Abstract
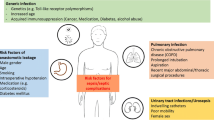
Ventriculostomy-associated infections, or ventriculitis, in critically ill patients are associated with considerable morbidity. Efficacious antibiotic dosing for the treatment of these infections may be complicated by altered antibiotic concentrations in the cerebrospinal fluid due to variable meningeal inflammation and antibiotic properties. Therefore, doses used to treat infections with a higher degree of meningeal inflammation (such as meningitis) may often fail to achieve equivalent exposures in patients with ventriculostomy-associated infections such as ventriculitis. This paper aims to review the disease burden, infection rates, and common pathogens associated with ventriculostomy-associated infections. This review also seeks to describe the disease- and drug-related factors that influence antibiotic distribution into cerebrospinal fluid and provide a critical appraisal of current dosing of antibiotics commonly used to treat these types of infections. A Medline search of relevant articles was conducted and used to support a review of cerebrospinal fluid penetration of vancomycin, including critical appraisal of the recent paper by Beach et al. recently published in this journal. We found that in the intensive care unit, ventriculostomy-associated infections are the most common and serious complication of external ventricular drain insertion and often result in prolonged patient stay and increased healthcare costs. Reported infection rates are extremely variable (between 0 and 45%), hindered by the inherent diagnostic difficulty. Both Gram-positive and Gram-negative organisms are associated with such infections and the rise of multi-drug-resistant pathogens means that effective treatment is an ongoing challenge. Disease factors that may need to be considered are reduced meningeal inflammation and the presence of critical illness; drug factors include physiochemical properties, degree of plasma–protein binding, and affinity to active transporter proteins present in the blood–cerebrospinal fluid barrier. The relationship between cerebrospinal fluid antibiotic exposures in the setting of ventriculostomy-associated infection and clinical response has not been fully elucidated for many of the antibiotics commonly used in its treatment. More thorough and clinically relevant investigations are needed to better define blood pharmacokinetic/pharmacodynamics targets and optimal therapeutic exposures for treatment of ventriculostomy-associated infections. It is hoped that this future research will be able to provide clearer recommendations for clinicians frequently faced with dosing-related dilemmas when treating patients with these challenging infections.


Similar content being viewed by others
References
Conen A, Fux CA, Vajkoczy P, Trampuz A. Management of infections associated with neurosurgical implanted devices. Expert Rev Anti Infect Ther. 2017;15(3):241–55.
Srihawan C, Castelblanco RL, Salazar L, Wootton SH, Aguilera E, Ostrosky-Zeichner L, et al. Clinical characteristics and predictors of adverse outcome in adult and pediatric patients with healthcare-associated ventriculitis and meningitis. Open Forum Infect Dis. 2016;3(2):ofw077.
Aarts MA, Brun-Buisson C, Cook DJ, Kumar A, Opal S, Rocker G, et al. Antibiotic management of suspected nosocomial ICU-acquired infection: does prolonged empiric therapy improve outcome? Intensive Care Med. 2007;33(8):1369–78.
Grgurich PE, Hudcova J, Lei Y, Sarwar A, Craven DE. Diagnosis of ventilator-associated pneumonia: controversies and working toward a gold standard. Curr Opin Infect Dis. 2013;26(2):140–50.
Fridkin S. Increasing prevalence of antimicrobial resistance in intensive care units. Crit Care Med. 2001;29(4):N64–8.
Seligman R, Ramos-Lima L, Oliveira V, Sanvicente C, Sartori J, Pacheco E. Risk factors for infection with multidrug-resistant bacteria in non-ventilated patients with hospital-acquired pneumonia. J Bras Pneumonol. 2013;39(3):339–48.
Nau R, Sorgel F, Eiffert H. Penetration of drugs through the blood–cerebrospinal fluid/blood–brain barrier for treatment of central nervous system infections. Clin Microbiol Rev. 2010;23(4):858–83.
Beach J, Perrot J, Turgeon R, Ensom MH. Penetration of vancomycin into the cerebrospinal fluid: a systematic review. Clin Pharmacokinet. 2017. doi:10.1007/s40262-017-0548-y.
Murphy RK, Liu B, Srinath A, Reynolds MR, Liu J, Craighead MC, et al. No additional protection against ventriculitis with prolonged systemic antibiotic prophylaxis for patients treated with antibiotic-coated external ventricular drains. J Neurosurg. 2015;122(5):1120–6.
Tunkel AR, Hasbun R, Bhimraj A, Byers K, Kaplan SL, Michael Scheld W, et al. 2017 Infectious Diseases Society of America’s clinical practice guidelines for healthcare-associated ventriculitis and meningitis. Clin Infect Dis. 2017. doi:10.1093/cid/ciw861.
Mounier R, Lobo D, Cook F, Fratani A, Attias A, Martin M, et al. Clinical, biological, and microbiological pattern associated with ventriculostomy-related infection: a retrospective longitudinal study. Acta Neurochir (Wien). 2015;157(12):2209–17 (discussion 17).
Citerio G, Signorini L, Bronco A, Vargiolu A, Rota M, Latronico N. External ventricular and lumbar drain device infections in ICU patients. Crit Care Med. 2015;43(8):1630–7.
Gutierrez-Gonzalez R, Boto GR, Perez-Zamarron A. Cerebrospinal fluid diversion devices and infection. A comprehensive review. Eur J Clin Microbiol Infect Dis. 2012;31(6):889–97.
Mayhall C, Archer N, Lamb V, Spadora A, Bagget J, Ward J, et al. Ventriculostomy-related infections: a prospective epidemiologic study. N Engl J Med. 1984;310(9):553–9.
Lozier A, Sciaca R, Romagnoli M, Connolly E. Ventriculostomy-related infections: a critical review of the literature. Neurosurgery. 2002;51(1):170–82.
Gozal YM, Farley CW, Hanseman DJ, Harwell D, Magner M, Andaluz N, et al. Ventriculostomy-associated infection: a new, standardized reporting definition and institutional experience. Neurocrit Care. 2014;21(1):147–51.
Bader M, Littlejohns L, Palmer S. Ventriculostomy and intracranial pressure monitoring: in search of a 0% infection rate. Heart Lung. 1995;24(2):166–72.
Ramanan M, Lipman J, Shorr A, Shankar A. A meta-analysis of ventriculostomy-associated cerebrospinal fluid infections. BMC Infect Dis. 2015;8(15):3.
Park P, Garton HJL, Kocan MJ, Thompson BG. Risk of infection with prolonged ventricular catheterization. Neurosurgery. 2004;55(3):594–601.
Hoogmoed J, van de Beek D, Coert BA, Horn J, Vandertop WP, Verbaan D. Clinical and laboratory characteristics for the diagnosis of bacterial ventriculitis after aneurysmal subarachnoid hemorrhage. Neurocrit Care. 2017;26(3):362–70.
World Health Organisation. Antimicrobial resistance: global report on surveillance. Geneva: World Health Organisation; 2014.
Wolburg H, Noell S, Mack A, Wolburg-Buchholz K, Fallier-Becker P. Brain endothelial cells and the glio-vascular complex. Cell Tissue Res. 2009;335(1):75–96.
Chodobski A, Zink BJ, Szmydynger-Chodobska J. Blood-brain barrier pathophysiology in traumatic brain injury. Transl Stroke Res. 2011;2(4):492–516.
Lutsar I, McCracken G, Friedland I. Antibiotic pharmacodynamics in cerebrospinal fluid. Clin Infect Dis. 1998;27(5):1117–27.
Poller B, Drewe J, Krahenbuhl S, Huwyler J, Gutmann H. Regulation of BCRP (ABCG2) and P-glycoprotein (ABCB1) by cytokines in a model of the human blood-brain barrier. Cell Mol Neurobiol. 2010;30(1):63–70.
Mouton J. General concepts of pharmacodynamics for anti-infective agents. In: Rotschafer J, Andes D, Rodvold K, editors. Antibiotic pharmacodynamics. New York: Springer; 2016.
Udy AA, Roberts JA, Lipman J. Implications of augmented renal clearance in critically ill patients. Nat Rev Nephrol. 2011;7(9):539–43.
Udy AA, Baptista JP, Lim NL, Joynt GM, Jarrett P, Wockner L, et al. Augmented renal clearance in the ICU: results of a multicenter observational study of renal function in critically ill patients with normal plasma creatinine concentrations. Crit Care Med. 2014;42(3):520–7.
Udy AA, Jarrett P, Lassig-Smith M, Stuart J, Starr T, Dunlop R, et al. Augmented renal clearance in traumatic brain injury: a single-center observational study of atrial natriuretic peptide, cardiac output, and creatinine clearance. J Neurotrauma. 2017;34(1):137–44.
Barling R, Selkon J. The penetration of antibiotics into cerebrospinal fluid and brain tissue. J Antimicrob Chemother. 1978;4:203–27.
Cotta MO, Roberts JA. Pharmacokinetic and pharmacodynamic principles of antimicrobials. In: Laundy M, Gilchrist M, Whitney L, editors. Antimicrobial stewardship. London: Oxford University Press; 2016.
Law V, Knox C, Djoumbou Y, Jewison T, Guo A, Liu Y, et al. DrugBank 4.0: shedding new light on drug metabolism. Nucleic Acids Res. 2014;42(1):D1091–7.
Tauber M, Doroshov C, Hackbarth C, Rusnak M, Drake T, Sande M. Antibacterial activity of beta-lactam antibiotics in experimental meningitis due to Streptococcus pneumoniae. J Infect Dis. 1984;149(4):568–74.
Norrby R. A review of the penetration of antibiotics into CSF and its clinical significance. Scand J Infect Dis. 1978;14(Suppl):296–309.
Scheld W, Brodeur J, Sande M, Alliegro G. Comparison of cefoperazone with penicillin, ampicillin, gentamicin, and chloramphenicol in the therapy of experimental meningitis. Antimicrob Agents Chemother. 1982;22:652–6.
Quagliarello V, Scheld W. Treatment of bacterial meningitis. N Engl J Med. 1997;336(10):708–16.
Iselius L. Protein binding of ampicillin. Path analysis of a twin material. Hereditas. 1978;89(1):132.
Clumeck N, Thys J, Vanhoof R, Vanderlinden M, Butzler J, Yourassowsky E. Amoxicillin entry into human cerebrospinal fluid: comparison with ampicillin. Antimicrob Agents Chemother. 1978;14(4):531–2.
Cherubin C, Eng R, Norrby R, Modai J, Humbert G, Overturf G. Penetration of newer cephalosporins into cerebrospinal fluid. Rev Infect Dis. 1989;11(4):526–48.
Nau R, Prange HW, Muth P, Mahr G, Menck S, Kolenda H, et al. Passage of cefotaxime and ceftriaxone into cerebrospinal fluid of patients with uninflamed meninges. Antimicrob Agents Chemother. 1993;37(7):1518–24.
Nakahara H, Oda T, Fukao E, Horiuchi I, Honma Y, Uchigata M. A case of meningococcal meningitis that was difficult to treat owing to concurrent ventriculitis. Rinsho Shinkeigaku. 2016;56(5):344–7.
Johan A, Hung L, Norlijah O. Salmonella enteritidis ventriculitis. Southeast Asian J Trop Med Public Health. 2013;44(3):456.
Chapman T, Perry C. Cefepime: a review of its use in the management of hospitalised patients with pneumonia. Am J Respir Med. 2003;2(1):75–107.
Lodise TP Jr, Rhoney DH, Tam VH, McKinnon PS, Drusano GL. Pharmacodynamic profiling of cefepime in plasma and cerebrospinal fluid of hospitalized patients with external ventriculostomies. Diagn Microbiol Infect Dis. 2006;54(3):223–30.
Lutsar I, Ahmed A, Friedland I, Trujillo M, Wubbel L, Olsen K, et al. Pharmacodynamics and bactericidal activity of ceftriaxone therapy in experimental cephalosporin-resistant pneumococcal meningitis. Antimicrob Agents Chemother. 1997;41(11):2414–7.
Andes D, Craig W. Pharmacokinetics and pharmacodynamics of antibiotics in meningitis. Infect Dis Clin North Am. 1999;13:595–618.
Krueger W, Schroeder T, Hutchinson M, Hoffman E, Dieterich H, Heininger A, et al. Pharmacokinetics of meropenem in critically ill patients with acute renal failure treated by continuous hemodiafiltration. Antimicrob Agents Chemother. 1998;42(9):2421–4.
Blassmann U, Roehr A, Frey O, Vetter-Kerkhoff C, Thon N, Hope W, et al. Cerebrospinal fluid penetration of meropenem in neurocritical care patients with proven or suspected ventriculitis: a prospective observational study. Crit Care. 2016;20:343.
Nau R, Lassek C, Kinzig-Schippers M, Thiel A, Prange HW, Fritz S. Disposition and elimination of meropenem in cerebrospinal fluid cultures of hydrocephalic patients with external ventriculostomy. Antimicrob Agents Chemother. 1998;42(8):2012–6.
Matsuda T, Ikawa K, Ikeda K, Morikawa N, Tsumura R, Shibukawa M, et al. LC method for the determination of meropenem in cerebrospinal fluid: application to therapeutic drug monitoring. Chromatographia. 2009;69(9–10):1031–4.
Chou Y, Yang Y, Chen J, Kuo C, Chen S. Quantification of meropenem in plasma and cerebrospinal fluid by micellar electrokinetic capillary chromatography and application in bacterial meningitis patients. J Chromatogr B Anal Technol Biomed Life. 2007;856(1):294–301.
Dagan R, Velghe L, Rodda J, Klugman K. Penetration of meropenem into the cerebrospinal fluid of patients with inflamed meninges. J Antimicrob Chemother. 1994;34(1):175–9.
Ulldemolins M, Roberts JA, Rello J, Paterson D, Lipman J. The effects of hypoalbuminaemia on optimizing antibacterial dosing in critically ill patients. Clin Pharmacokinet. 2011;50(2):99–110.
European Committee on Antimicrobial Susceptibility Testing. Breakpoint tables for interpretation of MICs and zone diameters. Växjö: EUCAST; 2017.
Ng K, Mabasa VH, Chow I, Ensom MH. Systematic review of efficacy, pharmacokinetics, and administration of intraventricular vancomycin in adults. Neurocrit Care. 2014;20(1):158–71.
Nau R, Scholz P, Sharifi S, Rohde S, Kolenda H, Prange HW. Netilmicin cerebrospinal fluid concentrations after an intravenous infusion of 400 mg in patients without meningeal inflammation. J Antimicrob Chemother. 1993;32:893–6.
Bailey D, Briggs J. Gentamicin and tobramycin binding to human serum in vitro. J Anal Toxicol. 2004;28:187–9.
Brunnemann S, Segal J. Amikacin serum protein binding in spinal cord injury. Life Sci. 1991;49(2):1–5.
LeBras M, Chow I, Mabasa VH, Ensom MHH. Systematic review of efficacy, pharmacokinetics, and administration of intraventricular aminoglycosides in adults. Neurocrit Care. 2016;25(3):492–507.
Kourtopoulos H, Holm S. Intraventricular treatment of Serratia marcescens meningitis with gentamicin: pharmacokinetic studies of gentamicin concentration in one case. Scand J Infect Dis. 1976;8:57–60.
Kaiser A, McGee Z. Aminoglycoside therapy of gram-negative bacillary meningitis. N Engl J Med. 1975;293:1215–20.
Barnes B, Wiederhold N, Micek S, Polish L, Ritchie D. Enterobacter cloacae ventriculitis successfully treated with cefepime and gentamicin: case report and review of the literature. Pharmacotherapy. 2003;23:537–42.
Wald S, McLaurin R. Cerebrospinal fluid antibiotic concentrations during treatment of shunt infections. J Neurosurg. 1980;52:41–6.
Mombelli G, Klastersky J, Coppens L, Daneau D, Nubourgh Y. Gram-negative bacillary meningitis in neurosurgical patients. J Neurosurg. 1983;59:634–41.
Ziai WC, Lewin JJ. Improving the role of intraventricular antimicrobial agents in the management of meningitis. Curr Opin Neurol. 2009;22(3):277–82.
Zhanel G. A review of new fluoroquinolones: focus on their use in respiratory tract infections. Treat Respir Med. 2006;5(6):437–65.
Peloquin CA, Hadad DJ, Molino LP, Palaci M, Boom WH, Dietze R, et al. Population pharmacokinetics of levofloxacin, gatifloxacin, and moxifloxacin in adults with pulmonary tuberculosis. Antimicrob Agents Chemother. 2008;52(3):852–7.
Gogos C, Maraziotis T, Papadakis N, Beerman D, Siamplis D, Bassaris H. Penetration of ciprofloxacin into human cerebrospinal fluid in patients with inflamed and non-inflamed meninges. Eur J Clin Microbiol Infect Dis. 1991;10(6):511–4.
Lipman J, Allworth A, Wallis S. Cerebrospinal fluid penetration of high doses of intravenous ciprofloxacin in meningitis. Clin Infect Dis. 2000;31(5):1131–3.
Pea F, Pavan F, Nascimben E, Benetton C, Scotton PG, Vaglia A, et al. Levofloxacin disposition in cerebrospinal fluid in patients with external ventriculostomy. Antimicrob Agents Chemother. 2003;47(10):3104–8.
Roberts JA, Cotta MO, Cojutti P, Lugano M, Della Rocca G, Pea F. Does critical illness change levofloxacin pharmacokinetics? Antimicrob Agents Chemother. 2015;60(3):1459–63.
Rayner C, Forrest A, Meagher A, Birmingham M, Schentag J. Clinical pharmacodynamics of linezolid in seriously ill patients treated in a compassionate use programme. Clin Pharmacokinet. 2003;42(15):1411–23.
Luque S, Grau S, Alvarez-Lerma F, Ferrández O, Campillo N, Horcajada JP, et al. Plasma and cerebrospinal fluid concentrations of linezolid in neurosurgical critically ill patients with proven or suspected central nervous system infections. Int J Antimicrob Agents. 2014;44(5):409–15.
Adembri C, Fallani S, Cassetta MI, Arrigucci S, Ottaviano A, Pecile P, et al. Linezolid pharmacokinetic/pharmacodynamic profile in critically ill septic patients: intermittent versus continuous infusion. Int J Antimicrob Agents. 2008;31(2):122–9.
Swoboda S, Ober MC, Lichtenstern C, Saleh S, Schwenger V, Sonntag HG, et al. Pharmacokinetics of linezolid in septic patients with and without extended dialysis. Eur J Clin Pharmacol. 2010;66(3):291–8.
Morata L, Cuesta M, Rojas J, Rodriguez S, Brunet M, Casals G, et al. Risk factors for a low linezolid trough plasma concentration in acute infections. Antimicrob Agents Chemother. 2013;57(4):1913–7.
Zoller M, Barbara M, Cyrill H, Neugebauer C, Döbbeler G, Nagel D, et al. Variability of linezolid concentrations after standard dosing in critically ill patients—a prospective observational study. Crit Care. 2014;18(4):R148.
Garcia B, Luque S, Roberts JA, Grau S. Pharmacokinetics and preliminary safety of high dose linezolid for the treatment of Gram-positive bacterial infections. J Infect. 2015;71(5):604–7.
Antachopoulos C, Karvanen M, Iosifidis E, Jansson B, Plachouras D, Cars O, et al. Serum and cerebrospinal fluid levels of colistin in pediatric patients. Antimicrob Agents Chemother. 2010;54(9):3985–7.
Jimenez-Mejias ME, Pichardo-Guerrero C, Marquez-Rivas FJ, Martin-Lozano D, Prados T, Pachon J. Cerebrospinal fluid penetration and pharmacokinetic/pharmacodynamic parameters of intravenously administered colistin in a case of multidrug-resistant Acinetobacter baumannii meningitis. Eur J Clin Microbiol Infect Dis. 2002;21(3):212–4.
Falagas M, Bliziotis I, Tam V. Intraventricular or intrathecal use of polymyxins in patients with Gram-negative meningitis: a systematic review of the available evidence. Int J Antimicrob Agents. 2007;29(1):9–25.
Paul M, Carmeli Y, Durante-Mangoni E, Mouton JW, Tacconelli E, Theuretzbacher U, et al. Combination therapy for carbapenem-resistant Gram-negative bacteria. J Antimicrob Chemother. 2014;69(9):2305–9.
Imberti R, Cusato M, Accetta G, Marino V, Procaccio F, Del Gaudio A, et al. Pharmacokinetics of colistin in cerebrospinal fluid after intraventricular administration of colistin methanesulfonate. Antimicrob Agents Chemother. 2012;56(8):4416–21.
Tunkel A, Hartman B, Kaplan S, Kaufman B, Roos K, Scheld M, et al. Practice guidelines for the management of bacterial meningitis. Clin Infect Dis. 2004;39(9):1267–84.
Kuhen E, Pfeifer G, Frenkell C. Penetration of fosfomycin into cerebrospinal fluid across non-inflamed and inflamed meninges. Infection. 1987;15(6):422–4.
Pfausler B, Spiss H, Dittrich P, Zeitlinger M, Schmutzhard E, Joukhadar C. Concentrations of fosfomycin in the cerebrospinal fluid of neurointensive care patients with ventriculostomy-associated ventriculitis. J Antimicrob Chemother. 2004;53(5):848–52.
Rafailidis P, Falagas M. Options for treating carbapenem-resistant Enterobacteriaceae. Curr Opin Infect Dis. 2014;27(6):479–83.
Ellard G, Humphries M, Allen B. Cerebrospinal fluid drug concentrations and the treatment of tuberculous meningitis. Am Rev Respir Dis. 1993;148(3):650–5.
Nahata M, Fan-Harvard P, Barson W, Bartkowski H, Kosnik E. Pharmacokinetics, cerebrospinal fluid concentration, and safety of intravenous rifampin in pediatric patients undergoing shunt placements. Eur J Clin Pharmacol. 1990;38(5):515–7.
Nau R, Prange HW, Menck S, Kolenda H, Visser K, Seydel J. Penetration of rifampicin into the cerebrospinal fluid of adults with uninflamed meninges. J Antimicrob Chemother. 1992;29(6):719–24.
Klugman K, Friedland I, Bradley J. Bactericidal activity against cephalosporin-resistant Streptococcus pneumoniae in cerebrospinal fluid of children with acute bacterial meningitis. Antimicrob Agents Chemother. 1995;39(9):1988–92.
Liu C, Bayer A, Cosgrove S, Daum R, Fridkin S, Gorwitz R, et al. Clinical practice guidelines by the Infectious Diseases Society of America for the treatment of methicillin-resistant Staphylococcus aureus infections in adults and children. Clin Infect Dis. 2011;52(3):e18–55.
Riser M, Bland C, Rudisil C, Bookstaver B. Cerebrospinal fluid penetration of high-dose daptomycin in suspected Staphylococcus aureus meningitis. Ann Pharmacother. 2010;44(11):1832–5.
Kullar R, Chin JN, Edwards DJ, Parker D, Coplin WM, Rybak MJ. Pharmacokinetics of single-dose daptomycin in patients with suspected or confirmed neurological infections. Antimicrob Agents Chemother. 2011;55(7):3505–9.
Mueller SW, Kiser TH, Anderson TA, Neumann RT. Intraventricular daptomycin and intravenous linezolid for the treatment of external ventricular-drain-associated ventriculitis due to vancomycin-resistant Enterococcus faecium. Ann Pharmacother. 2012;46(12):e35.
Rodvold KA, Gotfried MH, Cwik M, Korth-Bradley JM, Dukart G, Ellis-Grosse EJ. Serum, tissue and body fluid concentrations of tigecycline after a single 100 mg dose. J Antimicrob Chemother. 2006;58(6):1221–9.
Van Wart SA, Cirincione BB, Ludwig EA, Meagher AK, Korth-Bradley JM, Owen JS. Population pharmacokinetics of tigecycline in healthy volunteers. J Clin Pharmacol. 2007;47(6):727–37.
Lengerke C, Haap M, Mayer F, Kanz L, Kinzig M, Schumacher U, et al. Low tigecycline concentrations in the cerebrospinal fluid of a neutropenic patient with inflamed meninges. Antimicrob Agents Chemother. 2011;55(1):449–50.
Lauretti L, D’Alessandris QG, Fantoni M, D’Inzeo T, Fernandez E, Pallini R, et al. First reported case of intraventricular tigecycline for meningitis from extremely drug-resistant Acinetobacter baumannii. J Neurosurg. 2016;19:1–4.
Walti LN, Conen A, Coward J, Jost GF, Trampuz A. Characteristics of infections associated with external ventricular drains of cerebrospinal fluid. J Infect. 2013;66(5):424–31.
Li X, Wu Y, Sun S, Mei S, Wang J, Wang Q, et al. Population pharmacokinetics of vancomycin in postoperative neurosurgical patients. J Pharm Sci. 2015;104(11):3960–7.
Craig W. Basic pharmacodynamics of antibacterials with clinical applications to the use of beta-lactams, glycopeptides, and linezolid. Infect Dis Clin North Am. 2003;17(3):479–501.
Bowers D, Schilling A, Tam V. Aminoglycoside pharmacodynamics. In: Rotschafer J, Andes D, Rodvold K, editors. Antibiotic pharmacodynamics. New York: Springer; 2016. p. 199–229.
Rybak M. The pharmacokinetic and pharmacodynamic properties of vancomycin. Clin Infect Dis. 2006;42(S1):S35–9.
Andes D, van Ogtrop ML, Peng J, Craig WA. In vivo pharmacodynamics of a new oxazolidinone (linezolid). Antimicrob Agents Chemother. 2002;46(11):3484–9.
Wenzler E, Liao S, Rodvold K. Pharmacodynamics of daptomycin. In: Rotschafer J, Andes D, Rodvold K, editors. Antibiotic pharmacodynamics. New York: Springer; 2016. p. 317–43.
Rubino CM, Bhavnani SM, Forrest A, Dukart G, Dartois N, Cooper A, et al. Pharmacokinetics–pharmacodynamics of tigecycline in patients with community-acquired pneumonia. Antimicrob Agents Chemother. 2012;56(1):130–6.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no financial or other conflicts of interest in relation to this research and its publication.
Ethical approval
No clinical research was conducted for the purposes of publication of this article and no ethical approval was necessary.
Rights and permissions
About this article
Cite this article
Kumta, N., Roberts, J.A., Lipman, J. et al. Antibiotic Distribution into Cerebrospinal Fluid: Can Dosing Safely Account for Drug and Disease Factors in the Treatment of Ventriculostomy-Associated Infections?. Clin Pharmacokinet 57, 439–454 (2018). https://doi.org/10.1007/s40262-017-0588-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40262-017-0588-3