Abstract
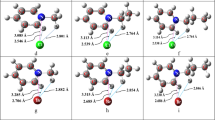
A systematic study on CoO n species for n = 1–5 in neutral and (mono)anionic forms has been performed using hybrid exchange-correlation functional in density functional theory. The ground-state structures are obtained, and their stabilities are analyzed against dissociation to O atom and O2 molecule. We find that Co can bind stably up to four O atoms, expanding its oxidation state to +8. The adiabatic electron affinities of CoO n and vertical detachment energies of \( {\text{CoO}}_{n}^{ - } \) reveal their superhalogen behavior for n ≥ 3 due to extra electron delocalization over O atoms. CoO n superhalogens can be used to form a new class of complex compounds. This idea is demonstrated by formation of stable LiCoO4 complex by interacting Li atom with CoO4 superhalogen.




Similar content being viewed by others
References
Hotop H, Lineberger WC (1985) J Phys Chem Ref Data 14:731
Gutsev GL, Boldyrev AI (1981) Chem Phys 56:277
Wang X-B, Ding C-F, Wang L-S, Boldyrev AI, Simons J (1999) J Chem Phys 110:4763
Bartlett N, Lohmann DH (1962) Proc Chem Soc (London) 3:115
Bartlett N (1962) Proc Chem Soc (London) 6:218
Gutsev GL, Boldyrev AI (1982) Chem Phys Lett 92:262
Gutsev GL (1991) Chem Phys Lett 184:305
Anusiewicz I, Skurski P (2002) Chem Phys Lett 358:426
Anusiewicz I, Sobczyk M, Dabkowska I, Skurski P (2003) Chem Phys 291:171
Graudejus O, Elder SH, Lucier GM, Shen C, Bartlett N (1999) Inorg Chem 38:2503
Lucier GM, Shen C, Elder SH, Bartlett N (1998) Inorg Chem 37:3829
Koirala P, Willis M, Kiran B, Kandalam AK, Jena P (2010) J Phys Chem C 114:16018
Srivastava AK, Misra N (2014) J Fluorine Chem 158:65
Ko YJ, Wang H, Pradhan K, Koirala P, Kandalam AK, Bowen KH, Jena P (2011) J Chem Phys 135:244312
Wu MM, Wang H, Ko YJ, Wang Q, Sun Q, Kiran B, Kandalam AK, Bowen KH, Jena P (2011) Angew Chem Int Ed 50:2568
Srivastava AK, Misra N (2014) Int J Quantum Chem 114:1513
Srivastava AK, Misra N (2014) Int J Quantum Chem 114:328
Srivastava AK, Misra N (2014) Int J Quantum Chem 114:521
Srivastava AK, Misra N (2014) Mol Phys 112:1963
Srivastava AK, Misra N (2014) Eur Phys J D 68:309
Gutsev GL, Rao BK, Jena P, Wang XB, Wang LS (1991) Chem Phys Lett 312:598
Gutsev GL, Khanna SN, Rao BK, Jena P (1999) Phys Rev A 59:3681
Srivastava AK, Misra N (2014) Mol Phys 112:1639
Becke AD (1993) J Chem Phys 98:5648
Lee C, Yang W, Parr RG (1988) Phys Rev B 37:785
Neese F (2009) Coord Chem Rev 253:526
Riley KE, Opt Holt BT, Merz KM Jr (2007) J Chem Theory Comput 3:407
Srivastava AK, Misra N (2014) Chem Phys Lett 612:302
M.J. Frisch, G.W. Trucks, H.B. Schlegel, G.E. Scuseria, M.A. Robb, J.R. Cheeseman, G. Scalmani, V. Barone, B. Mennucci, G.A. Petersson, H. Nakatsuji, M. Caricato, X. Li, H.P. Hratchian, A.F. Izmaylov, J. Bloino, G. Zheng, J.L. Sonnenberg, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, J.A. Montgomery Jr., J.E. Peralta, F. Ogliaro, M. Bearpark, J.J. Heyd, E. Brothers, K.N. Kudin, V.N. Staroverov, T. Keith, R. Kobayashi, J. Normand, K. Raghavachari, A. Rendell, J.C. Burant, S.S. Iyengar, J. Tomasi, M. Cossi, N. Rega, J.M. Millam, M. Klene, J.E. Knox, J.B. Cross, V. Bakken, C. Adamo, J. Jaramillo, R. Gomperts, R.E. Stratmann, O. Yazyev, A.J. Austin, R. Cammi, C. Pomelli, J.W. Ochterski, R.L. Martin, K. Morokuma, V.G. Zakrzewski, G.A. Voth, P. Salvador, J.J. Dannenberg, S. Dapprich, A.D. Daniels, O. Farkas, J.B. Foresman, J.V. Ortiz, J. Cioslowski, D.J. Fox, Gaussian 09, Revision B.01, Gaussian Inc., Wallingford, CT (2010)
Glendening ED, Badenhoop JK, Reed AE, Carpenter JE, Weinhold F (1996) NBO. 3.1 Program. Theoretical Chemistry Institute, University of Wisconsin, Madison
Reed AE, Weinstock RB, Weinhold F (1985) J Chem Phys 83:735
Srivastava AK, Misra N (2014) Comput Theor Chem 1047:1
Acknowledgments
A. K. Srivastava acknowledges Council of Scientific and Industrial Research (CSIR), New Delhi, India, for a research fellowship [Grant No. 09/107(0359)/2012-EMR-I].
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The manuscript has been prepared as per ethical guidelines. The authors declare no conflict of interests in the publication of this work.
Rights and permissions
About this article
Cite this article
Srivastava, A.K., Misra, N. Superhalogen properties of CoO n (n ≥ 3) species revealed by density functional theory. Theor Chem Acc 134, 93 (2015). https://doi.org/10.1007/s00214-015-1696-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00214-015-1696-5