 18:709–716
Xu Y, Masui T (2008) Application of AIM/CGE model to China. Eur J Oper Res 2217:00704–00712
Acknowledgments
Thanks the calculations in this paper at the GHPCC, Jiangnan University.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Diao, K.S., Wang, F. & Wang, H.J. Ab Initio Theoretical Study of the Interactions Between CFCl3 and SO2 . Bull Environ Contam Toxicol 84, 170–174 (2010). https://doi.org/10.1007/s00128-009-9912-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00128-009-9912-4