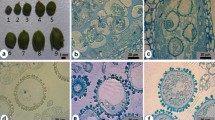
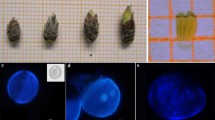
Abstract
Haploid technology is a valuable plant breeding tool for obtaining homozygosity particularly in woody plants. Hazelnut, the world’s sixth ranking nut tree crop is a monoecious, anemophilous species. It is characterized by a sporophytic incompatibility system that prevents production of homozygous plants with conventional methods, involving several self-pollination cycles. In this study, gametic embryogenesis, in particular isolated microspore culture, was tried with five genotypes. Two culture media were tested and four temperature stress treatments were applied to the isolated microspores that were cultivated at the vacuolated developmental stage. To our knowledge, this is the first report of early embryos being recovered from the five hazelnut genotypes via isolated microspore culture. Analysis of the embryo genotypes using SSR markers demonstrated the embryos had a single allele per each locus whereas the parent cultivar was heterozygous, indicating they developed from haploid microspores. The response to the culture treatments, was, however, genotype-dependent, as previously reported for male gamete embryogenesis in other fruit crops.







Similar content being viewed by others
References
Aleza P, Juárez J, Hernández M, Pina JA, Ollitrault P, Navarro L (2009) Recovery and characterization of a Citrus clementina Hort. ex Tan. ‘Clemenules’ haploid plant selected to establish the reverence whole Citrus genome sequence. BMC Plant Biol. doi 10.1189/1471-2229-9-110. http://www.biomedcentral.com/1471-2229/110
Bàràny I, Testillano PS, Mitykó J, Risueño MC (2001) The switch of the microspore developmental program in Capsicum involves HSP70 expression and leads to the production of haploid plants. Int J Dev Biol 45(S1):S39–S40
Bhowmik P, Dirpaul J, Ferrie AM (2011) A high throughput Brassica napus microspore culture system: influence of percoll gradient separation and bud selection on embryogenesis. Plant Cell Tissue Org Cult 106:95–107
Binarova P, Hause G, Cenklová V, Cordewener JH, Campagne ML (1997) A short severe heat shock is required to induce embryogenesis in late bicellular pollen of Brassica napus L. Sex Plant Reprod 10:200–208
Boccacci P, Akkak A, Bassil NV, Mehlenbacher SA, Botta R (2005) Characterization and evaluation of microsatellite loci in European hazelnut (Corylus avellana L.) and their transferability to other Corylus species. Mol Ecol Notes 5:934–937
Boccacci P, Botta R, Rovira M (2008) Genetic diversity of hazelnut (Corylus avellana L.) germplasm in Northeastern Spain. Hort Sci 43(3):667–672
Bueno MA, Gómez A, Sepúlveda F, Seguí JM, Testillano PS, Manzanera JA, Risueño MC (2003) Microspore-derived embryos from Quercus suber anthers mimic zygotic embryos and maintain haploidy in long-term anther culture. J Plant Physiol 160:953–960
Chiancone B, Germanà MA (2016) Microspore embryogenesis through anther culture in Citrus clementina Hort. ex Tan. In: Germanà MA, Lambardi M (eds) In vitro embryogenesis in higher plants., Methods in molecular biologySpringer, Berlin, pp 475–487
Chiancone B, Gniech Karasawa MM, Abdelgalel A, Germana’ MA (2013) Study on anther culture in hazelnut (Corylus avellana L.). In: European frontiers of plant reproduction research 2013. Final conference of the COST action FA0903 “Harnessing Plant Reproduction for Crop Improvement”. Oslo, Norway, 2–4 Oct 2013, pp 77–78
Chiancone B, Gniech Karasawa MM, Gianguzzi V, Mohamed AA, Bárány I, Testillano P, Torello Marinoni D, Botta R, Germanà MA (2015) Early embryo achievement through isolated microspore culture in Citrus clementina Hort. ex Tan., cvs. ‘Monreal Rosso’ and ‘Nules’. Front Plant Sci. 6:00413. http://dx.doi.org/10.3389/fpls.2015.00413 ISSN = 1664-462X
Chu C (1978) The N6 medium and its applications to anther culture of cereal crops. In: Proceedings of symposium on plant tissue culture. Science Press, Peking, pp 43–50
Chu CG, Xu SS, Friesen TL, Faris JD (2008) Whole genome mapping in wheat doubled haploid population using SSRs and TRAPs and the identification of QTL for agronomic traits. Mol Breed 22:251–266
Custers JBM, Cordewener JHG, NöLlen Y, Dons HJM, Van Lookeren Campagne MM (1994) Temperature controls both gametophytic and sporophytic development in microspore cultures of Brassica napus. Plant Cell Rep 13:267–271
Datta SK (2005) Androgenic haploids: factors controlling development and its application in crop improvement. Curr Sci 89(11):1870–1878
Dirks R, Van Dun K, Snoo De, Van Den Berg M, Lelivelt CL, Voermans W, Woudenberg L, De Wit JP, Reinink K, Schut JW (2009) Reverse breeding: a novel breeding approach based on engineered meiosis. Plant Biotechnol J 7:837–845
Doyle JJ, Doyle JL (1987) A rapid DNA isolation procedure for small quantities of fresh leaf tissue. Phytochem Bull 19:11–15
FAOSTAT (2015). http://faostat3.fao.org/
Ferrie AMR, Caswell KL (2010) Isolated microspore culture techniques and recent progress for haploid and doubled haploid plant production. Plant Cell Tiss Org Cult 104(3):301–309
Ferrie AM, Möllers C (2011) Haploids and doubled haploids in Brassica spp. for genetic and genomic research. Plant Cell Tiss Org Cult 104:375–386
Foster BP, Herberle-Bors E, Kasha KJ, Touraev A (2007) The resurgence of haploids in higher plants. Trends Plant Sci 12(8):368–375 ISSN 1360-1385. http://dx.doi.org/10.1016/j.tplants.2007.06.007
Germain E (1994) The reproduction of hazelnut (Corylus avellana L.): a review. In: III international congress on Hazelnut, Alba (CN), Italy, 14–18 Sep Acta Hort vol 351, pp 195–209
Germanà MA (2006) Doubled haploid production in fruit tree crops. Plant Cell Tiss Org Cult 86:131–146
Germanà MA (2009) Haploid and doubled haploids in fruit trees. In: Touraev A, Forster B, Jain M (eds) Advances in haploid production in higher plants. Springer, Berlin, pp 241–263
Germanà MA (2011a) Gametic embryogenesis and haploid technology as valuable support to plant breeding. Plant Cell Rep 30:839–857
Germanà MA (2011b) Anther culture for haploid and doubled haploid production. Plant Cell Tiss Org Cult 104:283–300
Germanà MA, Chiancone B (2003) Improvement of Citrus clementina Hort. Ex Tan. microspore-derived embryoid induction and regeneration. Plant Cell Rep 22:181–187
Germanà MA, Aleza P, Carrera E, Chen C, Chiancone B, Costantino G, Dambier D, Deng X, Federici TC, Froelicher Y, Guo W, Ibáñez V, Juárez J, Kwok K, Luro F, Machado AM, Naranjo AM, Navarro L, Ollitrault P, Ríos G, Roose LM, Talon M, Xu Q, Gmitter GF (2013) Cytological and molecular characterization of three gametoclones of Citrus clementina. BMC Plant Biol 13. ISSN: 1471-2229. doi:10.1186/1471-2229-13-129. http://www.biomedcentral.com/1471-2229/13/129
Germanà MA, Scarano MT, Crescimanno FG (1996) First results on isolated microspore culture of citrus. Proc Int Soc Citric 2:882–885
Giménez-Abián M, Rozalén A, Carballo J, Botella L, Pincheira J, López-Sáez J, de la Torre C (2004) HSP90 and checkpoint-dependent lengthening of the G2 phase observed in plant cells under hypoxia and cold. Protoplasma 223:191–196
Höfer M (2004) In vitro androgenesis in apple-improvement of the induction phase. Plant Cell Rep 22(6):365–370
Höfer M, Flachowsky H (2015) Comprehensive characterization of plant material obtained by in vitro androgenesis in apple. Plant Cell Tiss Organ Cult 122:617–628
Höfer M, Touraev A, Heberle-Bors E (1999) Induction of embryogenesis from isolated apple microspores. Plant Cell Rep 18:1012–1017
Hormaza H, Herrero M (1996) Male gametophytic selection as a plant-breeding tool. Sci Hort 65:321–333
Houssain T, Tausend P, Graham G, Ho J (2007) Registration of IBM2 SYN10 doubled haploid mapping population of maize. J Plant Regist 1:81
Kafkas S, Dogan Y (2009) Genetic characterization of hazelnut (Corylus avellana L.) cultivars from Turkey using molecular markers. Hort Sci 44(6):1557–1561
Karami O, Saidi A (2010) The molecular basis for stress-induced acquisition of somatic embryogenesis. Mol Biol Rep 36:2493–2507
Kumlehn J, Serazetdinova L, Hensel G, Becker DE, Lörz H (2006) Genetic transformation of barley (Hordeum vulgare L.) via infection of androgenetic pollen cultures with Agrobacterium tumefaciens. Plant Biotech J 4:251–261
Kunzel GL, Korzun L, Meister A (2000) Cytologically integrated physical restriction fragment length polymorphism maps for the barley genome based on translocation breakpoints. Genetics 154:397–412
Leeuwen H, Monfort A, Zhang H, Puigdomenech P (2003) Identification and characterization of a melon genomic region containing a resistance gene cluster from a constructed BAC library. Microcolinearity between Cucumis melo and Arabidopsis thaliana. Plant Mol Biol 51(5):703–718
Ma R, Yang-Dong G, Seppo P (2004) Comparison of anther and microspore culture in the embryogenesis and regeneration of rye (Secale cereale). Plant Cell Tissue Organ Cult 76:147–157
Martins S, Rovira M, Silva AP, Carnide (2010) Pollen incompatibility in Portuguese hazelnut landraces. In: XXVIII international horticultural congress on science and horticulture for people (IHC2010), Acta Horticulture, p 932
Nitsch C (1977) Culture of isolate microspore. In: Reinert J, Bajaj YPS (eds) Applied and fundamental aspects of plant cell, tissue and organ culture. Springer, Berlin, pp 268–278
Nitsch JP, Nitsch C (1969) Haploid plants from pollen grains. Science 163:85
Ochatt SJ, Revilla MA (2016) From stress to embryos: some of the problems for induction and maturation of somatic embryos. In: Germanà MA, Lambardi M (eds) In vitro embryogenesis in higher plants., Methods in molecular biologySpringer, Berlin, pp 523–535
Ochatt S, Pech C, Grewal R, Conreux C, Lulsdorf M, Jacas L (2009) Abiotic stress enhances androgenesis from isolated microspores of some legume species (Fabaceae). J Plant Physiol 166:1314–1328
Prem D, Solís M-T, Bárány I, Rodríguez-Sanz H, Risueño MC, Testillano PS (2012) A new microspore embryogenesis system under low temperature which mimics zygotic embryogenesis initials, expresses auxin and efficiently regenerates doubled-haploid plants in Brassica napus. BMC Plant Biol. doi:10.1186/1471-2229-12-127
Ribalta FM, Croser JS, Ochatt SJ (2012) Flow cytometry enables identification of sporophytic eliciting stress treatments in gametic cells. J Plant Physiol 169(1):104–110
Rovira M, Aleta N, Germain E, Arus P (1993) Inheritance and linkage relationships of ten isozyme genes in hazelnut. Theor Appl Genet 86:322–328
Seguí-Simarro JM, Nuez F (2008) Pathways to doubled haploidy: chromosome doubling during androgenesis. Cytogenet Genome Res 120:358–369
Shariatpanahi ME, Bal U, Heberle-Bors E, Touraev A (2006) Stresses applied for the re-programming of plant microspores towards in vitro embryogenesis. Physiol Plant 127:519–534
Soriano M, Li H, Boutilier K (2013) Microspore embryogenesis: establishment of the embryo identity and pattern in culture. Plant Reprod 26:181–193
Sopory S, Munshi M (1996) Anther culture. In: Mohan JM et al (eds) In vitro haploid production in higher plants, vol 1. Kluwer, Dordrecht, pp 145–176
Szareijo I, Foster BP (2006) Doubled haploidy and induced mutation. Euphytica 158:359–370
Talón M, Gmitter FG (2008) Citrus genomics. Int J Plant Genomics. doi:10.1155/2008/528361
Touraev A, Ilham A, Vicente O, Heberle-Bors E (1996a) Stress induced microspore embryogenesis from tobacco microspores: an optimized system for molecular studies. Plant Cell Rep 15:561–565
Touraev A, Indrianto A, Wratschko I, Vicente O, Heberle-Bors E (1996b) Efficient microspore embryogenesis in wheat (Triticum aestivum L.) induced by starvation at high temperatures. Sex Plant Reprod 9:209–215
Wang Z, Tarantino G, Yang D, Liu G, Tingey SV, Miao GH, Wang GL (2001) Rice ESTs with disease-resistance gene or defense-response gene like sequences mapped to regions containing major resistance genes or QTLs. Mol Genet Genomics 265:302–310
Zhang KP, Zhao L, Tian JC, Chen GF, Jiang XL, Liu B (2008) A genetic map constructed using a doubled haploid population derived from two elite Chinese common wheat varieties. J Integr Plant Biol 50(8):941–950
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gniech Karasawa, M.M., Chiancone, B., Gianguzzi, V. et al. Gametic embryogenesis through isolated microspore culture in Corylus avellana L.. Plant Cell Tiss Organ Cult 124, 635–647 (2016). https://doi.org/10.1007/s11240-015-0921-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-015-0921-1