Abstract
Airway management and ventilation are central to the resuscitation of the neurologically ill. These patients often have evolving processes that threaten the airway and adequate ventilation. Furthermore, intubation, ventilation, and sedative choices directly affect brain perfusion. Therefore, airway, ventilation, and sedation was chosen as an emergency neurological life support protocol. Topics include airway management, when and how to intubate with special attention to hemodynamics and preservation of cerebral blood flow, mechanical ventilation settings, and the use of sedative agents based on the patient’s neurological status.
Similar content being viewed by others
Introduction
Intubation of the acutely brain-injured patient can be a matter of life or death. Failure to intubate a patient with rapidly progressive neurological decline may result in respiratory arrest, secondary brain injury from hypoxia, acidosis, or elevated intracranial pressure (ICP), and severe aspiration pneumonitis or acute respiratory distress syndrome (ARDS).
Conversely, the process of induction and intubation can elevate intracranial hypertension when a mass lesion is present, complete a massive infarction when brain tissue is marginally perfused, and result in temporary loss of the neurological examination at a time when neurological and neurosurgical decision-making is required.
The goals of airway management in neurological patients are to maintain adequate (but not excessive) oxygenation and ventilation, preserve cerebral perfusion, and prevent aspiration. A neurological assessment prior to the administration of sedating and paralyzing medications should be performed to provide a functional baseline, whereby neurological and neurosurgical decision-making may ensue.
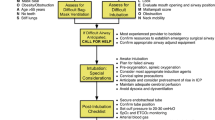
The emergency neurological life support (ENLS) suggested algorithm for the initial management of airway, ventilation, and sedation is shown in Fig. 1. Suggested items to complete within the first hour of evaluating a patient are shown in Table 1.
Assessing the Need for Intubation
Patients in severe respiratory distress or impending arrest should be intubated without delay. Additionally, a patient who cannot “protect his airway” because of depressed mental status or vomiting with aspiration may need tracheal intubation. Intubation has the potential for complications, creates significant hemodynamic disturbances, and should not be undertaken without a risk benefit assessment. However, it should not be delayed when necessary. The decision to intubate is influenced by factors specific to patient physiology, clinical environment, and the anticipated course of care.
In the prehospital or emergency department (ED) environment, a stuporous or comatose patient with an unknown diagnosis requiring extended transport, transfer, imaging, or invasive procedures may be most appropriately managed by intubation. The same patient with a known physiology, an anticipated stable or improving course, and no planned transportation may not require intubation and might be managed by a more conservative approach.
With these considerations in mind, there are four commonly accepted indications to intubate:
Failure to Oxygenate
This finding may be determined by pulse oximetry (limitations include regional or systemic hypoperfusion, severe anemia, and opaque nail polish), arterial blood gas analysis, or the patient’s visual appearance (cyanosis).
Failure to Ventilate
Ventilation is assessed by capnometry through nasal cannula monitoring (does not always correlate with pCO2, but provides a valuable tool for monitoring trends in ventilation) [1], arterial blood gas analysis, or gross visual appearance (excessive or inadequate work of breathing).
Failure to Protect the Airway
Airway protection determined by bulbar function, airway anatomy, quantity and quality of secretions, strength of cough reflex, and ability to swallow after suctioning [2]. The presence of a gag reflex is an inaccurate method of assessing airway protection [3].
Anticipated Neurological or Cardiopulmonary Decline Requiring Transport or Immediate Treatment
Anticipation of the trajectory of the patient’s condition can avoid rushed or emergent intubations and allow for appropriate preparation for the procedure.
Airway Assessment
Full assessment of the airway includes determination of the ease of bag-mask ventilation and the potential for a difficult intubation. Expectation of a difficult airway enables appropriate planning and use of available advanced airway equipment. This may include assistance from airway management specialists (e.g., call for anesthesia assistance), use of specialized devices, such as fiberoptic bronchoscopy, or mobilizing equipment for a cricothyrotomy). In certain circumstances availability of a laryngeal mask airway (LMA), may be warranted.
The “LEMON” pneumonic helps to predict the difficult airway [3]:
- L:
-
Look
- E:
-
Evaluate the mouth opening and airway position
- M:
-
Mallampati score
- O:
-
Obstruction
- N:
-
Neck mobility
The “MOANS” pneumonic predicts difficulty of bag-mask ventilation [3]:
- M:
-
Mask seal
- O:
-
Obesity/obstruction
- A:
-
Age > 55
- N:
-
No teeth
- S:
-
Stiff lungs
Decision Made to Intubate: Perform Neurological Assessment
Whenever possible, urgent management of the airway should coincide with a rapid but detailed neurological assessment. The examination can typically be conducted in 2 min or less.
The pre-sedation/pre-intubation neurologic exam establishes a baseline that is used to assess therapeutic interventions (e.g., patients with stroke, seizures, hydrocephalus, or other disorders) or may identify injuries that are at risk of progressing (e.g., unstable cervical spine fractures). The assessment identifies the type of testing required and may help to limit unnecessary interventions, such as radiological cervical spine clearance. In general, the pre-intubation neurological assessment is the responsibility of the team leader who is coordinating the resuscitation. Findings should be documented and communicated directly to the team that assumes care of the patient.
As demonstrated in Appendix 1, the pre-intubation neurological examination includes an assessment of:
-
Level of arousal, interaction, and orientation, as well as an assessment of simple cortical functions, such as vision, attention, and speech comprehension and fluency
-
Cranial nerve function
-
Motor function of each individual extremity
-
Tone & reflexes
-
Comment on subtle or gross seizure activity
-
Cervical spine instability
-
Sensory level in patients with suspected spinal cord injury
Intubating the Patient with Intracranial Pathology
Rapid sequence intubation (RSI) is the preferred method of securing the airway in patients with suspected elevated ICP. RSI limits elevation of ICP often associated with the physiologic responses to laryngoscopy [4–7]. The presence of coma should not justify proceeding without pharmacological agents, or administration of only a neuromuscular blocking agent without appropriate pre-treatment and induction agents. Although the patient may seem unresponsive, laryngoscopy and intubation often provoke reflexes that elevate ICP unless appropriate pre-treatment and induction agents are used [8].
Outcomes in patients with intracranial catastrophes are related to the maintenance of both brain perfusion and oxygenation. Consequently, close assessment and preservation of these two parameters are critical. Cerebral perfusion pressure (CPP) is the physiologic correlate for blood flow to the brain and is measured by the difference between the mean arterial pressure (MAP) and the ICP:
CPP = MAP − ICP
It is generally recommended that the ICP be maintained below 20 mmHg, MAP between 80 and 110 mmHg, and CPP at a minimum of 50 mmHg [9]. The needs of the individual patient are variable, and perfusion goals should consider these distinctions. Because the ICP may not be known at the time of urgent intubation, clinicians should anticipate elevated ICP in patients with a mass lesion, such as hematoma, hydrocephalus, tumor, or cerebral edema, and choose an appropriate BP target accordingly. When the airway is manipulated, two responses may exacerbate intracranial hypertension. The reflex sympathetic response (RSR) results in increased heart rate, increased blood pressure, and, consequently, increased ICP. The direct laryngeal reflex stimulates an increase in ICP independent of the RSR [3]. Although the RSR may be dangerous in a hypertensive patient, pre-treatment of the RSR is not indicated in a hypotensive patient with known or suspected increased ICP [10]. Elevations in ICP should be mitigated by minimizing airway manipulation (the most experienced person should perform the intubation) and administering medications.
Clinicians must recognize that many neurologically impaired patients have compromised cerebral blood flow (CBF) even with a normal ICP. For example, patients with ischemic stroke, vasospasm, and hypoxic-ischemic brain injury often have impaired auto-regulation and are each critically sensitive to decreases in blood pressure and CBF. In these patients, particular efforts should be made to maintain MAP and CPP goals. Vasodilators, which reverse physiological shunting, should be avoided in this patient population.
Common pre-medications used to prevent increased ICP during intubation include
Lidocaine
Administered intravenously at a dose of 1.5 mg/kg 60–90 s before intubation, lidocaine attenuates the direct laryngeal reflex. There is mixed evidence that it mitigates the RSR [3, 11]. It is not associated with drop in MAP.
Fentanyl
At doses of 2–3 μg/Kg, fentanyl attenuates the RSR associated with intubation, and is administered as a single pre-treatment dose over 30–60 s in order to reduce chances of apnea or hypoventilation before induction and paralysis [12]. It is generally not used in patients with incipient or actual hypotension, or those who are dependent on sympathetic drive to maintain an adequate blood pressure for cerebral perfusion.
Induction is performed using an agent that will not adversely affect CPP (see also Table 4).
Etomidate
Etomidate is a short-acting imidazole derivative that provides sedation and muscle relaxation with minimal hemodynamic effect. It is considered the most hemodynamically neutral induction agent of all commonly used induction agents and a drug of choice for patients with elevated ICP [13, 14].
Propofol
At a dose of 2 mg/kg intravenous (IV) push, propofol is an alternative induction agent. However, it is also a potent vasodilator that may cause hypotension and may not be appropriate for patients with threatened cerebral perfusion, unless concurrently administered with a vasopressor agent [15].
Thiopental
At a dose of 3 mg/kg IV push, thiopental confers cerebroprotective effect by decreasing the basal metabolic rate of oxygen utilization of the brain (CMRO2) and CBF, thus decreasing ICP. However, it is a potent venodilator and negative inotrope with a strong tendency to cause hypotension and reduce CPP, even in relatively hemodynamically stable patients [8].
Ketamine
Ketamine is a hemodynamically neutral dissociative agent administered at a dose of 2 mg/kg IV push. Historically, use of ketamine was generally avoided due to concerns related to ICP elevations. However, recent evidence suggests that when concurrent sedation is provided, it is safe in patients with elevated ICP. Its favorable hemodynamic profile may lead to more widespread use as an induction agent [16–18].
Succinylcholine
Succinylcholine is a depolarizing neuromuscular blocker often considered the agent of choice for intubation of acutely ill neurological patients with elevated ICP. It possesses a rapid onset and short duration of action. Although it has been associated with transient increases in ICP, the effect is not considered clinically significant [19]. Immobile and chronically ill neurologic patients are at risk for succinylcholine-induced hyperkalemia. This includes patients with prior brain or spinal cord injury and chronic neuromuscular disease, but also those with as little as 24–72 h of immobility [20]. Risk may be averted by using a non-depolarizing agent, such as rocuronium (at 1.2–1.4 mg/kg IV push) or the longer acting agents, pancuronium and vecuronium (at 0.1–0.2 mg/kg IV push).
ICP during intubation also rises due to body positioning and hypoventilation. Hypoventilation immediately causes increased pCO2, a potent acute cerebral vasodilator. When ICP is known or suspected to be elevated, the following approach is suggested (Fig. 2):
Intubation with elevated ICP [148]
-
The head of the bed should be brought flat as briefly as possible, and raised immediately following endotracheal tube placement. Reverse Trendelenburg positioning during intubation may be considered.
-
MAP must be preserved throughout the procedure, with a goal of 80–100 mmHg, but not lower than the pre-intubation blood pressure. If ICP is monitored, keep CPP > 50 mmHg.
-
If cervical spine injury is suspected, stabilize and immobilize the C-spine.
-
Pain, discomfort, agitation, and fear must be controlled with adequate analgesia and sedation.
-
Hypoventilation must be avoided, and quantitative end-tidal capnography monitoring is suggested in patients with elevated ICP or impaired cerebral perfusion, in whom dyscarbia would adversely affect cerebral blood flow.
-
Adequate pre-oxygenation with 100 % oxygen should be performed prior to induction, and the oxyhemoglobin saturation maintained >92 %. However, following successful intubation and hemodynamic stabilization, the FiO2 should be reduced to 0.5, or to the lowest fraction that assures a PaO2 in the range of 60–300 mmHg. Hypoxia increases ICP and can exacerbate brain injury. Please note that during hypothermic conditions, PaO2 and pCO2 should be corrected for acquisition temperature [21, 22].
Intubating the Patient with Brain Ischemia
In suspected or proven ischemic stroke, careful attention should be taken to avoid hypotension during induction and post-intubation. In the healthy state, the cerebrovascular circulation is well collateralized. During ischemic stroke, many patients possess an infarct core surrounded by a greater region of ischemic penumbra which can be demonstrated on perfusion computed tomography (CT) or magnetic resonance imaging (MRI). Under these circumstances, the ischemic penumbra consists of a region of vasodilated vessels, receiving maximal compensatory shunting from the adjacent cerebrovascular circulation. Hypertension and tachycardia often reflect a physiologic, not pathophysiologic, response to this ischemia and may be necessary to maintain perfusion of the ischemic territory.
Certain vasoactive agents may reverse regional vasoconstriction in normal areas of the brain that is necessary to maintain physiologic shunting of blood to the region of ischemia, even if they do not drop the systemic blood pressure or alter the global CPP. An episode of relative or actual hypotension can dramatically increase brain infarction size by “stealing” blood flow from the maximally dilated watershed territories between vascular distributions.
Brain ischemia is not limited to ischemic stroke but is also variably present in patients with vasospasm, traumatic brain injury (TBI), intracranial and extracranial cerebrovascular stenosis, and hypoxic-ischemic encephalopathy following resuscitation from cardiac arrest. Strong correlations between episodic hypotension and poor neurological outcome have been noted in the critical hours following resuscitation from TBI and cardiac arrest [23–26]. The intubating clinician should be aware of the risks of even a transient decrease in CBF and strive to maintain CBF and systemic vascular tone during airway management.
Additionally, brain ischemia is worsened by the impact of hyperventilation upon vascular tone. Normocapnia should be maintained during intubation, and early correlation of an arterial CO2 sample with ETCO2 is suggested to enable non-invasive tracking of ventilation [1, 27].
Intubating the Patient with Neuromuscular Weakness
Patients with neuromuscular weakness and acute or impending respiratory failure require a special approach to airway management. Respiratory failure in patients with neuromuscular disease is often associated with a weak cough and failure to clear secretions from the lower airways, leading to pneumonia, shunting, and an inability to meet ventilatory demands. Concerns over change in MAP with induction and intubation are much less often a concern in this population.
Although some patients with neuromuscular disease require immediate intubation, those with preserved bulbar function and reasonable functional ventilatory reserves may undergo a trial of non-invasive ventilation combined with airway clearance by the frequent use of chest physiotherapy and a cough-assist device [28–30].
Any patient with neuromuscular weakness that complains of dyspnea, should undergo an assessment of respiratory function that includes (see also the Acute Weakness protocol):
-
Arterial blood gas measurement
-
Serial pulmonary function testing to include negative inspiratory force (NIF) and vital capacity (FVC)
-
Assessment of bulbar function, neck strength, and cough
Candidates for intubation include patients with neuromuscular weakness and bulbar dysfunction, those who have a rapidly progressive course, and those who do not rapidly stabilize gas exchange and work of breathing with non-invasive ventilation [28]. Because of the potential for exacerbating weakness and prolonged effects of the medications, the administration of corticosteroids, muscle relaxants, or neuromuscular blocking agents is discouraged.
In myasthenia gravis, succinylcholine is safe but requires approximately 2.5 times the dose to get the same effects [31]. Non-depolarizing agents, such as rocuronium, are also safe but will have a prolonged duration [31]. In conditions, such as Guillain–Barre, succinylcholine can precipitate life-threatening hyperkalemia, and only non-depolarizing agents should be used. Rocuronium is safe in most cases of neuromuscular weakness at a dose of 0.6–1.2 mg/kg.
Intubating the Patient with Cervical Spine Injury
Cervical spine injury should be suspected during direct neck trauma or blunt head trauma resulting in loss of consciousness. During care of these patients, measures must be taken to protect the spinal cord during any movements or procedures, including intubation. Pre-intubation airway maneuvers, including jaw tilt and the bag-mask ventilation, cricoid pressure, and direct laryngoscopy itself, can all injure the spinal cord when cervical instability is present.
Basic principles of cervical spine stabilization have been developed and refined and can be reviewed in the American College of Surgeons’ Advanced Trauma Life Support (ATLS) course [30]. The use of cricoid pressure is no longer recommended during intubation, it definitely should not be implemented in patients with cervical spine injury, as it may cause posterior displacement of the cervical spine [32, 33].
While endotracheal intubation requires precautionary measures, its performance is often necessary to adequately address greater risks of hypoxia, hypoventilation, and large-volume aspiration. In-line spinal stabilization helps minimize risk of cervical injury when direct laryngoscopy is performed [34]. However, video and enhanced optical intubating devices are currently preferred when intubating patients at risk of cervical spine injury, since these allow ease of intubation with the patient’s cervical spine maintained in a neutral position.
Post-intubation and Ventilation
Basic Ventilator Settings
Immediately following intubation, respiratory and hemodynamic homeostasis must be restored. Except in situations of acute brain herniation, the goals of mechanical ventilation are:
-
Normalization of oxygenation utilizing the lowest FiO2 that will maintain hemoglobin saturation >94 %
-
Normalization of ventilation to achieve a systemic pH of 7.3–7.4, and pCO2 or ETCO2 to 30–40 mmHg
-
Normalization of the work of breathing
-
Prevention of ventilator-induced lung injury
In most circumstances, clinicians should default to volume-cycled ventilation at 6–8 cc/kg of ideal body weight and a respiratory rate of 12–14 per minute. However, these settings must take into account the patient’s minute ventilation prior to induction. Normal pCO2 is an appropriate target unless there is chronic hypercarbia (i.e., severe COPD or sleep-disordered breathing). In situations of chronic hypercarbia, the admission bicarbonate level should be used to estimate the “baseline” pCO2, and that level should subsequently be used as the target. When metabolic acidosis is present, ventilation should target normal pH.
Titrate Ventilation
Induced Hyperventilation: Ventilation, Carbon Dioxide Tension, and Clinical Outcome
Hyperventilation causes cerebral vasoconstriction and decreased CBF, while hypoventilation causes cerebral vasodilation and increased ICP [35]. Dysventilation (and especially hyperventilation) is associated with poor outcomes in TBI [36–39]. The Brain Trauma Foundation recommends targeting eucapnia in patients with brain trauma [9, 40].
However, the relationship between arterial and central pH and pCO2 is complex and incompletely understood. During concomitant metabolic acidosis and TBI, CNS pH and CBF are often preserved despite severe systemic acidosis due to the blood–brain barrier and the central nervous system (CNS) buffering capacity [41]. Alternatively, in patients with chronic respiratory acidosis, the set-point of cerebral CO2 reactivity changes. It is, therefore, recommended that mechanical ventilation be adjusted to correct the pH and not the pCO2, or that the estimated “pre-morbid” pCO2 target be used (see Table 2 below). This is a practical goal since ventilating these patients to “normal” pCO2 targets may be extremely difficult or impossible when obstructive lung disease is present.
Herniation: Intentional Hyperventilation to Treat Brain Herniation and Increased ICP
When a patient develops brain herniation with elevated ICP, hyperventilation is an appropriate temporizing intervention designed to acutely decrease ICP and prevent subsequent neuronal injury and death [42, 43]. Maximal cerebral vasoconstriction is achieved at a pCO2 near 20 mmHg. Therefore, hyperventilation below this level results in no further therapeutic advantage and may impede venous return to the heart, decrease blood pressure, and exacerbate cerebral hypoperfusion.
During hyperventilation, end-tidal CO2 monitoring (quantitative capnography) is suggested. As soon as other treatments to control ICP are in place (e.g., blood pressure support, osmotherapy, surgical decompression, hypothermia, metabolic therapy), hyperventilation should be weaned to restore brain perfusion [44].
Hyperventilation for increased ICP is not safe or effective when employed for a prolonged period [9, 45, 46]. Hyperventilation severely reduces CBF, increases the volume of ischemic tissue, and, may result in rebound elevation of ICP during weaning [47–49]. When prolonged (mild) hyperventilation is employed, it is strongly recommended that end-tidal CO2 and cerebral metabolic monitoring (jugular oximetry, CBF, brain tissue oxygen, or cerebral microdialysis) be used to verify the adequacy of tissue perfusion.
Acidemic and Alkalemic Hypocarbia: Potential for Suppression of Spontaneous Hyperventilation
There are two circumstances that should be considered in patients with spontaneous hypocarbia: those whose response to systemic metabolic acidosis accounts for their high ventilatory demand, and those (alkalotic) patients in whom ventilation exceeds systemic metabolic needs.
In patients whose ventilation is driven by metabolic acidosis, suppression of the respiratory drive with sedation or neuromuscular blockade is not recommended, unless direct measurement of brain chemistry suggests that hyperventilation is driving cerebral metabolic crisis. Under these circumstances, clinicians must find another means to buffer pH.
Mechanically ventilated TBI patients presenting with hypocarbia have worse outcomes than their normocarbic peers. However, non-intubated patients presenting with hypocarbia do not exhibit similar findings, suggesting that hypocarbia, in this setting, may be a physiologic response and should not be suppressed [37].
Despite decades of observation and consideration, little is known about alkalemic hypocarbia in patients with an acute brain injury. Alkalemic hypocarbia following brain injury may be theoretically explained by a variety of physiologic and pathophysiologic mechanisms, more than one of which may be present in an individual patient:
-
Brain tissue acidosis requiring acute hyperventilation as a buffer until CNS bicarbonate-generating compensatory mechanisms can catch up
-
Inadequately treated pain, anxiety, fear, or agitation
-
Fever
-
Auto-regulation of elevated ICP
-
Heme breakdown products or a lactic acid load in the ventricular system
-
Direct pressure on chemoreceptors present in the floor of the 4th ventricle
-
Physiologic dysregulation of the medullary respiratory rhythm generator, which has afferent inputs from the pons, mesencephalon, and higher cortical centers
A recent trial of patients with severe brain injury monitored for brain tissue oxygen showed brain tissue hypoxia worsened when EtCO2 values were reduced by spontaneous alkalemic hyperventilation, suggesting possible harm.[50] It is rarely known whether alkalemic hypocapnia is a physiologic or pathophysiologic process, and suppression of this respiratory activity is recommended only in response to evidence that hyperventilation is causing direct harm, either by inducing cerebral ischemia or indirectly by increased systemic metabolic demands and work of breathing.
Oxygenation and Outcomes
Hypoxia is a major source of secondary brain injury [51], and the injured and ischemic brain is particularly vulnerable to low oxygen levels. Similarly, supra-physiologic levels of oxygen provided to acutely ill patients have the potential to worsen reperfusion injury and outcomes [52, 53]. Hyperoxia drives the formation of reactive oxygen species, overwhelming antioxidants at sites of tissue injury; directly injures respiratory epithelium and alveoli inducing inflammation; drives hypercarbia; and leads to absorption atelectasis in the lung. Hyperoxia (PaO2 > 300 mmHg) immediately following resuscitation is independently associated with poor outcomes in TBI and cardiac arrest [53, 54], though not all published data are in accordance [55–57].
It is recommended that 100 % oxygen be provided for pre-oxygenation immediately prior to intubation, but that oxygen be immediately weaned following intubation to 50 %, or the lowest FiO2 that will support an oxyhemoglobin saturation of 95–100 %. This normoxic resuscitation strategy is recommended in the 2010 American Heart Association Guidelines for post-resuscitation care after cardiac arrest [55].
Oxygenation and Ventilation Monitoring
Oxygenation should be monitored by pulse oximetry or by arterial blood gas analysis when oximetry is suspected to be inaccurate. Conditions of poor perfusion to the extremities, acidosis, vasopressor use, anemia, carboxyhemoglobinemia and methemoglobinemia, and hypoxia all have the potential to compromise the accuracy of pulse oximetry measurements [58].
Ventilation is traditionally monitored by serial arterial blood gas analysis, though venous blood gas analysis may provide an adequate surrogate when arterial samples cannot be obtained. End-tidal quantitative capnography of exhaled gases provides an appealing continuous measurement and is extremely useful to monitor trends in ventilation. One study showed that severely hyperventilated head trauma patients (pCO2 <25 mmHg) in the prehospital environment had higher mortality, and that use of quantitative capnography by paramedics significantly decreased the incidence of hyperventilation [59]. A similar study in patients with major trauma showed a much higher incidence of “normocapnia” on hospital arrival when ETCO2 was monitored by medics.
Because ETCO2 measurements reflect not only ventilation but also systemic perfusion, the correlation between ETCO2 and pCO2 in the blood is variable, especially when severe physiologic derangements are present [60]. In an inpatient environment, ETCO2 measurements should always be correlated with an arterial pCO2 sample. ETCO2 and pCO2 may also vary significantly when lung disease and ventilation-perfusion mismatch are present [61].
Lung Injury
Patients with acute lung injury (ALI) or the ARDS are vulnerable to lung injury known variably as ventilator-induced lung injury (VILI) or ventilator-associated lung injury (VALI). ALI and ARDS represent a spectrum of disease characterized by bilateral parenchymal pulmonary infiltrates, significant hypoxia, absence of left ventricular dysfunction, and hyperacuity of onset [62]. These patients often require high levels of oxygen and elevated mean airway pressures to achieve adequate gas exchange.
Patients with ALI and ARDS often have high circulating levels of inflammatory mediators, which are associated with injury to other vital organs, and are exacerbated by injurious techniques of ventilation. VALI is thought to be caused by:
-
Barotrauma: induced by high ventilator pressures, particularly a high plateau pressure
-
Volutrauma: induced by higher tidal volumes, often despite low ventilator pressures
-
Atelectrauma: shearing injury due to recurrent opening and closing of alveolar sacs that may lack adequate surfactant
-
High inspired fraction of oxygen
-
High levels of circulating inflammatory cytokines
Many modes and techniques of ventilation have been proposed to manage the severe gas exchange abnormalities associated with ARDS, though only a few important issues are covered here.
Patients with ALI or ARDS should be ventilated using a strategy of low tidal volumes (6 cc/kg), low plateau pressures (<30 mmHg), adequate positive end expiratory pressure (PEEP) to prevent cyclic collapse of alveolar units, and inhaled oxygen fraction rapidly weaned to 0.6 or less.
Although the landmark study of low tidal volume mechanical ventilation [63] emphasizes permissive hypercarbia, elevation in carbon dioxide is a potent modulator of CBF and this strategy must be balanced with concerns for intracranial hypertension. Several small studies suggest that lung protective ventilation strategies causing mild hypercarbia in patients with elevated ICP may be tolerated [63, 64], but more data are needed before this may be considered safe in routine practice. Prone positioning seems to increase ICP [65], although several studies show that a small increase in ICP may be more than offset by dramatic improvements in oxygenation. [66, 67].
When lung compliance is low, airway pressure has the potential to be transmitted to the intrathoracic vessels and indirectly increase ICP. Although this physiology was once used to justify low-PEEP ventilation in patients with head injury, subsequent research has shown PEEP in brain injured patients to be well tolerated, especially when lung compliance is poor and adequate blood pressure is maintained [68, 69]. Ventilation without PEEP is discouraged, due to the likelihood of atelectrauma [70]. However, the relationship of airway pressure to ICP, CPP, and cerebral perfusion is of concern, and should be individually reviewed based on each patient’s physiology [71–73].
Sedation
Sedation use in the neurocritically ill has both benefits and drawbacks. Sedation may be needed to alleviate fear and anxiety, reduce ICP and cerebral oxygen consumption, facilitate intubation and tolerance of mechanical ventilation, or to reduce sympathetic nervous activity. Conversely, sedation makes accurate neurological examination, the cornerstone of clinical assessment, difficult or impossible. Acute changes in brain physiology become difficult to detect, and the accuracy of neuroprognostication is decreased [74, 75].
Sedation may cause vasodilation, reducing cerebral perfusion due to hypotension and also reversing physiologically advantageous shunting of blood into areas of ischemia. It is possible that prolonged deep sedation may worsen cognitive outcomes and contribute to muscle weakness. Even short-acting sedatives are known to accumulate in fatty tissues, causing effects beyond their intended duration. Therapeutic and procedural decision-making are contingent upon an accurate neurological assessment. Especially in the first hours of care, the sedation of patients with unstable intracranial pathophysiology should be minimized when it can safely be limited.
Balancing the side effects and needs of sedation are often challenging. Despite each of the competing interests, adequate consideration must be made for patient comfort.
Necessity of Sedation
Complications associated with under-sedation include ventilator dysynchrony, patient injury, agitation, anxiety, device removal, and elevated ICP [76–89]. Adequate sedation is paramount in all therapeutic algorithms for the treatment of increased ICP [80, 81], since psychomotor restlessness, pain, and autonomic stress all adversely affect ICP, CBF, CPP, and the cerebral metabolic rate for oxygen metabolism (CMRO2). In this respect, adequate analgesia and sedation make an essential contribution to preventing or limiting secondary brain damage. Nevertheless, there are insufficient data to confirm that sedation and analgesia per se improves neurological outcome [80]. Conversely, in a general intensive care unit (ICU) population, the use of excessive sedation and analgesia contributes to increased duration of mechanical ventilation, longer length of stay in the ICU and hospital [82–85], increased rates of depression, post-traumatic stress disorder, infections, and long-term neurocognitive impairments [85–87].
Randomized controlled trials demonstrate that protocols requiring decreases in sedative doses or daily interruption of sedative and analgesic drugs can reduce lengths of mechanical ventilation and ICU stay and reduce drug doses administered [82–84]. It is important to recognize that these trials did not include patients who required uninterrupted sedation, such as those with status epilepticus and refractory intracranial hypertension. Daily sedation lowering in these patient populations may not be therapeutically prudent.
Unless deep sedation or general anesthesia is desired, analgesia should precede sedation. Analgesia-based “sedation” is an evolving trend in general critical care, and a recent randomized trial suggests improved outcomes among mechanically ventilated patients receiving primarily opioid analgesia with no sedation [88]. Many patients with adequate pain control do not require sedation, and, conversely, most sedative medications provide no analgesia. Sedation without pain control may be an important cause of delirium. Infusion of short acting analgesics allows for interruption and neurological assessment at intervals.
Environmental stimuli are important triggers of anxiety and agitation. Providing a calm and reassuring environment, with attention to day-night cycles, restriction of noise, use of appropriate music, and/or the reassuring presence of friends and family may decrease agitation and anxiety [89]. These environmental measures are especially important when a dominant-hemisphere lesion causes aphasia and attempts at verbal communication generate agitated behaviors. The use of “sitters” to re-direct and re-orient confused or agitated patients is preferred to the use of sedating medications.
Patients with acute brain injury often have deficits in short-term memory, concentration, and emotional control, and their confusion may cause agitation. Confused patients require gentle and repeated re-orientation to situation and circumstances, which may obviate the need for sedation.
Common Sedatives in Neurological Intensive Care
Propofol
Propofol is among the best-studied sedative agents used in neurological critical care. Pharmacologically, its lipid formulation allows for rapid penetration of the blood–brain barrier, resulting in rapid onset and cessation of action. It has potent and immediate depressant effects on cerebral electrical and metabolic activity, and it does not require renal or hepatic metabolism for elimination. Disadvantages include robust vasodilating and hypotensive effects, considerable intravenous lipid load, and the potential for the rare, but frequently fatal, propofol infusion syndrome. This syndrome is characterized by acidosis, hepatic failure, hypertriglyceridemia, and elevated creating kinase level. Propofol infusion syndrome may be fatal and is more common in children and adults when used at higher doses. A recent study comparing propofol to dexmedetomidine sedation in the neurocritically ill found high (30 %) incidences of severe hypotension in both groups. Caution must be utilized with these medications when concerns for brain ischemia are present [90].
Remifentanil
Remifentanil hydrochloride is a mu-opioid agonist exhibiting analgesic effects with a rapid onset and a short duration of action. It is an agent which can be used a part of a combined sedative analgesic approach. A recent mechanistic review suggests that remifentanil may be a cost effective sedative alternative when ICU length of stay is considered [91]. Superiority over fentanyl has not been demonstrated [92].
Benzodiazepines
Midazolam is an appealing sedative option given the rapid onset of action and short duration of effect with bolus administration—making it an ideal agent for procedural sedation. Additionally, due to its potent gamma-aminobutyric acid (GABA) activity and relatively benign hemodynamic profile, midazolam is an important drug in refractory status epilepticus. As a long-term sedative for general ICU use, midazolam accumulates in adipose tissues, significantly prolonging duration of action unless interruptions or down-titration of dose are routinely utilized.
Bolus-dose midazolam is a good choice for intermittent agitation in a NICU population. Conversely, midazolam infusion has been associated with prolonged mechanical ventilation [93, 94]. Though most studies suggest that the impact of midazolam on hemodynamics is similar compared to dexmedetomidine or propofol, a recent report suggests less instability compared to dexmedetomidine [94].
Lorazepam is a longer acting benzodiazepine when used in the short term, but its duration of action is shorter than midazolam when infused for more than one to two days. The strong GABA activity of lorazepam suppresses electrical and metabolic brain activity. Unlike midazolam, lorazepam is formulated in propylene glycol, which can accumulate to toxic levels causing metabolic acidosis and kidney injury. At lorazepam infusion rates above 3 mg/hr or daily doses approaching 1 mg/kg, the osmolar gap should be followed, and alternative agents should be used if the osmolar gap rises above 10–12 mOsm/L [95, 96].
Dexmedetomidine
Dexmedetomidine is a centrally acting alpha agonist similar to clonidine, but more specific for the alpha-2 receptor. It is increasingly utilized for ICU sedation. Desirable properties include rapid onset and termination of activity, mild to moderate sedation without significant respiratory depressant action, analgesic effects, and less delirium than the benzodiazepines [95]. Undesirable properties include a high incidence of bradycardia and hypotension [94].
Barbiturates
Use of barbiturates as a sedative in the neurocritical care unit is limited due to its undesirable side effect profile. Immunosuppressant properties and negative inotropic effects are among the more concerning limitations. Barbiturates remain second-line therapy for the control of ICP after propofol. They remain in widespread use to control refractory status epilepticus, and their potent effects on cerebral metabolic and electrical activity make them an appealing class of agents for sedation in the NICU. Pentobarbital serves as a potent agent for deep sedation in patients with refractory status epilepticus or elevated ICP [97–99].
Sedation Targets and Monitoring
Sedation should be titrated to a validated sedation scale or use an electrophysiological endpoint when neuromuscular blockade is employed or burst-suppression is desired. A recent review of sedation assessment tools in the neurocritical care setting concluded that the SAS and Richmond Agitation Sedation Scale (RASS) are valid and useful for NICU patients, and the bispectral index (BIS) may have a role in monitoring deeply sedated patients in the NICU [100–103].
Regarding assessment of delirium, no assessment tools have been validated in neurocritical care patients, though several studies that used the Intensive Care Delirium Screening Checklist enrolled NICU patients [102, 104, 105]. Recent, highly insightful reviews of the topic are also available [106].
Several studies have addressed the sedation monitoring approaches and medication choices for NICU patients. One prospective, randomized trial of 67 mechanically ventilated adult patients sedated with propofol showed that the BIS monitor added to routine sedation scale monitoring significantly reduced the propofol dose and the time to awakening when compared to subjective scale monitoring alone [107].
Pediatric Considerations
Anatomical and physiological differences alter the approach to intubation and mechanical ventilation of children suffering from neurologic injury. While cervical spinal injury is uncommon in children, approximately half of all cervical spinal injuries are associated with simultaneous TBI [108]. Therefore, cervical spine precautions should be taken when intubating a child with suspected TBI. Criteria for tracheal intubation of children with TBI and other forms of acute brain injury include hypoxemia unresponsive to supplemental oxygen, apnea, hypercarbia (PaCO2 > 45 mmHg), Glasgow Coma Scale Sore (GCS) ≤8, rapid decrease in GCS, anisocoria >1 mm in the context of altered mental status, spinal injury compromising ventilation, abnormal airway reflexes, and any clinical signs of herniation syndrome [109]. A pediatric version of the GCS is recommended when evaluating children infants with acute brain injury.
Several anatomical differences between the pediatric and adult airway should be considered prior to intubating. Children have a proportionally larger tongue, more compliant epiglottis and upper airway tissues, and a prominent occiput that can easily be positioned by placing a small shoulder roll prior to intubation. The infant larynx is more anterior and cephalad (C3-4 vs. C4-5 in adults). The narrowest part occurs at the cricoid ring and gives a cone-shaped appearance that does not become cylindrical until approximately 8 years of age [110]. Pediatric Advanced Life Support Guidelines advise that oral intubation should be performed while maintaining spine immobilization using a cuffed ET tube in children with TBI [111]. Cuff pressures should not be >20 cm H2O due to the risk of mucosal ischemia [112]. A multi-center, randomized control trial demonstrated no increase in post-extubation stridor or long-term complications when using cuffed tubes [113]. For un-cuffed ET tube sizing in children (if cuffed is not available) use the age-based formula: 4 + (age in years/4) [114]. If inserting a regular cuffed ET tube, select one full size smaller than determined by the age-based formula [115]. If placing a micro-cuffed ET tube, select a tube one half size smaller than the age-based calculation [116]. A stylet bent in a hockey stick configuration can help reinforce the rigidity of small ET tubes and help direct them anteriorly through the glottis [117].
When intubating a child <2 years old, a straight laryngoscope blade directly lifting the epiglottis may be preferred because of the infant’s large and acutely angled epiglottis. Generally, a straight size 00 laryngoscope blade is appropriate for extremely premature infants, a size 0 for average-sized newborns, a size 1 for most infants beyond the immediate newborn period, and a size 2 blade for children over the age of two. If an appropriately sized ET tube is placed, the ideal depth can be achieved by inserting the tube until the centimeter marking at the lip is three times the internal diameter of the ET tube [118].
It is prudent to assume a full stomach and a cervical spinal injury when intubating a child with TBI. Endotracheal intubation should utilize a cerebral-protective rapid sequence induction with cricoid pressure and pre-oxygenation. Bag-valve-mask ventilation should not be attempted unless the child has apnea, hypoxemia, or signs of impending herniation [109]. However, the time to desaturation following pre-oxygenation is shorter in apneic infants compared to older children (<100 s), and a modified RSI technique with gentle pressure-limited mask ventilation (10–12 cm H2O) and 100 % oxygen may be used to avoid hypoxemia [119, 120]. This technique may also limit hypercarbia and keep small airways open without the risk of gastric inflation and related morbidity [121–123]. Cricoid pressure is routinely applied despite questionable evidence that it improves clinical outcomes [123–125].
Pre-treatment with lidocaine (1.5 mg/kg IV with max dose 100 mg) may be used but its administration should not delay emergent intubation [126]. Atropine (0.02 mg/kg IV with minimum dose 0.1 mg and max single dose 0.5 mg) is recommended in children ≤1 year old or children <5 years old receiving succinylcholine [127]. For hemodynamically unstable children, the combination of etomidate (0.2–0.6 mg/kg) and neuromuscular blockade with rocuronium (1 mg/kg) or vecuronium (0.3 mg/kg) IV is often used. The association between etomidate administration and clinically significant adrenal insufficiency is not well established in children but can be considered when selecting optimal medications for intubation. Succinylcholine is sometimes avoided because of the risk of malignant hyperthermia, possible ICP elevation [128], hyperkalemia, and life-threatening complications associated with unknown occult metabolic or neuromuscular disease [129, 130]. Fentanyl (2–4 µg/kg) or ketamine (1–2 mg/kg) IV are alternative sedatives, and recent pediatric studies show that ketamine does not increase ICP and may be neuroprotective [131–133]. If hemodynamically stable, midazolam (0.1–0.2 mg/kg) can be added to any of the above combinations.
Children <6 months old have immature liver metabolism, which places them at high risk for respiratory depression secondary to narcotic administration [134]. Continuous infusions of propofol, especially over a 24 h period have been associated with lethal cases of propofol infusion syndrome (metabolic acidosis and death). Therefore, continuous infusion of propofol is not recommended for pediatric patients with acute brain injury [135, 136]. Finally, while information is currently limited, concern exists regarding the potential neurotoxicity of sedatives on the developing brain [137]. The strongest evidence for this comes from animal models, with limited evidence in clinical studies [138–142].
After successful intubation, oxygen saturation of 100 % and normocarbia (35-39 mmHg) should be confirmed by arterial blood gas. Unless the child has signs of herniation, prophylactic hyperventilation (PaCO2 < 35 mmHg) should be avoided [143]. Adequate blood pressure must always be maintained when administering sedatives to assure adequate CPP. A CPP between 40 and 50 mmHg is recommended for children with severe TBI, with infants at the lower end of this range and adolescents at the upper end [144]. Many studies have demonstrated that a CPP ≤ 40 mmHg is associated with higher mortality and morbidity [145, 146]. However, optimal age appropriate CPP thresholds have not been established for TBI and other acute neurological diagnoses. Furthermore, abnormal cerebrovascular auto-regulation, which is more common in children <4 years old [147], makes establishing such thresholds difficult in the absence of advanced neuromonitoring.
Prophylactic interventions, such as hyperosmolar therapy and hyperventilation, are not recommended in the absence of signs or symptoms of herniation or neurological deterioration. In children with refractory intracranial hypertension, transient aggressive hyperventilation or barbiturate administration may be useful. Additional sedation and analgesia, together with prophylactic administration of lidocaine (1 mg/kg IV), may prevent blunt rises in ICP during ET tube suctioning.
Communication
References
Davis DP, Dunford JV, Ochs M, Park K, Hoyt DB. The use of quantitative end-tidal capnometry to avoid inadvertent severe hyperventilation in patients with head injury after paramedic rapid sequence intubation. J Trauma. 2004;56:808–14.
Coplin WM, Pierson DJ, Cooley KD, Newell DW, Rubenfeld GD. Implications of extubation delay in brain-injured patients meeting standard weaning criteria. Am J Respir Crit Care Med. 2000;161:1530–6.
Walls RM, Murphy MF. Manual of emergency airway management. 3rd ed. Philadelphia: Lippincott Williams & Wilkins; 2008.
Sagarin MJ, Barton ED, Chng YM, Walls RM. National Emergency Airway Registry I. Airway management by US and Canadian emergency medicine residents: a multicenter analysis of more than 6,000 endotracheal intubation attempts. Ann Emerg Med. 2005;46:328–36.
Li J, Murphy-Lavoie H, Bugas C, Martinez J, Preston C. Complications of emergency intubation with and without paralysis. Am J Emerg Med. 1999;17:141–3.
Sakles JC, Laurin EG, Rantapaa AA, Panacek EA. Airway management in the emergency department: a one-year study of 610 tracheal intubations. Ann Emerg Med. 1998;31:325–32.
Walls RM. Rapid-sequence intubation in head trauma. Ann Emerg Med. 1993;22:1008–13.
Bedford RF, Persing JA, Pobereskin L, Butler A. Lidocaine or thiopental for rapid control of intracranial hypertension? Anesth Analg. 1980;59:435–7.
Gabriel EJ, Ghajar J, Jagoda A, et al. Guidelines for prehospital management of traumatic brain injury. J Neurotrauma. 2002;19:111–74.
Weingart S. Additional thoughts on the controversy of lidocaine administration before rapid sequence intubation in patients with traumatic brain injuries. Ann Emerg Med. 2007;50:353.
Salhi B, Stettner E. In defense of the use of lidocaine in rapid sequence intubation. Ann Emerg Med. 2007;49:84–6.
Reynolds SF, Heffner J. Airway management of the critically ill patient: rapid-sequence intubation. Chest. 2005;127:1397–412.
Bergen JM, Smith DC. A review of etomidate for rapid sequence intubation in the emergency department. J Emerg Med. 1997;15:221–30.
Moss E, Powell D, Gibson RM, McDowall DG. Effect of etomidate on intracranial pressure and cerebral perfusion pressure. Br J Anaesth. 1979;51:347–52.
Hug CC Jr, McLeskey CH, Nahrwold ML, et al. Hemodynamic effects of propofol: data from over 25,000 patients. Anesth Analg. 1993;77:S21–9.
Bar-Joseph G, Guilburd Y, Tamir A, Guilburd JN. Effectiveness of ketamine in decreasing intracranial pressure in children with intracranial hypertension. J Neurosurg Pediatr. 2009;4:40–6.
Langsjo JW, Maksimow A, Salmi E, et al. S-ketamine anesthesia increases cerebral blood flow in excess of the metabolic needs in humans. Anesthesiology. 2005;103:258–68.
Bourgoin A, Albanese J, Wereszczynski N, Charbit M, Vialet R, Martin C. Safety of sedation with ketamine in severe head injury patients: comparison with sufentanil. Crit Care Med. 2003;31:711–7.
Kovarik WD, Mayberg TS, Lam AM, Mathisen TL, Winn HR. Succinylcholine does not change intracranial pressure, cerebral blood flow velocity, or the electroencephalogram in patients with neurologic injury. Anesth Analg. 1994;78:469–73.
Martyn JA, Richtsfeld M. Succinylcholine-induced hyperkalemia in acquired pathologic states: etiologic factors and molecular mechanisms. Anesthesiology. 2006;104:158–69.
Voicu S, Deye N, Malissin I, et al. Influence of α-stat and pH-stat blood gas management strategies on cerebral blood flow and oxygenation in patients treated with therapeutic hypothermia after out of hospital cardiac arrest: a crossover study. Crit Care Med. 2014;42(8):1849–61.
Eastwood GM, Suzuki S, Lluch C, Schneider AG, Bellomo R. A pilot assessment of alpha-stat vs pH-stat arterial blood gas analysis after cardiac arrest. J Crit Care. 2015;30(1):138–44.
Chesnut RM, Marshall SB, Piek J, Blunt BA, Klauber MR, Marshall LF. Early and late systemic hypotension as a frequent and fundamental source of cerebral ischemia following severe brain injury in the Traumatic Coma Data Bank. Acta Neurochir Suppl (Wien). 1993;59:121–5.
Prough DS, Lang J. Therapy of patients with head injuries: key parameters for management. J Trauma. 1997;42:S10–8.
Trzeciak S, Jones AE, Kilgannon JH, et al. Significance of arterial hypotension after resuscitation from cardiac arrest. Crit Care Med. 2009;37:2895–903.
Kilgannon JH, Roberts BW, Reihl LR, et al. Early arterial hypotension is common in the post-cardiac arrest syndrome and associated with increased in-hospital mortality. Resuscitation. 2008;79:410–6.
Oddo M, Bosel J, Participants in the International Multidisciplinary Consensus Conference on Multimodality Monitoring. Monitoring of brain and systemic oxygenation in neurocritical care patients. Neurocrit Care. 2014 [Epub ahead of print].
Walsh BK, Crotwell DN, Restrepo RD. Capnography/Capnometry during mechanical ventilation: 2011. Respir Care. 2011;56:503–9.
Seneviratne J, Mandrekar J, Wijdicks EF, Rabinstein AA. Noninvasive ventilation in myasthenic crisis. Arch Neurol. 2008;65:54–8.
Flandreau G, Bourdin G, Leray V, et al. Management and long-term outcome of patients with chronic neuromuscular disease admitted to the intensive care unit for acute respiratory failure: a single-center retrospective study. Respir Care. 2011;56:953–60.
Piastra M, Antonelli M, Caresta E, Chiaretti A, Polidori G, Conti G. Noninvasive ventilation in childhood acute neuromuscular respiratory failure: a pilot study. Respiration. 2006;73:791–8.
Abel M, Eisenkraft JB. Anesthetic implications of myasthenia gravis. Mt Sinai J Med. 2002;69:31–7.
Rice MJ, Mancuso AA, Gibbs C, Morey TE, Gravenstein N, Deitte LA. Cricoid pressure results in compression of the postcricoid hypopharynx: the esophageal position is irrelevant. Anesth Analg. 2009;109:1546–52.
Ellis DY, Harris T, Zideman D. Cricoid pressure in emergency department rapid sequence tracheal intubations: a risk-benefit analysis. Ann Emerg Med. 2007;50:653–65.
Bain AR, Smith KJ, Lewis NC, et al. Regional changes in brain blood flow during severe passive hyperthermia: effects of PaCO2 and extracranial blood flow. J Appl Physiol. 1985;115(5):653–9.
Dumont TM, Visioni AJ, Rughani AI, Tranmer BI, Crookes B. Inappropriate prehospital ventilation in severe traumatic brain injury increases in-hospital mortality. J Neurotrauma. 2010;27:1233–41.
Davis DP, Idris AH, Sise MJ, et al. Early ventilation and outcome in patients with moderate to severe traumatic brain injury. Crit Care Med. 2006;34:1202–8.
Davis DP, Stern J, Sise MJ, Hoyt DB. A follow-up analysis of factors associated with head-injury mortality after paramedic rapid sequence intubation. J Trauma. 2005;59:486–90.
Rangel-Castilla L, Lara LR, Gopinath S, et al. Cerebral hemodynamic effects of acute hyperoxia and hyperventilation after severe traumatic brain injury. J Neurotrauma. 2010;27(10):1853–63.
Badjatia N, Strongilis E, Prescutti M, et al. Metabolic benefits of surface counter warming during therapeutic temperature modulation. Crit Care Med. 2009;37:1893–7.
Wood EG, Go-Wingkun J, Luisiri A, Aceto T Jr. Symptomatic cerebral swelling complicating diabetic ketoacidosis documented by intraventricular pressure monitoring: survival without neurologic sequela. Pediatr Emerg Care. 1990;6:285–8.
Koenig MA, Bryan M, Lewin JL 3rd, Mirski MA, Geocadin RG, Stevens RD. Reversal of transtentorial herniation with hypertonic saline. Neurology. 2008;70:1023–9.
Qureshi AI, Geocadin RG, Suarez JI, Ulatowski JA. Long-term outcome after medical reversal of transtentorial herniation in patients with supratentorial mass lesions. Crit Care Med. 2000;28:1556–64.
Oertel M, Kelly DF, Lee JH, et al. Efficacy of hyperventilation, blood pressure elevation, and metabolic suppression therapy in controlling intracranial pressure after head injury. J Neurosurg. 2002;97:1045–53.
Muizelaar JP, Marmarou A, Ward JD, et al. Adverse effects of prolonged hyperventilation in patients with severe head injury: a randomized clinical trial. J Neurosurg. 1991;75(5):731–9.
Stoccchetti N, Maas AI, Chieregato A, van der Plas AA. Hyperventilation in head injury; a review. Chest. 2005;127:1812–27.
Coles JP, Fryer TD, Coleman MR, et al. Hyperventilation following head injury: effect on ischemic burden and cerebral oxidative metabolism. Crit Care Med. 2007;35:568–78.
Coles JP, Minhas PS, Fryer TD, et al. Effect of hyperventilation on cerebral blood flow in traumatic head injury: clinical relevance and monitoring correlates. Crit Care Med. 2002;30:1950–9.
Diringer MN, Videen TO, Yundt K, et al. Regional cerebrovascular and metabolic effects of hyperventilation after severe traumatic brain injury. J Neurosurg. 2002;96:103–8.
Carrera E, Schmidt JM, Fernandez L, et al. Spontaneous hyperventilation and brain tissue hypoxia in patients with severe brain injury. J Neurol Neurosurg Psychiatry. 2010;81(7):793–7.
Kilgannon JH, Jones AE, Shapiro NI, et al. Association between arterial hyperoxia following resuscitation from cardiac arrest and in-hospital mortality. JAMA. 2010;303:2165–71.
Balan IS, Fiskum G, Hazelton J, Cotto-Cumba C, Rosenthal RE. Oximetry-guided reoxygenation improves neurological outcome after experimental cardiac arrest. Stroke. 2006;37:3008–13.
Brucken A, Kaab AB, Kottmann K, et al. Reducing the duration of 100% oxygen ventilation in the early reperfusion period after cardiopulmonary resuscitation decreases striatal brain damage. Resuscitation. 2010;81:1698–703.
Kilgannon JH, Jones AE, Parrillo JE, et al. Relationship between supranormal oxygen tension and outcome after resuscitation from cardiac arrest. Circulation. 2011;123:2717–22.
Bellomo R, Bailey M, Eastwood GM, et al. Arterial hyperoxia and in-hospital mortality after resuscitation from cardiac arrest. Crit Care. 2011;15:R90.
Wang CH, Chang WT, Huang CH, et al. The effect of hyperoxia on survival following adult cardiac arrest: a systematic review and meta-analysis of observational studies. Resuscitation. 2014;85(9):1142–8.
Elmer J, Scutella M, Pullalarevu R, et al. The association between hyperoxia and patient outcomes after cardiac arrest: analysis of a high-resolution database. Intensive Care Med. 2015;41(1):49–57.
Peberdy MA, Callaway CW, Neumar RW, et al. Part 9: post-cardiac arrest care: 2010 American Heart Association Guidelines for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care. Circulation. 2010;122:S768–86.
Menchem CC. Pulse oximetry. Up To Date. 2012;1612.
Dyer BA, White AW Jr, Lee D, Elkins L, Slayton DJ. The relationship between arterial carbon dioxide tension and end-tidal carbon dioxide tension in intubated adults with traumatic brain injuries who required emergency craniotomies. Crit Care Nurs Q. 2013;36(3):310–5.
Helm M, Schuster R, Hauke J, Lampl L. Tight control of prehospital ventilation by capnography in major trauma victims. Br J Anaesth. 2003;90:327–32.
Hardman JG, Aitkenhead AR. Estimating alveolar dead space from the arterial to end-tidal CO(2) gradient: a modeling analysis. Anesth Analg. 2003;97:1846–51.
Ventilation with lower tidal volumes as compared with traditional tidal volumes for acute lung injury and the acute respiratory distress syndrome. The acute respiratory distress syndrome network. N Engl J Med. 2000;342:1301–8.
Petridis AK, Doukas A, Kienke S, et al. The effect of lung-protective permissive hypercapnia in intracerebral pressure in patients with subarachnoid haemorrhage and ARDS. A retrospective study. Acta Neurochir (Wien). 2010;152:2143–5.
Bennett SS, Graffagnino C, Borel CO, James ML. Use of high frequency oscillatory ventilation (HFOV) in neurocritical care patients. Neurocrit Care. 2007;7:221–6.
Roth C, Ferbert A, Deinsberger W, et al. Does prone positioning increase intracranial pressure? A retrospective analysis of patients with acute brain injury and acute respiratory failure. Neurocrit Care. 2014;21(2):186–92.
Reinprecht A, Greher M, Wolfsberger S, Dietrich W, Illievich UM, Gruber A. Prone positioning in subarachnoid hemorrhage patients with acute respiratory distress syndrome: effects on cerebral tissue oxygenation and intracranial pressure. Crit Care Med. 2003;31(6):1831–8.
Reinprecht A, Greher M, Wolfsberger S, Dietrich W, Illievich UM, Gruber A. Prone position in subarachnoid hemorrhage patients with acute respiratory distress syndrome: effects on cerebral tissue oxygenation and intracranial pressure. Crit Care Med. 2003;31:1831–8.
Caricato A, Conti G, Della Corte F, et al. Effects of PEEP on the intracranial system of patients with head injury and subarachnoid hemorrhage: the role of respiratory system compliance. J Trauma. 2005;58:571–6.
Muench E, Bauhuf C, Roth H, et al. Effects of positive end-expiratory pressure on regional cerebral blood flow, intracranial pressure, and brain tissue oxygenation. Crit Care Med. 2005;33:2367–72.
Muench E, Bauhuf C, Roth H, et al. Efefcts of positive end-expiratory pressure on regional cerebral blood flow, intracranial pressure, and brain tissue oxygenation. Crit Care Med. 2005;33(10):2367–72.
Schramm PI, Closhen D, Felkel M, et al. Influence of PEEP on cerebral blood flow and cerebrovascular autoregulation in patients with acute respiratory distress syndrome. J Neurosurg Anesthesiol. 2013;25(2):162–7.
Marik PE, Young A, Sibole S, Levitov A. The effect of APRV ventilation on ICP and cerebral hemodynamics. Neurocrit Care. 2012;17(2):219–23.
Koutsoukou A, Perraki H, Raftopoulou A, et al. Respiratory mechanics in brain-damaged patients. Intensive Care Med. 2006;32:1947–54.
Riker RR, Fugate JE, The Participants in the International Multi-disciplinary Consensus Conference on Multimodality Monitoring. Clinical monitoring scales in acute brain injury: assessment of coma, pain, agitation, and delirium. Neurocrit Care. 2014 [Epub ahead of print].
Samaniego EA, Mlynash M, Caulfield AF, Eyngorn I, Wijman CA. Sedation confounds outcome prediction in cardiac arrest survivors treated with hypothermia. Neurocrit Care. 2011;15:113–9.
Riker RR, Fraser GL. Altering intensive care sedation paradigms to improve patient outcomes. Anesthesiol Clin. 2011;29:663–74.
Jacobi J, Fraser GL, Coursin DB, et al. Clinical practice guidelines for the sustained use of sedatives and analgesics in the critically ill adult. Crit Care Med. 2002;30:119–41.
Fraser GL, Riker RR, Prato BS, Wilkins ML. The frequency and cost of patient-initiated device removal in the ICU. Pharmacotherapy. 2001;21:1–6.
Skoglund K, Enblad P, Marklund N. Effects of the neurological wake-up test on intracranial pressure and cerebral perfusion pressure in brain-injured patients. Neurocrit Care. 2009;11:135–42.
Brain TF, American Association of Neurological S, Congress of Neurological S, et al. Guidelines for the management of severe traumatic brain injury. VI. Indications for intracranial pressure monitoring. J Neurotrauma. 2007;24 Suppl 1:S37–44.
Citerio G, Cormio M. Sedation in neurointensive care: advances in understanding and practice. Curr Opin Crit Care. 2003;9:120–6.
Brook AD, Ahrens TS, Schaiff R, et al. Effect of a nursing-implemented sedation protocol on the duration of mechanical ventilation. Crit Care Med. 1999;27:2609–15.
Kress JP, Pohlman AS, O’Connor MF, Hall JB. Daily interruption of sedative infusions in critically ill patients undergoing mechanical ventilation. N Engl J Med. 2000;342:1471–7.
Treggiari MM, Romand JA, Yanez ND, et al. Randomized trial of light versus deep sedation on mental health after critical illness. Crit Care Med. 2009;37:2527–34.
Mehta S, Burry L, Martinez-Motta JC, et al. A randomized trial of daily awakening in critically ill patients managed with a sedation protocol: a pilot trial. Crit Care Med. 2008;36:2092–9.
Jones C, Backman C, Capuzzo M, Flaatten H, Rylander C, Griffiths RD. Precipitants of post-traumatic stress disorder following intensive care: a hypothesis generating study of diversity in care. Intensive Care Med. 2007;33:978–85.
Hopkins RO, Jackson JC. Long-term neurocognitive function after critical illness. Chest. 2006;130:869–78.
Al MJ, Hakkaart L, Tan SS, Bakker J. Cost-consequence analysis of remifentanil-based analgo-sedation vs. conventional analgesia and sedation for patients on mechanical ventilation in the Netherlands. Crit Care. 2010;14:R195.
Erdman MJ, Doepker BA, Gerlach AT, Phillips GS, Elijovich L, Jones GM. A comparison of severe hemodynamic disturbances between dexmedetomidine and propofol for sedation in neurocritical care patients. Crit Care Med. 2014;42(7):1696–702.
Strom T, Martinussen T, Toft P. A protocol of no sedation for critically ill patients receiving mechanical ventilation: a randomised trial. Lancet. 2010;375:475–80.
Spies C, Macguill M, Heymann A, et al. A prospective, randomized, double-blind, multicenter study comparing remifentanil with fentanyl in mechanically ventilated patients. Intensive Care Med. 2011;37:469–76.
Iyer VN, Hoel R, Rabinstein AA. Propofol infusion syndrome in patients with refractory status epilepticus: an 11-year clinical experience. Crit Care Med. 2009;37:3024–30.
Jakob SM, Ruokonen E, Grounds RM, et al. Dexmedetomidine vs midazolam or propofol for sedation during prolonged mechanical ventilation: two randomized controlled trials. JAMA. 2012;307:1151–60.
Yahwak JA, Riker RR, Fraser GL, Subak-Sharpe S. Determination of a lorazepam dose threshold for using the osmol gap to monitor for propylene glycol toxicity. Pharmacotherapy. 2008;28:984–91.
Horinek EL, Kiser TH, Fish DN, MacLaren R. Propylene glycol accumulation in critically ill patients receiving continuous intravenous lorazepam infusions. Ann Pharmacother. 2009;43:1964–71.
Grof TM, Bledsoe KA. Evaluating the use of dexmedetomidine in neurocritical care patients. Neurocrit Care. 2010;12:356–61.
Chen HI, Malhotra NR, Oddo M, Heuer GG, Levine JM, LeRoux PD. Barbiturate infusion for intractable intracranial hypertension and its effect on brain oxygenation. Neurosurgery. 2008;63:880–6.
Marshall GT, James RF, Landman MP, et al. Pentobarbital coma for refractory intra-cranial hypertension after severe traumatic brain injury: mortality predictions and one-year outcomes in 55 patients. J Trauma. 2010;69:275–83.
Teitelbaum JS, Ayoub O, Skrobik Y. A critical appraisal of sedation, analgesia and delirium in neurocritical care. Can J Neurol Sci. 2011;38:815–25.
Karabinis A, Mandragos K, Stergiopoulos S, et al. Safety and efficacy of analgesia-based sedation with remifentanil versus standard hypnotic-based regimens in intensive care unit patients with brain injuries: a randomised, controlled trial [ISRCTN50308308]. Crit Care. 2004;8:R268–80.
Riker RR, Picard JT, Fraser GL. Prospective evaluation of the Sedation-Agitation Scale for adult critically ill patients. Crit Care Med. 1999;27:1325–9.
Sessler CN, Gosnell MS, Grap MJ, et al. The Richmond Agitation-Sedation Scale: validity and reliability in adult intensive care unit patients. Am J Respir Crit Care Med. 2002;166:1338–44.
Deogaonkar A, Gupta R, DeGeorgia M, et al. Bispectral Index monitoring correlates with sedation scales in brain-injured patients. Crit Care Med. 2004;32:2403–6.
Bergeron N, Dubois MJ, Dumont M, Dial S, Skrobik Y. Intensive Care Delirium Screening Checklist: evaluation of a new screening tool. Intensive Care Med. 2001;27:859–64.
Van Rompaey B, Elseviers MM, Schuurmans MJ, Shortridge-Baggett LM, Truijen S, Bossaert L. Risk factors for delirium in intensive care patients: a prospective cohort study. Crit Care. 2009;13:R77.
Jumaa MA, Zhang F, Ruiz-Ares G, et al. Comparison of safety and clinical and radiographic outcomes in endovascular acute stroke therapy for proximal middle cerebral artery occlusion with intubation and general anesthesia versus the nonintubated state. Stroke. 2010;41:1180–4.
Brown RL, Brunn MA, Garcia VF. Cervical spine injuries in children: a review of 103 patients treated consecutively at a level 1 pediatric trauma center. J Pediatr Surg. 2001;36:1107.
Kochanek PM, Forbes ME, Ruppel RA, et al. Severe traumatic brain injury in infants and children. Pediatric critical care. 3rd ed. Philadelphia: Mosby; 2006. p. 1595–617.
Lucking S, Maffei F, Tamburro R, Thomas N. The approach to the critically ill infant. In: Lucking S, Maffei F, Tamburro R, Thomas N editors. Pediatric critical care study guide text and review. Chapter 33. London: Springer; 2012. p. 690–712.
Kleinman ME, Chameides L, Schexnayder SM, et al. Part 14: pediatric advanced life support: 2010 American Heart Association Guidelines for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care. Circulation. 2010;122:S876.
Galinski M, Tréoux V, Garrigue B, et al. Intracuff pressures of endotracheal tubes in the management of airway emergencies: the need for pressure monitoring. Ann Emerg Med. 2006;47:545.
Weiss M, Dullenkopf A, Fischer JE, et al. Prospective randomized controlled multi-centre trial of cuffed or uncuffed endotracheal tubes in small children. Br J Anaesth. 2009;103:867.
King BR, Baker MD, Braitman LE, et al. Endotracheal tube selection in children: a comparison of four methods. Ann Emerg Med. 1993;22:530.
Khine HH, Corddry DH, Kettrick RG, et al. Comparison of cuffed and uncuffed endotracheal tubes in young children during general anesthesia. Anesthesiology. 1997;86:627.
Wheeler M, Coté CJ, Todres ID. The pediatric airway. In: Coté C, Lerman J, Todres ID editors. A practice of anesthesia for infants and children. 4th ed. Philadelphia: Saunders-Elsevier; 2009. p. 237.
Levitan RM, Pisaturo JT, Kinkle WC, et al. Stylet bend angles and tracheal tube passage using a straight-to-cuff shape. Acad Emerg Med. 2006;13:1255.
Phipps LM, Thomas NJ, Gilmore RK, et al. Prospective assessment of guidelines for determining appropriate depth of endotracheal tube placement in children. Pediatr Crit Care Med. 2005;6:519.
Patel R, Lenczyk M, Hannallah RS, et al. Age and the onset of desaturation in apnoeic children. Can J Anaesth. 1994;41:771–4.
Weiss M, Gerber AC. Rapid sequence induction in children-it’s not a matter of time! Paediatr Anaesth. 2008;18:97–9.
Lawes EG, Campbell I, Mercer D. Inflation pressure, gastric insufflation and rapid sequence induction. Br J Anaesth. 1987;59:315–8.
Weiler N, Heinrichs W, Dick W. Assessment of pulmonary mechanics and gastric inflation pressure during mask ventilation. Prehospital Disaster Med. 1995;10:101–5.
Moynihan RJ, Brock-Utne JG, Archer JH, et al. The effect of cricoid pressure on preventing gastric insufflation in infants and children. Anesthesiology. 1993;78:652–6.
Landsmann I. Circoid pressure: indications and complications. Pediatr Anesth. 2004;14:43–7.
Brock-Utne JG. Is cricoid pressure necessary? Paediatr Anaesth. 2002;12:1–4.
Lev R, Rosen P. Prophylactic lidocaine use preintubation: a review. J Emerg Med. 1994;12:499–506.
Sagarin MJ, Chiang V, Sakles JC, et al. Rapid sequence intubation for pediatric emergency airway management. Pediatr Emerg Care. 2002;18:417.
Zelicof-Paul A, Smith-Lockridge A, Schnadower D, et al. Controversies in rapid sequence intubation in children. Curr Opin Pediatr. 2005;17:355.
Larach MG, Rosenberg H, Gronert GA, Allen GC. Hyperkalemic cardiac arrest during anesthesia in infants and children with occult myopathies. Clin Pediatr (Phila). 1997;36:9.
Gronert GA. Cardiac arrest after succinylcholine: mortality greater with rhabdomyolysis than receptor upregulation. Anesthesiology. 2001;94:523.
Bar-Joseph G, Guilburd Y, Tamir A, Guilburd JN. Effectiveness of ketamine in decreasing intracranial pressure in children with intracranial hypertension. J Neurosurg Pediatr. 2009;4(1):40–6.
Filanovsky Y, Miller P, Kao J. Myth: Ketamine should not be used as an induction agent for intubation in patients with head injury. CJEM. 2010;12(2):154–7.
Sehdev RS, Symmons DA, Kindl K. Ketamine for rapid sequence induction in patients with head injury in the emergency department. Emerg Med Australas. 2006;18:37–44.
Pokela ML, Olkkola KT, Seppala T, Koivisto M. Age-related morphine kinetics in infants. Dev Pharmacol Ther. 1993;20:26–34.
Bray RJ. Propofol infusion syndrome in children. Paediatr Anaesth. 1998;8:491–9.
Parke TJ, Stevens JE, Rice AS, et al. Metabolic acidosis and fatal myocardial failure after propofol infusion in children: five case reports. BMJ. 1992;305:613–6.
Nemergut ME, Aganga D. Flick RP. Anesthetic neurotoxicity: what to tell the parents? Paediatr Anesth. 2014;24(1):120–6.
Paule MG, Li M, Allen RR, et al. Ketamine anesthesia during the first week of life can cause long-lasting cognitive deficits in rhesus monkeys. Neurotoxicol Teratol. 2011;33:220–30.
Loepke AB, Soriano SG. An assessment of the effects of general anesthetics on developing brain structure and neurocognitive function. Anesth Analg. 2008;106:1681–707.
Jevtovic-Todorovic V, Hartman RE, Izumi Y, et al. Early exposure to common anesthetic agents causes widespread neurodegeneration in the developing rat brain and persistent learning deficits. J Neurosci. 2003;23(3):876–82.
Bittigau P, Sifringer M, Genz K, et al. Antiepileptic drugs and apoptotic neurodegeneration in the developing brain. Proc Natl Acad Sci USA. 2002;99(23):15089–94.
Pohl D, Bittigau P, Ishimaru MJ, et al. N-Methyl-D-aspartate antagonists and apoptotic cell death triggered by head trauma in developing rat brain. Proc Natl Acad Sci USA. 1999;96(5):2508–13.
Skippen P, Seear M, Poskitt K, et al. Effect of hyperventilation on regional cerebral blood flow in head-injured children. Crit Care Med. 1997;25:1402–9.
Kochanek PM, Carney N, Adelson PD, et al. Cerebral perfusion pressure. In: Guidelines for the acute medical management of severe traumatic brain injury in infants, children, and adolescents. 2nd ed. Chapter 8. Pediatr Crit Care Med. 2012;13 Suppl 1:S1–82.
Elias-Jones AC, Punt JA, Turnbull AE, et al. Management and outcome of severe head injuries in the Trent region 1985-90. Arch Dis Child. 1992;67:1430–5.
Downard C, Hulka F, Mullins RJ, et al. Relationship of cerebral perfusion pressure and survival in pediatric brain-injured patients. J Trauma. 2000;49:654–8.
Freeman SS, Udomphorn Y, Armstead WM, et al. Young age as a risk factor for impaired cerebral autoregulation after moderate to severe pediatric traumatic brain injury. Anesthesiology. 2008;108:588–95.
Seder DB, Mayer SA. Critical care management of subarachnoid hemorrhage and ischemic stroke. Clin Chest Med. 2009;30:103–22.
Author information
Authors and Affiliations
Corresponding author
Appendix 1: Pre-intubation Neurological Assessment Checklist
Appendix 1: Pre-intubation Neurological Assessment Checklist

Rights and permissions
About this article
Cite this article
Seder, D.B., Jagoda, A. & Riggs, B. Emergency Neurological Life Support: Airway, Ventilation, and Sedation. Neurocrit Care 23 (Suppl 2), 5–22 (2015). https://doi.org/10.1007/s12028-015-0164-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12028-015-0164-3