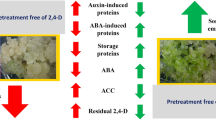
Abstract
Somatic embryogenesis is a biotechnological tool with high application potential in the in vitro propagation and regeneration of crop plants, such as sugarcane. However, decreasing embryogenic competence is observed in embryogenic callus with successive subcultures, especially when the callus are maintained in the presence of 2,4-dichlorophenoxyacetic acid (2,4-d). Thus, the aim of this work was to investigate the morphological and molecular changes associated with the decreasing embryogenic competence in sugarcane embryogenic callus under long-term culture with or without 2,4-d. Sugarcane embryogenic callus were subcultured with 10 μM 2,4-d every 21 days for eight subcultures, and during each subculture, the callus were matured. In addition, the embryogenic callus from the first subculture after induction (S1) and after six subcultures with (S6) or without 2,4-d [S6(-)] were matured, and their histomorphological and histochemical features, endogenous polyamine levels and proteomic profiles were examined. The callus cultured with 2,4-d for a long time period showed reduced embryogenic competence after six subcultures, while callus cultivated in culture medium without 2,4-d maintained high embryogenic competence. In contrast to the callus in S6, those in the first subculture (S1) and S6(-) presented prominent nuclei, a high nucleus/cytoplasm ratio. Long-term culture with 2,4-d also affected polyamine (PA) metabolism, leading to high concentrations of putrescine (Put) and spermidine (Spd) in particular, and the synthesis and regulation of proteins, such as late embryogenesis abundant protein, chitinase, oleosin, and heat shock proteins. Therefore, we demonstrate that long-term culture with 2,4-d decreases embryogenic competence in sugarcane embryogenic callus.
Key message
Loss in embryogenic competence in sugarcane callus by the long exposure to 2,4-d, is induced via changes in the synthesis/mobilization of reserves and protein abundance.






Similar content being viewed by others
References
Abrahamsson M, Valladares S, Larsson E, Clapham D, von Arnold S (2012) Patterning during somatic embryogenesis in Scots pine in relation to polar auxin transport and programmed cell death. Plant Cell Tissue Org Cult 109:391–400. https://doi.org/10.1007/s11240-011-0103-8
Almeida FA, Vale EM, Reis RS, Santa-Catarina C, Silveira V (2019) LED lamps enhance somatic embryo maturation in association with the differential accumulation of proteins in the Carica papaya L. ‘Golden’ embryogenic callus. Plant Physiol Biochem 143:109–118. https://doi.org/10.1016/j.plaphy.2019.08.029
Arruda P (2012) Genetically modified sugarcane for bioenergy generation. Curr Opin Biotechnol 23:315–322. https://doi.org/10.1016/j.copbio.2011.10.012
Aryal B et al (2019) ABCG36/PEN3/PDR8 is an exporter of the auxin precursor, indole-3-butyric acid, and involved in auxin-controlled development. Front Plant Sci 10:899. https://doi.org/10.3389/fpls.2019.00899
Bairu MW, Aremu AO, Van Staden J (2011) Somaclonal variation in plants: causes and detection methods. Plant Growth Regul 63:147–173. https://doi.org/10.1007/s10725-010-9554-x
Bradaï F, Pliego-Alfaro F, Sánchez-Romero C (2016) Long-term somatic embryogenesis in olive (Olea europaea L.): influence on regeneration capability and quality of regenerated plants. Sci Hortic 199:23–31. https://doi.org/10.1016/j.scienta.2015.12.010
Branca C, Torelli A, Fermi P, Altamura M, Bassi M (1994) Early phases in in vitro culture of tomato cotyledons: starch accumulation and protein pattern in relation to the hormonal treatment. Protoplasma 182:59–64. https://doi.org/10.1007/BF01403689
Breton D, Harvengt L, Trontin J-F, Bouvet A, Favre J-M (2006) Long-term subculture randomly affects morphology and subsequent maturation of early somatic embryos in maritime pine. Plant Cell Tissue Org Cult 87:95–108. https://doi.org/10.1007/s11240-006-9144-9
Brisibe EA, Miyake H, Taniguchi T, Maeda E (1994) Regulation of somatic embryogenesis in long-term callus cultures of sugarcane (Saccharum officinarum L.) New Phytol 126:301–307. https://doi.org/10.1111/j.1469-8137.1994.tb03949.x
Cangahuala-Inocente GC, Steiner N, Santos M, Guerra MP (2004) Morphohistological analysis and histochemistry of Feijoa sellowiana somatic embryogenesis. Protoplasma 224:33–40. https://doi.org/10.1007/s00709-004-0055-5
Cangahuala-Inocente GC, Silveira V, Caprestano CA, Floh EI, Guerra MP (2014) Dynamics of physiological and biochemical changes during somatic embryogenesis of Acca sellowiana. In Vitro Cell Dev Biol Plant 50:166–175
Che P, Love TM, Frame BR, Wang K, Carriquiry AL, Howell SH (2006) Gene expression patterns during somatic embryo development and germination in maize Hi II callus cultures. Plant Mol Biol 62:1–14. https://doi.org/10.1007/s11103-006-9013-2
Chugh A, Khurana P (2002) Gene expression during somatic embryogenesis—recent advances. Curr Sci 83:715–730
Cona A, Cenci F, Cervelli M, Federico R, Mariottini P, Moreno S, Angelini R (2003) Polyamine oxidase, a hydrogen peroxide-producing enzyme, is up-regulated by light and down-regulated by auxin in the outer tissues of the maize mesocotyl. Plant Physiol 131:803–813. https://doi.org/10.1104/pp.011379
Correia S, Vinhas R, Manadas B, Lourenço AS, Veríssimo P, Canhoto JM (2012) Comparative proteomic analysis of auxin-induced embryogenic and nonembryogenic tissues of the solanaceous tree Cyphomandra betacea (Tamarillo). J Proteome Res 11:1666–1675. https://doi.org/10.1021/pr200856w
de Carvalho Silva R, Luis ZG, Scherwinski-Pereira JE (2014) The histodifferentiation events involved during the acquisition and development of somatic embryogenesis in oil palm (Elaeis guineensis Jacq.) Plant Growth Regul 72:67–80. https://doi.org/10.1007/s10725-013-9837-0
De Jong AJ, Cordewener J, Schiavo FL, Terzi M, Vandekerckhove J, Van Kammen A, De Vries SC (1992) A carrot somatic embryo mutant is rescued by chitinase. Plant Cell 4:425–433. https://doi.org/10.1105/tpc.4.4.425
de Jong AJ et al (1995) Transient reduction in secreted 32 kD chitinase prevents somatic embryogenesis in the carrot (Daucus carota L.) variant ts11. Dev Genet 16:332–343. https://doi.org/10.1002/dvg.1020160406
Delbarre A, Muller P, Imhoff V, Guern J (1996) Comparison of mechanisms controlling uptake and accumulation of 2,4-dichlorophenoxy acetic acid, naphthalene-1-acetic acid, and indole-3-acetic acid in suspension-cultured tobacco cells. Planta 198:532–541. https://doi.org/10.1007/BF00262639
Distler U, Kuharev J, Navarro P, Levin Y, Schild H, Tenzer S (2014) Drift time-specific collision energies enable deep-coverage data-independent acquisition proteomics. Nat Methods 11:167–170. https://doi.org/10.1038/nmeth.2767
Distler U, Kuharev J, Navarro P, Tenzer S (2016) Label-free quantification in ion mobility–enhanced data-independent acquisition proteomics. Nat Protoc 11:795–812. https://doi.org/10.1038/nprot.2016.042
Dubrovina A, Kiselev K (2016) Age-associated alterations in the somatic mutation and DNA methylation levels in plants. Plant Biol 18:185–196. https://doi.org/10.1111/plb.12375
Eksomtramage T, Paulet F, Guiderdoni E, Glaszmann JC, Engelmann F (1992) Development of a cryopreservation process for embryogenic calluses of a commercial hybrid of sugarcane (Saccharum sp.) and application to different varieties. Cryo Lett 13:239–252
Feher A, Pasternak TP, Dudits D (2003) Transition of somatic plant cells to an embryogenic state. Plant Cell Tissue Org Cult 74:201–228. https://doi.org/10.1023/a:1024033216561
Ferreira LT, Araújo-Silva MM, Ulisses C, Camara TR, Willadino L (2017) Using LED lighting in somatic embryogenesis and micropropagation of an elite sugarcane variety and its effect on redox metabolism during acclimatization. Plant Cell Tissue Org Cult 128:211–221. https://doi.org/10.1007/s11240-016-1101-7
Fitch MM, Moore PH (1993) Long-term culture of embryogenic sugarcane callus. Plant Cell Tissue Org Cult 32:335–343. https://doi.org/10.1007/BF00042297
Fu C, Li L, Wu W, Li M, Yu X, Yu L (2012) Assessment of genetic and epigenetic variation during long-term Taxus cell culture. Plant Cell Rep 31:1321–1331. https://doi.org/10.1007/s00299-012-1251-y
Gahan PB (1984) Plant histochemistry and cytochemistry: an introduction. Experimental botany: a series of monographs, vol 18. Academic Press, London
Garcia C, Almeida AAF, Costa M, Britto D, Valle R, Royaert S, Marelli JP (2019) Abnormalities in somatic embryogenesis caused by 2,4-d: an overview. Plant Cell Tissue Org Cult. https://doi.org/10.1007/s11240-019-01569-8
Gliwicka M, Nowak K, Cieśla E, Gaj MD (2012) Expression of seed storage product genes (CRA1 and OLEO4) in embryogenic cultures of somatic tissues of Arabidopsis. Plant Cell Tissue Org Cult 109:235–245. https://doi.org/10.1007/s11240-011-0089-2
Gnanapragasam S, Vasil IK (1990) Plant-regeneration from a cryopreserved embryogenic cell-suspension of a commercial sugarcane hybrid (Saccharum sp). Plant Cell Rep 9:419–423. https://doi.org/10.1007/BF00232263
Gnanapragasam S, Vasil IK (1992) Cryopreservation of immature embryos, embryogenic callus and cell suspension cultures of gramineous species. Plant Sci 83:205–215. https://doi.org/10.1016/0168-9452(92)90080-6
Gomes HT, Bartos PMC, Silva CO, do Amaral LIV, Scherwinski-Pereira JE (2014) Comparative biochemical profiling during the stages of acquisition and development of somatic embryogenesis in African oil palm (Elaeis guineensis Jacq.) Plant Growth Regul 74:199–208. https://doi.org/10.1007/s10725-014-9901-4
Gong L et al (2004) Drosophila ventral furrow morphogenesis: a proteomic analysis. Development 131:643–656. https://doi.org/10.1242/dev.00955
Grafi G, Zemach A, Pitto L (2007) Methyl-CpG-binding domain (MBD) proteins in plants. Biochimica et Biophysica Acta (BBA) 1769:287–294. https://doi.org/10.1016/j.bbaexp.2007.02.004
Guo F et al (2013) Induced expression of AtLEC1 and AtLEC2 differentially promotes somatic embryogenesis in transgenic tobacco plants. PLoS ONE 8:e71714. https://doi.org/10.1371/journal.pone.0071714
Heringer AS et al (2015) Label-free quantitative proteomics of embryogenic and non-embryogenic callus during sugarcane somatic embryogenesis. PLoS ONE 10:e0127803. https://doi.org/10.1371/journal.pone.0127803
Heringer AS, Reis RS, Passamani LZ, de Souza-Filho GA, Santa-Catarina C, Silveira V (2017) Comparative proteomics analysis of the effect of combined red and blue lights on sugarcane somatic embryogenesis. Acta Physiol Plant 39:52. https://doi.org/10.1007/s11738-017-2349-1
Hernández G, Vázquez-Pianzola P, Sierra J, Rivera-Pomar R (2004) Internal ribosome entry site drives cap-independent translation of reaper and heat shock protein 70 mRNAs in Drosophila embryos. RNA 10:1783–1797. https://doi.org/10.1261/rna.7154104
Ijaz S, Rana I, Khan I, Saleem M (2012) Establishment of an in vitro regeneration system for genetic transformation of selected sugarcane genotypes. Genet Mol Res 11:512–530. https://doi.org/10.4238/2012.March.6.4
ISO (2017) International Sugar Organization (ISO). http://www.isosugar.org/sugarsector/sugar. Accessed 30 Aug 2017
Jimenez-Lopez JC, Zienkiewicz A, Zienkiewicz K, Alché JD, Rodríguez-García MI (2016) Biogenesis of protein bodies during legumin accumulation in developing olive (Olea europaea L.) seed. Protoplasma 253:517–530. https://doi.org/10.1007/s00709-015-0830-5
Karami O, Saidi A (2010) The molecular basis for stress-induced acquisition of somatic embryogenesis. Mol Biol Rep 37:2493–2507. https://doi.org/10.1007/s11033-009-9764-3
Kaur R, Kapoor M (2016) Plant regeneration through somatic embryogenesis in sugarcane. Sugar Tech 18:93–99. https://doi.org/10.1007/s12355-015-0380-3
Kumaravel M, Uma S, Backiyarani S, Saraswathi MS, Vaganan MM, Muthusamy M, Sajith KP (2017) Differential proteome analysis during early somatic embryogenesis in Musa spp. AAA cv. Grand Naine. Plant Cell Rep 36:163–178. https://doi.org/10.1007/s00299-016-2067-y
Landey RB et al (2015) Assessment of genetic and epigenetic changes during cell culture ageing and relations with somaclonal variation in Coffea arabica. Plant Cell Tissue Org Cult 122:517–531. https://doi.org/10.1007/s11240-015-0772-9
Lara-Chavez A, Egertsdotter U, Flinn BS (2012) Comparison of gene expression markers during zygotic and somatic embryogenesis in pine. In Vitro Cell Dev Biol Plant 48:341–354. https://doi.org/10.1007/s11627-012-9440-5
Lingle SE, Viator RP, Johnson RM, Tew TL, Boykin DL (2009) Recurrent selection for sucrose content has altered growth and sugar accumulation in sugarcane. Field Crops Res 113:306–311. https://doi.org/10.1016/j.fcr.2009.06.015
Mahmud I, Shrestha B, Boroujerdi A, Chowdhury K (2015) NMR-based metabolomics profile comparisons to distinguish between embryogenic and non-embryogenic callus tissue of sugarcane at the biochemical level. In Vitro Cell Dev Biol Plant 51:340–349. https://doi.org/10.1007/s11627-015-9687-8
Marsoni M, Bracale M, Espen L, Prinsi B, Negri AS, Vannini C (2008) Proteomic analysis of somatic embryogenesis in Vitis vinifera. Plant Cell Rep 27:347–356
Martin A et al (2000) Differences in the contents of total sugars, starch and sucrose in embryogenic and nonembryogenic calli from Medicago arborea L. Plant Sci 154:143–151. https://doi.org/10.1016/s0168-9452(99)00251-4
Martinez-Montero ME, Gonzalez-Arnao MT, Borroto-Nordelo C, Puentes-Diaz C, Engelmann F (1998) Cryopreservation of sugarcane embryogenic callus using a simplified freezing process. Cryo Lett 19:171–176
Martinez-Montero ME, Martinez J, Engelmann F (2008) Cryopreservation of sugarcane somatic embryos. Cryo Lett 29:229–242
Ming R et al (2006) Sugarcane improvement through breeding and biotechnology. In: Janick J (ed) Plant breeding reviews, vol 27. Wiley, New Jersey, pp 15–118. https://doi.org/10.1002/9780470650349.ch2
Moura EF, Ventrella MC, Motoike SY, de Sá Júnior AQ, Carvalho M, Manfio CE (2008) Histological study of somatic embryogenesis induction on zygotic embryos of macaw palm (Acrocomia aculeata (Jacq.) Lodd. ex Martius). Plant Cell Tissue Org Cult 95:175–184. https://doi.org/10.1007/s11240-008-9430-9
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol Plant 15:473–497. https://doi.org/10.1111/j.1399-3054.1962.tb08052.x
Noceda C, Salaj T, Pérez M, Viejo M, Cañal MJ, Salaj J, Rodriguez R (2009) DNA demethylation and decrease on free polyamines is associated with the embryogenic capacity of Pinus nigra Arn. cell culture. Trees 23:1285. https://doi.org/10.1007/s00468-009-0370-8
Passamani LZ, Bertolazi AA, Ramos AC, Santa-Catarina C, Thelen JJ, Silveira V (2018) Embryogenic competence acquisition in sugarcane callus is associated with differential H+ pump abundance and activity. J Proteome Res 17:2767–2779. https://doi.org/10.1021/acs.jproteome.8b00213
Pinto G, Silva S, Neves L, Araújo C, Santos C (2010) Histocytological changes and reserve accumulation during somatic embryogenesis in Eucalyptus globulus. Trees 24:763–769. https://doi.org/10.1007/s00468-010-0446-5
Pontaroli AC, Camadro EL (2005) Somaclonal variation in Asparagus officinalis plants regenerated by organogenesis from long-term callus cultures. Genet Mol Biol 28:423–430. https://doi.org/10.1590/S1415-47572005000300015
Reis RS, de Moura Vale E, Heringer AS, Santa-Catarina C, Silveira V (2016) Putrescine induces somatic embryo development and proteomic changes in embryogenic callus of sugarcane. J Proteomics 130:170–179. https://doi.org/10.1016/j.jprot.2015.09.029
Rival A et al (2013) Variations in genomic DNA methylation during the long-term in vitro proliferation of oil palm embryogenic suspension cultures. Plant Cell Rep 32:359–368. https://doi.org/10.1007/s00299-012-1369-y
Rocha DI, Pinto DLP, Vieira LM, Tanaka FAO, Dornelas MC, Otoni WC (2016) Cellular and molecular changes associated with competence acquisition during passion fruit somatic embryogenesis: ultrastructural characterization and analysis of SERK gene expression. Protoplasma 253:595–609. https://doi.org/10.1007/s00709-015-0837-y
Rode C, Lindhorst K, Braun H-P, Winkelmann T (2012) From callus to embryo: a proteomic view on the development and maturation of somatic embryos in Cyclamen persicum. Planta 235:995–1011. https://doi.org/10.1007/s00425-011-1554-1
Ruzicka K et al (2010) Arabidopsis PIS1 encodes the ABCG37 transporter of auxinic compounds including the auxin precursor indole-3-butyric acid. Proc Natl Acad Sci USA 107:10749–10753. https://doi.org/10.1073/pnas.1005878107
Sakhanokho HF, Ozias-Akins P, May OL, Chee PW (2005) Putrescine enhances somatic embryogenesis and plant regeneration in upland cotton. Plant Cell Tissue Org Cult 81:91–95. https://doi.org/10.1007/s11240-004-1541-3
Santa-Catarina C, Silveira V, Scherer GFE, Floh EIS (2007) Polyamine and nitric oxide levels relate with morphogenetic evolution in somatic embryogenesis of Ocotea catharinensis. Plant Cell Tissue Org Cult 90:93–101. https://doi.org/10.1007/s11240-007-9259-7
Satish L et al (2016) Influence of plant growth regulators and spermidine on somatic embryogenesis and plant regeneration in four Indian genotypes of finger millet (Eleusine coracana (L.) Gaertn). Plant Cell Tissue Org Cult 124:15–31
Sharma N, Sharma K, Gaur R, Gupta V (2011) Role of chitinase in plant defense. Asian J Biochem 6:29–37. https://doi.org/10.3923/ajb.2011.29.37
Silva FAS, Azevedo CAV (2016) The Assistat software version 7.7 and its use in the analysis of experimental data. Afr J Agric Res 11:3733–3740. https://doi.org/10.5897/AJAR2016.11522
Silveira V, Floh EIS, Handro W, Guerra MP (2004) Effect of plant growth regulators on the cellular growth and levels of intracellular protein, starch and polyamines in embryogenic suspension cultures of Pinus taeda. Plant Cell Tissue Org Cult 76:53–60. https://doi.org/10.1023/a:1025847515435
Silveira V, Vita A, Macedo A, Dias M, Floh E, Santa-Catarina C (2013) Morphological and polyamine content changes in embryogenic and non-embryogenic callus of sugarcane. Plant Cell Tissue Org Cult 114:351–364. https://doi.org/10.1007/s11240-013-0330-2
Singh G, Sandhu S, Meeta M, Singh K, Gill R, Gosal S (2008) In vitro induction and characterization of somaclonal variation for red rot and other agronomic traits in sugarcane. Euphytica 160:35–47. https://doi.org/10.1007/s10681-007-9531-2
Snyman SJ, Meyer GM, Koch AC, Banasiak M, Watt MP (2011) Applications of in vitro culture systems for commercial sugarcane production and improvement. In Vitro Cell Dev Biol Plant 47:234–249. https://doi.org/10.1007/s11627-011-9354-7
Sonenberg N, Hinnebusch AG (2009) Regulation of translation initiation in eukaryotes: mechanisms and biological targets. Cell 136:731–745. https://doi.org/10.1016/j.cell.2009.01.042
Stone SL et al (2001) LEAFY COTYLEDON2 encodes a B3 domain transcription factor that induces embryo development. Proc Natl Acad Sci USA 98:11806–11811. https://doi.org/10.1073/pnas.201413498
Su YH, Zhao XY, Liu YB, Zhang CL, O’Neill SD, Zhang XS (2009) Auxin-induced WUS expression is essential for embryonic stem cell renewal during somatic embryogenesis in Arabidopsis. Plant J 59:448–460. https://doi.org/10.1111/j.1365-313X.2009.03880.x
Uarrota VG, Maraschin M (2015) Metabolomic, enzymatic, and histochemical analyzes of cassava roots during postharvest physiological deterioration. BMC Res Notes 8:648. https://doi.org/10.1186/s13104-015-1580-3
Vondráková Z, Eliášová K, Vágner M, Martincová O, Cvikrová M (2015) Exogenous putrescine affects endogenous polyamine levels and the development of Picea abies somatic embryos. Plant Growth Regul 75:405–414. https://doi.org/10.1007/s10725-014-0001-2
Wang A-Q, Dong W-Q, Wei Y-W, Huang C-M, He L-F, Yang L-T, Li Y-R (2009) Transformation of sugarcane with ACC oxidase antisense gene. Sugar Tech 11:39–43. https://doi.org/10.1007/s12355-009-0007-7
Weckx S, Inzé D, Maene L (2019) Tissue culture of oil palm: finding the balance between mass propagation and somaclonal variation. Front Plant Sci 10:722. https://doi.org/10.3389/fpls.2019.00722
Yang X, Zhang X (2010) Regulation of somatic embryogenesis in higher plants. Crit Rev Plant Sci 29:36–57
Yemm E, Willis A (1954) The estimation of carbohydrates in plant extracts by anthrone. Biochem J 57:508–514. https://doi.org/10.1042/bj0570508
Zhang J et al (2018) Allele-defined genome of the autopolyploid sugarcane Saccharum spontaneum L. Nat Genet 50:1565–1573. https://doi.org/10.1038/s41588-018-0237-2
Zhao J, Wang B, Wang X, Zhang Y, Dong M, Zhang J (2015) iTRAQ-based comparative proteomic analysis of embryogenic and non-embryogenic tissues of Prince Rupprecht’s larch (Larix principis-rupprechtii Mayr). Plant Cell Tissue Org Cult 120:655–669. https://doi.org/10.1007/s11240-014-0633-y
Zi J et al (2013) Stress responsive proteins are actively regulated during rice (Oryza sativa) embryogenesis as indicated by quantitative proteomics analysis. PLoS ONE 8:e74229. https://doi.org/10.1371/journal.pone.0074229
Acknowledgements
This research was supported by funds from the Fundação de Amparo à Pesquisa do Estado do Rio de Janeiro-FAPERJ (Proc. E26/211.690/2015; Proc. E26/203.311/2017) and Conselho Nacional de Desenvolvimento Científico e Tecnológico-CNPq (305415/2016-6) to VS. This study was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior—Brasil (CAPES)—Finance Code 001. LZP, VPMA and EMV thank CAPES for a fellowship, and RSS and KSR thank FAPERJ for a fellowship.
Author information
Authors and Affiliations
Contributions
LZP and VS designed the study; LZP and RSR carried out the experiments; LZP, EMV, KRS and CSC performed the histomorphological and histochemical analyses; LZP, VPMA and CSC performed the free polyamine determination; LZP, RSR and VS performed the proteomic analysis; and all authors read, reviewed and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Communicated by Bart Panis.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Fig. S1
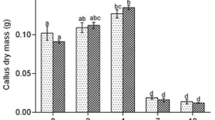
Number of somatic embryos (SEs) formed by callus in the different subcultures in the presence of 2,4-d after 28 days of maturation. Means followed by different letters are significantly different (P < 0.05) according to the Student–Newman–Keuls test. S1–S8 are callus from the first to eighth subculture with 2,4-d (n = 9, coefficient of variation = 19.35%) (TIFF 819 kb)
Table S1
Complete list of identified proteins (XLSX 1698 kb)
Rights and permissions
About this article
Cite this article
Passamani, L.Z., Reis, R.S., Vale, E.M. et al. Long-term culture with 2,4-dichlorophenoxyacetic acid affects embryogenic competence in sugarcane callus via changes in starch, polyamine and protein profiles. Plant Cell Tiss Organ Cult 140, 415–429 (2020). https://doi.org/10.1007/s11240-019-01737-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-019-01737-w