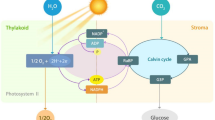
Abstract
The green alga Chlorella ohadii was isolated from a desert biological soil crust, one of the harshest environments on Earth. When grown under optimal laboratory settings it shows the fastest growth rate ever reported for a photosynthetic eukaryote and a complete resistance to photodamage even under unnaturally high light intensities. Here we examined the energy distribution along the photosynthetic pathway under four light and carbon regimes. This was performed using various methodologies such as membrane inlet mass spectrometer with stable O2 isotopes, variable fluorescence, electrochromic shift and fluorescence assessment of NADPH level, as well as the use of specific inhibitors. We show that the preceding illumination and CO2 level during growth strongly affect the energy dissipation strategies employed by the cell. For example, plastid terminal oxidase (PTOX) plays an important role in energy dissipation, particularly in high light- and low-CO2-grown cells. Of particular note is the reliance on PSII cyclic electron flow as an effective and flexible dissipation mechanism in all conditions tested. The energy management observed here may be unique to C. ohadii, as it is the only known organism to cope with such conditions. However, the strategies demonstrated may provide an insight into the processes necessary for photosynthesis under high-light conditions.








Similar content being viewed by others
References
Alboresi A, Storti M, Morosinotto T (2019) Balancing protection and efficiency in the regulation of photosynthetic electron transport across plant evolution. New Phytol 221:105–109. https://doi.org/10.1111/nph.15372
Allahverdiyeva Y, Isojarvi J, Zhang P, Aro E-M (2015) Cyanobacterial oxygenic photosynthesis is protected by flavodiiron proteins. Life 5:716–743. https://doi.org/10.3390/life5010716
Ananyev F, Gates C, Kaplan A, Dismukes GC (2017) Photosystem II-cyclic electron flow powers exceptional photoprotection and record growth in the microalga Chlorella ohadii. Biochem Biophys Acta Bioenerg 1858:873–883. https://doi.org/10.1016/j.bbabio.2017.07.001
Arnon DI (1949) Copper enzymes in isolated chloroplasts. Polyphenol oxidase in beta Vulgaris. Plant Physiol 24:1–15. https://doi.org/10.1104/pp.24.1.1
Aro E-M, Virgin I, Andersson B (1993) Photoinhibition of photosystem II. Inactivation, protein damage and turnover. Biochim Biophys Acta 1143:113–134
Bailleul B, Berne N, Murik O, Petroutsos D, Prihoda J, Tanaka A, Villanova V, Bligny R, Flori S, Falconet D, Krieger-Liszkay A, Santabarbara S, Rappaport F, Joliot P, Tirichine L, Falkowski PG, Cardol P, Bowler C, Finazzi G (2015) Energetic coupling between plastids and mitochondria drives CO2 assimilation in diatoms. Nature 524:366–369. https://doi.org/10.1038/nature14599
Bailleul B, Cardol P, Breyton C, Finazzi G (2010) Electrochromism: a useful probe to study algal photosynthesis. Photosynth Res 106:179–189
Barber J, Andersson B (1992) Too much of a good thing: light can be bad for photosynthesis. Trends Biochem Sci 17:61–66
Beardall J, Raven JA (2020) Acquisition of inorganic carbon by microalgae and cyanobacteria. Microbial photosynthesis. Springer, Singapore. https://doi.org/10.1007/978-981-15-3110-1_8
Bersanini L, Allahverdiyeva Y, Battchikova N, Heinz S, Lespinasse M, Ruohisto E, Mustila H, Nickelsen J, Vass I, Aro E-M (2017) Dissecting the photoprotective mechanism encoded by the flv4-2 operon: a distinct contribution of Sll0218 in photosystem II stabilization. Plant Cell Environ 40:378–389
Bonente G, Pippa S, Castellano S, Bassi R, Ballottari M (2012) Acclimation of Chlamydomonas reinhardtii to different growth irradiances. J Biol Chem 287:5833–5847. https://doi.org/10.1074/jbc.M111.304279
Burlacot A, Sawyer A, Cuiné S, Auroy-Tarrago P, Blangy S, Happe T, Peltier G (2018) Flavodiiron-mediated O2 photoreduction links H2 production with CO2 fixation during the anaerobic induction of photosynthesis. Plant Physiol. https://doi.org/10.1104/pp.18.00721
Campbell DA, Tyystjärvi E (2012) Parameterization of photosystem II photoinactivation and repair. Biochim Biophys Acta Bioenerg 1817:258–265
Cardona T, Sedoud A, Cox N, Rutherford AW (2012) Charge separation in Photosystem II: a comparative and evolutionary overview. Biochim Biophys Acta-Bioenerg 1817:26–43. https://doi.org/10.1016/j.bbabio.2011.07.012
Choubeh RR, Bar-Eyal L, Paltiel Y, Keren N, Struik PC, van Amerongen H (2020) Photosystem II core quenching in desiccated Leptolyngbya ohadii. Photosynth Res 143:13–18. https://doi.org/10.1007/s11120-019-00675-0
Croce R, van Amerongen H (2017) The complex that conquered the land. Science. https://doi.org/10.1126/science.aao4191
Eisenstadt D, Ohad I, Keren N, Kaplan A (2008) Changes in the photosynthetic reaction center II in the diatom Phaeodactylum tricornutum result in non-photochemical fluorescence quenching. Environ Microbiol 10:1997–2007
Erickson E, Wakao S, Niyogi KK (2015) Light stress and photoprotection in Chlamydomonas reinhardtii. Plant J 82:449–465. https://doi.org/10.1111/tpj.12825
Feilke K, Ajlani G, Krieger-Liszkay A (2017) Overexpression of plastid terminal oxidase in Synechocystis sp PCC 6803 alters cellular redox state. Philos Trans Royal Soc B-Biol Sci. https://doi.org/10.1098/rstb.2016.0379
Finazzi G, Johnson GN, Dall’Osto L, Zito F, Bonente G, Bassi R, Wollman FA (2006) Nonphotochemical quenching of chlorophyll fluorescence in Chlamydomonas reinhardtii. Biochemistry 45:1490–1498
Fischer BB, Hideg E, Krieger-Liszkay A (2013) Production, detection, and signaling of singlet oxygen in photosynthetic organisms. Antioxid Redox Signal 18:2145–2162
Foyer CH, Ruban AV, Nixon PJ (2017) Enhancing photosynthesis in crop plants: targets for improvement’. Philos Trans R Soc B. https://doi.org/10.1098/rstb.2016.0374
Fufezan C, Rutherford AW, Krieger-Liszkay A (2002) Singlet oxygen production in herbicide-treated photosystem II. FEBS Lett 532:407–410. https://doi.org/10.1016/s0014-5793(02)03724-9
Fukuzawa H, Ogawa T, Kaplan A (2012) The Uptake of CO2 by cyanobacteria and microalgae. In: Eaton Rye JJ, Tripathy BC, Sharkey TD (eds) Photosynthesis: plastid biology, energy conversion and carbon assimilation. Advances in photosynthesis and respirati, vol 34. Springer, Berlin, pp 625–650. https://doi.org/10.1007/978-94-007-1579-0_25
Giordano M, Beardall J, Raven JA (2005) CO2 concentrating mechanisms in algae: mechanisms, environmental modulation, and evolution. Ann Rev Plant Biol 56:99–131
Girolomoni L, Cazzaniga S, Pinnola A, Perozeni F, Ballottari M, Bassi R (2019) LHCSR3 is a nonphotochemical quencher of both photosystems in Chlamydomonas reinhardtii. Proc Natl Acad Sci USA 116:4212–4217. https://doi.org/10.1073/pnas.1809812116
Gollan PJ, Lima-Melo Y, Tiwari A, Tikkanen M, Aro E-M (2017) Interaction between photosynthetic electron transport and chloroplast sinks triggers protection and signalling important for plant productivity. Philos Trans R Soc B. https://doi.org/10.1098/rstb.2016.0390
Hagemann M, Kaplan A (2020) Is the structure of CO2-hydrating complex I compatible with the cyanobacterial CO2-concentrating mechanism? Plant Physiol. https://doi.org/10.1104/pp.20.00220
Havurinne V, Mattila H, Antinluoma M, Tyystjärvi E (2019) Unresolved quenching mechanisms of chlorophyll fluorescence may invalidate MT saturating pulse analyses of photosynthetic electron transfer in microalgae. Physiol Plant 166:365–379. https://doi.org/10.1111/ppl.12829
Helman Y, Barkan E, Eisenstadt D, Luz B, Kaplan A (2005) Fractionation of the three stable oxygen isotopes by oxygen-producing and oxygen-consuming reactions in photosynthetic organisms. Plant Physiol 138:2292–2298
Helman Y, Tchernov D, Reinhold L, Shibata M, Ogawa T, Schwarz R, Ohad I, Kaplan A (2003) Genes encoding A-type flavoproteins are essential for photoreduction of O2 in cyanobacteria. Curr Biol 13:230–235
Hossain MS, Dietz K-J (2016) Tuning of redox regulatory mechanisms, reactive oxygen species and redox homeostasis under salinity stress. Front Plant Sci. https://doi.org/10.3389/fpls.2016.00548
Ifuku K, Shikanai T (2016) Regulation of photosynthetic electron transport via supercomplex formation in the thylakoid membrane, vol https://doi.org/https://doi.org/10.1201/b19087. Redox proteins in supercomplexes and signalosomes. CRC Press, Boca Raton
Jahns P, Holzwarth AR (2012) The role of the xanthophyll cycle and of lutein in photoprotection of photosystem II. Biochim Biophys Acta-Bioenerg 1817:182–193. https://doi.org/10.1016/j.bbabio.2011.04.012
Josse EM, Alcaraz JP, Labouré AM, Kuntz M (2003) In vitro characterization of a plastid terminal oxidase (PTOX). Eur J Biochem 270:3787–3794. https://doi.org/10.1046/j.1432-1033.2003.03766.x
Kangasjarvi S, Tikkanen M, Durian G, Aro EM (2014) Photosynthetic light reactions–an adjustable hub in basic production and plant immunity signaling. Plant Physiol Biochem 81:128–134
Kaplan A, Reinhold L (1999) The CO2 concentrating mechanisms in photosynthetic microorganisms. Annu Rev Plant Physiol Plant Mol Biol 50:539–570
Kaye Y, Huang WC, Clowez S, Saroussi S, Idoine A, Sanz-Luque E, Grossman AR (2019) The mitochondrial alternative oxidase from Chlamydomonas reinhardtii enables survival in high light. J Biol Chem 294:1380–1395. https://doi.org/10.1074/jbc.RA118.004667
Keren N, Krieger-Liszkay A (2011) Photoinhibition: molecular mechanisms and physiological significance. Physiol Plant 142:1–5
Krupnik T, Kotabová E, van Bezouwen LS, Mazur R, Garstka M, Nixon PJ, Barber J, Kaňa R, Boekema EJ, Kargul J (2013) A reaction centre-dependent photoprotection mechanism in a highly robust photosystem II from an extremophilic red alga Cyanidioschyzon merolae. J Biol Chem 288:23529–23542
Li ZR, Wakao S, Fischer BB, Niyogi KK (2009) Sensing and responding to excess light. Annu Rev Plant Biol 60:239–260
Miyake C, Yonekura K, Kobayashi Y, Yokota A (2002) Cyclic electron flow within PSII functions in intact chloroplasts from spinach leaves. Plant Cell Physiol 43:951–957
Mukherjee A, Lau CS, Walker CE, Rai AK, Prejean CI, Yates G, Emrich-Mills T, Lemoine SG, Vinyard DJ, Mackinder LCM, Moroney JV (2019) Thylakoid localized bestrophin-like proteins are essential for the CO2 concentrating mechanism of Chlamydomonas reinhardtii. Proc Natl Acad Sci USA. https://doi.org/10.1073/pnas.1909706116
Murik O, Tirichine L, Prihoda J, Thomas Y, Araujo WL, Allen AE, Fernie AR, Bowler C (2019) Downregulation of mitochondrial alternative oxidase affects chloroplast function, redox status and stress response in a marine diatom. New Phytol 221:1303–1316. https://doi.org/10.1111/nph.15479
Ohad I, Berg A, Berkowicz SM, Kaplan A, Keren N (2011) Photoinactivation of photosystem II: is there more than one way to skin a cat? Physiol Plant 142:79–86
Ohad I, Raanan H, Keren N, Tchernov D, Kaplan A (2010) Light-Induced changes within photosystem II protects Microcoleus sp. in biological desert sand crusts against excess light. PLoS ONE 5(6):e11000
Peers G, Truong TB, Ostendorf E, Busch A, Elrad D, Grossman AR, Hippler M, Niyogi KK (2009) An ancient light-harvesting protein is critical for the regulation of algal photosynthesis. Nature 462:518-U215
Petroutsos D, Tokutsu R, Maruyama S, Flori S, Greiner A, Magneschi L, Cusant L, Kottke T, Mittag M, Hegemann P, Finazzi G, Minagawa J (2016) A blue-light photoreceptor mediates the feedback regulation of photosynthesis. Nature. https://doi.org/10.1038/nature19358
Price GD (2011) Inorganic carbon transporters of the cyanobacterial CO2 concentrating mechanism. Photosynth Res 109:47–57. https://doi.org/10.1007/s11120-010-9608-y
Raven JA, Beardall J, Giordano M (2014) Energy costs of carbon dioxide concentrating mechanisms in aquatic organisms. Photosynth Res 121:111–124. https://doi.org/10.1007/s11120-013-9962-7
Rehman AU, Cser K, Sass L, Vass I (2013) Characterization of singlet oxygen production and its involvement in photodamage of Photosystem II in the cyanobacterium Synechocystis PCC 6803 by histidine-mediated chemical trapping. Biochim Biophys Acta-Bioenerg 1827:689–698
Roberty S, Bailleul B, Berne N, Franck F, Cardol P (2014) PSI Mehler reaction is the main alternative photosynthetic electron pathway in Symbiodinium sp., symbiotic dinoflagellates of cnidarians. New Phytol 204:81–91. https://doi.org/10.1111/nph.12903
Ruban AV (2016) Nonphotochemical chlorophyll fluorescence quenching: Mechanism and fffectiveness in protecting plants from photodamage. Plant Physiol 170:1903–1916. https://doi.org/10.1104/pp.15.01935
Rumeau D, Peltier G, Cournac L (2007) Chlororespiration and cyclic electron flow around PSI during photosynthesis and plant stress response. Plant Cell and Environ 30(9):1041–1051
Rutherford AW, Paterson DR, Mullet JE (1981) A light-induced spin-polarized triplet detected by EPR in Photosystem II reaction centers. Biochim Biophys Acta 635:205–214
Sacksteder CA, Kramer DA (2000) Dark-interval relaxation kinetics (DIRK) of absorbance changes as a quantitative probe of steady-state electron transfer. Photosy Res 66:145–158
Saroussi S, Karns DAJ, Thomas DC, Bloszies C, Fiehn O, Posewitz MC, Grossman AR (2019) Alternative outlets for sustaining photosynthetic electron transport during dark-to-light transitions. Proc Natl Acad Sci USA. https://doi.org/10.1073/pnas.1903185116
Scholz M, Gäbelein P, Xue H, Mosebach L, Bergner SV, Hippler M (2019) Light-dependent N-terminal phosphorylation of LHCSR3 and LHCB4 are interlinked in Chlamydomonas reinhardtii. Plant J 99:877–894. https://doi.org/10.1111/tpj.14368
Shimakawa G, Ishizaki K, Tsukamoto S, Tanaka M, Sejima T, Miyake C (2017) The liverwort, Marchantia, drives alternative electron flow using a flavodiiron protein to protect PSI. Plant Physiol 173:1636–1647. https://doi.org/10.1104/pp.16.01038
Sonoike K (2011) Photoinhibition of photosystem I. Physiol Plant 142:56–64. https://doi.org/10.1111/j.1399-3054.2010.01437.x
Srivastava A, Strasser RJ, Govindjee (1999) Greening ***of peas: parallel measurements of 77 K emission spectra, OJIP chlorophyll a fluorescence transient, period four oscillation of the initial fluorescence level, delayed light emission, and P700. Photosynthetica 37:365–392. https://doi.org/10.1023/a:1007199408689
Stirbet A, Govindjee (2011) On the relation between the Kautsky effect (chlorophyll a fluorescence induction) and photosystem II: basics and applications of the OJIP fluorescence transient. J Photochem Photobiol B 104:236–257. https://doi.org/10.1016/j.jphotobiol.2010.12.010
Stirbet A, Govindjee (2012) Chlorophyll a fluorescence induction: a personal perspective of the thermal phase, the J—I—P rise. Photosynth Res 113:15–61. https://doi.org/10.1007/s11120-012-9754-5
Tchernov D, Hassidim M, Luz B, Sukenik A, Reinhold L, Kaplan A (1997) Sustained net CO2 evolution during photosynthesis by marine microorganisms. Curr Biol 7:723–728
Tchernov D, Silverman J, Luz B, Reinhold L, Kaplan A (2003) Massive light-dependent cycling of inorganic carbon between photosynthetic microorganisms and their surroundings. Photosynth Res 77:95–103
Treves H, Murik O, Kedem I, Eisenstadt D, Meir S, Rogachev I, Szymanski J, Keren N, Orf I, Tiburcio AF, Alcázar R, Aharoni A, Kopka J, Kaplan A (2017) Metabolic flexibility underpins growth capabilities of the fastest growing alga. Curr Biol 27:2559–2567. https://doi.org/10.1016/j.cub.2017.07.014
Treves H, Raanan H, Finkel MO, Berkowicz SM, Keren N, Kaplan A (2013) A newly isolated Chlorella sp. from desert sand crusts exhibits a unique resistance to excess light intensity. FEMS Microbiol Ecol 86:373–380. https://doi.org/10.1111/1574-6941.12162
Treves H, Raanan H, Kedem I, Murik O, Keren N, Zer H, Berkowicz SM, Giordano M, Norici A, Shotland Y, Ohad I, Kaplan A (2016) The mechanisms whereby the green alga Chlorella ohadii, isolated from desert soil crust, exhibits unparalleled photodamage resistance. New Phytol 210:1229–1243. https://doi.org/10.1111/nph.13870
Treves T, Siemiatkowska B, Luzarowska U, Murik O, Pozo NF, Moraes TA, Erban A, Armbruster U, Brotman Y, Kopka J, Rensing S, Szymanski J, Stitt M (2020) Multi-omics analysis reveals mechanisms of complete resistance to extreme illumination levels in a green alga isolated from desert soil crusts. Nat Plants. https://doi.org/10.1038/s41477-020-0729-9
Vanlerberghe GC, Martyn GD, Dahal K (2016) Alternative oxidase: a respiratory electron transport chain pathway essential for maintaining photosynthetic performance during drought stress. Physiol Plant 157:322–337. https://doi.org/10.1111/ppl.12451
Vass I (2012) Molecular mechanisms of photodamage in the photosystem II complex. Biochim Biophys Acta 1817:209–217
Vinyard DJ, Ananyev GM, Dismukes GC (2013) Photosystem II: the reaction center of oxygenic photosynthesis. Annu Rev Biochem 82:577–606
Wada S, Yamamoto H, Suzuki Y, Yamori W, Shikanai T, Makino A (2018) Flavodiiron protein substitutes for cyclic electron flow without competing CO2 assimilation in rice. Plant Physiol 176:1509–1518. https://doi.org/10.1104/pp.17.01335
Zito F, Kuras R, Choquet Y, Kossel H, Wollman FA (1997) Mutations of cytochrome b6 in Chlamydomonas reinhardtii disclose the functional significance for a proline to leucine conversion by petB editing in maize and tobacco. Plant Mol Biol 31:79–86
Acknowledgements
This research was supported by the Israel Science Foundation Grants to IY and AK and from the Israeli Ministry for Science and Technology to AK. We thank Dr. Haim Treves for many fruitful discussions and Dr. Judy Lieman-Hurwitz for technical assistance.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Figure S1
: QA reoxidation achieved at 4 minutes of dark acclimation. C. ohadii cultures grown in LC/LL were placed in measurement apparatus and kept in dark for 15 minutes. Then they were exposed to HL (2000 μmol photons m-2 s-1 red light) for 4 minutes, followed by an additional 15 minute of dark. PAM saturating pulses were obtained 4 and 15 minutes into each dark phase to test for differences in QA reoxidation between these times. In both F0 (dark bars) and Fm (light bars) parameters, no significant difference is found between 4 and 14 minutes of the first dark phase (LL Dark 4, LL Dark 14), or between 4 and 15 minute of the second dark phase (HL Dark 4, HL Dark 15) (lowest P value was 0.067). Error bars are ± standard error, n=3 (TIF 181 kb)
Figure S2: ECS at prolonged light-off
. Normally, ΔΨ/ΔpH ratio is derived from the ration between the minimum level and the steady-state dark level. However, in this organism these two value are very close, probably indicating that this analysis is not suitable here. A similar image is derived in LC cultures (TIF 315 kb)
Table S1: Parameters derived from MIMS measurements
. Average values of ≥4 measurements in each condition. Standard error of the mean given in parentheses. Please note that these are raw results, results shown in Fig. 2 are normalized to dark respiration (TIF 278 kb)
Table S2: Parameters derived from OJIP analysis
of fast fluorescence rise kinetics. a) Average values of 4 measurements in each condition. b) Results of students T-test between relevant pairs of treatments, values below 0.05 emphasized in bold. Please note that for both LL conditions, nOG treatment has hardly any effect. (TIF 688 kb)
(TIF 418 kb)
Rights and permissions
About this article
Cite this article
Kedem, I., Milrad, Y., Kaplan, A. et al. Juggling Lightning: How Chlorella ohadii handles extreme energy inputs without damage. Photosynth Res 147, 329–344 (2021). https://doi.org/10.1007/s11120-020-00809-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11120-020-00809-9