Abstract
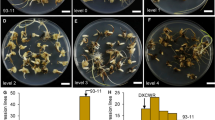
A new cold tolerant germplasm resource named glutinous rice 89-1 (Gr89-1, Oryza sativa L.) can overwinter using axillary buds, with these buds being ratooned the following year. The overwintering seedling rate (OSR) is an important factor for evaluating cold tolerance. Many quantitative trait loci (QTLs) controlling cold tolerance at different growth stages in rice have been identified, with some of these QTLs being successfully cloned. However, no QTLs conferring to the OSR trait have been located in the perennial O. sativa L. To identify QTLs associated with OSR and to evaluate cold tolerance. 286 F12 recombinant inbred lines (RILs) derived from a cross between the cold tolerant variety Gr89-1 and cold sensitive variety Shuhui527 (SH527) were used. A total of 198 polymorphic simple sequence repeat (SSR) markers that were distributed uniformly on 12 chromosomes were used to construct the linkage map. The gene ontology (GO) annotation of the major QTL was performed through the rice genome annotation project system. Three main-effect QTLs (qOSR2, qOSR3, and qOSR8) were detected and mapped on chromosomes 2, 3, and 8, respectively. These QTLs were located in the interval of RM14208 (35,160,202 base pairs (bp))–RM208 (35,520,147 bp), RM218 (8,375,236 bp)–RM232 (9,755,778 bp), and RM5891 (24,626,930 bp)–RM23608 (25,355,519 bp), and explained 19.6%, 9.3%, and 11.8% of the phenotypic variations, respectively. The qOSR2 QTL displayed the largest effect, with a logarithm of odds score (LOD) of 5.5. A total of 47 candidate genes on the qOSR2 locus were associated with 219 GO terms. Among these candidate genes, 11 were related to cell membrane, 7 were associated with cold stress, and 3 were involved in response to stress and biotic stimulus. OsPIP1;3 was the only one candidate gene related to stress, biotic stimulus, cold stress, and encoding a cell membrane protein. After QTL mapping, a total of three main-effect QTLs—qOSR2, qOSR3, and qOSR8—were detected on chromosomes 2, 3, and 8, respectively. Among these, qOSR2 explained the highest phenotypic variance. All the QTLs elite traits come from the cold resistance parent Gr89-1. OsPIP1;3 might be a candidate gene of qOSR2.




Similar content being viewed by others
References
Andaya VC, Mackill DJ (2003) Mapping of QTLs associated with cold tolerance during the vegetative stage in rice. J Exp Bot 54:2579
Andaya VC, Tai TH (2007) Fine mapping of the qCTS4 locus associated with seeding cold tolerance in rice (Oryza sativa L.). Mol Breed 20:349–358
Biswas MK, Chai L, Mayer C, Xu Q, Guo WW, Deng XX (2012) Exploiting BAC-end sequences for the mining, characterization and utility of new short sequences repeat (SSR) markers in citrus. Mol Biol Rep 39:5373–5386
Biswas PS, Khatun H, Das N, Sarker MM, Anisuzzaman M (2017) Mapping and validation of QTLs for cold tolerance at seedling stage in rice from an indica cultivar Habiganj Boro VI (Hbj.BVI). 3 Biotech 7:359
Chen DZ, Zhong PA, Xiao YQ, Huang YJ, Xie JK (2002) Identification of QTLs for cold tolerance at seedling stage in Dongxiang wild rice by SSR markers. Acta Agriculturae Universitatis Jiangxiensis 24:753–757
Deng JY, Chen GH, Liu B, Liu J (2009) Progress on the studies of physiological and genetic mechanisms of cold tolerance in rice. Guangdong Agric Sci 5:25–28
Deng XS, Wu KQ, Liu Y, Tang AJ, Luo AC, Du C, Zhao ZW (2015) Breeding and application of rice restorer line Kehui 36 with cold tolerance. Hybrid Rice 5:10–11
Fujino K, Sekiguchi H, Sato T, Kiuchi H, Nonoue Y, Takeuchi Y, Ando T, Lin SY, Yano M (2004) Mapping of quantitative trait loci controlling low-temperature germinability in rice (Oryza sativa L.). Theor Appl Genet 108:794–799
Han LZ, Yuan DL, Xuan YS, Pu ZZ, Gao XZ (2004) QTLs analysis of cold tolerance during early growth period for rice. Rice Sci 11:245–250
Hu Y, Wang YZ (2005) Mapping of QTL controlling seedling cold tolerance using recombinant inbred lines of rice (Oryza sativa L.). J Wuhan Bot Res 23:211–215
Kim SI, Andaya CB, Goyal SS, Tai TH (2008) The rice OsLpa1 gene encodes a novel protein involved in phytic acid metabolism. Theor Appl Genet 117:769–779
Lincoln S, Daly M, Lander E (1992) Constructing genetic linkage maps with MAPMAKER/EXP 3.0. Genomics 1:174–181
Liu CT, Wang W, Mao BG, Chu CC (2018) Cold stress tolerance in rice: physiological changes, molecular mechanism, and future prospects. Hereditas 3:171–185
Lu G, Wu FQ, Wu W, Wang HJ, Zheng XM, Zhang Y, Chen X, Zhou K, Jin M, Cheng Z, Li X, Jiang L, Wang H, Wan J (2014) Rice LTG1 is involved in adaptive growth and fitness under low ambient temperature. Plant J 78:468–480
Ma Y, Dai XY, Xu YY, Luo W, Zheng XM (2015) COLD1 confers chilling tolerance in rice. Cell 160:1209–1221
Mao DH, Chen CY (2012) Colinearity and similar expression pattern of rice DREB1s reveal their functional conservation in the cold-responsive pathway. PLoS ONE 7:e47275
Mao D, Yu L, Chen D (2015) Multiple cold resistance loci confer the high cold tolerance adaptation of Dongxiang wild rice (Oryza rufipogon) to its high-latitude habitat. Theor Appl Genet 128:1359–1371
McCouch SR, Teytelman L, Xu YB, Lobos KB, Clare K, Walton M, Fu BY, Maghirang R, Li ZK, Xing YZ, Zhang QF, Kono I, Yano M, Fjellstrom R, DeClerrck G, Schneider D, Cartinhour S, Ware D, Stein L (2002) Development of 2240 new SSR markers for rice (Oryza sativa L.). DNA Res 9:199–207
Meng L, Li HH, Zhang LY, Wang JK (2015) QTL IciMapping: integrated software for genetic linkage map construction and quantitative trait locus mapping in biparental populations. Crop J 3:269–283
Qian Q, Zeng DL, He P, Zheng XW, Chen Y, Zhu LH (1999) QTL analysis of the rice seedling cold tolerance in a double haploid population derived from anther of a hybrid between Indica and Japonica rice. Chin Sci Bull 44:2402–2408
Qiao YL, Han LZ, An YP, Zhang YY, Cao GL, Koh HJ (2004) Molecular mapping of QTLs for cold tolerance at the budburst period in rice. Agric Sci China 11:1–6
Qiao JL, Liang C, Sun ZX, Xing YZ, Li J, Xu XY, Mei HW, Luo LJ (2007) A major QTL associated with cold tolerance at seedling stage in rice (Oryza sativa L.). Euphytica 158:87–94
Rao YC, Yang YL, Huang LC, Pan JW, Ma BJ, Qian Q, Zeng DL (2013) Research progress on cold stress in rice. Mol Plant Breed 11:443–450
Rogers OS, Bendich AJ (1988) Extraction of DNA from plant tissue. In: Gelvin SB, Schilperoof RA (eds) Plant molecular biology manual. Kluwer Academic Publishers, Dordrecht, pp 1–10
Sakurai J, Ishikawa F, Yamaguchi T, Uemura M, Maeshima M (2005) Identification of 33 rice aquaporin genes and analysis of their expression and function. Plant Cell Physiol 46:1568–1577
Suh JP, Roh JH, Cho YC, Han SS, Kim YG, Jena K, Shinozaki K (2009) The Pi40 gene for durable resistance to rice blast and molecular analysis of Pi40-advanced backcross breeding Lines. Phytopathology 99:243–250
Wang QY, Guan YC, Wu YR, Chen HL, Chen F, Chu CC (2008) Overexpression of a rice OsDREB1F gene increases salt, drought, and low temperature tolerance in both Arabidopsis and rice. Plant Mol Biol 67:589–602
Wu LL, Wu J, Liu YX, Gong XD, Xu JL, Lin DZ, Dong YJ (2016) The rice pentatricopeptide repeat gene TCD10 is needed for chloroplast development under cold stress. Rice 9:67
Wuriyanghan H, Zhang B, Cao WH, Ma B, Lei G, Liu YF, Wei W, Wu HJ, Zhang JS (2009) The ethylene receptor ETR2 delays floral transition and affects starch accumulation in rice. Plant Cell 21:1473–1494
Xiao N, Huang WN, Li AH, Gao Y, Li YH, Pan CH, Ji H, Zhang XX, Dai Y, Dai ZY, Chen JM (2015) Fine mapping of the qLOP2 and qPSR2-1 loci associated with chilling stress tolerance of wild rice seedlings. Theor Appl Genet 128:173–185
Xiao N, Gao Y, Qian H, Gao Q, Wu Y, Zhang D, Wang Z, Zhang X, Yu L, Li Y, Pan C, Liu G, Zhou C, Jiang M, Huang N, Chen Z, Liang C, Dai Z, Chen J, Li A (2018) Identification of genes related to cold tolerance and a functional allele that confers cold tolerance. Plant Physiol. https://doi.org/10.1104/pp.18.00209
Xu S, Tao Y, Yang Z, Chu J (2002) A simple and rapid methods used for silver staining and gel preservation. Hereditas 24:335–336
Xu ML, Chen SY, Mo X, Yan HM (2014) Progresses on cloning of cold tolerance related genes in rice. Life Sci Res 18:162–166
Yang ZM, Li DY, Mao DH, Liu X, Ji CJ, Li XB, Zhao XF, Cheng ZK, Chen CY, Zhu LH (2013) Overexpression of micro RNA 319 impacts leaf morphogenesis and leads to enhanced cold tolerance in rice (Oryza sativa L.). Plant Cell Environ 36:2207–2218
Yang TF, Zhang SH, Zhao JL, Huang ZH, Zhang GQ, Liu B (2015) Meta-analysis of QTLs underlying cold tolerance in rice (Oryza sativa L.). Mol Plant Breed 13:1–15
Zhang ZH, Su L, Wei L (2005) A major QTL conferring cold tolerance at the early seedling stage using recombinant inbred lines of rice (Oryza sativa L.). Plant Sci 168:527–534
Zhang ZY, Li JH, Li F, Liu HH, Yang WS, Chong K, Xu YY (2017) OsMAPK3 phosphorylates OsbHLH002/OsICE1 and inhibits its ubiquitination to activate OsTPP1 and enhances rice chilling tolerance. Dev Cell 43:731–743
Zhao ZW, Liu JF, Qi JS, Jin L, Li SG (2007) Genetic analysis on overwintering character of glutinous 89-1. Agric Sci China 6:1035–1042
Zhao ZW, Liu JF, Qi JS, Chen KF, Ju YJ, Cai JJ (2008) Study on cold tolerance of one share of specific rice resource in different growth periods. J Anhui Agric Sci 36:929–957
Zhao ZW, Luo AC, Le T, Li SG (2012a) Identification of quantitative trait locus for overwintering germinability in rice (Oryza sativa L.). J Integr Agric 11:1767–1774
Zhao MH, Zhang WZ, Ma DR, Chen WH (2012b) Riview of the cold tolerance gene studies on rice. J Shenyang Agric Univ 43:711–718
Zheng JX, Ma ZF, Song JD, Liu C, Li YT, Huang DH, Wei SL, Zhang YX, Mi K, Huang JY, Chen M, Meng JR, Li RB, Chen BS (2011) Identification and mapping of QTLs for cold tolerance at the seedling stage in common wild rice (Oryza rufipogon). China J Rice Sci 25:52–58
Acknowledgements
This research was supported by the National Natural Science Foundation of China (31670326), the key Natural Science Foundation of Chongqing (CSTC2015jcyjBX0091), the Program for Innovative Research Team in University, Chongqing (CXTDX201601018).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Deng, X., Gan, L., Liu, Y. et al. Locating QTLs controlling overwintering seedling rate in perennial glutinous rice 89-1 (Oryza sativa L.). Genes Genom 40, 1351–1361 (2018). https://doi.org/10.1007/s13258-018-0731-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13258-018-0731-z