Abstract
Key message
Phytophthora infestans resistant somatic hybrids of S. × michoacanum (+) S. tuberosum and autofused 4 x S. × michoacanum were obtained. Our material is promising to introgress resistance from S. × michoacanum into cultivated potato background.
Abstract
Solanum × michoacanum (Bitter.) Rydb. (mch) is a wild diploid (2n = 2x = 24) potato species derived from spontaneous cross of S. bulbocastanum and S. pinnatisectum. This hybrid is a 1 EBN (endosperm balance number) species and can cross effectively only with other 1 EBN species. Plants of mch are resistant to Phytophthora infestans (Mont) de Bary. To introgress late blight resistance genes from mch into S. tuberosum (tbr), genepool somatic hybridization between mch and susceptible diploid potato clones (2n = 2x = 24) or potato cultivar Rywal (2n = 4x = 48) was performed. In total 18,775 calli were obtained from postfusion products from which 1,482 formed shoots. The Simple Sequence Repeat (SSR), Cleaved Amplified Polymorphic Sequences (CAPS) and Random Amplified Polymorphic DNA (RAPD) analyses confirmed hybrid nature of 228 plants and 116 autofused 4x mch. After evaluation of morphological features, flowering, pollen stainability, tuberization and ploidy level, 118 somatic hybrids and 116 autofused 4x mch were tested for late blight resistance using the detached leaf assay. After two seasons of testing three somatic hybrids and 109 4x mch were resistant. Resistant forms have adequate pollen stainability for use in crossing programme and are a promising material useful for introgression resistance from mch into the cultivated potato background.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Phytophthora infestans (Mont.) de Bary is one of the most important pathogens of the cultivated potato Solanum tuberosum L. (tbr) worldwide. It is responsible for the late blight disease, which results in 10–15 % reduction of the global production of potato tubers (Kamoun and Smart 2005; Park et al. 2009). Identification of new sources of resistance to P. infestans and introgression of novel resistance genes from wild potato species into the tetraploid potato genepool is a way to achieve the progress in breeding of the cultivated potatoes resistant to late blight (Zimnoch-Guzowska et al. 2003; Guo et al. 2010). Due to differences in the ploidy level or in the Endosperm Balance Number (EBN) many wild potato species cannot be used as late blight resistance donors in crosses with tbr (Borgato et al. 2007; Tiwari et al. 2010). According to the EBN hypothesis, some species, including Solanum has a specific EBN value, called effective ploidy, determined from crosses with standard species. Successful crosses require a 2:1 maternal:paternal EBN ratio in the hybrid endosperm (Johnston and Hanneman 1980). Somatic hybridization is an alternative method to classical sexual hybridization (Oberwalder et al. 2000; Wielgat and Wasilewska 2001). It enables obtaining variable materials with higher level of resistance and preserved biodiversity in the nuclear and cytoplasmic genomes (Thach et al. 1993; Davey et al. 2005). Somatic hybridization has a potential for introgression of mono- and polygenic traits from wild Solanum species into tbr genepool (Gavrilenko et al. 2003). Resistant to P. infestans somatic hybrids has been obtained between tbr and S. circaeifolium (Mattheij et al. 1992), S. pinnatisectum (Thieme et al. 1997; Polzerová et al. 2011), S. bulbocastanum (Helgeson et al. 1998), S. nigrum (Horsman et al. 2001), S. berthaultii (Bidani et al. 2007), S. tarnii (Thieme et al. 2008) and S. cardiophyllum (Thieme et al. 2010). Somatic hybrids obtained using S. bulbocastanum (Helgeson et al. 1998), S. nigrum (Horsman et al. 2001), S. tarnii (Thieme et al. 2008) and S. commersoni (Carputo et al. 2000) were backcrossed sexually to potato cultivars and exploited in potato breeding programs.
Solanum × michoacanum (Bitter.) Rydb. (mch) is a wild, 1EBN diploid (2n = 2x = 24) nothospecies, a relative to potato, which occurs on the area of Morelia in Michoacán State of Mexico (Hawkes 1990). In natural habitat it grows in damp grassy fields and among rocks, elevated 2,000–2,100 m above the sea level. Mch is tuber-bearing species originating from a spontaneous cross of S. bulbocastanum and S. pinnatisectum (Hawkes 1990) and it is morphologically intermediate between them. Mch is known as the source of resistance to P. infestans of both leaflets and tubers (Jakuczun and Wasilewicz-Flis 2004a; Zoteyeva et al. 2004, 2012). Clones suitable for the potato chips production were found within mch (Jakuczun and Wasilewicz-Flis 2004b). Mch clones resistant to P. infestans were selected (Jakuczun and Wasilewicz-Flis 2004a; Smyda et al. 2011). The P. infestans resistance gene Rpi-mch1 was mapped on potato chromosome VII (Śliwka et al. 2012).
The present paper reports production of somatic hybrids mch (+) tbr and autofused 4x mch plants via electrofusion, which can be potential sources of late blight resistance in the cultivated potato breeding.
Materials and methods
Plant material
The parental forms used in electrofusion were: two clones of mch: 99-12/8 (mch/8) and 99-12/39 (mch/39), three diploid potato clones: DG 81-68, DG 88-89 and dH Bard, and the cultivar (cv.) Rywal. Clones mch/8 and mch/39 were resistant to P. infestans, which was determined before in laboratory detached leaf test in 4 years of evaluation. They derived from the accession VIR5763 of mch, which was received from the N. I. Vavilov Research Institute of Plant Industry (VIR) potato collection, St. Petersburg, Russia. DG 81-68, DG 88-89, dH Bard and cv. Rywal were susceptible to late blight and were chosen from the collection of IHAR-PIB, Młochów. DG 81-68 and DG 88-89 were hybrids of tbr, S. chacoense and S. yungasense. In addition, DG 88-89 had S. gourlayi in origin. They were male fertile, producing functional 2n male gametes, and useful for protoplast fusion with other diploids (Przetakiewicz et al. 2007). Dihaploid dH Bard derived from cv. Bard expressed male sterility, however, it can function well as a seed parent. cv. Rywal was resistant to PVY (Szajko et al. 2008). Plants of parental forms were maintained in vitro and propagated by nodal subculture on hormone-free MS medium (Murashige and Skoog 1962), 2 % sucrose, 0.75 % agar and 0.115 % NH4NO3. Selected forms were virus and bacteria-free and originated from in vitro gene bank. In order to establish in vitro plants of selected parental forms, the presence of common viruses of potato, bacterial and PSTV infection was checked in plants grown in greenhouse conditions. Pathogen-free forms were established in vitro by cutting apical buds and decontaminated with sterilizing agents. Plant material was immersed in the 70 % ethanol for 30 s and then immersed in 2 % solution of sodium hypochlorite for 4 min. Then material was washed with sterile distilled water three times for 5 min each time.
Protoplast isolation, fusion, and culture
Protoplast isolation and fusion were done in Julius Kühn-Institute (JKI), Gross Lüsewitz, Germany according to protocols described by Thieme et al. (2008). About 1 g of leaflets from 3 to 4 weeks old in vitro plants was used for isolation of mesophyll protoplasts according to Mőllers et al. (1992). The enzyme solution containing 0.2 % macerozyme (Serva, Heidelberg, Germany) and 0.8 % cellulase (Serva, Heidelberg, Germany) was applied for digestion of the cellular walls. The purified protoplasts of mch and tbr at a density of 1 × 106 pp/ml were mixed in a ratio of 1:1. 200 μl of this mixture was placed in lamellar fusion chamber. Fusion was achieved by applying first 18–20 V of an AC-field of 800 kHz for aligning the protoplasts, followed by two DC pulses of 120 V amplitude and 15 μs duration with a break of 2 s and AC-field of 10–20 s. Fusion of protoplasts was carried out using the CFA 500 electrofusion equipment of Krüss Company, Hamburg. Afterwards, modified VKM-medium (Binding and Nehls 1977) was added to suspension of fused protoplasts. The cultures were kept in a culture chamber at 25 °C in the dark. The microcalluses forming in the suspension were transferred to Cul-medium (Haberlach et al. 1985) and exposed to 16 h/day illumination (fluorescent light intensity: 55.5 μmol/m2/s1) at 25 °C. After 4 weeks, calluses were transferred on the RJM regeneration medium (Masson et al. 1988). Only one shoot was excised from each callus and rooted on the MS medium. One in vitro copy was preserved for each plant, second copy was planted in pot and propagated in the greenhouse. Plants were covered by glass cap for first 2 weeks of growing in the greenhouse.
DNA analysis
Genomic DNA was extracted from 200 mg fresh, young leaves of greenhouse-grown plants using the GeneElute™ Plant Genomic DNA Miniprep Kit (Sigma). The hybrid nature of somatic hybrids was confirmed by SSR, CAPS and RAPD markers.
SSR markers
The hybrid nature of plants from mch/39 (+) DG 81-68 fusion combination was proved by three SSR markers: ST13ST (localized on chromosome V by Sandbrink et al. 2000), STI 057 (localized on chromosome IX by Feingold et al. 2005) and STM 1049 (mapped to chromosome I, by Ghislain et al. 2004) based on Provan et al. (1996) protocol. PCR amplicons were separated on 6 % polyacrylamide denaturing gel in Sequin-Gen GT sequencing cell (Bio-Rad Laboratories, Inc.) and visualized by silver-staining method (Thieme et al. 2008).
CAPS and RAPD markers
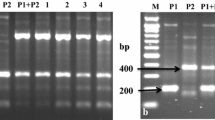
The CAPS marker C2_At5g51970 (Tomato-EXPEN 2000, SGN 2012) and RAPD primer OPA11 (CAATCGCCGT, Sigma-Aldrich, St. Louis, MO, USA) were useful for detection of somatic hybrids and autofused plants. CAPS marker C2_At1g53670 (Tomato-EXPEN 2000, SGN 2012) linked to the Rpi-mch1 gene on chromosome VII was used to verify resistant fusion plants originating from the clone mch/8, which was a resistant parent of mapping population (Śliwka et al. 2012). The presence of the marker C2_At1g53670 was tested in mch/39 parental form as well. PCR reactions for C2_At5g51970 and C2_At1g53670 were performed in a G-Storm thermocycler in 20 μl of a reaction mixture containing: 10× buffer including 20 mM MgCl2, 0.5 mM of each dNTP, 12.5 mM primers, 0.05 U/μl DreamTaq polymerase (Fermentas Life Sciences, Thermo Fischer Scientific Inc) and 20 ng DNA template. Amplification was performed with initial denaturation at 94 °C for 30 s followed by 40 cycles at 94 °C for 45 s, 55 °C for 45 s, 72 °C for 1 min 30 s and one final extension at 72 °C for 10 min. Digestions of the amplicons with restriction endonuclease MspI (Fermentas Life Sciences, Thermo Fischer Scientific Inc.) and RsaI (Fermentas Life Sciences, Thermo Fischer Scientific Inc.) were performed according to producers’ protocol for C2_At5g51970 and C2_At1g53670, respectively. Product sizes of selected markers are presented in Table 2.
Amplification of OPA11 was performed in a G-Storm thermocycler in 20 μl of a reaction mixture containing: 10× buffer including 20 mM MgCl2, 0.5 mM of each dNTP, 12.5 mM primer, 0.05 U/μl U DreamTaq polymerase (Fermentas Life Sciences, Thermo Fischer Scientific Inc) and 30 ng DNA template. The PCR parameters were: 94 °C for 30 s followed by 45 cycles at 92 °C for 15 s, 36 °C for 25 s, 72 °C for 74 s and one final extension at 72 °C for 5 min.
The amplification products of CAPS and RAPD markers were separated in 1.5 % agarose gel during electrophoresis in 1× TBE buffer (Tris-Borate-EDTA) and stained with ethidium bromide. 1 kb DNA ladder (Invitrogen) was used as a molecular marker.
Morphological, physiological and cytological characterization
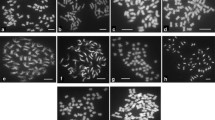
The most vigorous plants were multiplied in vitro and transferred to the greenhouse. For each formed plantlet first copy was kept in vitro and after root formation a second one was planted in pot in the greenhouse. In vitro plants with abnormalities in the growth or low vitality were eliminated. Phenotypes of vigorous greenhouse-grown plants were compared to parental forms and characterized in terms of habit, shape of leaves and whole plants, flowering and tuberization. The ploidy level mostly was evaluated by counting chloroplasts in the guard cells and for chosen group by the use of flow cytometry (Thieme et al. 1997). The mean number of chloroplasts in the pair of guard cells is assumed for diploids 11.2 (range 7.5–14.0), for triploids 14.4 (range 10.7–19.0) and for tetraploid forms 19.7 (range 16.0–25.7), respectively (Rothacker and Junges 1966). Pollen fertility was estimated using an indirect lactofuchsin method based on percentage of regularly shaped and stained pollen grains (Janssen and Hermsen 1976).
Late blight resistance assessment
The resistance tests of post-fusion plants, their parental forms and standard cultivars were performed in 3 years—2009, 2010 and 2011. In 2009 and 2010 resistance to P. infestans was tested on greenhouse plants grown from in vitro plants and in 2011 on plants grown from tubers. Greenhouse-grown plants were assessed in a laboratory test for resistance to foliage blight using detached leaf test (Fig. 1). Respective parental forms and standard cultivars were always tested together with post-fusion plants. Tests were performed at two different dates in two replicates. The single replicate was represented by one leaf in which 1–6 leaflets were evaluated based on 1–9 scale (where 9 = the most resistant). In total about 24 leaflets were tested for each genotype during season. A genotype with mean infection score ≥6 was assumed to be resistant to P. infestans based on research done on the mch by Śliwka et al. (2012). In 2009 and 2010 plants grown in the greenhouse directly from in vitro were tested with two P. infestans isolates MP847 and MP921. In 2011 plants were propagated from tubers harvested in previous season and tests were performed with the isolate MP921. The isolates of P. infestans MP847 and MP921 originated from pathogen collection of IHAR-PIB, Młochów. Those isolates were of A2 sexual type and Ia mitochondrial type, sensitive to metalaxyl, of complex race: 1. 3. 4. 7. 10. 11 (Śliwka et al. 2012). Inoculation was done by spraying (Kuhl et al. 2001) with 50 sporangia/μl suspension prepared according to Zarzycka (2001).
Results
Protoplast fusion
In total, 18,775 calli were produced from post-fusion products in the all eight combinations. From those calli in total 1,482 in vitro plants were regenerated (Table 1). Out of this group 344 plants were selected for further experiments.
Identification of somatic hybrids by molecular markers and selection of 4x autofused mch
A number of obtained somatic hybrids and autofused 4x mch are presented in Table 1. In order to identify somatic hybrids and 4x mch, all putative ones from eight fusion combinations were analyzed using three SSR, two CAPS markers and one RAPD marker (Table 2). The hybrid nature of 123 plants from mch/39 (+) DG 81-68 fusion combination was confirmed in JKI using three SSR markers: ST13ST (122 hybrids), STI057 (123 hybrids), STM1049 (122 hybrids). Second set of 59 plants from this combination was tested in IHAR-PIB with CAPS and RAPD markers C2_At5g51970 (29) and OPA11 (29 hybrids) and next 29 hybrids were identified. In two fusion combinations of mch/8 (+) DG 81-68 and mch/8 (+) DG 88-89 somatic hybrids were not obtained. From the fusion combination of mch/8 (+) dH Bard, out of 25 analyzed plants four were identified as hybrids. In mch/8 (+) cv. Rywal, only three out of 122 analyzed plants were of expected hybrid nature. The number of somatic hybrids from next three combinations mch/39 (+) DG 88-89, mch/39 (+) dH Bard and mch/39 (+) cv. Rywal was 1 (out of 42 tested), 56 (out of 153 tested) and 12 (out of 31 tested), respectively (Table 3). In total 228 somatic hybrids were identified across eight combinations studied. An example of hybrid identification by a CAPS marker is demonstrated in Fig. 2. In addition, 116 autofused forms of 4x mch were obtained both from mch/8 and mch/39 parents. Seven somatic hybrids from two combinations with mch/8 parent were tested using C2_At1g53670 marker linked to Rpi-mch1 gene (Śliwka et al. 2012). One from seven analyzed hybrids had the marker; however, all the seven were susceptible to late blight. The presence of the marker C2_At1g53670 was not found in the second parental form mch/39, thus this marker was not used to verify resistant fusion products originating from the clone mch/39.
Morphological, physiological and cytological characterization
Morphological observations indicated two distinct groups of plants: one with intermediate phenotype, between parental forms and a second one similar to mch parent. Among plants with intermediate phenotype, some deformations of whole plants, leaves and tubers or various colours of flowers were observed; however, plants and tubers without these deformations were often observed (Fig. 3). Plants similar to mch parents were more luxuriant and had bigger leaves with widen leaflets in comparison to wild forms. Within set of 228 confirmed hybrids, 160 hybrids were tetraploids and 68 were with ploidy level higher than 4x (6x or 8x) (Table 1), what was confirmed by counting of chloroplasts in the guard cells or by flow cytometry. Among 118 tuberizing hybrids, 103 ones were 4x and the ploidy level of 15 forms were more than 4x (they originated from fusion of mch to 4x cv. Rywal). The separate set was represented by 116 autofused 4x mch, which was evidenced by morphological traits and molecular markers. Majority of somatic hybrids and 4x mch flowered intensively. Among them, 85 of 4x somatic hybrids and 113 of autofused 4x mch produced pollen grains which were successfully stained. Stainability of pollen grains of resistant somatic hybrids and 4x mch was between 40–60 and 40–70 %, respectively.
Late blight resistance test
In 2009 and 2010 plants tested for late blight resistance were replanted in the greenhouse from in vitro. In 2011 tested plants grown from tubers. In 2009–2011 mean resistance to P. infestans of parental clones of mch/8 and mch/39 was scored as 8.1 and 8.0, respectively, with individual scores ranging from 4.0 to 9.0 (Table 4). The mean scores of resistance to late blight of susceptible clones DG 81-68, DG 88-89, dH Bard and cv. Rywal in 2009–2011 were 2.3, 2.1, 1.3, and 1.4, accordingly with a range 1.0–6.0 for individual readings (Table 4). In 2009–2011 in total 118 defined somatic hybrids and 116 autofused 4x mch were tested for resistance to late blight (Table 5). After two seasons of testing three somatic hybrids and 109 autofused 4x mch were defined as resistant to P. infestans. The mean scores of resistance to P. infestans of three resistant somatic hybrids were: 7.0, 7.6, and 6.9 which were slightly lower than the scores of their resistant parents. Resistance of remaining susceptible somatic hybrids ranged between 1.0 and 5.0. Mean resistance to late blight of autofused 91 4x mch/8 and 18 4x mch/39 plants was 7.5 and 7.8, respectively with a range between 5.0 and 9.0 (Table 4).
Discussion
Many wild potato species are potential sources of resistance to P. infestans (Hawkes 1990). At present, there is a deficit of potato cultivars resistant to late blight because new and more aggressive pathogenic races of P. infestans often overcome monogenic resistance (Thieme et al. 2010). Introduction of late blight resistance genes from more distantly related germplasm of wild species into the cultivated potato could be the most effective and environment friendly way to defeat P. infestans. Late blight resistance genes from S. demissum, S. chacoense, and S. phureja were successfully transferred to cultivated potato via sexual crosses (reviewed by Heřmanová et al. 2007). Majority of wild potato species are diploids (2n = 2x = 24) with 1 or 2 EBN. Unfortunately, 1EBN diploid species are not crossable to tbr due to different ploidy level and EBN. Hence, 1EBN forms cannot be directly used for widening the cultivated potato genepool, which is 4EBN. Because of genetic incompatibilities and the reproductive isolation between wild and cultivated potato species, genetic improvement through interspecific hybridization is difficult and time consuming. However, sexual discordances might be bypassed in several ways: through cloning of the R gene and its transfer via cisgenesis or transgenesis into potato genepool, applying protoplast fusion or changing ploidy level by doubling of chromosome number (Park et al. 2009). Many resistance Rpi genes from wild species have been identified (Park et al. 2009). About 20 Rpi genes have been cloned (Śliwka and Zimnoch-Guzowska 2012) and part of them were transferred to the cultivated potato through the cisgenic approach, like: RB/Rpi-blb1 (Song et al. 2003; van der Vossen et al. 2003), Rpi-vnt1.1 (Foster et al. 2009) and Rpi-mcq1 (GMO notification B/GB/10/R29/01). Somatic hybridization is an alternative way to such R genes transfer. However, there are several bottle necks in application of somatic hybridization technique to the enrichment of breeding genepool. In somatic hybridization nuclear and cytoplasmic genomes of wild potato species are fused to potato genepool and not only the gene of interest as it is in genetic transformation. Thus, somatic fusion method is slower than genetic transformation because of necessity of elimination of unwanted traits from somatic hybrids through series of backcrosses to tbr background (Thieme et al. 1997; Laferriere et al. 1999). Genetic instability, lack or poor of fertility and very low crossability of produced somatic hybrids to cultivated genepool are the major problems in routine application of this technique in breeding schemes (Mattheij et al. 1992; Rokka et al. 1995; Nyman and Waara 1997; Carputo et al. 1998; Orczyk et al. 2003). Nevertheless, there are many examples of successful introgression of some agronomically important traits to somatic hybrids. Somatic hybrids resistant to P. infestans, Erwinia carotovora, Ralstonia solanacea, PLRV, PVX, PVY, nematode and somatic hybrids with frost tolerance and higher capacity to cold acclimation or with higher starch content and reduced concentration of glycoalkaloids are known (reviewed by Orczyk et al. 2003).
Post-fusion mixture besides products of heterofusion (heterokaryons) contains both fusion components and products of homofusion (homokaryons). Those additional products of somatic hybridization are eliminated in first steps of this process as a result of conscious selection of heterokaryons. Fusions of protoplasts of wild plants result in production of wild forms with higher ploidy level. Electrofusion and PEG induced fusion are equally effective methods (Orczyk et al. 2003) and the result of protoplast fusion mainly depends on efficient fusion protocol. Przetakiewicz et al. (2007) produced somatic hybrids between diploid interspecific hybrids using PEG method. Szczerbakowa et al. (2010) applied this approach to obtain the first set of 13 somatic hybrids between clone mch/8 and clone DG 81-68. In the presented study about 15 % of regenerated plants were somatic hybrids and about 8 % were autofused 4x mch via electrofusion method. Remaining 77 % plants were 2x mch, 2x tbr or 4x tbr and were eliminated. In the group of somatic hybrids characterized by intermediate phenotype, between parental forms some deformations of whole plants, leaves and tubers or various colours of flowers were observed. It could be caused by several factors: additive or non-additive effects of parental genes, new interactions between nuclear and cytoplasmic genomes or dosage of parental genome (Cardi 1998). The ploidy level was estimated by counting the chloroplasts in guard cells (Carrasco et al. 2000; Szczerbakowa et al. 2010) and by flow cytometry (Rokka et al. 1995; Thieme et al. 1997) and these two methods allowed us to identify a set of expected tetraploids, hexaploids and hybrids with higher chromosome number.
Based on published data by Śliwka et al. (2012), the presence of the Rpi-mch1 gene (on chromosome VII of mch/8 parent) was tested using C2_At1g53670 CAPS marker linked to this gene (at a distance of 5.7 cM) in seven, assessed as susceptible to P. infestans, somatic hybrids originated from mch/8 parent. Among them one susceptible hybrid had a marker. This might have resulted from chromosome elimination/recombination or silencing of the gene (Orczyk et al. 2003). Lack of the marker in remaining six plants might have resulted from the loss of the chromosome VII or its fragment with Rpi-mch1 gene in the electrofusion process, what is a frequent phenomenon in somatic hybrids’ genome (Śliwka et al. 2012). To explain this situation research has to be conducted on characterization of the chromosomal structure of obtained somatic hybrids, what is in our future plans.
It is known that nuclear–cytoplasmic interactions are important factors in male fertility (Nyman and Waara 1997; Orczyk et al. 2003). Although a number of somatic hybrids produced a lot of flowers, it was difficult to obtain a backcross generation with cultivated potato. Some of the obtained somatic hybrids have been fertile and could be backcrossed sexually to cultivated potato. However, such forms are genetically instable and very often cannot be used in further breeding exploitation. In our work the frequency of stained pollen grains of the somatic hybrids ranged between 10 and 85 %. Similar data were noted by Thieme et al. (2010). In our study flowering was abundant, 85 somatic hybrids flowered and produced stainable pollen grains and three resistant somatic hybrids had fertile pollen grains adequate for using them as pollinators. All 116 autofused 4x mch flowered and 113 of them produced stainable pollen grains. 4x mch could potentially be crossed with tbr plants, because one of the crossing barriers was eliminated.
Somatic hybrids produced in our study varied in morphology and resistance to P. infestans. This phenomenon can be partly explained by various ploidy levels of regenerants and new composition of somatic hybrids’ genome. The range of late blight resistance observed in somatic hybrids forms and 4x mch was from 1.0 to 9.0. Two classes of resistance were distinguished after resistance tests: resistant (mean scores ≥6) and susceptible forms (mean scores <6) (Table 5). The resistance level of three identified resistant somatic hybrids was enhanced, but slightly lower than resistance level of mch parents and similar or lower than resistance of two sets of autofused 4x mch. Only three from 118 tested somatic hybrids were resistant to P. infestans, while in the group of autofused 4x mch all forms (in total 109 after 2 years of testing) were resistant (Tables 4, 5). Resistance of assessed forms was confirmed in tests in 2012. In previous study Szczerbakowa et al. (2010) identified one resistant somatic hybrid mch (+) tbr (with mean score = 6.3) out of 10 evaluated. This resistant clone did not flower and could not be used in backcrossing programme. It is a question for our further studies why among identified somatic hybrids the frequency of those with good expression of resistance to late blight was so low.
Work is currently in progress to introgress resistance to P. infestans both from resistant somatic hybrids and autofused 4x mch into cultivated potato by sexual crosses. We also assume possibility of cloning of the Rpi-mch1 gene for further use in cisgenic approach. In order to confirm results of laboratory test for resistance to foliage blight we plan to assess late blight resistance under field conditions. Pyramiding of R genes is common tendency in many breeding programs, since it can provide high and durable resistance to P. infestans. Mch is one of interesting and valuable source of resistance to P. infestans. This species has not been used in breeding so far. A resistance gene Rpi-mch1 (Śliwka et al. 2012), against late blight was mapped on chromosome VII of the resistant parent mch/8. On potato chromosome VII only two resistance genes were mapped: Rpi-mch1 (Śliwka et al. 2012) and Rpi1 (Kuhl et al. 2001). Rpi-mch1 is located on different chromosomes than other R genes (except Rpi1) and it can demonstrate distinct structure, mechanism of action or it can react to different effectors of pathogen. Mch can be valuable component in pyramiding of R genes via sexual crosses or cisgenic approach.
Abbreviations
- CAPS:
-
Cleaved amplified polymorphic sequences
- EBN:
-
Endosperm balance number
- IHAR-PIB:
-
Plant Breeding and Acclimatization Institute-National Research Institute
- JKI:
-
Julius Kühn-Institute
- PLRV:
-
Potato leafroll virus
- PSTV:
-
Potato spindle tuber viroid
- PVX:
-
Potato virus X
- PVY:
-
Potato virus Y
- RAPD:
-
Random amplified polymorphic DNA
- SSR:
-
Simple sequence repeat
References
Bidani A, Nouri-Ellouz O, Lakhoua L, Sihachakr D, Cheniclet C, Mahjoub A, Drira N, Gargouri-Bouzid R (2007) Interspecific potato somatic hybrids between Solanum berthaultii and Solanum tuberosum L. showed recombinant plastome and improved tolerance to salinity. Plant Cell Tiss Org Cult 91:179–189
Binding H, Nehls R (1977) Regeneration of isolated protoplasts to plants in Solanum dulcamone L. Z Pflanzenphysiol 85:279–280
Borgato L, Cobicella C, Pisani F, Furini A (2007) Production and characterization of arboreous and fertile Solanum melongena + Solanum marginatum somatic hybrids plants. Planta 226:961–969
Cardi T (1998) Multivariate analysis of variation among Solanum commersonii (+) S. tuberosum somatic hybrids with different ploidy levels. Euphytica 99:35–41
Carputo D, Garreffa P, Mazzei M, Monti L, Cardi T (1998) Fertility of somatic hybrids Solanum commersonii (2x, 1EBN) (+) S. tuberosum haploid (2x, 2EBN) in intra- and inter-EBN crosses. Genome 41:776–781
Carputo D, Basile B, Cardi T, Frusciante L (2000) Erwinia resistance in backcross progenies of Solanum tuberosum x S. tarijense and S. tuberosum (+) S. commersonii hybrids. Potato Res 43:135–142
Carrasco A, Ruiz De Galarreta JI, Rico A, Ritter E (2000) Transfer of PLRV resistance from Solanum verrucosum Schlechdt to potato (S. tuberosum L.) by protoplast electrofusion. Potato Res 43:31–42
Davey MR, Anthony P, Power JB, Lowe KC (2005) Plant protoplast technology: current status. Acta Physiol Plant 27:117–129
Feingold S, Lloyd J, Norero N, Bonierbale M, Lorenzen J (2005) Mapping and characterization of new EST-derived microsatellites for potato (Solanum tuberosum L.). Theor Appl Genet 111:456–466
Foster SJ, Park TH, Pel M, Brigneti G, Śliwka J, Jagger L, van der Vossen E, Jones J (2009) Rpi-vnt1.1, a Tm-2 2 homolog from Solanum venturii, confers resistance to potato late blight. Mol Plant Microbe Interact 22:589–600
Gavrilenko T, Thieme R, Heimbach U, Thieme T (2003) Fertile somatic hybrids of Solanum etuberosum (+) dihaploid Solanum tuberosum and their backcrossing progenies: relationships of genome dosage with tuber development and resistance to potato virus Y. Euphytica 131:323–332
Ghislain M, Spooner DM, Rodriguez F, Villamón F, Núnez J, Vásquez C, Waugh R, Bonierbale M (2004) Selection of highly informative and user-friendly microsatellites (SSRs) for genotyping of cultivated potato. Theor Appl Genet 108:881–890
Guo X, Xie C, Cai X, Song B, He L, Liu J (2010) Meiotic behavior of pollen mother cells in relation to ploidy level of somatic hybrids between Solanum tuberosum and S. chacoense. Plant Cell Rep 29:1277–1285
Haberlach GT, Cohen BA, Reichert NA, Baer A, Towill LE, Helgeson JP (1985) Isolation, culture, and regeneration of protoplasts from potato and several related Solanum species. Plant Sci 39:67–74
Hawkes JG (1990) The potato, evolution, biodiversity and genetic resources. Belhaven Press, London
Helgeson JP, Pohlman JD, Austin S, Haberlach GT, Wielgus SM, Ronis D, Zambolim L, Tooley P, McGrath JM, James RV, Stevenson WR (1998) Somatic hybrids between Solanum bulbocastanum and potato: a new source of resistance to late blight. Theor Appl Genet 96:738–742
Heřmanová V, Bárta J, Čurn V (2007) Wild potato species: characterization and biological potential for potato breeding. Czech J Genet Plant Breed 43:73–81
Horsman K, Gavrilenko T, Bergervoet M, Huigen DJ, Joe ATW, Jacobsen E (2001) Alteration of the genomic composition of Solanum nigrum (+) potato backcross derivatives by somatic hybridization: selection of fusion hybrids by DNA measurements and GISH. Plant breed 120:201–207
Jakuczun H, Wasilewicz-Flis I (2004a) New sources of potato resistance to Phytophthora infestans at the diploid level. Plant Breed Seed Sci 50:137–145
Jakuczun H, Wasilewicz-Flis I (2004b) Ziemniak diploidalny źródłem cech jakościowych w hodowli. Zesz Prob Nauk Roln 500:127–136 (in Polish)
Janssen AWB, Hermsen JGTh (1976) Estimating pollen fertility in Solanum species and haploids. Euphytica 25:577–586
Johnston SA, Hanneman RE Jr (1980) Support of the endosperm balance number hypothesis utilizing some tuber-bearing Solanum species. Am Potato J 57:7–14
Kamoun S, Smart CD (2005) Late blight of potato and tomato in the genomics era. Plant Dis 89:692–699
Kuhl JC, Hanneman RE, Havey MJ (2001) Characterization and mapping of Rpi1, a late-blight resistance locus from diploid (1EBN) Mexican Solanum pinnatisectum. Mol Genet Genomics 265:977–985
Laferriere LT, Helgeson JP, Allen C (1999) Fertile Solanum tuberosum + S. commersonii somatic hybrids as sources of resistance to bacterial wilt caused by Ralstonia solanacearum. Theor Appl Genet 98:1272–1278
Masson JM, Lecerf M, Rousselle P, Perennec P, Pellettier G (1988) Plant regeneration from protoplasts of diploid potato derived from crosses of Solanum tuberosum with wild Solanum species. Plant Sci 53:167–176
Mattheij WM, Eijlander R, de Koning JRA, Louwes KM (1992) Interspecific hybridization between the cultivated potato Solanum tuberosum subspecies tuberosum L. and the wild species S. circaeifolium subsp. circaeifolium Bitter exhibiting resistance to Phytophthora infestans (Mont.) de Bary and Globodera palliada (Stone) Behrens. 1. Somatic hybrids. Theor Appl Genet 83:459–466
Mőllers C, Zhang S, Wenzel G (1992) Influence of silver thiosulfate on potato protoplast culture. Plant Breed 108:12–18
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15:473–497
Nyman M, Waara S (1997) Characterisation of somatic hybrids between Solanum tuberosum and its frost-tolerant relative Solanum commersonii. Theor Appl Genet 95:1127–1132
Oberwalder B, Schilde-Rentschler L, Löffelhardt-Ruoß B, Ninnemann H (2000) Differences between hybrids of Solanum tuberosum L. and Solanum circaeifolium Bitt. obtained from symmetric and asymmetric fusion experiments. Potato Res 43:71–82
Orczyk W, Przetakiewicz J, Nadolska-Orczyk A (2003) Somatic hybrids of Solanum tuberosum—application to genetics and breeding. Plant Cell Tiss Org Cult 74:1–13
Park TH, Vleeshouwers VGAA, Jacobsen E, van Der Vossen E, Visser RGF (2009) Molecular breeding for resistance to Phytophthora infestans (Mont.) de Bary in potato (Solanum tuberosum L.): a perspective of cisgenesis. Plant Breed 128:109–117
Polzerová H, Patzak J, Greplová M (2011) Early characterization of somatic hybrids from symmetric protoplast electrofusion of Solanum pinnatisectum Dun. and Solanum tuberosum L. Plant Cell Tiss Organ Cult 104:163–170
Provan J, Kumar A, Shepherd L, Powell W, Wangh R (1996) Analysis of intra-specific somatic hybrids of potato (Solanum tuberosum) using simple sequence repeats. Plant Cell Rep 16:196–199
Przetakiewicz J, Nadolska-Orczyk A, Kuć D, Orczyk W (2007) Tetraploid somatic cybrids of potato (Solanum tuberosum L.) obtained from diploid breeding lines. Cell Mol Biol Lett 12:253–267
Rokka V-M, Valkonen JPT, Pehu E (1995) Production and characterization of haploids derived from somatic hybrids between Solanum brevidens and S. tuberosum through anther culture. Plant Sci 112:85–95
Rothacker DJ, Junges W (1966) Untersuchungen zur Erzeugung und Auslese dihaploider Sämlinge bei Solanum tuberosum L. Eur Potato J 9:99–110
Sandbrink JM, Colon LT, Wolters PJCC, Stiekema WJ (2000) Two related genotypes of Solanum microdontum carry different segregating alleles for field resistance to Phytophthora infestans. Mol Breed 6:215–225
SGN (SOL Genomics Network) Database (2012). http://solgenomics.net/
Śliwka J, Zimnoch-Guzowska E (2012) Resistance to late blight in potato. In: Tuberosa R, Varshney RK (eds) Genomics applications in plant breeding. Wiley, USA (in press)
Śliwka J, Jakuczun H, Chmielarz M, Hara-Skrzypiec A, Tomczyńska I, Kilian A, Zimnoch-Guzowska E (2012) A resistance gene against potato late blight originating from Solanum × michoacanum maps to potato chromosome VII. Theor Appl Genet 124:397–406
Smyda P, Jakuczun H, Dębski K, Śliwka J, Thieme R, Nachtigall M, Wasilewicz-Flis I, Zimnoch-Guzowska E (2011) Production and characterization of potato somatic hybrids between Solanum michoacanum (Bitter.) Rydb. resistant to Phytophthora infestans and S. tuberosum L. The 18th Triennial conference of the European Association for potato research. Oulu, Finland, 24–29 July: Abstracts 191
Song J, Bradeen JM, Naess SK, Raasch JA, Wielgus SM, Haberlach GT, Liu J, Kuang H, Austin-Phillips S, Buell CR, Helgeson JP, Jiang J (2003) Gene RB cloned from Solanum bulbocastanum confers broad spectrum resistance to potato late blight. Proc Natl Acad Sci USA 100:9128–9133
Szajko K, Chrzanowska M, Witek K, Strzelczyk-Żyta D, Zagórska D, Gebhardt C, Hennig J, Marczewski W (2008) The novel gene Ny-1 on potato chromosome IX confers hypersensitive resistance to Potato virus Y and is an alternative to Ry genes in potato breeding for PVY resistance. Theor Appl Genet 116:297–303
Szczerbakowa A, Tarwacka J, Oskiera M, Jakuczun H, Wielgat B (2010) Somatic hybridization between the diploids of S. × michoacanum and S. tuberosum. Acta Physiol Plant 32:867–873
Thach NQ, Frei U, Wenzel G (1993) Somatic fusion for combining virus resistances in Solanum tuberosum L. Theor Appl Genet 85:863–867
Thieme R, Darsow U, Gavrilenko T, Dorokhov D, Tiemann H (1997) Production of somatic hybrids between S. tuberosum L. and late blight resistant Mexican wild potato species. Euphytica 97:189–200
Thieme R, Rakosy-Tican E, Gavrilenko T, Antonova O, Schubert J, Nachtigall M, Heimbach U, Thieme T (2008) Novel somatic hybrids (Solanum tuberosum L. + Solanum tarnii) and their fertile BC1 progenies express extreme resistance to potato virus Y and late blight. Theor Appl Genet 116:691–700
Thieme R, Rakosy-Tican E, Nachtigall M, Schubert J, Hammann T, Antonova O, Gavrilenko T, Heimbach U, Thieme T (2010) Characterization of the multiple resistance traits of somatic hybrids between Solanum cardiophyllum Lindl. and two commercial potato cultivars. Plant Cell Rep 29:1187–1201
Tiwari JK, Sarkar PD, Pandey SK, Gopal J, Kumar SR (2010) Molecular and morphological characterization of somatic hybrids between Solanum tuberosum L. and S. etuberosum Lindl. Plant Cell Tiss Org Cult 103:175–187
van der Vossen E, Sikkema A, Hekkert BL, Gros J, Stevens P, Muskens M, Wouters D, Pereira A, Stiekema W, Allefs S (2003) An ancient R gene from wild potato species Solanum bulbocastanum confers broad-spectrum resistance to Phytophthora infestans in cultivated potato and tomato. Plant J 36:867–882
Wielgat B, Wasilewska LD (2001) Somatic hybridization between wild and cultivated Solanum species. Biotechnology 2:9–15
Zarzycka H (2001) Evaluation of resistance to Phytophthora infestans in detached leaflet assay. Preparation of the inoculum, vol 10a. Monografie i Rozprawy Naukowe IHAR, Radzików, pp 75–77
Zimnoch-Guzowska E, Lebecka R, Kryszczuk A, Maciejewska U, Szczerbakowa A, Wielgat B (2003) Resistance to Phytophthora infestans in somatic hybrids of Solanum nigrum L. and diploid potato. Theor Appl Genet 107:43–48
Zoteyeva N, Chrzanowska M, Evstratova L, Fasulati S, Yusupov T (2004): Resistance to diseases and pests in wild potato accessions. Catalogue of VIR’s collection. St. Petersburg 88 pp. (in Russian) Issue 761
Zoteyeva N, Chrzanowska M, Flis B, Zimnoch-Guzowska E (2012) Resistance to pathogens of the potato accessions from the collection of N.I. Vavilov Institute of Plant Industry (VIR). Am J Pot Res 89:277–293
Acknowledgments
This work was financed by The National Centre of Research and Development in Poland, grant: PBZ-MNiSW-2/3/2006/28.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by R. Rose.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution License which permits any use, distribution, and reproduction in any medium, provided the original author(s) and the source are credited.
About this article
Cite this article
Smyda, P., Jakuczun, H., Dębski, K. et al. Development of somatic hybrids Solanum × michoacanum Bitter. (Rydb.) (+) S. tuberosum L. and autofused 4x S. × michoacanum plants as potential sources of late blight resistance for potato breeding. Plant Cell Rep 32, 1231–1241 (2013). https://doi.org/10.1007/s00299-013-1422-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-013-1422-5