Abstract
Background
Tumor-induced osteomalacia is a paraneoplastic syndrome resulting in renal phosphate wasting and decreased bone mineralization. Phosphaturic mesenchymal tumors represent a rare etiology of tumor-induced osteomalacia. Nonspecific symptoms of fatigue, bone pain, and musculoskeletal weakness make the diagnosis elusive and lead to a delay in surgical treatment.
Questions/purposes
In this case series, the following three questions were asked: (1) How do the clinical presentation and features of phosphaturic mesenchymal tumors delay the diagnosis? (2) What is the clinical course after surgical treatment of phosphaturic mesenchymal tumors? (3) How frequently do phosphaturic mesenchymal tumors recur and are there factors associated with recurrence?
Methods
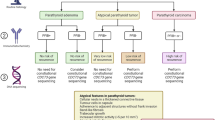
This study retrospectively reviewed the cases of five adults diagnosed and treated for phosphaturic mesenchymal tumors. Patients were identified through an internal orthopaedic oncology database with clinical, surgical, and histologic data obtained through a systematic chart review.
Results
Five patients presented with a long-standing history of osteomalacia, generalized fatigue, pain, and weakness before the diagnosis was reached at an average of 7.2 years (range, 2–12 years) after initial symptom onset. The diagnosis appeared to be delayed owing to the cryptic medical presentation, difficulty in locating tumor by imaging, and confirming histologic appearance. Two patients treated with wide surgical resection did not experience recurrence compared with three patients who did show recurrent signs and symptoms after marginal excision. A postoperative increase in fibroblast-derived growth factor-23 was associated with recurrent disease.
Conclusions
Although uncommon, the diagnosis of phosphaturic mesenchymal tumor should be considered in any patient who presents with hypophosphaturic osteomalacia and no other physiologic cause. Definitive treatment is early, wide surgical resection.
Level of Evidence
Level IV, therapeutic study. See the Instructions for Authors for a complete description of levels of evidence.



Similar content being viewed by others
References
Bahrami A, Weiss SW, Montgomery E, Horvai AE, Jin L, Inwards CY, Folpe AL. RT-PCR analysis for FGF23 using paraffin sections in the diagnosis of phosphaturic mesenchymal tumors with and without known tumor induced osteomalacia. Am J Surg Pathol. 2009;33:1348–1354.
Carpenter TO. Oncogenic osteomalacia: a complex dance of factors. N Engl J Med. 2003;348:1705–1708.
Chong WH, Molinolo AA, Chen CC, Collins MT. Tumor-induced osteomalacia. Endocr Relat Cancer. 2011;18:R53–77.
Duet M, Kerkeni S, Sfar R, Bazille C, Liote F, Orcel P. Clinical impact of somatostatin receptor scintigraphy in the management of tumor-induced osteomalacia. Clin Nucl Med. 2008;33:752–756.
Fletcher CDM, Bridge JA, Hogendoorn P, Mertens F, eds. WHO Classification of Tumours of Soft Tissue and Bone. Geneva, Switzerland: WHO Press; 2013.
Folpe AL, Fanburg-Smith JC, Billings SD, Bisceglia M, Bertoni F, Cho JY, Econs MJ, Inwards CY, Jan de Beur SM, Mentzel T, Montgomery E, Michal M, Miettinen M, Mills SE, Reith JD, O’Connell JX, Rosenberg AE, Rubin BP, Sweet DE, Vinh TN, Wold LE, Wehrli BM, White KE, Zaino RJ, Weiss SW. Most osteomalacia-associated mesenchymal tumors are a single histopathologic entity: an analysis of 32 cases and a comprehensive review of the literature. Am J Surg Pathol. 2004;28:1–30.
Ito N, Shimizu Y, Suzuki H, Saito T, Okamoto T, Hori M, Akahane M, Fukumoto S, Fujita T. Clinical utility of systemic venous sampling of FGF23 for identifying tumours responsible for tumour-induced osteomalacia. J Intern Med. 2010;268:390–394.
Jan de Beur SM, Levine MA. Molecular pathogenesis of hypophosphatemic rickets. J Clin Endocrinol Metab. 2002;87:2467–2473.
Jan de Beur SM, Streeten EA, Civelek AC, McCarthy EF, Uribe L, Marx SJ, Onobrakpeya O, Raisz LG, Watts NB, Sharon M, Levine MA. Localisation of mesenchymal tumours by somatostatin receptor imaging. Lancet. 2002;359:761–763.
Jonsson KB, Zahradnik R, Larsson T, White KE, Sugimoto T, Imanishi Y, Yamamoto T, Hampson G, Koshiyama H, Ljunggren O, Oba K, Yang IM, Miyauchi A, Econs MJ, Lavigne J, Juppner H. Fibroblast growth factor 23 in oncogenic osteomalacia and X-linked hypophosphatemia. N Engl J Med. 2003;348:1656–1663.
Kumar R. Tumor-induced osteomalacia and the regulation of phosphate homeostasis. Bone. 2000;27:333–338.
McCance RA. Osteomalacia with Looser’s nodes (Milkman’s syndrome) due to a raised resistance to vitamin D acquired about the age of 15 years. Q J Med. 1947;16:33–46.
Mekinian A, Ladsous M, Balavoine AS, Carnaille B, Aubert S, Soudan B, Wemeau JL. Curative surgical treatment after inefficient long-acting somatostatin analogues therapy of a tumor-induced osteomalacia. Presse Med. 2011;40:309–313.
Nguyen BD, Wang EA. Indium-111 pentetreotide scintigraphy of mesenchymal tumor with oncogenic osteomalacia. Clin Nucl Med. 1999;24:130–131.
Ogose A, Hotta T, Emura I, Hatano H, Inoue Y, Umezu H, Endo N. Recurrent malignant variant of phosphaturic mesenchymal tumor with oncogenic osteomalacia. Skeletal Radiol. 2001;30:99–103.
Paglia F, Dionisi S, Minisola S. Octreotide for tumor-induced osteomalacia. N Engl J Med. 2002;346:1748–1749; author reply 1748–1749.
Prader A, Illig R, Uehlinger E, Stalder G. [Rickets following bone tumor][in German]. Helv Paediatr Acta. 1959;14:554–565.
Rhee Y, Lee JD, Shin KH, Lee HC, Huh KB, Lim SK. Oncogenic osteomalacia associated with mesenchymal tumour detected by indium-111 octreotide scintigraphy. Clin Endocrinol (Oxf). 2001;54:551–554.
Rosen CH, ed. Primer on the Metabolic Bone Diseases and Disorders of Mineral Metabolism. Ames, IA, Wiley-Blackwell; 2013.
Seufert J, Ebert K, Muller J, Eulert J, Hendrich C, Werner E, Schuuze N, Schulz G, Kenn W, Richtmann H, Palitzsch KD, Jakob F. Octreotide therapy for tumor-induced osteomalacia. N Engl J Med. 2001;345:1883–1888.
Shimada T, Mizutani S, Muto T, Yoneya T, Hino R, Takeda S, Takeuchi Y, Fujita T, Fukumoto S, Yamashita T. Cloning and characterization of FGF23 as a causative factor of tumor-induced osteomalacia. Proc Natl Acad Sci U S A. 2001;98:6500–6505.
Sidell D, Lai C, Bhuta S, Barnes L, Chhetri DK. Malignant phosphaturic mesenchymal tumor of the larynx. Laryngoscope. 2011;121:1860–1863.
Terek RM, Nielsen GP. Case records of the Massachusetts General Hospital: weekly clinicopathological exercises. Case 29-2001. A 14-year-old with abnormal bones and a sacral mass. N Engl J Med. 2001;345:903–908.
Uramoto N, Furukawa M, Yoshizaki T. Malignant phosphaturic mesenchymal tumor, mixed connective tissue variant of the tongue. Auris Nasus Larynx. 2009;36:104–105.
Author information
Authors and Affiliations
Corresponding author
Additional information
Each author certifies that he or she, or a member of his or her immediate family, has no funding or commercial associations (eg, consultancies, stock ownership, equity interest, patent/licensing arrangements, etc) that might pose a conflict of interest in connection with the submitted article.
All ICMJE Conflict of Interest Forms for authors and Clinical Orthopaedics and Related Research editors and board members are on file with the publication and can be viewed on request.
Each author certifies that his or her institution approved the human protocol for this investigation that all investigations were conducted in conformity with ethical principles of research, and that informed consent for participation in the study was obtained.
About this article
Cite this article
Ledford, C.K., Zelenski, N.A., Cardona, D.M. et al. The Phosphaturic Mesenchymal Tumor: Why is Definitive Diagnosis and Curative Surgery Often Delayed?. Clin Orthop Relat Res 471, 3618–3625 (2013). https://doi.org/10.1007/s11999-013-3178-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11999-013-3178-1