Abstract
Liquid biofuels like biodiesel and bioethanol are crucial in the transition to low-carbon and high-energy alternatives to fossil fuels. One significant by-product of biodiesel production is glycerol, which accounts for about 10% of the total conversion output. While waste glycerol poses challenges due to its impurities and contaminants, it also holds potential as a metabolic resource for essential cellular components in microorganisms. Crude glycerol production is reviewed, highlighting relevance in current biodiesel technologies and its biochemical composition. To efficiently utilize waste glycerol, co-valorization with low-cost substrates through biocircular platforms using various microorganisms or insects for second and third-generation oxy-biofuels has been explored. Among these, the black soldier fly larvae have demonstrated higher competitiveness for lipid contents (35–43%), making them a promising organism for recycling waste glycerol into biodiesel production, alongside microalgae and oleaginous yeast. The microbial biodiesel productivity from oleaginous yeast is notably higher (3546 kg ha−1 y−1) than soybean biodiesel (562 kg ha−1 y−1), while microalgal biodiesel productivity surpasses palm biodiesel by more than 25 times. Remarkably, black soldier fly larvae biodiesel productivity was reported to be ~ 1.7 times higher than microalgae and an impressive ~ 43 times higher than palm biodiesel. Despite their potential for biodiesel production, waste glycerol from biodiesel industry still represents a challenge because of high impurities, high viscosity, and limited direct applications in existing processes. To further enhance energy sustainability and address the challenge of waste glycerol, biocircular platforms are discussed for waste glycerol utilization with domestic wastewater sludge, lignocellulosic biomass, and protein-rich wastes. These platforms offer opportunities to create other sustainable agricultural products while minimizing their environmental footprint.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The necessity to explore sustainable feedstocks for biofuel production has been recognized by the depletion of natural resources brought on by excessive consumption of non-renewable fossil fuels and the rising global energy demand (Farghali et al. 2023a; Osman et al. 2021a, b Elsayed et al. 2018). The primary focus of energy strategies is maximizing the efficient utilization of non-edible and affordable renewable resources to produce various biofuels. These include gaseous fuels like biomethane and hydrogen and liquid fuels like bioethanol, biodiesel, and crude bio-oil. The aim is to mitigate adverse environmental effects while simultaneously bolstering sustainable energy security at a reasonable cost (Abomohra et al. 2020; Osman et al. 2023a, b). Biodiesel and bioethanol have emerged as highly viable alternatives for diesel and gasoline engines among the new biofuel options. Their carbon–neutral nature, high energy density, biodegradability, and safe handling characteristics closely resemble the performance of petroleum-derived fuels, indicating their potential to replace fossil fuels (Xu et al. 2019; Osman et al. 2023b). However, during biodiesel synthesis through transesterification reaction, waste (crude) glycerol accumulates as a by-product, containing impurities and contaminants that impede downstream processing and raise pollution concerns (Wang et al. 2019; Ge et al. 2022; Li et al. 2022; Farghali et al. 2023b). As a by-product of this process, waste glycerol is generated at approximately 1–1.4 tons per 10 tons of fatty acid methyl esters (biodiesel) as the main product (Zhang et al. 2016). Furthermore, crude glycerol is generated as a by-product from oleo-chemical processes like saponification and hydrolysis. Therefore, the biodiesel sector is experiencing rapid growth, leading to a significant increase in waste glycerol exports in the market. Since 1995, these exports have risen by 10% annually and are expected to reach a substantial volume of 7.66 million tons by 2022 (Zhao et al. 2023).
The oversupply of waste glycerol has led to an unusual drop in its price, reaching less than the US dollar (USD) 100 per ton (Sun et al. 2022). This decline is linked to the negative correlation between glycerol prices and the growth in biodiesel production. For small- and medium-sized companies in the oleo-chemicals or biodiesel industries, dealing with this oversupply has become a pressing concern. There are two options to address this issue. The first is purification through chemical treatment routes involving the addition of acids as catalysts at high temperatures, which comes with a cost of up to USD 50.85 per kg (Zhao et al. 2023). This process generates environmentally toxic by-products, making it economically unviable on an industrial scale. The second option is the biological conversion route by selected microorganisms, which is more reliable, eco-friendlier, utilizes less energy, and produces more bioproducts (Elahinik et al. 2022). The microbial heterofermentative process of waste glycerol produces ethanol and hydrogen, making it a promising renewable energy source. Ethanol derived from this process can be utilized as a base substance, an additive to gasoline, or a precursor for biodiesel synthesis, reducing the dependence on methanol as a petroleum-based source (Sunarno et al. 2020). Considering the low price of raw glycerol, the selling price of ethanol made from glycerol is approximate to be USD 0.27 per liter, making it a highly economical and profitable option compared to sugarcane-based ethanol production (0.55 USD per liter) (Posada and Cardona 2010). Additionally, glycerol is effectively utilized by various organisms through metabolic processes, transforming glycerol into intermediate metabolites used to synthesize essential components, including fatty acids and lipids (Xue et al. 2017; Abomohra et al. 2018).
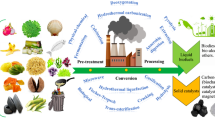
Glycerol also has the feasibility to be explored as a solitary carbon substrate (precursor) for lipids biosynthesis and biodiesel production by oleaginous yeast (Kumar et al. 2021), microalgae (Abomohra et al. 2018), and black soldier fly larvae (Almutairi et al. 2023). In this context, second- and third-generation oxy-biofuels (bioethanol and biodiesel) production from incorporating waste glycerol with various biowastes, including domestic wastewater sludge, lignocellulosic biomass, and protein-rich wastes using an array of microorganisms and insects, has not yet been comprehensively reviewed. Addressing this gap, this review aims to present the existing production processes of crude glycerol and its advanced valorization routes, as shown in Fig. 1. The review thoroughly provides the appropriate co-valorization technologies of waste glycerol with inexpensive substrates by different microorganisms/insects to maximize its benefits. The critical points between the biochemical composition of waste glycerol and current biodiesel technologies are briefly described. The potential interactions linking glycerol metabolic processes and lipid biosynthesis in oleaginous yeast, microalgae, and black soldier fly larvae are presented. In addition, the innovations in co-valorized waste glycerol with low-cost substrates for second- and third-generation oxy-biofuels are comprehensively discussed. Lastly, the review illustrates the integration strategies for waste glycerol utilization in conjunction with other feedstocks, including domestic wastewater sludge, lignocellulosic biomass, and protein-rich wastes. These strategies would offer potential solutions to the energy crisis and the creation of sustainable agricultural products, all while mitigating their environmental impact.
Crude glycerol production processes, composition, and valorized products. Crude glycerol is a by-product of three primary processes: transesterification, saponification, and hydrolysis of lipids, to produce biodiesel, soap, and fatty acids, respectively. Its composition typically includes glycerol, water, and non-glycerol organic substances like methanol and fatty acids, along with ash. The valorization of crude glycerol presents opportunities to produce energy and value-added agri-products
Current technologies of waste glycerol production
Glycerol is a versatile compound derived from vegetable and animal oils/fats, as well as it can be obtained through various processes. As depicted in Fig. 2, the generation of crude glycerol as a by-product from triacylglycerols is commonly associated with three main processes, namely soap production involving the addition of alkali (sodium hydroxide), fatty acid production through hydrolysis with steam water, and fatty methyl ester production via transesterification reaction with methanol and catalyst, as discussed in the following sections. These processes result in crude waste glycerol with a glycerol content ranging from 65 to 80% (Chilakamarry et al. 2021b).
The production of crude glycerol by-product from triacylglycerols by saponification, hydrolysis, and transesterification showing and estimation for the main components. Saponification involves the addition of alkali, such as sodium (NaOH) or potassium hydroxide (KOH), to produce soap, while transesterification uses methanol and a catalyst to synthesize fatty methyl esters. Hydrolysis, on the other hand, employs steam water to break down triacylglycerol into glycerol and fatty acids. These processes yield crude glycerol, which finds applications in various industries. Average concentrations percentage of crude glycerol produced from different processes (Valerio et al. 2015). The glycerol content in the crude glycerol increases significantly as the production process progresses from transesterification (60–75%) to saponification (83–84%) and further to hydrolysis (88–90%). On the other hand, the ash content decreases notably as the process advances from transesterification (10.0–19.0%) to saponification (8.5–9.5%), reaching its lowest level in hydrolysis (0.7–1.0%). The water content shows a gradual increase from transesterification (< 10%) to saponification (6.0–7.0%) and then to hydrolysis (8.0–9.0%). Non-glycerol organic matter (NG-OM) exhibits a decreasing trend from transesterification (< 40%) to saponification (3.0–4.0%) and finally to hydrolysis (0.7–1.0%).
Saponification
Soap production from vegetable or animal lipids involves the hydrolysis of their triglyceride contents. Free hydroxide breaks the ester bonds, generating fatty acids and glycerol (Wan Isahak et al. 2015), this chemical transformation is referred to as saponification. During this process, alkaline hydrolysis of triacylglycerols produces fatty acid salts (soap) and yields glycerol as a high-purity by-product. Saponification effectively removes lipid deposits, including microbial cell lipids, and acts as an antimicrobial agent (Chilakamarry et al. 2021a).
Hydrolysis
The primary method for producing glycerol from animal fats and plant oils involves the hydrolysis of triacylglycerols, resulting in both glycerol and fatty acids. According to stoichiometry, this hydrolysis process uses three moles of water to break down the tri-ester linkages in a single mole of triacylglycerol, leading to the formation of three moles of fatty acids and a single mole of glycerol. Several common hydrolysis reactions can achieve this conversion, including the Twitchell, Eisenlohr, and Colgate-Emery processes (Casali et al. 2021). Among these, the Colgate-Emery process stands out for commercial applications due to its simplicity. In this process, the hydrolysis of triglycerides takes place at 250 °C and 50 bar for 2–3 h (Nitbani et al. 2020).
Transesterification
The surging energy demand has triggered a revolution in biodiesel production, with various sources being utilized, ranging from oleaginous microorganisms and black soldier fly larvae to higher plants such as rapeseed, palm, and sunflowers (Asopa et al. 2022; Eraky et al. 2022). Chemically, biodiesel is the alkyl ester form of fatty acids. It is generated through transesterifying triacylglycerols with alcohol in the existence of either an acid or alkali catalyst. According to Peters et al. (2022), the transesterification reaction yields 10% by weight of glycerol and 90% by weight of methyl ester (biodiesel). Alkali catalysts have been proven to increase biodiesel yield, and the choice of catalyst depends on the composition of fatty acids in the oil. In comparison with the utilization of acid catalysts, alkaline-catalyzed transesterification has several advantages, such as a quick reaction rate, shorter reaction time, and easier setup (Chilakamarry et al. 2021b; Pan et al. 2022). The transesterification process involving triacylglycerols and three moles of methanol occurs in three steps facilitated by a catalyst. In the first step, methanol reacts with triacylglycerols, forming diglycerides, and methyl esters. Subsequently, methanol combines with diglycerides to produce monoglycerides and methyl esters. In the last reaction, monoglycerides react with methanol, producing glycerol and methyl esters (Bagnato et al. 2017). This technique is frequently used to create methyl esters and glycerol from triacylglycerols.
In summary, the biodiesel production industry is experiencing a revolution in response to increasing energy demands. Various sources, including oleaginous microorganisms and higher plants, are utilized for this purpose. The production process involves transesterification, where triglycerides react with alcohol in the presence of acid or alkali catalysts, forming methyl ester (biodiesel) and glycerol. Overall, alkali catalysts have proven advantageous, offering relatively faster reaction rates and simpler setups than acid catalysts.
Physicochemical properties of waste glycerol
Common impurities found in crude glycerol include water, ashes, soaps (formed from saponified fatty acids or fatty acid salts), and other organic substances like fatty acids, alcohols, and aldehydes. The main components of crude glycerol originate from the three main aforementioned processes: saponification, hydrolysis, and transesterification (Fig. 2). The composition of waste glycerol from transesterification and the principal reaction in biodiesel synthesis depend on the catalyst and the specific oil used, where it produces the lowest amount of impurities. However, methanol in the reaction mixture may elevate the non-glycerol organic compounds. This phenomenon is attributed to the high methanol-to-oil ratio (3:1) used in the reaction. Even after the crude glycerol separation process, residual methanol can still be detected (Kumar et al. 2019b). Recently, biodiesel synthesis has become a significant source of crude glycerol, accounting for 10% of the biodiesel yield. Overall, the chemical constitution of the resulting crude glycerol varies according to transesterification conditions and the catalysts used. Hu et al. (2012) investigated the composition of crude glycerol derived from five biodiesel samples produced from soybean oil. Crude glycerol was fractionated into glycerol, non-glycerol compounds, water, and ash. The glycerol content in the samples ranged from 22.9 to 63%, while water and ash content varied from 1.0 to 28.7% and 2.7 to 5.7%, respectively. The non-glycerol components present in the crude glycerol included methanol, fatty acid methyl esters, free fatty acids, and soap. Among these, soap and methanol were the most prevalent, accounting for 20.5–31.4% and 6.2–12.6%, respectively. Fatty acid methyl esters were recorded in a wide range, from 0.5 to 28.8%, while glycerides and free fatty acids represented 0.4–7.0% and 1.0–3.0%, respectively.
Hansen et al. (2009) examined the composition of several crude glycerol samples obtained from different biodiesel sources. The glycerol content exhibited a wide range, with values varying from 38 to 96%, highlighting its significance as the primary constituent. Moreover, non-glycerol components were detected, with methanol accounting for 14% and ash present in more than 29% of the crude glycerol samples. Similarly, de Carvalho et al. (2012) studied the chemical composition of waste glycerol derived from two distinct sources during biodiesel synthesis: soybean oil and a blend of animal fat with soybean oil (at a ratio of 4:1). These samples were obtained from biopar diagnosis, a biofuel company in Brazil, and exhibited an ash content of 4% and glycerol content of 55%. Comparing the crude glycerol obtained from the mixed substrate to that derived from soybean oil showed slightly higher levels of organic non-glycerol substances, methanol, and total fatty acids in the latter. Johnson and Rehmann (2016) studied the constitution of waste glycerol generated through the transesterification reaction using sunflower oil as a feedstock. The waste glycerol consisted of 30% glycerol, 50% methanol, 13% soap, 2% moisture, 2–3% salts, and 2–3% miscellaneous contaminants. Another study focused on synthesizing biodiesel from soybean oil and found that the resulting crude glycerol contained 70–85% glycerol (Mu et al. 2006). It was confirmed that there was a minimal variation in glycerol content across various feedstock (Thompson and He 2006). However, it is essential to mention that these experiments were conducted on a laboratory scale rather than in an industrial environment.
The composition of waste glycerol is closely related to its physicochemical properties. The presence of moisture and non-glycerol components, such as methanol and fatty acids, contributes to waste glycerol’s reduced density and viscosity compared to pure glycerol. Additionally, these components can cause the crude glycerol to be dark brownish (Valerio et al. 2015; Saidi et al. 2020). Hu et al. (2012) found that the density of crude glycerol originating from soybean biodiesel was 1.01–1.3 g.cm–3. Similarly, Guerfali et al. (2020) reported the density to be 1.07 ± 0.1 g.cm–3, while pure glycerol has a density of 1.26–1.31 g.cm–3. The dynamic viscosity of the crude glycerol was approximately 95 ± 5.3 centipoise, lower than that of pure glycerol (628 ± 12.2 centipoise). This is caused by some lighter impurities, such as methanol, fatty acids, and water, that are present in crude glycerol (Guerfali et al. 2020). The transesterification reaction in an alkaline medium contributes to the recorded alkaline pH of crude glycerol, which typically ranges from 9.4 to 10.0. In contrast, pure glycerol is slightly acidic, with a pH of around 6.5 (Bharti et al. 2021). The presence of alkaline residuals in crude glycerol can be removed to recover a pH level similar to that of pure glycerol (Hu et al. 2012).
According to Guerfali et al. (2020), the elemental analysis of crude glycerol sample revealed a high carbon content of 65.2 wt%, surpassing the range of 24 wt% to 37.7 wt% for different types of crude glycerol produced from various esterified oils as reported by Thompson and He (2006). Moreover, the carbon content in crude glycerol samples from other industries varied significantly, ranging from 24.3 to 54.2%. This variation is attributed to the downstream processing methods used, which can influence the availability of carbon molecules, such as glycerol, methanol, free fatty acids, and residues of triacylglycerols (Hu et al. 2012). The presence of inorganic carbon in the waste glycerol samples is not taken into account since it is below 1%. Despite the significant variability in the carbon content of crude glycerol, the nitrogen content remains consistently low in various types of crude glycerol samples, ranging from 0.03 to 1.2% by weight (Uprety et al. 2017).
As shown in Table 1, the conversion of soybean oil results in a wide range of glycerol content (22.9–63%) and methanol content (6.2–12.6%), with low levels of free fatty acids (< 3%) and varying amounts of water, ash, soap, and other impurities. Sunflower oil-derived biodiesel production showed a glycerol content of 30%, higher methanol content of 50%, and minimal free fatty acids, along with small quantities of water, ash, soap, and impurities. Commercial biodiesel samples have varying glycerol content of 15.4–30.2% and relatively consistent methanol content of 14.4–15.5%, with low levels of free fatty acids, water, and ash and varying soap content. Biodiesel derived from an unspecified vegetable oil source exhibits a high glycerol content of 64.5% and a lower methanol content of 5.9%. The biodiesel samples also contain substantial levels of free fatty acids, water, ash, soap, non-glycerol organic compounds, and trace amounts of suspended matter and other impurities. This data offers valuable insights into the composition and impurity levels of the biodiesel samples, facilitating the evaluation and comparison of their overall quality.
In summary, the physicochemical properties of waste glycerol originating from biodiesel production vary based on the specific processes involved. The glycerol content notably increases from transesterification to saponification and hydrolysis. Moreover, biodiesel derived from different sources displays different levels of glycerol, methanol, free fatty acids, water, ash, soap, and other impurities, offering valuable insights to evaluate biodiesel quality.
Bioethanol production from waste glycerol by microbial fermentation
Process development for converting crude glycerol into bioethanol provides an eco-friendly approach to handle this by-product effectively. By utilizing crude glycerol as a feedstock instead of conventional sources like sugar, molasses, or lignocellulosic biomass, the production cost of ethanol can be reduced by approximately 40% (Yazdani and Gonzalez 2007). Moreover, employing mixed cultures with higher microbial diversity proved beneficial over pure cultures, especially when dealing with crude glycerol that contains variable impurities (Vikromvarasiri et al. 2016). This approach offers a sustainable and efficient means of harnessing bioethanol from a waste material while optimizing the economic viability of the process.
Several microbes, including Enterobacter aerogenes, Escherichia coli, Anaerobium acetethylicum, and Klebsiella pneumoniae, have been widely used in bioethanol production processes due to their capability to utilize glycerol as a carbon source under aerobic or anaerobic fermentation. Hydrogen generation, accompanied by ethanol production, provides another high-value-added biofuel. Vikromvarasiri et al. (2016) explored those mixed cultures of anaerobic sludge originating from wastewater treatment systems that were highly efficient in digesting waste glycerol to produce ethanol in batch fermenters. Microbial community analysis of the sludge showed the presence of Proteobacteria, Firmicutes, and Bacteroidetes. Interestingly, only a small portion of glycerol was transformed into organic acids, such as acetate and propionate, through the oxidative pathway. Additionally, it was suggested that the reduction pathway could convert some glycerol into 1,3-propanediol and 3-hydroxypropionic acid (Agrawal et al. 2023).
Figure 3 illustrates the glycerol fermentation pathway into value-added products, which include ethanol, butanol, succinate, and volatile fatty acids, in addition to hydrogen and carbon dioxide. Glycerol is transformed into phosphoenol pyruvate, followed by pyruvate, creating a loop route that can synthesize succinic acid. These metabolic pathways are essential for controlling fluxes during fermentation of glucose or glycerol. Unlike glycolysis, glycerol fermentation does not produce a net increase in adenosine triphosphate. These variations have a variety of bioenergetic implications, such as maintaining a decreased pH and creating a lower proton-motive force than glucose fermentation (Trchounian et al. 2013). Pyruvate dehydrogenase complex and pyruvate formatelyase synthesize acetyl-co-enzyme A from pyruvate. However, it is anticipated that formatelyase plays a substantial role, given the high hydrogenated nicotinamide adenine dinucleotide/nicotinamide adenine dinucleotide (non-hydrogenated) ratios seen during glycerol fermentation and the inhibitory effect of hydrogenated nicotinamide adenine on pyruvate dehydrogenase complex. Acetyl-co-enzyme A is transformed into ethanol in the last stages of the biosynthetic pathway for bioethanol in Escherichia coli by a two-step process involving acetaldehyde or alcohol dehydrogenase (Sawers and Clark 2004).
The glycerol fermentation pathway into value-added products. Glycerol can be converted into valuable by-products, including ethanol, butanol, succinate, and volatile fatty acids, i.e., formate, acetate, lactate, and butyrate. Hydrogen and carbon dioxide are co-generated from the process. Adenosine triphosphate = ATP, adenosine di-phosphate = ADP, nicotinamide adenine dinucleotide = NAD+, hydrogenated nicotinamide adenine dinucleotide = NADH, hydrogen = H, carbon dioxide = CO2, acetyl CoA = acetyl-co-enzyme A
Recently, Anaerobium acetethylicum GluBS11 has been used as a potentially beneficial bacterium for remediating glycerol-rich wastewaters and producing ethanol (Patil et al. 2017). It was found to naturally have a high tolerance toward increased glycerol levels. In addition, Escherichia coli has been employed to produce ethanol because it is easily manipulated genetically and recognized as adaptable to industrial applications. Compared to aerobic fermentation, anaerobic or microaerobic fermentation produces an equivalent amount of ethanol while using less energy (without aeration) (Zhang et al. 2016). The hypothetical yield of ethanol and hydrogen (or formate) from glycerol fermentation is one mol each of ethanol and hydrogen (or formic acid) per mol of glycerol utilized according to Lee et al. (2020) using the equations of the reactions 1 and 2.
This implies that 50% of the glycerol is hypothetically transformed to ethanol, 2.2% to hydrogen, and 47.8% to carbon dioxide, or 50% of the glycerol is theoretically transformed to ethanol and 50% to formic acid. The gaseous effluent, like hydrogen and carbon dioxide, is collected and pumped via a washing tank filled with sodium hydroxide to remove the carbon dioxide. After fermentation, the broth is distilled to improve the ethanol, and the biomass is condensed and used as animal feed.
Table 2 describes the various microbes that produce bioethanol from crude glycerol. The studies have demonstrated that the ethanol produced from crude glycerol is an excellent carbon source for microbial utilization under aerobic and anaerobic fermentation processes. For instance, Enterobacter aerogenes TISTR 1468 generated ethanol at an average concentration of 12.33 g/L, yielding 0.62 g/g glycerol (Sunarno et al. 2019). Similarly, Klebsiella aerogenes caused an ethanol titer of 15.89 g/L, with an ethanol productivity of 0.487 g/L/h (Lee et al. 2020). With a noteworthy productivity rate as high as 0.61 g/L/h, Kluyvera cryocrescens S26 produced an excellent ethanol production of 27 g/L (Choi et al. 2011). These results highlight the potential of utilizing crude glycerol as a feedstock for efficient ethanol production by these microorganisms, making it a valuable resource for sustainable biotechnological applications.
Cofré et al. (2016) examined the digestion of waste glycerol as the sole carbon source by Escherichia coli MG1655. Fermentation at 34 °C and pH 7.6 was the optimum condition for bioethanol generation; the primary glycerol concentration of 10 g/L was suggested for optimal bioethanol yield (0.40 g/g glycerol). However, optimal hydrogen productivity requires higher glycerol concentration (20–25 g/L) (Chaudhary et al. 2012). Among different heterofermentative bacteria, Enterobacter aerogenes TISTR 1468 demonstrated the highest bioethanol production of 0.94 g/g glycerol under microaerobic conditions (Sunarno et al. 2020). Nevertheless, it was observed that Enterobacter aerogenes experienced glycerol inhibition at concentrations exceeding 20 g/L, which elucidates the advantage of using a fed-batch culture over a batch process (Sunarno et al. 2020).
In summary, previous studies showed a marked improvement in ethanol production by anaerobiosis in batch fermentation through limited oxygen (microaerobic), which was better than anaerobic fermentation conditions. In addition, utilizing waste glycerol as a feedstock for bioethanol production can significantly reduce the production cost, making it a promising resource for sustainable biotechnological applications. However, impurities, microbial strains, and process optimization challenges need to be addressed for further process improvement.
Approaches for biosynthesis of bio-lipids from glycerol by oleaginous organisms
Glycerol has proven to be a versatile and cost-effective building block, widely utilized as a substrate for the biosynthesis of lipids in various oleaginous species, including multicellular insects and single-celled microorganisms such as microalgae, yeast, and bacteria. These organisms have demonstrated a relatively high yield of exploratory lipids when utilizing glycerol as a substrate. Recent literature highlights the significance of different factors like media composition, environmental conditions, and biosystem design in influencing the scale-up and optimization of lipid production using waste glycerol as a substrate (Lazar et al. 2018; De Bhowmick et al. 2015). The significant progress in the lipid biosynthesis field, along with the crucial role of glycerol metabolism in various species cultivated on (waste) glycerol, is noteworthy and has received much interest in recent years. Glycerol metabolism begins when glycerol kinase converts it into glycerol-3-phosphate. The latter is a critical building block for lipid/triacylglycerol synthesis, which can be found in all oleaginous organisms (Xue et al. 2017). The sites where it is activated to glycerol-3-phosphate and transported among cells, in addition to the regulatory activities of related enzymes or genes on how lipids are metabolized, have been thoroughly investigated (Xue et al. 2017). Figure 4 illustrates a comprehensive metabolic and biochemical pathway presenting glycerol utilization by yeast, microalgae, and insect larvae for triacylglycerol biosynthesis. Specifically, the metabolic pathway of glycerol follows de novo for lipogenesis through converting the glycerol-3-phosphate precursor/intermediates molecule before transmitting it into de novo synthesis of triacylglycerol. The process starts with the entry of glycerol into the cells, which is then converted into glycerol-3-phosphate. During glycolysis, glycerol-3-phosphate is further metabolized into pyruvate. Inside the mitochondria, pyruvate is converted into citrate, which serves as a precursor for subsequent steps. The transformation of citrate into malonyl CoA takes place, and this intermediate plays a crucial role in fatty acid synthesis process, which occurs in generally the microalgal plastids (Xu et al. 2019) or the cytosol of yeasts/insect larvae (Zhu et al. 2019). In this process, malonyl co-enzyme A is transformed into various types of fatty acids and their respective acyl forms are then combined with glycerol-3-phosphate to generate triacylglycerol. Synthesis of triacylglycerol occurs in the endoplasmic reticulum of yeast and algae or the cytosol of larvae. Finally, the synthesized triacylglycerol molecules are stored in lipid droplets. This pathway highlights the intricate biochemical reactions and metabolic engineering involved in converting glycerol into valuable triacylglycerols, demonstrating the potential of these organisms in utilization of waste glycerol for sustainable lipid production.
A simplified metabolic pathway of triacylglycerol production from glycerol by microorganisms, including yeast and microalgal cells (A) and insect larvae (B). The glycerol enters the cells and undergoes a series of metabolic steps, including glycerol-3-phosphate formation, glycolysis, and fatty acid synthesis, producing triacylglycerol molecules stored as lipid droplets
In summary, glycerol is a crucial and cost-effective building block for lipid biosynthesis in various oleaginous species, including bacteria, yeast, microalgae, insects, and fungi. The metabolic pathway involves converting glycerol to glycerol-3-phosphate, which plays a crucial role in triacylglycerol synthesis, leading to the storage of synthesized triacylglycerol molecules into lipid droplets. These insights have significant implications for biofuel and lipid-based industries, displaying the potential of waste glycerol utilization for sustainable lipid production.
Holistic recycling of lignocellulosic biomass with waste glycerol for lipids and bioethanol
Various oleaginous microbes can transform carbon sources from glucose, glycerol, and organic acids into droplet forms of lipids in the biomass as a proportion of 20 to 60% per dry weight, which can be mainly preserved as fat (Elsayed et al. 2022a). These lipids, also called single-cell oil or microbial lipids, are formed during the stationary growth phase when nitrogen is limited and a carbon source is abundant (Zhou et al. 2021). The highest feasible ratio is 100 g of glucose to 20–22 g of lipids, which indicates that 5 tons of glucose are required to generate one ton of lipids (Ratledge and Cohen 2008). In this way, a techno-economic analysis of single-cell oil generation using glucose as a medium demonstrated that while conventional biodiesel was priced at USD 1.2/L, the unit production price of biodiesel employing single-cell oil from glucose as a substrate was expected to be USD 5.9/kg (Kumar et al. 2019a). This indicates that biodiesel synthesis using single-cell oil technology from glucose is still not economically feasible (Lazar et al. 2018). Therefore, recycling waste streams from lignocellulosic biomass by waste glycerol combined with the recognition of new microbes will lead to more effective and economical lipid production through this route.
Recently, more research has been conducted on the screening potential of oleaginous microbes grown on lignocellulosic biomass hydrolysates as a low-cost feedstock for single-cell oil accumulation (Patel et al. 2020; Guerfali et al. 2020). However, there are still particular technical challenges that have to be addressed when cultivating oleaginous microorganisms on lignocellulosic biomass hydrolysates for biodiesel generation, such as lignocellulosic biomass recalcitrance, inhibitors toxicity, low lipid content, and low lipid productivity. Therefore, using appropriate technologies for pretreatment and detoxification through lignin removal could be potentially feasible for industrial production of single-cell oil from lignocellulosic biomass in the near future. To obtain a high sugar yield in the lignocellulosic biomass hydrolysates, various pretreatment techniques including chemical, e.g., sulfuric acid and sodium hydroxide, or hydrothermal, e.g., hot water and steam explosion, have been suggested to fractionate the lignocellulosic biomass (Madadi et al. 2023). However, these methods can only extract lignin, hemicellulose, or both lignin and hemicellulose in an unmixable phase, which requires further steps for their separation. Fortunately, a glycerol-based organosolv pretreatment has been developed, which has the potential to produce single-cell oil, bioethanol, and native-like lignin, as shown in Fig. 5. This process employs the glycerolysis reaction to alterations holo-cellulosic structures to improve their hydrolyzability. Another appealing feature is that it was discovered that lignin and glycerol interact under acidic conditions via α-etherification of β-aryl ethers, protecting internal lignin integrity and producing native-like lignin, especially abundant β–O–4 bonds for subsequent monolignol products and bio-aromatic applications (Hassanpour et al. 2020a). After lignin separation, the pretreated fibers with highly digestible cellulose are suitable for bioethanol production (> 90%) by ethanolic fermentation (Hassanpour et al. 2020a). In addition, the pretreated liquor contains processed glycerol, which can be used to synthesize single-cell oil by oleaginous yeast fermentation without dilution steps for detoxification (Fig. 5).
Glycerol-based organosolv pretreatment to convert lignocellulosic biomass to native-like lignin, ethanol, and single-cell oil. The pretreatment of lignocellulosic biomass involves the utilization of glycerol in combination with acidic or alkali catalysts. This treatment is followed by separating the biomass into lignin, liquid, and solid fractions to enhance the valorization of the pretreated biomass. The liquid fraction can be subjected to fermentation to produce single-cell oil, while the solid fraction can be fermented to produce ethanol. This approach maximizes the utilization and conversion of the pretreated biomass into valuable products such as single-cell oil and ethanol
To increase the efficiency of glycerol pretreatment, a tiny amount of catalysts, such as acid and alkali, is supplied to reduce process temperatures and time (Hassanpour et al. 2020b). In this regard, Zhang et al. (2013) found that pretreatment of sugarcane bagasse with acidified glycerol (containing 1.2% hydrochloric acid at 130 °C for 1 h) resulted in a high glucose yield (92.0%). In addition, Wu et al. (2019) reported that compared to pure glycerol, crude glycerol pretreatment enhanced the corncob yield of glucose by 42.6%. Guerfali et al. (2020) also found that the nitrogen content in waste glycerol was 0.02% by weight; hence, oleaginous yeast cells tend to produce more neutral lipids under nitrogen-limiting conditions, creating favorable lipid accumulation. In addition, waste glycerol contains several elements, including potassium, sulfur, magnesium, and calcium, which can be an added advantage for yeast growth (Qin et al. 2017). As a result, waste glycerol is typically generated as a by-product in biodiesel refineries and can be adopted directly as a practical pretreatment process.
Table 3 describes the results of glycerol-based organosolv pretreatment for simultaneous assimilation of hydrolysates with residual glycerol for microbial lipid production from pretreated sugarcane bagasse (Hassanpour et al. 2019), switchgrass (Chen et al. 2018), and rice straw (Tang et al. 2020b). Chen et al. (2018) found that switchgrass pretreated liquor generated from multiple cycles of acidified-glycerol pretreatment could be utilized for fermentation by Rhodotorula toruloides, producing 8.1 g/L lipids. However, this approach consisted of washing and solid–liquid separation, which would raise operating and capital costs. Alternatively, Tang et al. (2020a, b) presented Cryptococcus curvatus lipid fermentation on glycerol-ferric chloride pretreatment supernatant after lignin recovery. The highest lipid titer of 7.2 g/L was noted. Interestingly, a recent study by Hassanpour et al. (2020b) developed a two-step pretreatment for effective enzymatic and lipid production. The fed-batch fermentation of Rhodotorula toruloides RP 15 growing on this hydrolysate with residual glycerol resulted in lipid yield and content of 26.7 g/L and 0.24 g/g, respectively.
It was found that incorporating glycerol-based pretreatment and co-fermentation of hydrolysate sugars with remaining glycerol by Rhodotorula toruloides to synthesize microbial lipids has an advantage. It can decrease water and nutrient utilization while increasing the whole process’s efficiency and economics compared to the conversion of pretreated lignocellulosic biomass solely (Hassanpour et al. 2020b). In addition, Cai et al. (2019) revealed that supplementing pretreated hydrolysate with 7.5% glycerol increased cellulose digestibility by 11% above the control enzymatic hydrolysis in water (no buffer). Hemicellulose is frequently acetylated; some organic acids, like acetic acid, are typically present in lignocellulosic hydrolysates. Based on metabolic models assumptions, the co-fermenting of acetic acid with additional carbon sources, such as glucose, xylose, and glycerol, achieved higher theoretical lipid yields, indicating that co-fermentation by glycerol/acetic acid could be a useful route for single-cell oil synthesis (Zhou et al. 2021). Fontanille et al. (2012) evaluated the co-fermented acetic acid with glycerol by Yarrowia lipolytica MUCL 28849 employed in a fed-batch fermentation mode in two stages. Adding acetic acid after eliminating glycerol yielded a biomass of 41.0 g/L and lipid production of 16.0 g/L. This process confirmed the possibility of using waste glycerol as an efficient link between the conventional biodiesel industry and lignocellulosic lipid-based yeast cell factories.
In summary, oleaginous yeast can convert glucose, glycerol, and organic acids into lipid droplets, constituting over 20% of their cellular dry weight and stored as fat in their components. Nevertheless, the high costs associated with single-cell oil production challenge its economic viability. To address this, researchers are exploring the use of lignocellulosic biomass to identify high-value microorganisms that can tolerate waste glycerol impurities, thereby enhancing lipid productivity. Promising research focuses on the mass production of single-cell oil, bioethanol, and native lignin through cost-effective and scalable glycerol-based pretreatment methods. These endeavors hold the potential to overcome economic barriers and pave the way for sustainable and economically feasible production of valuable products from glycerol-rich sources.
Bio-lipids production through co-cultivation of yeast and microalgae
The concept of co-cultivation of ectosymbiotic oleaginous species (yeast and algae) has been introduced as a synergistic technique by employing their dynamic symbiotic interactions and metabolic abilities to increase the efficiency of resource recovery and lipid productivity (Dias et al. 2019). This technique was considered for great application in lipid production from waste glycerol utilization for biofuel production (Kitcha and Cheirsilp 2014).
In this technique, co-culturing experiments demonstrated the relevance of the symbiotic interaction between oleaginous species (yeast and algae) for enhancing lipid production. The beneficial effects of the mixed yeast–algae symbiotic co-culture performance over mono-culture are displayed in Fig. 6. (1) In the co-culture system, nitrogen or phosphate starvation begins earlier, increasing lipid accumulation. (2) Synergistic effects on gas balance, nutrient exchange, and pH auto-regulation have been observed in the co-culture system, providing a more suitable environment for cellular growth and lipid accumulation. (3) Microalgae could generate dissolved carbon into the cultivation system, which would harm photosynthesis; however, yeast can utilize it. (4) Specific secondary metabolites released by algae would have a positive effect on the proliferation of yeast.
Symbiotic relationship in a co-culture of microalgae and yeast. Microalgae play a crucial role in providing yeast with oxygen and carbon in the form of extracellular polymeric substances produced through photosynthesis. In return, yeast supports microalgae by supplying them with carbon dioxide to aid in their photosynthesis process. This symbiotic relationship helps stabilize the pH balance in the environment. Microalgae utilizes the carbon dioxide and organic acids produced by yeast, while carbon dioxide and residual organic acids from microalgae help to prevent excessive alkalinity. As a result, this symbiotic interaction between microalgae and yeast promotes a harmonious and balanced environment for both organisms
In the context of the yeast–algae consortium, a partner yeast can break down various carbohydrates into monosaccharides, which are utilized by the algae for their growth. Additionally, the presence of carbon dioxide in the medium is converted by microalgae into bicarbonate, which is subsequently absorbed by the microalgae, releasing OH– ions and causing the culture system to become alkaline (Arora et al. 2019). Autotrophic microalgae in the consortium play a crucial role in supplying more oxygen, which is essential for the growth of heterotrophic yeast (Iasimone et al. 2018). Furthermore, the microalga may play a vital function in consuming potentially toxic substances present in the mixed culture that could otherwise harm the growth of the yeast (Dias et al. 2022). However, one challenge in this consortium is the alteration of light exposure for the microalgal cell population due to the proliferation of the partner yeast. This issue can be addressed by adjusting the inoculum ratio of yeast to algae or by implementing sequential culture systems (Dias et al. 2019).
Table 4 describes the various co-cultivation of microalgae and oleaginous yeast that produce lipids from crude glycerol or mixed wastewater. The studies have demonstrated that the lipid produced from crude glycerol is an excellent carbon source for mixed co-culture. For instance, Kitcha and Cheirsilp (2014) pioneered the development of a mixed co-culture involving microalgae (Chlorella spp.) and yeast (Trichosporonoides spathulate). They employed various inoculum ratios in a 5-L stirred bioreactor to address the treatment of waste glycerol. The study hypothesized that increasing the microalgae ratio (> 1:1) led to higher final algal cell counts; however, the specific productivity of these cells was lower. This phenomenon was attributed to the shading effect caused by the high density of yeast cells, which subsequently hindered the growth of microalgae. Furthermore, the study demonstrated that co-encapsulation of algal and yeast cells within alginate beads facilitated the development of lipid-like free cells. Consequently, this encapsulation technology not only enabled lipid production but also simplified the subsequent harvesting process.
Qin et al. (2018) evaluated a mixed culture of microalgae (Chlorella vulgaris) with yeast (Yarrowia lipolytica) at a 1:1 alga/yeast ratio to treat the mixed liquid digestate/ glycerol. They found that adding the yeast inocula to the culture during the algal lag phase could improve lipid productivity and elevate the algal assimilable organic substrate in the culture. Further, they noticed generated biomass at an average concentration of 1.62 g/L with a lipid content of 18.98%. Cheirsilp et al. (2012) explored the possibility of improving lipid synthesis from pure glycerol with urea using a symbiotic mixed culture of Rhodotorula glutinis and Chlorella vulgaris. The results showed a substantial improvement in total biomass (3.20 g/L) and lipid content (34.40%) when these two species were co-cultivated. Another study found that Spirulina platensis co-cultivating with Rhodotorula glutinis significantly increased the production of both biomass and lipid yield. This co-culture system showed high efficiency for the removal of nitrogen and chemical oxygen requirements, with removal efficiencies of 35% and 73%, respectively (Xue et al. 2010). Likewise, a mixed culture of Rhodosporidium toruloides and Chlorella pyrenoidosa improved lipid content (63.45%) when these two species were co-cultivated on mixed wastewaters (Ling et al. 2014). Thus, co-culturing the microalgae and oleaginous yeast can effectively recycle nitrogen, phosphorous, and organic carbon compounds from the wastewaters riched in crude glycerol as compared to monocultures.
In summary, the microalgae-yeast consortium shows great promise for sustainable and effective utilization of industrial wastes while producing valuable products like lipids and biodiesel and mitigating pollution. The symbiotic interaction between microalgae and yeast allows oxygen production, carbon dioxide utilization, and effective pollutant removal. This co-culturing approach addresses the limitations of heterotrophic yeast and offers a cost-effective and sustainable strategy for treating waste glycerol with wastewater, reducing energy consumption for aeration, and lowering carbon dioxide emissions from traditional biological treatment systems. While numerous studies have demonstrated enhanced lipid synthesis through the synergistic interaction of microalgae and oleaginous yeast, further research is necessary to develop metabolic models with better optimal growth conditions. Factors such as species selection, growth media, algae-to-yeast inoculum ratio, carbon–nitrogen ratio, illumination level, aeration, incubation period, and temperature need to be investigated to maximize the benefits of this co-cultivation approach. By advancing our understanding of these aspects, we can unlock the full potential of this consortium for sustainable waste glycerol treatment and biofuel production.
Holistic approaches of waste glycerol utilization with wastewater for biodiesel production by yeast or microalgae
Worldwide, domestic wastewater sludge as a by-product of municipal wastewater treatment is generated daily in significant quantities. To prevent further pollution, it is crucial to manage this sludge appropriately. Traditionally, wastewater sludge has been considered as a biowaste and dealt mainly with landfilling and incineration methods. However, recent advancements in biological and thermal technologies have identified wastewater sludge as a valuable bioresource (Chen et al. 2021). These emerging technologies have revealed that wastewater sludge contains increased organic content and serves as a reusable substrate for the growth of microorganisms like yeast and microalgae. Enriched with nutrients such as soluble carbon, nitrogen, phosphorus, and trace elements, it represents a cost-effective medium for microorganism development (Han et al. 2016). In a study conducted by Kumar et al. (2021), it was found that wastewater sludge contains ample trace elements and nitrogen, which, when combined with crude glycerol as a carbon source, significantly enhanced the growth of the oleaginous yeast Yarrowia lipolytica. The research demonstrated that within 72 h of culturing the yeast in a mixture of crude glycerol and diluted sludge, an impressive amount of lipids (31.44 g/L) was produced.
Co-valorization of crude glycerol (carbon source) and wastewater sludge (nutrient source) for the biosynthesis of lipids using oleaginous yeast or microalgae can establish a dual-purpose solution to significantly lower the cost of producing biodiesel while simultaneously recycling these two types of wastes eco-friendly. In addition, Zhang et al. (2016) evaluated the uses of waste glycerol for the production of different biofuels. The study showed that ethanol and hydrogen production from crude glycerin was less feasible regarding conversion efficiency, net energy output, and other technical issues than biodiesel production via oleaginous yeast fermentation, which achieved a positive net energy balance. Therefore, converting waste glycerol into lipids using yeast or microalgae and subsequently transforming these lipids into biodiesel is a more advantageous pathway than ethanol production. When comparing biodiesel production costs, utilizing bio-lipids derived solely from sludge as the medium yields a competitive selling price of approximately USD 0.78/L. This price is competitive with fossil diesel (USD 0.88/L) and plant oil (USD 1.0/L) biodiesel (Zhang et al. 2014). Alternatively, producing biodiesel from the oleaginous yeast Trichosporon oleaginosus cultivated with waste glycerol and sludge as the medium could achieve a net energy output and reduce the global warming potential (Chen et al. 2021). This study’s projected biodiesel production costs range from USD 630 to USD 860 per ton, depending on ammonium chloride addition. The study assumed that biodiesel synthesis from co-fermented waste glycerol and sludge stands as a highly competitive and economically viable option, especially when compared to the current market price of USD 900 per ton (Chen et al. 2021). Using oleaginous yeast for co-valorizing sludge and crude glycerol demonstrated comparability to using yellow grease for biodiesel production.
Another possible route that is more effective for co-valorizing wastewater sludge and waste glycerol using microalgae has been found. Utilizing algal biomass stands out as one of the most viable long-term solutions to the world’s energy crisis and sustainability, particularly the issue of using fossil fuels (Wang et al. 2018; Abomohra and Hanelt 2022). The minimal requirements for algal growth include carbon dioxide, sunlight, organic matter, and water. Microalgae cells, like oleaginous yeast, can be cultivated in non-agricultural land using a variety of biowaste such as waste glycerol and domestic wastewater sludge (Abomohra et al. 2022a, b; Beyer et al. 2023). As a result, the concept of “algae for green fuels” has recently acquired attention regarding sustainability and circular bioeconomy (Xu et al. 2019; Wang et al. 2020). Many species showed a wide range of oil concentrations, i.e., 20–60% dry weight, which could increase under stress conditions (Elsayed and Abomohra 2022). In addition, one ton of dry microalgal biomass needs 2.05 tons of carbon dioxide at a baseline cultivation scenario (Rosenberg et al. 2011), which, in terms of carbon dioxide bio-fixation, is considered to be 10–50 times more efficient than higher plants (Elsayed and Abomohra 2022). An earlier investigation assessed the ability of carbon dioxide from flue gases to produce microalgae and reported that it was possible to reduce carbon dioxide emissions by 90% overall (Rosenberg et al. 2011). This finding confirms that cultivating microalgae along with bio-sequestration of carbon dioxide is an attractive technique for sustainable biodiesel production.
Choi and Yu (2015) assessed three microalgae, Botryococcus braunii, Chlorella vulgaris, and Scenedesmus sp., cultivated under mixotrophic conditions in various waste glycerol levels to increase cellular growth and lipid content. Compared to autotrophic growth, these strains showed a higher biomass yield when grown on crude glycerol, where 5 g/L glycerol achieved the maximum biomass concentration for these species. Supplementation of 5 g/L glycerol into the growth medium stimulated cellular growth and yielded the highest level of lipid accumulation of the algal biomass. The strains represent prospective resources for the biodiesel industry since the fatty acid profiles met the requisite biodiesel standards. In addition, Grama et al. (2016) confirmed that waste glycerol could increase the microalgal (Dactylococcus dissociatus MT1) biomass production photoheterotrophy while concurrently lowering the requirement for cell culture gas exchange. These findings suggest that adding waste glycerol could enhance the biomass output while decreasing the energy needed for aeration. In addition, Abomohra et al. (2018) introduced an innovative technique using Scenedesmus obliquus to enhance algal biodiesel productivity. They achieved this by cultivating the microalga in a medium that included various substitutions of waste glycerol and hydrolysate of lipid-free algal biomass. Among these substitutions, 5 g/L waste glycerol demonstrated the most significant treatment for lipid synthesis, with a maximum lipid productivity of 59.66 mg/L. This approach presents a promising and revolutionary zero-waste system for cultivating microalgae using waste resources from microalgal biodiesel production. By adopting this technique, biodiesel production can be enhanced while simultaneously addressing the disposal of these two types of waste.
Figure 7 shows three possible routes to reuse waste glycerol derived from algae biodiesel through using it as a carbon source in microalgae cultivation or for alcohol production that enables waste-free biorefinery. In this way, Martín and Grossmann (2014) developed a mathematical model to optimize the route design and operation of integrated units that utilize waste glycerol to produce methanol or ethanol as part of the manufacturing of fully renewable biodiesel from algal biomass. The waste glycerol can be converted into methanol via two processes, viz. the reforming in steam reforming or autoreforming technologies of glycerol to syngas (hydrogen/carbon monoxide), followed by the conversion of this syngas processed into methanol. The purified methanol is utilized to transesterifying the algal lipid into fatty acid methyl ester (FAME). The second method involves co-fermenting waste glycerol with glucose obtained from algal carbohydrates to produce ethanol. The refined ethanol is utilized to transesterifying the algal lipids into fatty acid ethyl ester (FAEE). When comparing the integration of waste glycerol to methanol or ethanol production in terms of integrated features, using waste glycerol for methanol production reduces reliance on fossil fuels by 50% without an increase in biofuel yield, while using it for ethanol production increases biofuel production by 50% without the need for materials derived from fossil fuels, lower production costs, and freshwater usage. The main disadvantage is higher investment costs and energy consumption due to ethanol dehydration relative to methanol production. The use of ethanol as transesterifying agent has the advantage of reducing the need for raw materials derived from fossil fuels while achieving bioenergy self-sufficiency.
Diagrammatic routes of reuse waste glycerol derived from algae biodiesel by: (1) The waste glycerol can be returned into the microalgal cultivation as a carbon source to enhance lipid and carbohydrate production. (2) Waste glycerol is reformed by steam reforming technology to generate syngas and methanol. (3) Waste glycerol is fermented with glucose obtained from algal carbohydrates to produce ethanol. The algal lipid fraction can produce biodiesel, either using ethanol in the form of a fatty acid ethyl ester (FAEE) or methanol in the form of a fatty acid methyl ester (FAME) as the transesterification agent
Xu et al. (2019) evaluated energy recovery by producing two forms of biofuels (biodiesel and bioethanol) using Scenedesmus obliquus in a dual valorization process by lipid extraction for biodiesel followed by fermentation of lipid-free algal biomass for ethanol production. The study was conducted through the recycling of waste glycerol as a carbon source for the mixotrophic growth of the microalga to enhance the lipids and carbohydrates in the algae cells. The biomass treated with 2.5% waste glycerol demonstrated an improvement in maximum bioethanol productivity, up to 0.185 g L−1 h−1, as a result of the buildup of carbohydrates under the waste glycerol. However, further fermentation following lipid extraction increased the highest bioethanol output over the whole cell from wastewater and lipid-free biomass by 32.3% and 15.1%, respectively. The integrated route, which involved growing Scenedesmus obliquus in waste glycerol-enriched medium and applying sequential fermentation of the remaining biomass after lipid extraction, produced the highest gross energy output of 21.4 gigajoules per ton dry microalgae and had the highest rate of energy recovery at 62.9%. These results provide a cutting-edge practical integrated route for recycling waste glycerol with wastewater using Scenedesmus obliquus to produce two liquid biofuels, improving resource conversion efficiency. Thus, the optimal algal composition can be established concurrently with the combined synthesis of biodiesel and bioethanol, with a portion of the bioethanol serving as a transesterifying agent.
In summary, domestic wastewater sludge represents a significant biowaste on a global scale, necessitating careful management to prevent secondary pollution. Recent developments have identified wastewater sludge as a valuable nutrient-rich bioresource, providing a cost-efficient substrate for microorganisms like yeast and microalgae to produce single-cell oil. By co-valorizing wastewater sludge with crude glycerol, the biodiesel production cost can be substantially reduced, rendering the process economically feasible. Furthermore, microalgae offer a promising and environmentally friendly route for biodiesel production through carbon dioxide bio-fixation and re-utilization of biodiesel by-products, including lipid-free biomass and waste glycerol, making them an attractive option for sustainable biodiesel production.
Insect for achieving high biodiesel from waste glycerol
Insects can store energy in their biomass cells as triglycerides (fat) and glycogen during their larval stage development (Zhu et al. 2019). This has led to the emergence of saprophagous insects as a novel and efficient recycling biotechnology for bioconverting various organic biowastes into valuable lipids through their metabolic processes (Elsayed et al. 2020). Among the fastest-growing and most promising insect species, the black soldier fly larvae (BSFL), Hermetia illucens, stands out, especially for its high-lipid content, making it an ideal candidate for a new generation of bioenergy sources. Notably, the diet’s composition significantly influences the final product of BSFL biomass, the amount of lipids accumulated, and the quality of BSFL biodiesel. By providing a balanced diet rich in organic acids, glycerol, protein, and carbohydrates, these features can be tailored to optimize the bioconversion process, leading to a remarkable improvement in their efficiency and a reduction in the overall production cost (Elsayed et al. 2020).
In this way, Elsayed et al. (2022b) utilized the black soldier fly larvae to bio-recycle chicken manure combined with various amounts of rapeseed straw, followed by biodiesel and protein generation, as a novel solid waste disposal strategy. Applying a 20% rapeseed straw ratio across various treatments demonstrated the highest fiber digestibility, improved larval biomass in a shorter time, and increased lipid productivity. The study showed that insect farming biotechnology can efficiently recover elements and nutrients in the form of energy or animal feed. In addition, the black soldier fly larvae have been studied for co-conversion of protein-rich wastes (rapeseed cake) with crude glycerol for built-up lipids in larvae and biodiesel production (Almutairi et al. 2023). The study revealed that the mixture acts synergistically with waste glycerol, immediately assimilating into the fatty acid biosynthesis pathway to accumulate lipids, while rapeseed cake is converted to promote larval development. Applying 5.0% waste glycerol significantly increased the areal productivity, larval biomass gain rate, and biodiesel productivity above the control by 13.0%, 17.2%, and 57.7%, respectively. In addition, the results of the study reported that with rearing larvae on rapeseed cake, the crude larval lipid content reached 35%, whereas with supplemented 5% waste glycerol, the crude lipid content increased dramatically, reaching 43.8%.
Table 5 summarizes the potential productivities of lipids and biodiesel with land use from triglyceride feedstocks. Based on the above study, black soldier fly larvae demonstrate competitive lipid contents, ranging from 35 to 43% compared to terrestrial oil crops and microbial lipid production from oleaginous yeast or microalgae. Interestingly, microbial biodiesel productivity from oleaginous yeast is higher, at 3546 kg ha−1 y−1, which is 6.3-fold of the yield of soybean biodiesel (562 kg ha−1 y−1) (Soccol et al. 2017). Similarly, microalgal biodiesel productivity exceeds palm biodiesel by more than 25-fold. However, BSFL biodiesel productivity is 1.72 times higher than microalgae and an impressive 43 times higher than palm biodiesel. With the inclusion of 5.0% glycerol in the larvae’s diet, the improvement in larval lipid levels allows for a remarkable increase in BSFL biodiesel productivity, reaching 204,819 kg ha−1 y−1, which represents a 22.8% increase compared to without glycerol addition (Almutairi et al. 2023). This highlights the insect larvae’s superior biomass and biodiesel areal productivity compared to microalgae, oleaginous yeast, and other oil crops. Moreover, as compared to converting yeast or algal lipids to biodiesel, BSFL biodiesel will be a more cost-effective technology since it minimizes cost and time in harvesting, drying, and lipids extraction. Regarding land use, BSFL-based biodiesel offers also advantages such as easy rearing, high biodiesel and protein productivity, lower competition, and reduced resource requirements for energy, water, and farmland. These factors make BSFL-based biodiesel a promising and acceptable strategy for mitigating the land-use change impacts. Overall, the findings from Table 5 underscore the potential of black soldier fly larvae as an appealing platform for sustainable large-scale biodiesel and protein production through waste glycerol reintegration strategies. This approach holds promise in contributing to a more environmentally friendly and resource-efficient biofuel production process.
In summary, saprophagous insects, mainly black soldier fly larvae, exhibit remarkable efficiency in converting various biowastes into lipids and glycogen stored within their cells, presenting a promising source for biodiesel production and waste utilization. Black soldier fly larvae and microalgae demonstrate higher biodiesel productivity than other crops, making them attractive alternatives to replace fossil fuels. By combining protein-rich biomass, such as rapeseed cake, with waste glycerol, the lipid content and biomass yield of black soldier fly larvae can be significantly enhanced. This innovative waste-free biorefinery approach offers biodiesel-based industries a sustainable and eco-friendly strategy to address environmental concerns.
Estimation of biodiesel properties of oleaginous species
There are many kinds of saturated and unsaturated fatty acids majorly accumulated by oleaginous organisms. Fatty acid compositional profiles were varied with oleaginous organisms, cultivation conditions, and medium composition. Insects, yeast, and microalgae represent the main oleaginous organisms that can utilize waste glycerol to generate fatty acids that are subsequently converted into biodiesel through the transesterification process. As shown in Table 6, the waste glycerol addition can enhance the compositional profile of the fatty acids and, hence, the subsequent biodiesel properties. Almutairi et al. (2023) reported that supplementation of 5% waste glycerol in the cultivation media of black soldier fly larvae caused a significant increase in fatty acid synthesis compared to the control media, which lacked glycerol. The predominant fatty acids were lauric acid (C12:0) and oleic acid (C18:1), which recorded 35.05% and 39.97% in 5% glycerol media. It was noticed that the glycerol supplementation has no significant effect on the total saturated, monounsaturated, and polyunsaturated fatty acids.
Various oleaginous yeasts demonstrate higher biodiesel productivity than oilseed crops, as mentioned in the previous section. In this regard, Rhodosporidium toruloides is a well-known oleaginous yeast with a high efficiency of fatty acid synthesis from different carbon sources (Soccol et al. 2017; Carota et al. 2020). Although sugarcane juice and urea were enough to produce fatty acids via R. toruloides, glycerol with a combination of orange peel enhanced the fatty acid synthesis. The palmitic acid (C16:0) and oleic acid (C18:1) were the predominant fatty acids produced by R. toruloides. Glycerol–orange peel medium enhanced the palmitic acid (C16:0) production to 23.45% compared to the sugarcane–urea media, which recorded 21.5%, while it is not optimal for oleic acid (C18:1), which was 54.99%. Sugarcane juice and urea were suitable for the production of oleic acid (C18:1), recording 62.1% (Soccol et al. 2017; Carota et al. 2020). In addition, many oleaginous microalgae, including Chlorella vulgaris, Chlorella pyrenoidosa, and Scenedesmus sp., have been widely applied for lipid and biodiesel production due to their capability to cultivate under mixotrophic conditions in various waste glycerol levels (Abomohra et al. 2018; Xu et al. 2019). Recently, Rana and Prajapati (2021) investigated fatty acid synthesis via Chlorella pyrenoidosa cultivated on synthetic wastewater with and without waste glycerol. They found that linolenic acid (C18:3) was the dominant fatty acid produced without waste glycerol, recording 39.78%, while the addition of 3 g/L glycerol enhanced the fatty acid productivity. The productivity of palmitic acid (C16:0) and oleic acid (C18:1) enhanced after supplementation of 3 g/L glycerol to the synthetic wastewater media to be 26.37% and 33.38%. However, the total saturated fatty acids fraction originating from black soldier fly larvae was higher than the total saturated fatty acids fractions originating from yeast and microalgae; this finding highlights the higher insect’s efficiency in biodiesel production.
Biodiesel productivity and its characteristics depend on the fatty acids profile. Several biodiesel parameters, as mentioned in Table 6, can be expected from the empirical models based on the fatty acid profile (Hoekman et al. 2012). Among these parameters, the cetane number is the most common indicator of the biodiesel quality, while the higher cetane number’s biodiesel causes the engine to work easily and smoothly. According to the American Society for Testing and Materials Standard and the European Standard (EN 14214), the cetane number should be above 47 and 51, respectively (Zhao et al. 2023). The biodiesel originating from the black soldier fly larvae has the highest cetane number and, hence, the highest quality compared to the biodiesel originating from yeast and microalgae. Moreover, the black soldier fly larvae-derived biodiesel was compared to rapeseed oil-based biodiesel within the criteria for European Standard 14,214 and American Society for Test and Materials D6751 (Ishak and Kamari 2019). The findings confirmed that BSFL biodiesel proved to be comparable to the international standards and to that of rapeseed oil-derived biodiesel. The cetane number of the biodiesel originated from Rhodosporidium toruloides cultivated on synthetic wastewater without glycerol accounted 57.06 (Soccol et al. 2017), and this is similar to other studies mentioned that the cetane number of biodiesel produced by Rhodosporidium toruloides was 56.3, 56.9, and 57.6 using media containing molasses of 357 g/L sucrose + 152 g/L glucose + 121 g/L fructose, sugarcane bagasse hydrolysate, and corn stover hydrolysate, respectively (Vieira et al. 2014; Fei et al. 2016). On the other side, cetane number increased in biodiesel originating from Chlorella pyrenoidosa after supplementation of the waste glycerol. The degree of unsaturation and the oxidative stability are significant parameters indicating the auto-oxidation rate of the biodiesel. The higher oxidative stability of insect’s biodiesel than yeast’s and microalgal biodiesel supported the longer shelf life of biodiesel originating from insects (Zhao et al. 2023). In contrast to oxidative stability, the higher cloud point is not preferred in biodiesel due to the difficult application in cold regions; therefore, the lower cloud point of insect’s biodiesel increases its application potential, while the yeast and microalgae generate restricted applied biodiesel, especially in cold regions (Hoekman et al. 2012).
In conclusion, insects, yeast, and microalgae can convert waste glycerol into fatty acids and subsequent biodiesel, with glycerol addition enhancing biodiesel production and their characteristics. The oleaginous yeast Rhodosporidium toruloides and microalgae Chlorella pyrenoidosa also benefited from glycerol-based media for enhanced fatty acid synthesis. The total saturated fatty acids fraction originating from black soldier fly larvae was higher than that from yeast and microalgae. Black soldier fly larvae-derived biodiesel had the greatest quality, as characterized by cetane number, oxidative stability, and cloud point, followed by Rhodosporidium toruloides and Chlorella pyrenoidosa-derived biodiesels.
Biocircular platforms and perspectives
To promote public health, minimize ecological burden, and create new job opportunities, it is imperative to explore and implement sustainable waste glycerol management strategies through circular agriculture and economy biosystems (Eraky et al. 2022). Figure 8 illustrates suggested reintegration approaches for waste glycerol, employing cascade valorization routes with both liquid and solid wastes, utilizing oleaginous yeast, microalgae, and black soldier fly larvae, aligning with the principles of circular bioeconomy. The liquid and solid fractions containing essential nutrients can be effectively reused or remediated by the suggested organisms, facilitating the production of lipids (for biodiesel) and other high-value by-products that can find applications in agriculture. This approach enhances the energy and food security and minimizes the negative ecological impacts. Various sources of biowastes, such as domestic wastewater sludge, lignocellulosic biomass, and protein-rich wastes, can be synergistically combined with waste glycerol to generate lipids and other valuable products. The symbiotic co-culture technique of microalgae with oleaginous yeast is a successful example of organisms that can treat waste glycerol in conjunction with liquid fractions from wastewater sludge or lignocellulosic hydrolysis. This integrated recovery process efficiently harnesses nutrients, bioenergy, and clean water, representing a promising advancement in waste glycerol management within the circular bioeconomy framework.
Valorization of waste glycerol by the reintegration strategies with microalgae, yeast, and black soldier fly larvae. The reintegration strategy involves cascading valorizing waste glycerol with liquid wastes (such as sludge wastewater or lignocellulosic hydrolysates) using a co-culture technique of microalgae with oleaginous yeast. Solid wastes (protein-rich wastes) are also processed using black soldier fly larvae. In this process, glycerol serves as the carbon source for the oleaginous organisms, while other nutrients are obtained from the target wastes. These methods can produce valuable bioproducts like biodiesel, protein feed, and crop fertilizer from suggested agro-waste and organisms. This approach fosters environmentally friendly and more sustainable circular agriculture systems
On the other hand, the growing global demand for animal-based products has led to a significant increase in protein-rich wastes, including rapeseed cake or animal by-products. However, these wastes pose environmental challenges due to disease spread and the emission of harmful gases like ammonia. Hence, developing efficient and eco-friendly techniques for converting these wastes is crucial. In this context, we propose a novel approach using black soldier fly larvae to recycle the solid fractions of protein-rich wastes, incorporating waste glycerol into the process. This integrated system allows for protein feed, biodiesel, and crop fertilizer production. The conversion of protein-rich wastes with waste glycerol by black soldier fly larvae leads to a well-balanced diet for the larvae, resulting in increased lipid content and productivity. This holistic recycling route not only offers a sustainable solution for managing protein-rich waste and waste glycerol from industrial animal and biodiesel by-products but also aids in enhanced resource recovery and reduced greenhouse gas emissions. Consequently, it aligns with several Sustainable Development Goals and supports achieving low-environmental footprints. Overall, the suggested cascade valorization routes provide a versatile approach for decision-makers to choose the most suitable waste glycerol remediation/recycling technique based on their objectives, the type of waste, and investment considerations. This comprehensive approach contributes to sustainable waste management, resource utilization, and environmental protection.
Further studies are required to conduct energetic and mass balance assessments to confirm the feasibility of the aforementioned suggested reintegration biorefinery routes. Future investigations should focus on assessing the sustainability aspects of waste-free biorefinery from multiple perspectives, including environmental, economic, and thermodynamic aspects, using advanced sustainability assessment tools such as life cycle assessment and techno-economic analysis (Osman et al. 2021a). By employing life cycle assessment and techno-economic analysis, future studies can identify the most efficient, productive, and sustainable routes for producing valuable bioproducts from the suggested waste materials, including biodiesel, protein feed, and crop fertilizer. Economic analysis is crucial in evaluating the economic viability and competitiveness of the waste glycerol-based biorefinery, as well as its produced products, compared to conventional alternatives. Finally, a comprehensive assessment that integrates life cycle assessment with economic analysis will provide valuable insights into the waste glycerol-based biorefinery’s environmental impacts, energy efficiency, and economic viability. This approach will enable informed decision-making, optimize resource utilization, and ensure the sustainability metrics and economic competitiveness of the suggested reintegration strategies through cascade valorization of waste glycerol with liquid and solid wastes for biodiesel production as the main product.
In summary, waste glycerol can be effectively utilized in biocircular platforms along with nutrient-rich liquid and solid waste fractions from various sources, such as domestic wastewater sludge, lignocellulosic biomass, and protein-rich waste, to generate biodiesel and other valuable by-products. Oleaginous yeast and microalgae co-culture efficiently treats waste glycerol, facilitating integrated nutrient recovery and clean water generation. Furthermore, black soldier fly larvae present an innovative approach to recycling protein-rich wastes with waste glycerol, increasing lipid content and productivity while reducing greenhouse gas emissions. Advanced sustainability assessments like life cycle assessment and techno-economic analysis can be applied to confirm the feasibility of these waste-free biorefinery strategies and ensure the efficient and sustainable production of valuable by-products. By integrating these assessments, the environmental and economic viability of the waste glycerol-based biorefinery can be validated, enabling informed decision-making and optimizing resource utilization for a more sustainable and productive biodiesel and by-product production process.
Conclusion
This review addressed the state-of-the-art studies and biotechnological achievements of reusing crude (waste) glycerol derived from biodiesel production. Crude glycerol, a valuable by-product of biodiesel production technologies, has garnered considerable attention for its economic potential. Biodiesel production contributes to around 10% of the total crude glycerol produced. The production of high-value products from crude glycerol was thoroughly addressed in this review. Studies have shown that Escherichia coli and Klebsiella pneumoniae can efficiently utilize crude glycerol to produce ethanol and other valuable products such as volatile fatty acids and hydrogen. Moreover, converting crude glycerol into bio-lipids presents a novel route through oleaginous organisms like yeast, microalgae, and saprophagous insects, which holds potential for a low-carbon bioeconomy and sustainable biodiesel practices. The synergistic approach involves co-valorizing crude glycerol and domestic wastewater or lignocellulosic hydrolysates using oleaginous yeast or microalgae, producing lipid droplets. In addition, the symbiotic relationship between oleaginous yeast and microalgae results in a significant increase in biomass and lipid productivity. Finally, the black soldier fly larvae exhibit remarkable efficiency in converting protein-rich wastes incorporated with waste glycerol into high-value-added products such as biodiesel, animal feeds, and fertilizers.
References
Abomohra A, Hanelt D (2022) Recent advances in micro-/nanoplastic (MNPs) removal by microalgae and possible integrated routes of energy recovery. Microorganisms 10:240010–242400. https://doi.org/10.3390/MICROORGANISMS10122400
Abomohra A, Eladel H, El-Esawi M et al (2018) Effect of lipid-free microalgal biomass and waste glycerol on growth and lipid production of Scenedesmus obliquus: innovative waste recycling for extraordinary lipid production. Biores Technol 249:992–999
Abomohra AE-F, Elsayed M, Esakkimuthu S et al (2020) Potential of fat, oil and grease (FOG) for biodiesel production: a critical review on the recent progress and future perspectives. Prog Energy Combust Sci 81:100868
Abomohra A, Li M, Faisal S et al (2022b) Maximizing nitrogen removal and lipid production by microalgae under mixotrophic growth using response surface methodology: towards enhanced biodiesel production. Fermentation 8:682
Agrawal D, Budakoti M, Kumar V (2023) Strategies and tools for the biotechnological valorization of glycerol to 1, 3-propanediol: challenges, recent advancements and future outlook. Biotechnol Adv 66:108177. https://doi.org/10.1016/J.BIOTECHADV.2023.108177
Almutairi AW, Abomohra A, Elsayed M (2023) A closed-loop approach for enhanced biodiesel recovery from rapeseed biodiesel-based byproducts through integrated glycerol recycling by black soldier fly larvae. J Cleaner Prod 409:137236
Arora N, Patel A, Mehtani J et al (2019) Co-culturing of oleaginous microalgae and yeast: paradigm shift towards enhanced lipid productivity. Environ Sci Pollut Res 26:16952–16973
Asad-ur-Rehman, Saman Wijesekara RG, Nomura N et al (2008) Pre-treatment and utilization of raw glycerol from sunflower oil biodiesel for growth and 1,3-propanediol production by Clostridium butyricum. J Chem Technol Biotechnol 83:1072–1080. https://doi.org/10.1002/JCTB.1917
Asopa RP, Bhoi R, Saharan VK (2022) Valorization of glycerol into value-added products: a comprehensive review on biochemical route. Bioresour Technol Rep 20
Bagnato G, Iulianelli A, Sanna A, Basile A (2017) Glycerol production and transformation: a critical review with particular emphasis on glycerol reforming reaction for producing hydrogen in conventional and membrane reactors. Membranes 7
Barnebey HL, Brown AC (1948) Continuous fat splitting plants using the colgate-emery process. J Am Oil Chemists’ Soc. https://doi.org/10.1007/BF02579733
Beyer AS, Meier J, Jiménez-Muñoz M et al (2023) New microalgae media formulated with completely recycled phosphorus originating from agricultural sidestreams. J Appl Phycol 1:1–16. https://doi.org/10.1007/S10811-023-03005-Z/TABLES/6
Bharti MK, Chalia S, Thakur P et al (2021) Nanoferrites heterogeneous catalysts for biodiesel production from soybean and canola oil: a review. Environ Chem Lett 19:3727–3746. https://doi.org/10.1007/S10311-021-01247-2
Cai G, Moghaddam L, O’Hara IM, Zhang Z (2019) Microbial oil production from acidified glycerol pretreated sugarcane bagasse by Mortierella isabellina. RSC Adv 9:2539–2550
Carota E, Petruccioli M, D’Annibale A, Crognale S (2020) Mixed glycerol and orange peel-based substrate for fed-batch microbial biodiesel production. Heliyon 6(9):e04801. https://doi.org/10.1016/j.heliyon.2020.e04801
Casali B, Brenna E, Parmeggiani F et al (2021) Enzymatic methods for the manipulation and valorization of soapstock from vegetable oil refining processes. Sustain Chem. https://doi.org/10.3390/suschem2010006
Chaudhary N, Ngai MO, Simpson B (2012) Comparison of glucose, glycerol and crude glycerol fermentation by escherichia coli K12. J Bioprocess Biotech 1:1. https://doi.org/10.4172/2155-9821.S1-001
Cheirsilp B, Kitcha S, Torpee S (2012) Co-culture of an oleaginous yeast Rhodotorula glutinis and a microalga Chlorella vulgaris for biomass and lipid production using pure and crude glycerol as a sole carbon source. Annal Microbiol 62:987–993. https://doi.org/10.1007/S13213-011-0338-Y
Chen Z, Reznicek WD, Wan C (2018) Deep eutectic solvent pretreatment enabling full utilization of switchgrass. Biores Technol 263:40–48
Chen J, Zhang X, Tyagi RD (2021) Impact of nitrogen on the industrial feasibility of biodiesel production from lipid accumulated in oleaginous yeast with wastewater sludge and crude glycerol. Energy. https://doi.org/10.1016/j.energy.2020.119343
Chilakamarry CR, Mimi Sakinah AM, Zularisam AW et al (2021a) Technological perspectives for utilisation of waste glycerol for the production of biofuels: a review. Elsevier, Amsterdam
Chilakamarry CR, Sakinah AMM, Zularisam AW, Pandey A (2021b) Glycerol waste to value added products and its potential applications. Syst Microbiol Biomanuf 1
Choi H-J, Yu S-W (2015) Influence of crude glycerol on the biomass and lipid content of microalgae. Biotechnol Biotechnol Equip 29:506–513
Choi WJ, Hartono MR, Chan WH, Yeo SS (2011) Ethanol production from biodiesel-derived crude glycerol by newly isolated Kluyvera cryocrescens. Appl Microbiol Biotechnol 89:1255–1264. https://doi.org/10.1007/S00253-010-3076-3/FIGURES/5
Cofré O, Ramírez M, Gómez JM, Cantero D (2016) Pilot scale fed-batch fermentation in a closed loop mixed reactor for the biotransformation of crude glycerol into ethanol and hydrogen by Escherichia coli MG1655. Biomass Bioenerg 91:37–47
de Carvalho PLO, Moreira I, Martins EN et al (2012) Crude glycerine in diets for piglets. Revista Brasileira De Zootecnia 41:1654–1661. https://doi.org/10.1590/S1516-35982012000700014
Dias C, Santos J, Reis A, da Silva TL (2019) Yeast and microalgal symbiotic cultures using low-cost substrates for lipid production. Bioresour Technol Rep 7:100261
Dias C, Santos JAL, Reis A, da Silva TL (2022) Impact of brewery wastewater inhibitors in pure and mixed cultures of the yeast Rhodosporidium toruloides NCYC 921 and the microalga Tetradesmus obliquus ACOI 204/07. Biochem Eng J 185:108518
Elahinik A, Haarsma M, Abbas B et al (2022) Glycerol conversion by aerobic granular sludge. Water Res. https://doi.org/10.1016/j.watres.2022.119340
Elsayed M, Abomohra AE-F (2022) Sequential algal biofuel production through whole biomass conversion. Handbook of Algal Biofuels. Elsevier, Amsterdam, pp 385–404
Elsayed M, Abomohra AE-FF, Ai P et al (2018) Biorefining of rice straw by sequential fermentation and anaerobic digestion for bioethanol and/or biomethane production: Comparison of structural properties and energy output. Biores Technol 268:183–189. https://doi.org/10.1016/j.biortech.2018.07.130
Elsayed M, Ran Y, Ai P et al (2020) Innovative integrated approach of biofuel production from agricultural wastes by anaerobic digestion and black soldier fly larvae. J Clean Prod 263:121495. https://doi.org/10.1016/j.jclepro.2020.121495
Elsayed M, El-Mashad HM, Mahmoud-Aly M (2022a) Microbial-mediated lignocellulose conversion to biodiesel. Waste-to-Energy. Springer, Berlin, pp 449–477
Elsayed M, Li W, Abdalla N et al (2022b) Innovative approach for rapeseed straw recycling using black solider fly larvae: towards enhanced energy recovery. Renew Energy 188:211–222
Eraky M, Elsayed M, Qyyum MA et al (2022) A new cutting-edge review on the bioremediation of anaerobic digestate for environmental applications and cleaner bioenergy. Environ Res 213:113708
Farghali M, Osman AI, Mohamed IMA et al (2023a) Strategies to save energy in the context of the energy crisis: a review. Environ Chem Lett 214(21):2003–2039. https://doi.org/10.1007/S10311-023-01591-5
Farghali M, Mohamed IM, Hassan D et al (2023b) Kinetic modeling of anaerobic co-digestion with glycerol: implications for process stability and organic overloads. Biochem Eng J 199:109061
Fei Q, O’Brien M, Nelson R, et al (2016) Enhanced lipid production by Rhodosporidium toruloides using different fed-batch feeding strategies with lignocellulosic hydrolysate as the sole carbon source. Biotechnol Biofuels 9(1). https://doi.org/10.1186/s13068-016-0542-x
Fontanille P, Kumar V, Christophe G, Nouaille R, Larroche C (2012) Bioconversion of volatile fatty acids into lipids by the oleaginous yeast Yarrowia lipolytica. Biores Technol 114:443–449. https://doi.org/10.1016/j.biortech.2012.02.091
Fukuzumi K, Koyama Y (1957) Fat splitting by the twitchell process at low temperature. J Am Oil Chem Soc. https://doi.org/10.1007/BF02638076
De Bhowmick G, Koduru L, Sen R (2015) Metabolic pathway engineering towards enhancing microalgal lipid biosynthesis for biofuel application—a review. Renew Sustain Energy Rev 1(50):1239–1253. https://doi.org/10.1016/J.RSER.2015.04.131
Ge JC, Wu G, Yoo B-O, Choi NJ (2022) Effect of injection timing on combustion, emission and particle morphology of an old diesel engine fueled with ternary blends at low idling operations. Energy 253:124150
Grama BS, Agathos SN, Jeffryes CS (2016) Balancing photosynthesis and respiration increases microalgal biomass productivity during photoheterotrophy on glycerol. ACS Sustain Chem Eng 4:1611–1618
Guerfali M, Ayadi I, Sassi HE et al (2020) Biodiesel-derived crude glycerol as alternative feedstock for single cell oil production by the oleaginous yeast Candida viswanathii Y-E4. Ind Crops Prod. https://doi.org/10.1016/j.indcrop.2020.112103
Han SFS-F, Jin W, Tu R et al (2016) Optimization of aeration for biodiesel production by Scenedesmus obliquus grown in municipal wastewater. Bioprocess Biosyst Eng 39:1073–1079. https://doi.org/10.1007/s00449-016-1585-x
Hansen CF, Hernandez A, Mullan BP et al (2009) A chemical analysis of samples of crude glycerol from the production of biodiesel in Australia, and the effects of feeding crude glycerol to growing-finishing pigs on performance, plasma metabolites and meat quality at slaughter. Anim Prod Sci 49:154–161. https://doi.org/10.1071/EA08210
Hassanpour M, Cai G, Gebbie LK et al (2019) Co-utilization of acidified glycerol pretreated-sugarcane bagasse for microbial oil production by a novel Rhodosporidium strain. Eng Life Sci 19:217–228
Hassanpour M, Abbasabadi M, Moghaddam L et al (2020a) Mild fractionation of sugarcane bagasse into fermentable sugars and β-O-4 linkage-rich lignin based on acid-catalysed crude glycerol pretreatment. Biores Technol 318:124059
Hassanpour M, Abbasabadi M, Strong J et al (2020b) Scale-up of two-step acid-catalysed glycerol pretreatment for production of oleaginous yeast biomass from sugarcane bagasse by Rhodosporidium toruloides. Biores Technol 313:123666
Hoekman SK, Broch A, Robbins C et al (2012) Review of biodiesel composition properties and specifications. Renew Sustain Energy Rev 16(1):143–169. https://doi.org/10.1016/j.rser.2011.07.143
Hu S, Luo X, Wan C, Li Y (2012) Characterization of crude glycerol from biodiesel plants. J Agric Food Chem 60:5915–5921. https://doi.org/10.1021/JF3008629/ASSET/IMAGES/MEDIUM/JF-2012-008629_0004.GIF
Iasimone F, Zuccaro G, D’Oriano V et al (2018) Combined yeast and microalgal cultivation in a pilot-scale raceway pond for urban wastewater treatment and potential biodiesel production. Water Sci Technol 77:1062–1071
Ishak S, Kamari A (2019) Biodiesel from black soldier fly larvae grown on restaurant kitchen waste. Environ Chem Lett 17:1143–1150
Johnson EE, Rehmann L (2016) The role of 1,3-propanediol production in fermentation of glycerol by Clostridium pasteurianum. Biores Technol 209:1–7. https://doi.org/10.1016/J.BIORTECH.2016.02.088
Kitcha S, Cheirsilp B (2014) Enhanced lipid production by co-cultivation and co-encapsulation of oleaginous yeast Trichosporonoides spathulata with microalgae in alginate gel beads. Appl Biochem Biotechnol 173:522–534
Kumar LR, Kumar S, Zhang X, Tyagi RD (2019a) Energy balance for biodiesel production processes using microbial oil and scum. Biores Technol 272:379–388. https://doi.org/10.1016/j.biortech.2018.10.071
Kumar LR, Yellapu SK, Tyagi RD, Zhang X (2019b) A review on variation in crude glycerol composition, bio-valorization of crude and purified glycerol as carbon source for lipid production. Biores Technol 293:122155
Kumar LR, Yellapu SK, Tyagi RD, Drogui P (2021) Microbial lipid and biodiesel production from municipal sludge fortified with crude glycerol medium using pH-based fed-batch strategy. J Environ Chem Eng 9:105032
Lazar Z, Liu N, Stephanopoulos G (2018) Holistic approaches in lipid production by Yarrowia lipolytica. Trends Biotechnol 36:1157–1170
Lee JH, Yoo HY, Lee SK et al (2020) Significant impact of casein hydrolysate to overcome the low consumption of glycerol by Klebsiella aerogenes ATCC 29007 and its application to bioethanol production. Energy Convers Manage 221:113181. https://doi.org/10.1016/J.ENCONMAN.2020.113181
Li M, Alotaibi MKH, Li L, Abomohra AE-F (2022) Enhanced waste glycerol recycling by yeast for efficient biodiesel production: Towards waste biorefinery. Biomass Bioenerg 159:106410
Ling J, Nip S, Cheok WL et al (2014) Lipid production by a mixed culture of oleaginous yeast and microalga from distillery and domestic mixed wastewater. Biores Technol 173:132–139
Madadi M, Elsayed M, Sun F et al (2023) Sustainable lignocellulose fractionation by integrating p-toluenesulfonic acid/pentanol pretreatment with mannitol for efficient production of glucose, native-like lignin, and furfural. Bioresour Technol 371:128591
Martín M, Grossmann I (2014) Integrating glycerol to methanol vs. glycerol to ethanol within the production of biodiesel from algae. Computer Aided Chemical Engineering. Elsevier, Amsterdam, pp 85–90
Mu Y, Teng H, Zhang DJ et al (2006) Microbial production of 1,3-propanediol by Klebsiella pneumoniae using crude glycerol from biodiesel preparations. Biotech Lett 28:1755–1759. https://doi.org/10.1007/s10529-006-9154-z
Nitbani FO, Tjitda PJP, Nurohmah BA, Wogo HE (2020) Preparation of fatty acid and monoglyceride from vegetable oil. J Oleo Sci 69:277–295. https://doi.org/10.5650/jos.ess19168
Nwachukwu RES, Shahbazi A, Wang L et al (2013) Optimization of cultural conditions for conversion of glycerol to ethanol by Enterobacter aerogenes S012. AMB Express 3:1–10
Oh BR, Seo JW, Heo SY et al (2011) Efficient production of ethanol from crude glycerol by a Klebsiella pneumoniae mutant strain. Biores Technol 102:3918–3922. https://doi.org/10.1016/J.BIORTECH.2010.12.007
Osman AI, Mehta N, Elgarahy AM et al (2021a) Conversion of biomass to biofuels and life cycle assessment: a review. Environ Chem Lett 19:4075–4118
Osman AI, Qasim U, Jamil F et al (2021b) Bioethanol and biodiesel: bibliometric mapping, policies and future needs. Renew Sustain Energy Rev 152:111677
Osman AI, Elgarahy AM, Eltaweil AS et al (2023a) Biofuel production, hydrogen production and water remediation by photocatalysis, biocatalysis and electrocatalysis. Environ Chem Lett 21:1315–1379
Osman AI, Lai ZY, Farghali M et al (2023b) Optimizing biomass pathways to bioenergy and biochar application in electricity generation, biodiesel production, and biohydrogen production. Environ Chem Lett 21:1–67
Pan H, Xia Q, Wang Y et al (2022) Recent advances in biodiesel production using functional carbon materials as acid/base catalysts. Fuel Process Technol 237:107421. https://doi.org/10.1016/J.FUPROC.2022.107421
Patel A, Karageorgou D, Rova E et al (2020) An overview of potential oleaginous microorganisms and their role in biodiesel and omega-3 fatty acid-based industries. Microorganisms. https://doi.org/10.3390/microorganisms8030434
Patil Y, Junghare M, Müller N (2017) Fermentation of glycerol by Anaerobium acetethylicum and its potential use in biofuel production. Microb Biotechnol 10:203–217
Peters MA, Alves CT, Wang J, Onwudili JA (2022) Subcritical water hydrolysis of fresh and waste cooking oils to fatty acids followed by esterification to fatty acid methyl esters: detailed characterization of feedstocks and products. ACS Omega. https://doi.org/10.1021/acsomega.2c05972
Posada JA, Cardona CA (2010) Design and analysis of fuel ethanol production from raw glycerol. Energy 35:5286–5293
Qian X, Zhou X, Chen L et al (2020) Bioconversion of volatile fatty acids into lipids by the oleaginous yeast Apiotrichum porosum DSM27194. Fuel. https://doi.org/10.1016/j.fuel.2020.119811
Qin L, Liu L, Zeng A-P, Wei D (2017) From low-cost substrates to single cell oils synthesized by oleaginous yeasts. Biores Technol 245:1507–1519
Qin L, Liu L, Wang Z et al (2018) Efficient resource recycling from liquid digestate by microalgae-yeast mixed culture and the assessment of key gene transcription related to nitrogen assimilation in microalgae. Biores Technol 264:90–97
Ran Y, Elsayed M, Eraky M et al (2022) Sequential production of biomethane and bioethanol through the whole biorefining of rice straw: analysis of structural properties and mass balance. Biomass Convers Biorefinery 1:1–13. https://doi.org/10.1007/s13399-022-02548-4
Rana MS, Prajapati SK (2021) Stimulating effects of glycerol on the growth phycoremediation and biofuel potential of Chlorella pyrenoidosa cultivated in wastewater. Environ Technol Innov 24:102082. https://doi.org/10.1016/j.eti.2021.102082
Ratledge C, Cohen Z (2008) Microbial and algal oils: Do they have a future for biodiesel or as commodity oils? Lipid Technol 20:155–160. https://doi.org/10.1002/lite.200800044
Rosenberg JN, Mathias A, Korth K et al (2011) Microalgal biomass production and carbon dioxide sequestration from an integrated ethanol biorefinery in Iowa: a technical appraisal and economic feasibility evaluation. Biomass Bioenerg 35:3865–3876
Saidi M, Gohari MH, Ramezani AT (2020) Hydrogen production from waste gasification followed by membrane filtration: a review. Environ Chem Lett 18:1529–1556. https://doi.org/10.1007/S10311-020-01030-9
Sawers RG, Clark DP (2004) Fermentative pyruvate and acetyl-coenzyme a metabolism. EcoSal plus 1:10–1128
Soccol CR, Neto CJD, Soccol VT et al (2017) Pilot scale biodiesel production from microbial oil of Rhodosporidium toruloides DEBB 5533 using sugarcane juice: performance in diesel engine and preliminary economic study. Biores Technol 223:259–268
Sun C, Ren H, Sun F et al (2022) Glycerol organosolv pretreatment can unlock lignocellulosic biomass for production of fermentable sugars: present situation and challenges. Biores Technol 344:126264. https://doi.org/10.1016/j.biortech.2021.126264
Sunarno JN, Prasertsan P, Duangsuwan W et al (2019) Biodiesel derived crude glycerol and tuna condensate as an alternative low-cost fermentation medium for ethanol production by Enterobacter aerogenes. Ind Crops Prod 138:111451. https://doi.org/10.1016/J.INDCROP.2019.06.014
Sunarno JN, Prasertsan P, Duangsuwan W et al (2020) Improve biotransformation of crude glycerol to ethanol of Enterobacter aerogenes by two-stage redox potential fed-batch process under microaerobic environment. Biomass Bioenerg 134:105503
Tang DYY, Yew GY, Koyande AK et al (2020a) Green technology for the industrial production of biofuels and bioproducts from microalgae: a review. Environ Chem Lett 18:1967–1985
Tang S, Dong Q, Fang Z et al (2020b) Microbial lipid production from rice straw hydrolysates and recycled pretreated glycerol. Biores Technol 312:123580
Thapa LP, Lee SJ, Yang X et al (2015) Improved bioethanol production from metabolic engineering of Enterobacter aerogenes ATCC 29007. Process Biochem 50:2051–2060
Thompson JC, He BB (2006) Characterization of crude glycerol from biodiesel production from multiple feedstocks. Appl Eng Agric 22:261–265. https://doi.org/10.13031/2013.20272
Trchounian K, Blbulyan S, Trchounian A (2013) Hydrogenase activity and proton-motive force generation by Escherichia coli during glycerol fermentation. J Bioenerg Biomembr 45:253–260. https://doi.org/10.1007/S10863-012-9498-0/METRICS
Tu R, Jin W, Wang M et al (2016) Improving of lipid productivity of the biodiesel promising green microalga Chlorella pyrenoidosa via low-energy ion implantation. J Appl Phycol 28:2159–2166
Uprety BK, Dalli SS, Rakshit SK (2017) Bioconversion of crude glycerol to microbial lipid using a robust oleaginous yeast Rhodosporidium toruloides ATCC 10788 capable of growing in the presence of impurities. Energy Convers Manage 135:117–128. https://doi.org/10.1016/J.ENCONMAN.2016.12.071
Valerio O, Horvath T, Pond C et al (2015) Improved utilization of crude glycerol from biodiesel industries: synthesis and characterization of sustainable biobased polyesters. Ind Crops Prod. https://doi.org/10.1016/j.indcrop.2015.10.019
Vieira JPF, Ienczak JL, Rossell CEV, et al (2014) Microbial lipid production: screening with yeasts grown on Brazilian molasses. Biotechnol Lett 36(12):2433–2442. https://doi.org/10.1007/s10529-014-1624-0
Vikromvarasiri N, Haosagul S, Boonyawanich S, Pisutpaisal N (2016) Microbial dynamics in ethanol fermentation from glycerol. Int J Hydrogen Energy. https://doi.org/10.1016/j.ijhydene.2016.04.188
Da Silva VZ, Ourique LJ, de David C, Ayub MA (2020) Construction of recombinant klebsiella pneumoniae to increase ethanol production on residual glycerol fed-batch cultivations. Appl Biochem Biotechnol 192(4):1147–1162. https://doi.org/10.1007/S12010-020-03397-5/TABLES/3
Wang S, Uzoejinwa BB, Abomohra A et al (2018) Characterization and pyrolysis behavior of the green microalga Micractinium conductrix grown in lab-scale tubular photobioreactor using Py-GC/MS and TGA/MS. J Anal Appl Pyrol 135:340–349
Wang S, Shang H, Abomohra A, Wang Q (2019) One-step conversion of microalgae to alcohols and esters through co-pyrolysis with biodiesel-derived glycerol. Energy Convers Manage 198:111792
Wang S, Hu S, Shang H et al (2020) Study on the co-operative effect of kitchen wastewater for harvest and enhanced pyrolysis of microalgae. Bioresour Technol 317:123983. https://doi.org/10.1016/j.biortech.2020.123983
Wan Isahak WN, Che Ramli ZA, Ismail M, Mohd Jahim J, Yarmo MA (2015) Recovery and purification of crude glycerol from vegetable oil transesterification. Sep Purif Rev. https://doi.org/10.1080/15422119.2013.851696
Wu Y, Jiang L, Lin Y et al (2019) Novel crude glycerol pretreatment for selective saccharification of sugarcane bagasse via fast pyrolysis. Biores Technol 294:122094. https://doi.org/10.1016/j.biortech.2019.122094
Xiao Y, Xiao G, Varma A (2013) A universal procedure for crude glycerol purification from different feedstocks in biodiesel production: experimental and simulation study. In: Industrial and Engineering Chemistry Research
Xu S, Elsayed M, Ismail GA et al (2019) Evaluation of bioethanol and biodiesel production from Scenedesmus obliquus grown in biodiesel waste glycerol: a sequential integrated route for enhanced energy recovery. Energy Convers Manage 197:111907. https://doi.org/10.1016/j.enconman.2019.111907
Xue F, Miao J, Zhang X, Tan T (2010) A new strategy for lipid production by mix cultivation of Spirulina platensis and Rhodotorula glutinis. Appl Biochem Biotechnol 160:498–503
Xue LL, Chen HH, Jiang JG (2017) Implications of glycerol metabolism for lipid production. Prog Lipid Res 68:12–25. https://doi.org/10.1016/J.PLIPRES.2017.07.002
Yazdani SS, Gonzalez R (2007) Anaerobic fermentation of glycerol: a path to economic viability for the biofuels industry. Elsevier Current Trends, Amsterdam
Zhang Z, Wong HH, Albertson PL et al (2013) Laboratory and pilot scale pretreatment of sugarcane bagasse by acidified aqueous glycerol solutions. Biores Technol 138:14–21
Zhang X, Yan S, Tyagi RD et al (2014) Wastewater sludge as raw material for microbial oils production. Appl Energy 135:192–201
Zhang X, Yan S, Tyagi RD et al (2016) Energy balance of biofuel production from biological conversion of crude glycerol. J Environ Manage 170:169–176
Zhao M, Wang Y, Zhou W et al (2023) Co-valorization of crude glycerol and low-cost substrates via oleaginous yeasts to micro-biodiesel: status and outlook. Renew Sustain Energy Rev 180:113303
Zhou W, Wang Y, Zhang J et al (2021) A metabolic model of Lipomyces starkeyi for predicting lipogenesis potential from diverse low-cost substrates. Biotechnol Biofuels 14:148
Zhu Z, Rehman K, Yu Y et al (2019) De novo transcriptome sequencing and analysis revealed the molecular basis of rapid fat accumulation by black soldier fly (Hermetia illucens, L.) for development of insectival biodiesel. Biotechnol Biofuels. https://doi.org/10.1186/s13068-019-1531-7
Acknowledgements
The current work was supported by Chengdu University’s start-up funds (No. 2081923028). The authors wish to acknowledge the support of the Bryden Centre project (Project ID VA5048). The Bryden Centre project is supported by the European Union’s INTERREG VA Programme, managed by the Special EU Programmes Body (SEUPB).
Funding
The authors have not disclosed any funding.
Author information
Authors and Affiliations
Contributions
ME contributed to conceptualization and writing and review. ME was involved in data gathering and writing and review. AIO contributed to review and review and editing. JW was involved in writing and review. MF contributed to writing and review and editing. AKR was involved in figure editing and review. IHY contributed to data gathering. DH was involved in writing and review and editing. AA contributed to writing and review and editing.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper. The views and opinions expressed in this review do not necessarily reflect those of the European Commission or the Special EU Programs Body (SEUPB).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Elsayed, M., Eraky, M., Osman, A.I. et al. Sustainable valorization of waste glycerol into bioethanol and biodiesel through biocircular approaches: a review. Environ Chem Lett 22, 609–634 (2024). https://doi.org/10.1007/s10311-023-01671-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10311-023-01671-6