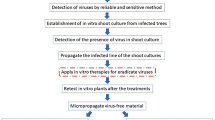
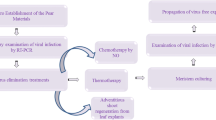
Abstract
Producing virus-free plants is required for disease management, the importation of innovative varieties from other nations, the transmission of breeding materials among regions, and the preservation of plant germplasm. Two commercial pear cultivars (Spadona and Mellina) as well as an apple genotype (Red flesh-Shahrood) were checked for six viral disease, employing thermotherapy (0, 7, 14, and 21 days at 38 °C), ribavirin (0, 20, 40, and 80 mg.L−1), and sodium nitroprusside (0, 10, 17, 25, 50, and 70 µM) followed by subculturing of different apical meristem culture sizes (less than 0.2 mm, 0.2 to 0.7 mm, and larger than 0.7 mm). Before meristem culture, seedlings were exposed to heat and chemicals. When compared to thermotherapy, ribavirin 40 mg.L−1 treatment was shown to be significantly more effective against mixed virus infections of the Red flesh genotype with 64.45%. SNP treatments were also more effective than thermotherapy and ribavirin-based chemotherapy; all grown apple and pear explants from SNP treatments were free of the ASPV virus. Additionally, the influence of dissected meristem size was notable; when therapies were combined, meristems smaller than 0.2 mm had a higher average rate of virus elimination than other samples. As soon as a year had passed after the trials concluded, the virus-free samples were reproduced, rooted and transplanted into pots for further propagation and establishment of the mother orchard.





Similar content being viewed by others
Data availability
Upon reasonable request, the corresponding author will provide the data that supports the conclusions of this study.
References
Aijaz, A., & Singh, G. (2007). Production of Indian citrus ringspot virus free plants of Kinnow employing chemotherapy coupled with shoot tip grafting. Journal of Central European Agriculture, 8(1), 1–8. https://doi.org/10.5513/jcea.v8i1.426
Barba, M., Ilardi, V., & Pasquini, G. (2015). Control of pome and stone fruit virus diseases. Advances in Virus Research, 91, 47–83. https://doi.org/10.1016/bs.aivir.2014.11.001
Basso, M. F., Fajardo, T. V. M., Eiras, M., Ayub, R. A., & Nickel, O. (2010). Molecular detection and identification of virus associated with symptomatic and asymptomatic grapevines. Rural Science, 40, 2249–2255. https://doi.org/10.1590/S0103-84782010001100001
Chiari, A., & Mark, P. B. (2002). Meristem culture and virus eradication in Alstroemeria. Plant Cell, Tissue and Organ Culture, 68, 49–55. https://doi.org/10.1023/A:1012947401640
Cieslinska, M. (2007). Application of thermotherapy and chemotherapy in vitro for elimination of some viruses infecting fruit trees and small fruits. Journal of Fruit and Ornamental Plant Research, 15, 117–124.
Cieslinska M. (2000). Elimination of Apple chlorotic leaf spot virus (ACLSV) from pear by in vitro thermotherapy and chemotherapy. VIII International Symposium on Pear, 596. https://doi.org/10.17660/ActaHortic.2002.596.80
Deng, X. Y., Hong, N., Hu, H. J., & Wang, G. P. (2004). Detection of latent viruses in Pyrus pyrifolia by IC-RT-PCR and TC-RT-PCR. Journal of Fruit Science, 21, 569–572.
Dessoky, E. S., Attia, O. A., Ismail, A. I., & El-Sharnouby, M. E. (2018). Production of Virus-Free Peach (Prunus persica L. Batsch) Plants cv. Balady Grown in Taif, by Meristem Culture and Thermotherapy. Bioscience Research, 15(1), 124–132.
Food and Agriculture Organization. (2020). FAOSTAT, from http://www.fao.org/statistics/en
Fu, L. J., Shi, K., Gu, M., Zhou, Y. H., Dong, D. K., Liang, W. S., Song, F. M., & Yu, J. Q. (2010). Systemic induction and role of mitochondrial alternative oxidase and nitric oxide in a compatible tomato–tobacco mosaic virus interaction. Molecular Plant-Microbe Interactions, 23, 39–48. https://doi.org/10.1094/MPMI-23-1-0039
Gambino, G., Bondaz, J., & Gribaudo, I. (2006). Detection and elimination of viruses in callus, somatic embryos and regenerated plantlets of grapevine. European Journal of Plant Pathology, 114, 397–404. https://doi.org/10.1007/s10658-006-0004-6
Grimova, L., Winkowska, L., Konrady, M., & Rysanek, P. (2016). Apple mosaic virus. Phytopathologia Mediterrane, 55(1), 1–19.
Gupta, K. J., Fernie, A. R., Kaiser, W. M., & van Dongen, J. T. (2011). On the origins of nitric oxide. Trends in Plant Science, 16, 160–168. https://doi.org/10.1016/j.tplants.2010.11.007
Hadidi, A., & Barba, M. (2011). Economic impact of pome and stone fruit viruses and viroids. Virus and Virus like Diseases of Pome and Stone Fruits, 1(8). https://doi.org/10.1094/9780890545010.001
Hassan, M., Myrta, A., & Polak, J. (2006). Simultaneous detection and identification of four pome fruit viruses by one-tube pentaplex RT-PCR. Journal of Virological Methods, 133(2), 124–129.
Hu, G., Hong, N., Wang, L., Hu, H., & Wang, G. (2012). Efficacy of virus elimination from in vitro-cultured sand pear (Pyrus pyrifolia) by chemotherapy combined with thermotherapy. Crop Protection, 37, 20–25. https://doi.org/10.1016/j.cropro.2012.02.017
Hu, G., Dong, Y., Zhang, Z., Fan, X., Ren, F., & Zhou, J. (2015a). Virus elimination from in vitro apple by thermotherapy combined with chemotherapy. Plant Cell, Tissue and Organ Culture (PCTOC), 121, 435–443. https://doi.org/10.1007/s11240-015-0714-6
Hu, G. J., Zhang, Z. P., Dong, Y. F., Fan, X. D., Ren, F., & Zhu, H. J. (2015b). Efficiency of virus elimination from potted apple plants by thermotherapy coupled with shoot-tip grafting. Australasian Plant Pathology, 44, 167–173.
James, D. (2000). Long term assessment of the effects of in vitro chemotherapy as a tool for Apple stem grooving virus elimination. XVIII International Symposium on Virus and Virus-like Diseases of Temperate Fruit Crops-Top Fruit Diseases, (2000) 550. https://doi.org/10.17660/ActaHortic.2001.550.69
Jossey, S., & Babadoost, M. (2008). Occurrence and distribution of pumpkin and squash viruses in Illinois. Plant Disease, 92(1), 61–68.
Karimpour, S., Davarynejad, G., Zakiaghl, M., & Safarnejad, M. R. (2020). In Vitro Thermotherapy and Thermo-Chemotherapy Approaches to Eliminate Some Viruses in Pyrus communis L. cv. ‘Natanz.’ Journal of Agricultural Science and Technology, 22(6), 1645–1653.
Kazemi, N., Kermani, M. J., & Habashi, A. A. (2019b). Sodium nitroprusside stimulates micropropagation and TDZ induces adventitious shoots regeneration in red flesh apple Malus niedzwetzkyana Koehne Dieck ex. Journal of Horticultural Research, 27(2), 23–30. https://doi.org/10.2478/johr-2019-0014
Kazemi, N., Zaree, N. F., Habashi, A. A., & Asadi, W. (2019a). Molecular Assessment of Chemotherapy and Meristem Culture Efficiency for Production of Seven Cultivars of Virus-Free Pear (Pyrus communis L.). Journal of Crops Improvement, 21(1), 107–118. (In Farsi). https://doi.org/10.22059/jci.2018.267050.2095
Kazemi, N., Habashi, A. A., & Asadi, W. (2019c). Evaluation of Combined Treatments of Thermotherapy and Apical Meristem Culture Efficiency on Virus Elimination from in vitro Shootlets of Red Flesh Apple (Malus pumila Mill.). Journal of horticulture science, 33(3), 499–509. (In Farsi). https://doi.org/10.22067/jhorts4.v33i3.77252
Kazemi, N., Nahandi, F. Z., Habashi, A. A., & Masoomi-Aladizgeh, F. (2020a). Comparing the efficiency of conventional and novel methods of virus elimination using molecular techniques. European Journal of Plant Pathology 1–11. https://doi.org/10.1007/s10658-020-02048-z.
Kazemi, N., Zaree, N. F., Habashi, A. A., & Dadpour, M. R. (2020b). Molecular assessment of thermotherapy and meristem culture efficiency on some viruses eradication from important commercial pear cultivars (Pyrus Communis L.). Iranian Journal of Horticultural Science, 50(4), 983–922. (In Farsi). https://doi.org/10.22059/ijhs.2019.267056.1516
Kers, J. A., Wach, M. J., Krasnoff, S. B., Widom, J., Cameron, K. D., Bukhalid, R. A., Gibson, D. M., Crane, B. R., & Loria, R. (2004). Nitration of a peptide phytotoxin by bacterial nitric oxide synthase. Nature, 429(6987), 79. https://doi.org/10.1038/nature02504
Komorowska, B., Malinowski, T., & Michalczuk, L. (2010). Evaluation of several RT-PCR primer pairs for the detection of Apple stem pitting virus. Journal of Virological Methods, 168, 242–247. https://doi.org/10.1016/j.jviromet.2010.04.024
Manganaris, G. A., Economou, A. S., Boubourakas, I. N., & Katis, N. I. (2003). Elimination of PPV and PNRSV through thermotherapy and meristem-tip culture in nectarine. Plant Cell Reports, 22, 195–200.
Masoomi-Aladizgeh, F., Jabbari, L., Khayam Nekouei, R., & Aalami, A. (2016). A simple and rapid system for DNA and RNA isolation from diverse plants using handmade kit. Protocol Exchange. https://doi.org/10.21203/rs.2.1347/v2
Moreau, M., Lindermayr, C., Durner, J., & Klessig, D. F. (2010). NO synthesis and signaling in plants–where do we stand? Physiologia Plantarum, 138, 372–383. https://doi.org/10.1111/j.1399-3054.2009.01308.x
Murashige, T., & Skoog, F. (1962). A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiologia Plantarum, 15, 473–497. https://doi.org/10.1111/j.1399-3054.1962.tb08052.x
Palukaitis, P., Groen, S. C., & Carr, J. P. (2013). The Rumsfeld paradox: Some of the things we know that we don’t know about plant virus infection. Current Opinion in Plant Biology, 16, 513–519. https://doi.org/10.1016/j.pbi.2013.06.004
Panattoni, A., Luvisi, A., & Triolo, E. (2013). Elimination of viruses in plants: twenty years of progress. Spanish Journal of Agricultural Research, 1, 173–188. https://doi.org/10.5424/sjar/2013111-3201
Paprstein, F., Sedlak, J., Svobodova, L., Polak, J., & Gadiou, S. (2013). Results of in vitro chemotherapy of apple cv. Fragrance-Short communication. Horticultural Science, 40, 186–190.
Paunovic, S., Ruzic, D., Vujovic, T., Milenkovic, S., & Jevremovic, D. (2007). In vitro production of Plum pox virus-free plums by chemotherapy with ribavirin. Biotechnology & Biotechnological Equipment, 21, 417–421. https://doi.org/10.1080/13102818.2007.10817486
Peck, T. C., Fuchs, H., & Whitton, M. C. (2010, March). Improved redirection with distractors: A large-scale-realwalking locomotion interface and its effect on navigation in virtual environments. In 2010 IEEE Virtual Reality Conference (VR) (pp. 35–38). IEEE.
Prokhnevsky, A. I., Peremyslov, V. V., Napuli, A. J., & Dolja, V. V. (2002). Interaction between long-distance transport factor and Hsp70-related movement protein of beet yellows virus. Journal of Virology, 76, 11003–11011. https://doi.org/10.1128/JVI.76.21.11003-11011.2002
Quecini, V., Lopes, M. L., Pacheco, F. T. H., & Ongarelli, M. D. G. (2008). Ribavirin, a guanosine analogue mammalian antiviral agent, impairs tomato spotted wilt virus multiplicationin tobacco cell cultures. Archives of Phytopathology and Plant Protection, 41, 1–13. https://doi.org/10.1080/03235400600628047
Quoirin, M., & Lepoivre, P. (1977). Improved media for in vitro culture of Prunus sp. Symposium on Tissue Culture for Horticultural Purposes, 78. https://doi.org/10.17660/ActaHortic.1977.78.54
Ramgareeb, S., Snyman, S. J., Antwerpen, T. V., & Rutherford, R. S. (2010). Elimination of virus and rapid propagation of disease-free sugarcane (Saccharum spp. cultivar NCo376) using apical meristem culture. Plant Cell, Tissue and Organ Culture, 100, 175–181. https://doi.org/10.1007/s11240-009-9634-7
Retheesh, S. T., & Bhat, A. I. (2010). Simultaneous elimination of Cucumber mosaic virus and Cymbidium mosaic virus infecting Vanilla planifolia through meristem culture. Crop Protection, 29, 1214–1217. https://doi.org/10.1016/j.cropro.2010.05.017
Sareila, O., Hohkuri, M., Wahlroos, T., & Susi, P. (2004). Role of Viral Movement and Coat Proteins and RNA in Phloem-dependent Movement and Phloem Unloading of Tobamoviruses. Journal of Phytopathology, 152, 622–629. https://doi.org/10.1111/j.1439-0434.2004.00905.x
Scheler, C., Durner, J., & Astier, J. (2013). Nitric oxide and reactive oxygen species in plant biotic interactions. Current opinion in Plant Biology, 16, 534–539. https://doi.org/10.1016/j.pbi.2013.06.020
Schlicht, M., & Kombrink, E. (2013). The role of nitric oxide in the interaction of Arabidopsis thaliana with the biotrophic fungi, Golovinomyces orontii and Erysiphe pisi. Frontiers in Plant Science, 4, 351. https://doi.org/10.3389/fpls.2013.00351
Simpkins, I., Walkey, D. G. A., & Neely, H. A. (1981). Chemical suppression of virus in cultured plant tissues. Annals of Applied Biology, 99, 161–169. https://doi.org/10.1111/j.1744-7348.1981.Tb05143.x
Song, F. M., & Goodman, R. M. (2001). Activity of nitric oxide is dependent on, but is partially required for function of salicylic acid in the signaling pathway in tobacco systemic acquired resistance. Molecular Plant-Microbe Interactions, 14, 1458–1462. https://doi.org/10.1094/MPMI.2001.14.12.1458
Sun, Q., Sun, H., & Bell, R. L. (2009). Effect of polyvinyl alcohol on in vitro rooting capacity of shoots in pear clones (Pyrus communis L.) of different ploidy. Plant Cell, Tissue and Organ Culture, 99, 299–304. https://doi.org/10.1007/s11240-009-9604-0
Tan, R. R., Wang, L. P., Hong, N., & Wang, G. P. (2010). Enhanced efficiency of virus eradication following thermotherapy of shoot-tip cultures of pear. Plant Cell, Tissue and Organ Culture, 101, 229–235. https://doi.org/10.1007/s11240-010-9681-0
Tiziano, C., Folwell, R. J., Wandschneider, P., Eastwell, K. C., & Howell, W. E. (2003). Economic implications of a virus prevention program in deciduous tree fruits in the US. Crop Protection, 22, 1149–1156. https://doi.org/10.1016/S0261-2194(03)00156-X
Volk, G. M., Thomas, C. C., Norelli, J., Brown, S. K., Fazio, G., Peace, C., McFerson, J., Zhong, G. Y., & Bretting, P. (2015). The vulnerability of US apple (Malus) genetic resources. Genetic Resources and Crop Evolution, 62, 765–794. https://doi.org/10.1007/s10722-014-0194-2
Wach, M. J., Kers, J. A., Krasnoff, S. B., Loria, R., & Gibson, D. M. (2005). Nitric oxide synthase inhibitors and nitric oxide donors modulate the biosynthesis of thaxtomin A, a nitrated phytotoxin produced by Streptomyces spp. Nitric Oxide, 12, 46–53. https://doi.org/10.1016/j.niox.2004.11.004
Wang, L. P., Wang, G. P., Hong, N., Tang, R. R., & Deng, X. Y. (2006). Effect of thermotherapy on elimination of Apple stem grooving virus and Apple chlorotic leaf spot virus for in vitro-cultured pear shoot tips. HortScience, 41, 729–732. https://doi.org/10.1016/j.cropro.2010.08.003
Wang, Q. C., Cuellar, W. J., Rajamäki, M., Hirata, Y., & Valkonen, J. P. T. (2008). Combined thermotherapy and cryotherapy for efficient virus eradication: Relation of virus distribution, subcellular changes, cell survival and viral RNA degradation in shoot tips. Molecular Plant Pathology, 9, 237–250. https://doi.org/10.1111/j.1364-3703.2007.00456.X
Acknowledgements
It is with gratitude that we acknowledge and appreciate the staff of the Agricultural Biotechnology Research Institute of Iran (ABRII) Department of Tissue and Cell Culture for their expertise and assistance.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
There is no conflict of interest to declare.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kazemi, N., Abdollahi, H., Habashi, A.A. et al. Pome fruit-virus interactions using combined therapies and meristem culture. Eur J Plant Pathol 168, 279–296 (2024). https://doi.org/10.1007/s10658-023-02754-4
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10658-023-02754-4