Abstract
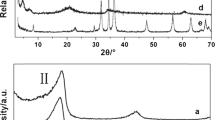
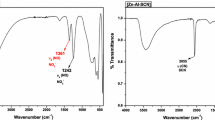
Intercalation behaviour of layered-double hydroxide (LDH) with short-chain intercalants (\({-}(\hbox {CH}_{2})_{n}{-}, n<9\)) is significantly difficult and less reported than with long-chain intercalants. The present study reports an efficient way to intercalate LDH with short-chain intercalants (\(n=4\) and 8) and investigates the effect of layer charge on intercalation behaviour of LDHs. Short-chain anionic surfactants were successfully intercalated with synthetic LDHs \([\hbox {Zn}_{1-x}\hbox {Al}_{x}(\hbox {OH})_{2}\hbox {NO}_{{3}}{\cdot } n\hbox {H}_{2}\hbox {O},\,x=0.2{-}0.33]\) by an ion-exchange intercalation technique in a slightly acidic medium (\(\hbox {pH}=5.4\)). The adverse effect of a carbonate anion was avoided by performing the ion-exchange intercalation in slightly acidic medium (\(\hbox {pH}=5.4\)). It was found that basal spacing \((d_{003})\) and experimental organic loading of intercalated LDH (O-LDH) increase monotonically with increasing anion-exchange capacity of LDH and intercalant chain length. The evolution of intercalated LDH (O-LDH) structures with increasing intercalant chain length and layered charge has been deciphered by correlating basal spacing of O-LDHs (by X-ray powder diffraction), organic loading data (by thermogravimetric analysis) and molecular conformation of O-LDHs (by Fourier-transform infrared spectroscopy) within the LDH gallery. Successful intercalation of LDH with these short-chain intercalants in slightly acidic medium has not been reported previously.







Similar content being viewed by others
References
Motokura K, Nishimura D, Mori K, Mizugaki T, Ebitani K and Kaneda K 2004 J. Am. Chem. Soc. 126 5662
Zou X, Goswami A and Asefa T 2013 J. Am. Chem. Soc. 135 17242
Shu Y, Yin P, Wang J, Liang B, Wang H and Guo L 2014 Ind. Eng. Chem. Res. 53 3820
Chakraborty C, Dana K and Malik S 2011 J. Phys. Chem. C 115 1996
Bi B, Xu L, Xu B B and Liu X Z 2011 Appl. Clay Sci. 54 242
Starukh G, Rozovik O and Oranska O 2016 Nanoscale Res. Lett. 11 10
Prasanna S R, Rao A P and Kamath P V 2006 J. Colloid Interface Sci. 304 292
Radha A K, Kamath P V and Shivakumara C 2007 J. Phys. Chem. B 111 3411
Miyata S and Kumara T 1973 Chem. Lett. 2 843
Costa F R, Leuteritz A, Wagenknecht U, Jehnichen D, Haussler L and Heinrich G 2008 Appl. Clay Sci. 38 153
Crepaldi E L, Pavan P C and Valim J B 1999 Chem. Commun. 2 155
Ayala-Luis K B, Koch C B and Hansen H C B 2010 Appl. Clay Sci. 48 334
Costa F R, Leuteritz A, Wagenknecht U, Landwehr M A D, Jehnichen D, Haeussler L et al 2009 Appl. Clay Sci. 44 7
Crepaldi E L, Pavan P C, Tronto J and Valim J B 2002 J. Colloid Interface Sci. 248 429
Pavan P C, Crepaldi E L and Valim J B 2000 J. Colloid Interface Sci. 229 346
Refait P, Drissi S H, Pytkiewicz J and Genin J M R 1997 Corros. Sci. 39 1699
Zhang H, Zhang F Z, Ren L L, Evans D G and Duan X 2004 Mater. Chem. Phys. 85 207
Pavan P C, Gomes G D and Valim J B 1998 Microporous Mesoporous Mater. 21 659
Prevot V, Forano C and Besse J P 1998 Inorg. Chem. 37 4293
Pavan P C, Crepaldi E L, Gomes G D and Valim J B 1999 Colloids Surf. A 154 399
Antonyraj C A, Koilraj P and Kannan S 2010 Chem. Commun. 46 1902
Shamim M and Dana K 2016 Thermochim. Acta 632 64
Ganguly S, Dana K, Mukhopadhyay T K and Ghatak S 2011 Clays Clay Miner. 59 13
Moyo L, Nhlapo N and Focke W W 2008 J. Mater. Sci. 43 6144
Nejati K and Rezvani Z 2012 J. Exp. Nanosci. 7 412
Ogawa M and Hiramine M 2014 Cryst. Growth Des. 14 1516
Xing F-F, Ni Z-M, Wang P, Pan G-X, Xia S-J and Wang L-G 2007 Acta Chim. Sin. 65 2738
Choy J-H, Kwak S-Y, Jeong Y-J and Park J-S 2000 Angew. Chem. Int. Ed. 39 4041
Nalawade P, Aware B, Kadam V J and Hirlekar R S 2009 J. Sci. Ind. Res. 68 267
Anbarasan R, Lee W D and Im S S 2005 Bull. Mater. Sci. 28 145
Barahuie F, Hussein M Z, Fakurazi S and Zainal Z 2014 Int. J. Mol. Sci. 15 7750
Li S, Lu J, Xu J, Dang S, Evans D G and Duan X 2010 J. Mater. Chem. 20 9718
Williams G R, Dunbar T G, Beer A J, Fogg A M and O’Hare D 2006 J. Mater. Chem. 16 1222
Ruan X, Huang S, Chen H and Qian G 2013 Appl. Clay Sci. 72 96
Ganguly S, Dana K, Mukhopadhyay T K and Ghatak S 2011 J. Therm. Anal. Calorim. 105 199
Bai Z M, Wang Z Y, Zhang T G, Fu F and Yang N 2013 Appl. Clay Sci. 75–76 22
Clearfield A, Kieke M, Kwan J, Colon J L and Wang R C 1991 J. Inclusion Phenom. Mol. Recognit. Chem. 11 361
Sarkar M, Dana K and Ghatak S 2011 J. Mol. Struct. 1005 161
Jankovič L, Madejová J, Komadel P, Jochec-Mošková D and Chodák I 2011 Appl. Clay Sci. 51 438
Wei M, Pu M, Guo J, Han J B, Li F, He J et al 2008 Chem. Mater. 20 5169
Babakhani S, Talib Z A, Hussein M Z and Ahmed A A A 2014 Int. J. Spectrosc. 2014 1
Lagaly G 1976 Angew. Chem. 15 575
Meyn M, Beneke K and Lagaly G 1990 Inorg. Chem. 29 5201
Acknowledgements
The research work was funded by CSIR under GLASSFIB project and one of the authors (MS) acknowledges the ‘SRF-GATE’ research fellowship granted to him by CSIR, New Delhi, India.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Shamim, M., Dana, K. Intercalation of \(\hbox {LDH NO}_{{3}}\) with short-chain intercalants. Bull Mater Sci 42, 25 (2019). https://doi.org/10.1007/s12034-018-1704-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12034-018-1704-1