Abstract
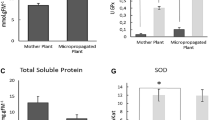
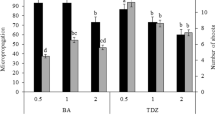
The medicinal plant Piper crassinervium is a source of bioactive compounds with potential use in agrochemical and pharmaceutical industries. However, its propagation is slow and influenced by the composition of the culture medium and growth conditions. In the genus Piper, tissue oxidation limits the biomass and recovery of bioactive compounds such as phenols. We evaluated the effect of medium formulations, sucrose, growth regulators, medium consistency, explant type, gas exchange and irradiance levels on growth and phenols and total flavonoids content in P. crassinervium in vitro plants. Murashige and Skoog (MS) and DKW/Juglans (DKW/J) media induced the highest production of chlorophyll and phenolic compounds; however, the regeneration rate of the explants in the DKW/J medium was the lowest (~75%) in comparison with the other media (90%). The maximum biomass production was achieved when half-strength MS (½MS) liquid medium was used; while the optimum sucrose level depended on the medium salt concentration used. Notably, the irradiance altered the accumulation of biomass and phenolic compounds. The presence of one remaining leaf in the nodal segments, used as explants, produced taller plants in ½MS liquid medium, combined or not with 2-iP. The optimal growth conditions for biomass and phenolic compound production in P. crassinervium in vitro plants established were: nodal explant with a preexisting leaf cultured on ½MS stationary liquid medium, 15 g L−1 sucrose, using lids with one PTFE 0.45-µm-pore size membrane and an irradiance of 100 µmol m−2 s−1.
Key message
Medium formulation, sucrose concentration, irradiance levels, gas exchange and explants with preexisting leaf improved the in vitro propagation and phenolic compounds production in Piper crassinervium.





Similar content being viewed by others
References
Ahmad N, Abbasi BH, Fazal H, Khan MA, Afridi MS (2014) Effect of reverse photoperiod on in vitro regeneration and piperine production in Piper nigrum L. C R Biol 337:19–28. https://doi.org/10.1016/j.crvi.2013.10.011
Alvarenga ICA, Pacheco FV, Silva ST, Bertolucci SKV, Pinto JEBP (2015) In vitro culture of Achillea millefolium L.: quality and intensity of light on growth and production of volatiles. Plant Cell Tissue Organ Cult 122:299–308. https://doi.org/10.1007/s11240-015-0766-7
Anand A, Rao CS (2000) A rapid in vitro propagation protocol for Piper barberi Gamble, a critically endangered plant. In Vitro Cell Dev Biol–Plant 36:61–64. https://doi.org/10.1007/s11627-000-0014-6
Araujo L, Bispo WS, Rios VS, Fernandes SA, Rodrigues FA (2015) Induction of the phenylpropanoid pathway by acibenzolar-s-methyl and potassium phosphite increases mango resistance to Ceratocystis fimbriata infection. Plant Dis 99:447–459. https://doi.org/10.1094/PDIS-08-14-0788-RE
Badr A, Angers P, Desjardins Y (2011) Metabolic profiling of photoautotrophic and photomixotrophic potato plantlets (Solanum tuberosum) provides new insights into acclimatization. Plant Cell Tissue Organ Cult 107:13–24. https://doi.org/10.1007/s11240-011-9951-5
Balbuena TS, Santa-Catarina C, Silveira V, Kato MJ, Floh EIS (2009) In vitro morphogenesis and cell suspension culture establishment in Piper solmsianum C. DC. (Piperaceae). Acta Bot Brasilica 23:274–281. https://doi.org/10.1590/S0102-33062009000100029
Baque MA, Lee EJ, Paek KY (2010) Medium salt strength induced changes in growth, physiology and secondary metabolite content in adventitious roots of Morinda citrifolia: the role of antioxidant enzymes and phenylalanine ammonia lyase. Plant Cell Rep 29:685–694. https://doi.org/10.1007/s00299-010-0854-4
Batista DS, Dias LLC, Rêgo MMD, Saldanha CW, Otoni WC (2017) Flask sealing on in vitro seed germination and morphogenesis of two types of ornamental pepper explants. Cienc Rural 47:1–6. https://doi.org/10.1590/0103-8478cr20150245
Bhat SR, Kackar A, Chandel KPS (1992) Plant regeneration from callus cultures of Piper longum L. by organogenesis. Plant Cell Rep 11:525–528. https://doi.org/10.1007/BF00236270
Cáceres A, Kato MJ (2014) Importance of a multidisciplinary evaluation of Piper genus for development of new natural products in Latin America. Int J Phytocos Nat Ingred 1:1–7. https://doi.org/10.15171/ijpni.2014.04
Cavalcante M (2002) Cultivo da pimenta longa (Piper hispidinervum) na Amazônia Ocidental. Embrapa Acre. Sistema de Produção, 29 p
Clemente JM, Martinez HEP, Pedrosa AW, Poltronieri Neves Y, Cecon PR, Jifon JL (2018) Boron, copper, and zinc affect the productivity, cup quality, and chemical compounds in coffee beans. J Food Qual. https://doi.org/10.1155/2018/7960231
Correia D, Gonçalves AN, Couto HYZ, Ribeiro MC (1995) Efeito do meio de cultura líquido e sólido no crescimento e desenvolvimento de gemas de Eucalyptus grandis. x Eucalyptus urophylla na multiplicação in vitro. Instituto de Pesquisas e Estudos Florestais 48:107–116
Costa CT, de Almeida MR, Ruedell CM, Schwambach J, Maraschin FDS, Fett-Neto AG (2013) When stress and development go hand in hand: main hormonal controls of adventitious rooting in cuttings. Front Plant Sci 4:133. https://doi.org/10.3389/fpls.2013.00133
Cruz CD (2013) Genes: a software package for analysis in experimental statistics and quantitative genetics. Acta Sci Agron 35:271–276. https://doi.org/10.4025/actasciagron.v35i3.21251
Cuenca B, Sánchez C, Aldrey A, Bogo B, Blanco B, Correa B, Vidal N (2017) Micropropagation of axillary shoots of hybrid chestnut (Castanea sativa x C. crenata) in liquid medium in a continuous immersion system. Plant Cell Tissue Organ Cult 131:307–320. https://doi.org/10.1007/s11240-017-1285-5
Cui HY, Baque MA, Lee EJ, Paek KY (2013) Scale-up of adventitious root cultures of Echinacea angustifolia in a pilot-scale bioreactor for the production of biomass and caffeic acid derivatives. Plant Biotechnol Rep 7:297–308. https://doi.org/10.1007/s11816-012-0263-y
Danelutte AP, Costantin MB, Delgado GE, Braz-Filho R, Kato MJ (2005) Divergence of secondary metabolism in cell suspension cultures and differentiated plants of Piper cernuum and P. crassinervium. J Braz Chem Soc 16:1425–1430. https://doi.org/10.1590/S0103-50532005000800022
Delgado-Paredes GE, Kato MJ, Vásquez-Dueñas N, Minchala-Patiño J, Rojas-Idrogo C (2012) Cultivo de tejidos de Piper sp. (Piperaceae): Propagación, organogénesis y conservación de germoplasma in vitro. Rev Colomb Biotecnol 14:49–60
Dias MI, Sousa MJ, Alves RC, Ferreira IC (2016) Exploring plant tissue culture to improve the production of phenolic compounds: a review. Ind Crops Prod 82:9–22. https://doi.org/10.1016/j.indcrop.2015.12.016
Dousseau S, Alvarenga AAD, Alves E, Chaves IDS, Souza EDS, Alves JDS (2011) Physiological, morphological and biochemical characteristics of the sexual propagation of Piper aduncum (Piperaceae). Rev Bras Bot 34:297–305. https://doi.org/10.1590/S0100-84042011000300005
Driver JA, Kuniyuki AH (1984) In vitro propagation of Paradox walnut rootstock [Juglans hindsii x Juglans regia, tissue culture]. HortScience 19:507–509
Farzadfar S, Zarinkamar F, Hojati M (2017) Magnesium and manganese affect photosynthesis, essential oil composition and phenolic compounds of Tanacetum parthenium. Plant Physiol Biochem 112:207–217. https://doi.org/10.1016/j.plaphy.2017.01.002
Figueiredo RA, Sazima M (2004) Pollination ecology and resource partitioning in neotropical Pipers. In: Dyer LA, Palmer ADN (eds) Piper: a model genus for studies of phytochemistry, ecology, and evolution, 1st edn. Kluwer Academic/Plenum Publishers, New York, pp 33–57
Flores P, Carvajal M, Cerdá A, Martínez V (2001) Salinity and ammonium/nitrate interactions on tomato plant development, nutrition, and metabolites. J Plant Nutr 24:1561–1573. https://doi.org/10.1081/PLN-100106021
Fu Y, Li H, Yu J, Liu H, Cao Z, Manukovsky NS, Liu H (2017) Interaction effects of light intensity and nitrogen concentration on growth, photosynthetic characteristics and quality of lettuce (Lactuca sativa L. Var. youmaicai). Sci Hortic 214:51–57. https://doi.org/10.1016/j.scienta.2016.11.020
Ghimire BK, Kim HY, Seong ES, Yoo JH, Lee JH, Park SK, Kim S-H, Yu C-Y, Chung IM (2018) Establishment of culturing conditions and assessment of antioxidant activity and somaclonal variation in the adventitious root suspension cultures of Oplopanax elatus Nakai. Acta Physiol Plant 40:51. https://doi.org/10.1007/s11738-018-2625-8
Giri L, Dhyani P, Rawat S, Bhatt ID, Nandi SK, Rawal RS, Pande V (2012) In vitro production of phenolic compounds and antioxidant activity in callus suspension cultures of Habenaria edgeworthii: a rare Himalayan medicinal orchid. Ind Crops Prod 39:1–6. https://doi.org/10.1016/j.indcrop.2012.01.024
Gomes EN, Krinski D (2018) Piper crassinervium Kunth vegetative propagation: influence of substrates and stem cuttings positions. Appl Res Agrotechnol 11:51–59. https://doi.org/10.5935/PAeT.V11.N3.05
Guo W, Nazim H, Liang Z, Yang D (2016) Magnesium deficiency in plants: an urgent realistic problem. Crop J 4:83–91. https://doi.org/10.1016/j.cj.2015.11.003
Gupta PK, Timmis R (2005) Mass propagation of conifer trees in liquid cultures-progress towards commercialization. Plant Cell Tissue Organ Cult 81:339–346. https://doi.org/10.1007/1-4020-3200-5_30
Hermans C, Verbruggen N (2005) Physiological characterization of Mg deficiency in Arabidopsis thaliana. J Exp Bot 56:2153–2161. https://doi.org/10.1093/jxb/eri215
Hoang NN, Kitaya Y, Morishita T, Endo R, Shibuya T (2017) A comparative study on growth and morphology of wasabi plantlets under the influence of the micro-environment in shoot and root zones during photoautotrophic and photomixotrophic micropropagation. Plant Cell Tissue Organ Cult 130:255–263. https://doi.org/10.1007/s11240-017-1219-2
Isah T, Umar S, Mujib A, Sharma MP, Rajasekharan PE, Zafar N, Frukh A (2018) Secondary metabolism of pharmaceuticals in the plant in vitro cultures: strategies, approaches, and limitations to achieving higher yield. Plant Cell Tissue Organ Cult 132:239–265. https://doi.org/10.1007/s11240-017-1332-2
Jaramillo MA, Callejas R, Davidson C, Smith JF, Stevens AC, Tepe EJ (2008) A phylogeny of the tropical genus Piper using ITS and the chloroplast intron psbJ–petA. Syst Bot 33:647–660. https://doi.org/10.1600/036364408786500244
Kato MJ, Furlan M (2007) Chemistry and evolution of the Piperaceae. Pure Appl Chem 79:529–538. https://doi.org/10.1351/pac200779040529
Kozai T (2010) Photoautotrophic micropropagation environmental control for promoting photosynthesis. Propag Ornam Plants 10:188–204
Lago JHG, Kato MJ (2007) 3α,4α-Epoxy-2-piperidone, a new minor derivative from leaves of Piper crassinervium Kunth (Piperaceae). Nat Prod Res 21:910–914. https://doi.org/10.1080/14786410601130711
Lloyd G, McCown B (1981) Commercially-feasible micropropagation of mountain laurel, Kalmia latifolia, by use of shoot-tip culture. Int Plant Propag Soc 30:421–427
Lopes AA, López SN, Regasini LO, Junior JMB, Ambrosio DL, Kato MJ, Bolzani VS, Cicarelli B, Furlan M (2008) In vitro activity of compounds isolated from Piper crassinervium against Trypanosoma cruzi. Nat Prod Res 22:1040–1046. https://doi.org/10.1080/14786410802243271
López SN, Lopes AA, Batista JM Jr, Flausino O Jr, da Silva Bolzani V, Kato MJ, Furlan M (2010) Geranylation of benzoic acid derivatives by enzymatic extracts from Piper crassinervium (Piperaceae). Bioresour Technol 101:4251–4260. https://doi.org/10.1016/j.biortech.2010.01.041
Lucini L, Borgognone D, Rouphael Y, Cardarelli M, Bernardi J, Colla G (2016) Mild potassium chloride stress alters the mineral composition, hormone network, and phenolic profile in artichoke leaves. Front Plant Sci 7:948. https://doi.org/10.3389/fpls.2016.00948
Madhusudhanan K, Rahiman BA (2000) The effect of activated charcoal supplemented media to browning of in vitro cultures of Piper species. Biol Plant 43:297–299. https://doi.org/10.1023/A:100272903
Magangana TP, Stander MA, Makunga NP (2018) Effect of nitrogen and phosphate on in vitro growth and metabolite profiles of Stevia rebaudiana Bertoni (Asteraceae). Plant Cell Tissue Organ Cult 134:141–151. https://doi.org/10.1007/s11240-018-1407-8
Martins V, Garcia A, Costa C, Sottomayor M, Gerós H (2018) Calcium-and hormone-driven regulation of secondary metabolism and cell wall enzymes in grape berry cells. J Plant Physiol 231:57–67. https://doi.org/10.1016/j.jplph.2018.08.011
Matsuura HN, Malik S, Costa F, Yousefzadi M, Mirjalili MH, Arroo R, Bhambra AS, Strnad M, Bonfill M, Fett-Neto AG (2018) Specialized plant metabolism characteristics and impact on target molecule biotechnological production. Mol Biotechnol 60:169–183. https://doi.org/10.1007/s12033-017-0056-1
McGranahan GH, Driver JA, Tulecke W (1987) Tissue culture of Juglans. In: Bonga JM, Durzan DJ (eds) Cell and tissue culture in forestry. Forestry Sciences, vol. 24–26. Springer, Dordrecht, pp 261–271
Mesén F, Newton AC, Leakey RR (1997) Vegetative propagation of Cordia alliodora (Ruiz & Pavon) Oken: the effects of IBA concentration, propagation medium and cutting origin. For Ecol Manag 92:45–54. https://doi.org/10.1016/S0378-1127(96)03960-6
Modarres M, Bahabadi SE, Yazdi MET (2018) Enhanced production of phenolic acids in cell suspension culture of Salvia leriifolia Benth. using growth regulators and sucrose. Cytotechnology 70:741–750. https://doi.org/10.1007/s10616-017-0178-0
Mohamed MAH, Ibrahim TA (2012) Enhanced in vitro production of Ruta graveolens L. coumarins and rutin by manitol and ventilation. Plant Cell Tissue Organ Cult 111:335–343. https://doi.org/10.1007/s11240-012-0199-5
Morandim-Giannetti ADA, Pin AR, Santo Pietro NA, de Oliveira HC, Mendes-Giannini MJS, Alecio AC, Kato MJ, de Oliveira JE, Furlan M (2010) Composition and antifungal activity against Candida albicans, Candida parapsilosis, Candida krusei and Cryptococcus neoformans of essential oils from leaves of Piper and Peperomia species. J Med Plant Res 47:1810–1814. https://doi.org/10.5897/JMPR09.303
Mosquera OM, González LM, Cortes YJ, Camargo JC (2015) Caracterización fitoquímica, determinación del contenido de lignina y la actividad antioxidante de los culmos de Guadua angustifolia Kunth. Rev Facultad Ciencias Básicas 11:124–135
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15:473–497. https://doi.org/10.1111/j.1399-3054.1962.tb08052.x
Musarurwa HT, Koegelenberg L, Makunga NP (2012) Chemical variation in essential oil profiles detected using headspace solid-phase microextraction gas chromatography spectrometry in response to potassium, nitrogen, and water available to micropropagated plants of Salvia stenophylla (Burch. ex Benth.). J Plant Growth Regul 31:207–220. https://doi.org/10.1007/s00344-011-9232-x
Newton AC, Muthoka PN, Dick JM (1992) The influence of leaf area on the rooting physiology of leafy stem cuttings of Terminalia spinosa Engl. Trees 6:210–215. https://doi.org/10.1007/BF00224338
Nirmal Babu K, Divakaran M, Yamuna G, Ravindran PN, Peter KV (2016) Protocols for improvement of black pepper (Piper nigrum L.) utilizing biotechnological tools. In: Jain SM (ed) Protocols for in vitro cultures and secondary metabolite analysis of aromatic and medicinal plants, 2nd edn. Humana Press, New York, pp 367–385
Panhwar QA, Naher UA, Radziah O, Shamshuddin J, Razi IM, Dipti SS, Aghamolki MK (2015) Quality and antioxidant activity of rice grown on alluvial soil amended with Zn, Cu and Mo. S Afr J Bot 98:77–83. https://doi.org/10.1016/j.sajb.2015.01.021
Parmar VR, Jasrai YT (2018) Improved production of Oroxylum indicum (L.) Kurz by altering the salts of the medium. In Vitro Cell Dev Biol–Plant 54:79–86. https://doi.org/10.1007/s11627-017-9863-0
Pérez-López U, Sgherri C, Miranda-Apodaca J, Micaelli F, Lacuesta M, Mena-Petite A, Quartacci MF, Muñoz-Rueda A (2018) Concentration of phenolic compounds is increased in lettuce grown under high light intensity and elevated CO2. Plant Physiol Biochem 123:233–241. https://doi.org/10.1016/j.plaphy.2017.12.010
Perigo CV, Torres RB, Bernacci LC, Guimarães EF, Haber LL, Facanali R, Vieira MAR, Quecini V, Marques MOM (2016) The chemical composition and antibacterial activity of eleven Piper species from distinct rainforest areas in Southeastern Brazil. Ind Crops Prod 94:528–539. https://doi.org/10.1016/j.indcrop.2016.09.028
Pinheiro MVM, Martins FB, Xavier A, Otoni WC (2013) Trocas gasosas influenciam na morfogênese in vitro de duas cultivares de oliveira (Olea europaea L.). Rev Árvore 37:19–29
Poorter H, Nagel O (2000) The role of biomass allocation in the growth response of plants to different levels of light, CO2, nutrients and water: a quantitative review. Aust J Plant Physiol 27:595–607. https://doi.org/10.1071/PP99173_CO
Quiala E, Cañal MJ, Meijón M, Rodríguez R, Chávez M, Valledor L, de Feria M, Barbón R (2012) Morphological and physiological responses of proliferating shoots of teak to temporary immersion and BA treatments. Plant Cell Tissue Organ Cult 109:223–234. https://doi.org/10.1007/s11240-011-0088-3
Quijano-Abril M, Callejas-Posada R, Miranda-Esquivel DR (2006) Areas of endemism and distribution patterns for Neotropical Piper species (Piperaceae). J Biogeogr 33:1266–1278. https://doi.org/10.1111/j.1365-2699.2006.01501.x
Quoirin M, Lepoivre P (1977) Etude de milieux adaptes aux cultures in vitro de Prunus. Acta Hortic 78:437–442
Rani D, Dantu PK (2012) Direct shoot regeneration from nodal, internodal and petiolar segments of Piper longum L. and in vitro conservation of indexed plantlets. Plant Cell Tissue Organ Cult 109:9–17. https://doi.org/10.1007/s11240-011-0068-7
Rugini E (1984) In vitro propagation of some olive (Olea europaea L.) cultivars with different root ability, and medium development using analytical data from developing shoots and embryos. Sci Hortic 24:123–134. https://doi.org/10.1016/0304-4238(84)90143-2
Sáez PL, Bravo LA, Latsague MI, Toneatti MJ, Sánchez-Olate M, Ríos DG (2013) Light energy management in micropropagated plants of Castanea sativa, effects of photoinhibition. Plant Sci 201:12–24. https://doi.org/10.1016/j.plantsci.2012.11.008
Saidani F, Giménez R, Aubert C, Chalot G, Betrán JA, Gogorcena Y (2017) Phenolic, sugar and acid profiles and the antioxidant composition in the peel and pulp of peach fruits. J Food Compos Anal 62:126–133. https://doi.org/10.1016/j.jfca.2017.04.015
Saldanha CW, Otoni CG, Azevedo JLF, Dias LLC, Rêgo MM, Otoni WC (2012) A low-cost alternative membrane system that promotes growth in nodal cultures of Brazilian ginseng [Pfaffia glomerata (Spreng.) Pedersen]. Plant Cell Tissue Organ Cult 110:413–422. https://doi.org/10.1007/s11240-012-0162-5
Santos RP, Cruz ACF, Iarema L, Kuki KN, Otoni WC (2008) Protocolo para extração de pigmentos foliares em porta-enxertos de videira micropropagados. Rev Ceres 55:356–364
Savio LEB, Astarita LV, Santarém ER (2012) Secondary metabolism in micropropagated Hypericum perforatum L. grown in non-aerated liquid medium. Plant Cell Tissue Organ Cult 108:465–472. https://doi.org/10.1007/s11240-011-0058-9
Schneider CA, Rasband WS, Eliceiri KW (2012) NIH Image to ImageJ: 25 years of image analysis. Nat Methods 9:671–675. https://doi.org/10.1038/nmeth.2089
Schulten A, Krämer U (2017) Interactions between copper homeostasis and metabolism in plants. Prog Bot 79:111–146. https://doi.org/10.1007/124_2017_7
Sencie-Tarazona A, Rojas-Idrogo C, Kato M, Delgado-Paredes GE (2015) In vitro rapid clonal propagation in static liquid medium and acclimatization of Piper solmsianum and P. tuberculatum. Int J Pure Appl Biosci 3:1–10
Silva ST, Pacheco FV, Alvarenga ICA, Pinto JEBP, Bertolucci SKV, Ferreira CP (2014) Optimization of the protocol for the in vitro cultivation of Piper aduncum L. Am J Plant Sci 5:3474–3482. https://doi.org/10.4236/ajps.2014.523363
Singleton VL, Rossi JA (1965) Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am J Enol Vitic 16:144–158
Stevens ME, Pijut PM (2018) Rapid in vitro shoot multiplication of the recalcitrant species Juglans nigra L. In Vitro Cell Dev Biol–Plant 54:309–317. https://doi.org/10.1007/s11627-018-9892-3
Szopa A, Kokotkiewicz A, Bednarz M, Luczkiewicz M, Ekiert H (2017) Studies on the accumulation of phenolic acids and flavonoids in different in vitro culture systems of Schisandra chinensis (Turcz.) Baill. using a DAD-HPLC method. Phytochem Lett 20:462–469. https://doi.org/10.1016/j.phytol.2016.10.016
Tchoundjeu Z, Avana ML, Leakey RRB, Simons AJ, Assah E, Duguma B, Bell JM (2002) Vegetative propagation of Prunus africana: effects of rooting medium, auxin concentrations and leaf area. Agrofor Syst 54:183–192. https://doi.org/10.1023/A:1016049004139
Tognolini M, Barocelli E, Ballabeni V, Bruni R, Bianchi A, Chiavarini M, Impicciatore M (2006) Comparative screening of plant essential oils: phenylpropanoid moiety as basic core for antiplatelet activity. Life Sci 78:1419–1432. https://doi.org/10.1016/j.lfs.2005.07.020
Valentin-Silva A, Staggemeier VG, Batalha MA, Guimarães E (2018) What factors can influence the reproductive phenology of Neotropical Piper species (Piperaceae) in a semi-deciduous seasonal forest? Botany 96:675–684. https://doi.org/10.1139/cjb-2018-0029
Wang Y, Gao L, Wang Z, Liu Y, Sun M, Yang D, Wei C, Shan Y, Xia T (2012) Light-induced expression of genes involved in phenylpropanoid biosynthetic pathways in callus of tea (Camellia sinensis (L.) O. Kuntze). Sci Hortic 133:72–83. https://doi.org/10.1016/j.scienta.2011.10.017
Wellburn AR (1994) The spectral determination of chlorophylls a and b, as well as total carotenoids, using various solvents with spectrophotometers of different resolution. J Plant Physiol 144:307–313. https://doi.org/10.1016/S0176-1617(11)81192-2
Yamaguchi LF, Lago JHG, Tanizaki TM, Di Mascio P, Kato MJ (2006) Antioxidant activity of prenylated hydroquinone and benzoic acid derivatives from Piper crassinervium. Kunth. Phytochemistry 67:1838–1843. https://doi.org/10.1016/j.phytochem.2006.03.001
Zhishen J, Mengcheng T, Jianming W (1999) The determination of flavonoid contents in mulberry and their scavenging effects on superoxide radicals. Food Chem 64:555–559. https://doi.org/10.1016/S0308-8146(98)00102-2
Zhou W, Chen Y, Xu H, Liang X, Hu Y, Jin C, Lu L, Lin X (2018) Short-term nitrate limitation prior to harvest improves phenolic compound accumulation in hydroponic-cultivated lettuce (Lactuca sativa L.) without reducing shoot fresh weight. J Agric Food Chem 66:10353–10361. https://doi.org/10.1021/acs.jafc.8b02157
Zhu H, Li X, Zhai W, Liu Y, Gao Q, Liu J, Ren L, Chen H, Zhu Y (2017) Effects of low light on photosynthetic properties, antioxidant enzyme activity, and anthocyanin accumulation in purple pak-choi (Brassica campestris ssp. Chinensis Makino). PLoS ONE 12:e0179305. https://doi.org/10.1371/journal.pone.0179305
Zoratti L, Karppinen K, Luengo Escobar A, Häggman H, Jaakola L (2014) Light-controlled flavonoid biosynthesis in fruits. Front Plant Sci 5:534. https://doi.org/10.3389/fpls.2014.00534
Acknowledgements
The authors are thankful to Prof. Oscar Marino Mosquera and MSc. Yeny Juliana Cortes of the Grupo de Biotecnología—Productos Naturales (GB-PN) [Escuela de Tecnología Química, Universidad Tecnológica de Pereira (UTP), Pereira, Risaralda, Colombia] for their guidance in the establishment of chemical analysis protocols; to Dr. Adriano Valentin-Silva (Graduate Program of Biological Sciences (Botany), Institute of Biosciences, UNESP—São Paulo State University, Botucatu, SP, Brazil) for his assistance in plant identification and collection of seeds of P. crassinervium and to Dr. Manuel Alejandro Ix Balam for the schematic drawing of the Fig. 1. The financial support was provided by Fundacão de Amparo à Pesquisa do Estado de Minas Gerais—Brazil (FAPEMIG), Conselho Nacional de Desenvolvimento Científico e Tecnológico—Brazil (CNPq) and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior—Brasil (CAPES—Finance Code 001). WCO and SAF are supported by Research Fellowships from CNPq.
Author information
Authors and Affiliations
Contributions
AMRR, JVSS and JVMF raised the in vitro plants for the experiments and performed the experiments. AMRR, JVSS, JVMF, TDS, KC and DVF performed growth and photosynthetic pigments analysis. AMRR, JVSS, JVMF and DVF performed extractions and phytochemical analysis. AMRR, DSB and MVMP performed statistical analyses. AMRR, DSB, MVMP, DVF, WCO and SAF designed the research, interpreted the data and wrote the paper.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there are no conflicts of interest.
Additional information
Communicated by Silvia Moreno.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.

11240_2019_1585_MOESM2_ESM.jpg
Online Resource 2 In vitro plant growth variables of Piper crassinervium after 35 days of culture under different salt medium composition (a – f), culture medium consistency and sealing systems (g – l) and salt concentration of MS medium (m – r). (MS: Murashige and Skoog; DKW/J: DKW/Junglans; QL: Quoirin and Lepoivre; WPM: McCown’s Woody Plant; JADSm: JADS without the antioxidant polyvinylpyrrolidone (PVP); SL: Stirring liquid medium; StL: Stationary liquid medium; S: Semi-solid medium; 0M and 1M: Autoclavable rigid polypropylene lid without membrane and cover with 1 membrane; ½MS: MS medium with half the concentration of macro and micronutrients. Means followed by the same lowercase letter did not differ significantly among treatments (Tukey’s test at 5%). Error bars indicate the standard error among replicates). (JPG 5045 KB)

11240_2019_1585_MOESM3_ESM.jpg
Online Resource 3 In vitro plant growth variables of Piper crassinervium after 35 days of culture under irradiance of 45, 70, 100 and 150 µmol m-2 s-1 (a – f) and from stem segments without, with half and one preexisting leaf, respectively (0L, ½L and 1L) (g – l). (Means followed by the same lowercase letter do not differ significantly among treatments (Tukey’s test at 5%). Error bars indicate the standard error among the replicates). (JPG 3678 KB)
11240_2019_1585_MOESM4_ESM.doc
Online Resource 4 Growth variables of Piper crassinervium plantlet grown in vitro under different photoperiods. (DOC 40 KB)
11240_2019_1585_MOESM5_ESM.doc
Online Resource 5 Growth variables of Piper crassinervium plantlet grown in vitro with different growth regulators. (DOC 41 KB)

11240_2019_1585_MOESM6_ESM.jpg
Online Resource 6 In vitro plant growth variables of Piper crassinervium after 35 days of in vitro culture in ½MS liquid medium supplemented with 2-iP (2, 5 and 10 μM) (a - i) and ½OM and ½MS medium in combination with sucrose (7.5, 15 and 30 g L-1) (j - o). (2-iP: N6-(2-isopentenyl) adenine; ½OM and ½MS: Rugini Olive medium (OM) and Murashige and Skoog (MS) medium with half the concentration of macro and micronutrients. Means followed by the same lowercase letter do not differ significantly between 2-iP and sucrose concentrations; Averages followed by the same capital letter do not differ when comparing means ½OM and ½MS (Tukey’s test at 5%). Error bars indicate the standard error among the replicates). (JPG 4897 KB)
Rights and permissions
About this article
Cite this article
Ríos-Ríos, A.M., da Silva, J.V.S., Fernandes, J.V.M. et al. Micropropagation of Piper crassinervium: an improved protocol for faster growth and augmented production of phenolic compounds. Plant Cell Tiss Organ Cult 137, 495–509 (2019). https://doi.org/10.1007/s11240-019-01585-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-019-01585-8