Abstract
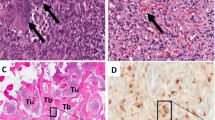
Tumor-induced osteomalacia (TIO) is a paraneoplastic syndrome resulting in renal phosphate wasting and decreased bone mineralization. TIO is usually induced by small, slowly growing tumors of mesenchymal origin (phosphaturic mesenchymal tumor mixed connective tissue variant [PMTMCT]). Nonspecific symptoms including fatigue, bone pain, and musculoskeletal weakness make the diagnosis elusive and often lead to a delay in treatment. The prognosis of TIO is excellent following complete resection of the neoplasm, which leads to the rapid and complete reversal of all symptoms. If the tumor cannot be detected, treatment relies on supplementation with phosphate and active vitamin D compounds. Subsequent radiotherapy in case of incompletely resected tumors or definitive radiotherapy in unresectable tumors is an important treatment option to avoid recurrence or metastasis even though this occurs rarely. Due to the risk of recurrence or late metastases, long-term monitoring is required even in TIO patients diagnosed with a benign tumor.

Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Drezner MK. Tumor-induced osteomalacia. Rev Endocr Metab Disord. 2001;2(2):175–86.
de Jan Beur SM. Tumor-induced osteomalacia. JAMA: J Am Med Assoc. 2005;294(10):1260–7.
White KE, Larsson TE, Econs MJ. The roles of specific genes implicated as circulating factors involved in normal and disordered phosphate homeostasis: frizzled related protein-4, matrix extracellular phosphoglycoprotein, and fibroblast growth factor 23. Endocr Rev. 2006;27(3):221–41.
Kaul M, Silverberg M, DiCarlo E, et al. Tumor-induced osteomalacia. Clin Rheumatol. 2007;26(9):1575–9.
Rendina D, de Filippo G, Tauchmanovà L, et al. Bone turnover and the osteoprotegerin–RANKL pathway in tumor-induced osteomalacia: a longitudinal study of five cases. Calcif Tissue Int. 2009;85(4):293–300.
Carpenter T. The expanding family of hypophosphatemic syndromes. J Bone Miner Metab. 2012;30(1):1–9.
Folpe AL, Fanburg-Smith JC, Billings SD, et al. Most osteomalacia-associated mesenchymal tumors are a single histopathologic entity: an analysis of 32 cases and a comprehensive review of the literature. Am J Surg Pathol. 2004;28 (1).
Woo VL, Landesberg R, Imel EA, et al. Phosphaturic mesenchymal tumor, mixed connective tissue variant, of the mandible: report of a case and review of the literature. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2009;108(6):925–32.
Mori Y, Ogasawara T, Motoi T, et al. Tumor-induced osteomalacia associated with a maxillofacial tumor producing fibroblast growth factor 23: report of a case and review of the literature. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2010;109(3):e57–63.
Chong WH, Molinolo AA, Chen CC, et al. Tumor-induced osteomalacia. Endocr-Relat Cancer. 2011;18(3):R53–77. Worth reading overview about the literature and the differential diagnosis of TIO.
Chong WH, Yavuz S, Patel SM, et al. The importance of whole body imaging in tumor-induced osteomalacia. J Clin Endocrinol Metab. 2011;96(12):3599–600.
Manger B, Schett G. Paraneoplastic syndromes in rheumatology. Nat Rev Rheumatol. 2014;10(11):662–70.
Rowe PSN. The wrickkened pathways of FGF23, MEPE and PHEX. Crit Rev Oral Biol Med. 2004;15(5):264–81.
Imanishi Y, Hashimoto J, Ando W, et al. Matrix extracellular phosphoglycoprotein is expressed in causative tumors of oncogenic osteomalacia. J Bone Miner Metab. 2012;30(1):93–9.
Leaf DE, Pereira RC, Bazari H, et al. Oncogenic osteomalacia due to FGF23-expressing colon adenocarcinoma. J Clin Endocrinol Metab. 2013;98(3):887–91.
Honda R, Kawabata Y, Ito S, et al. Phosphaturic mesenchymal tumor, mixed connective tissue type, non-phosphaturic variant: report of a case and review of 32 cases from the Japanese published work. J Dermatol. 2014;41(9):845–9. Excellent overview about the recently published literature.
Ledford CK, Zelenski NA, Cardona DM, et al. The phosphaturic mesenchymal tumor: why is definitive diagnosis and curative surgery often delayed? Clin Orthop Relat Res. 2013;471(11):3618–25.
Duet M, Kerkeni S, Sfar R, et al. Clinical impact of somatostatin receptor scintigraphy in the management of tumor-induced osteomalacia. Clin Nucl Med. 2008;33 (11).
Haeusler G, Freilinger M, Dominkus M, et al. Tumor-induced hypophosphatemic rickets in an adolescent boy—clinical presentation, diagnosis, and histological findings in growth plate and muscle tissue. J Clin Endocrinol Metab. 2010;95(10):4511–7.
Houang M, Clarkson A, Sioson L, et al. Phosphaturic mesenchymal tumors show positive staining for somatostatin receptor 2A (SSTR2A). Hum Pathol. 2013;44(12):2711–8.
Hautmann AH, Schroeder J, Wild P, et al. Tumor-induced osteomalacia: increased level of FGF-23 in a patient with a phosphaturic mesenchymal tumor at the tibia expressing periostin. Case Rep Endocrinol. 2014;2014:729387. Interesting case report with new findings in immunohistochemical stainings and review of the recently published literature.
Wilkins GE, Granleese S, Hegele RG, et al. Oncogenic osteomalacia: evidence for a humoral phosphaturic factor. J Clin Endocrinol Metab. 1995;80(5):1628–34.
Shelekhova K, Kazakov D, Hes O, et al. Phosphaturic mesenchymal tumor (mixed connective tissue variant): a case report with spectral analysis. Virchows Arch. 2006;448(2):232–5.
Stone MD, Quincey C, Hosking DJ. A neuroendocrine cause of oncogenic osteomalacia. J Pathol. 1992;167(2):181–5.
Malhotra G, Agrawal A, Jambhekar NA, et al. The search for primary tumor in a patient with oncogenic osteomalacia: F-18 FDG PET resolves the conundrum. Clin Nucl Med. 2010;35 (11).
Agrawal K, Bhadada S, Mittal BR, et al. Comparison of 18F-FDG and 68Ga DOTATATE PET/CT in localization of tumor causing oncogenic osteomalacia. Clin Nucl Med. 2015;40(1):e6–e10.
Breer S, Brunkhorst T, Beil FT, et al. 68Ga DOTA-TATE PET/CT allows tumor localization in patients with tumor-induced osteomalacia but negative 111In-octreotide SPECT/CT. Bone. 2014;64:222–7.
Fukumoto S. Diagnostic modalities for FGF23-producing tumors in patients with tumor-induced osteomalacia. Endocrinol Metab. 2014;29(2):136–43.
Caudell JJ, Ballo MT, Zagars GK, et al. Radiotherapy in the management of giant cell tumor of bone. Int J Radiat Oncol Biol Phys. 2003;57(1):158–65.
Kinoshita Y, Fukumoto S. [Anti-FGF23 antibody therapy for patients with tumor-induced osteomalacia]. Clin Calcium. 2014;24(8):1217–22.
Seufert J, Ebert K, Müller J, et al. Octreotide therapy for tumor-induced osteomalacia. N Engl J Med. 2001;345(26):1883–8.
Ogose A, Hotta T, Emura I, et al. Recurrent malignant variant of phosphaturic mesenchymal tumor with oncogenic osteomalacia. Skeletal Radiol. 2001;30(2):99–103.
MORIMOTO T, TAKENAKA S, HASHIMOTO N, et al. Malignant phosphaturic mesenchymal tumor of the pelvis: a report of two cases. Oncol Lett. 2014;8(1):67–71.
Mendenhall WM, Zlotecki RA, Scarborough MT, et al. Giant cell tumor of bone. Am J Clin Oncol. 2006;29 (1).
Tarasova VD, Trepp-Carrasco AG, Thompson R, et al. Successful treatment of tumor-induced osteomalacia due to an intracranial tumor by fractionated stereotactic radiotherapy. J Clinical Endocrinol Metab. 2013.
Feigenberg SJ, Marcus Jr RB, Zlotecki RA, et al. Radiation therapy for giant cell tumors of bone. Clin Orthop Relat Res. 2003;411:207–16.
Miszczyk L, Wydmański J, Spindel J. Efficacy of radiotherapy for giant cell tumor of bone: given either postoperatively or as sole treatment. Int J Radiat Oncol Biol Phys. 2001;49(5):1239–42.
Malone S, O’Sullivan B, Catton C, et al. Long-term follow-up of efficacy and safety of megavoltage radiotherapy in high-risk giant cell tumors of bone. Int J Radiat Oncol Biol Phys. 1995;33(3):689–94.
Ruka W, Rutkowski P, Morysiński T, et al. The megavoltage radiation therapy in treatment of patients with advanced or difficult giant cell tumors of bone. Int J Radiat Oncol Biol Phys. 2010;78(2):494–8.
Nair MK, Jyothirmayi R. Radiation therapy in the treatment of giant cell tumor of bone. Int J Radiat Oncol Biol Phys. 1999;43(5):1065–9.
Ghia AJ, Allen PK, Mahajan A, et al. Intracranial hemangiopericytoma and the role of radiation therapy: a population based analysis. Neurosurgery. 2013;72(2):203–9.
Sonabend AM, Zacharia BE, Goldstein H, et al. The role for adjuvant radiotherapy in the treatment of hemangiopericytoma: a Surveillance, Epidemiology, and End Results analysis. J Neurosurg. 2014;120(2):300–8.
Jalali R, Srinivas C, Nadkarni TD, et al. Suprasellar haemangiopericytoma—challenges in diagnosis and treatment. Acta Neurochir (Wien). 2008;150(1):67–71.
Guthrie BL, Ebersold MJ, Scheithauer BW, et al. Meningeal hemangiopericytoma: histopathological features, treatment, and long-term follow-up of 44 cases. Neurosurgery. 1989;25(4):514–22.
Compliance with Ethics Guidelines
Conflict of Interest
Anke H. Hautmann, Matthias G. Hautmann, Oliver Kölbl, Wolfgang Herr, and Martin Fleck declare no conflicts of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is part of the Topical Collection on Orphan Diseases
Rights and permissions
About this article
Cite this article
Hautmann, A.H., Hautmann, M.G., Kölbl, O. et al. Tumor-Induced Osteomalacia: an Up-to-Date Review. Curr Rheumatol Rep 17, 37 (2015). https://doi.org/10.1007/s11926-015-0512-5
Published:
DOI: https://doi.org/10.1007/s11926-015-0512-5